2,4,5-Trichlorophenoxyacetic Acid: Legacy, Risks, and Evolving Realities
Historical Development
2,4,5-Trichlorophenoxyacetic acid, better known as 2,4,5-T, started gathering attention in the 1940s during the push to boost agricultural production in North America and Europe. Chemists wanted a way to control broadleaf weeds that threatened food crops, so they turned to synthetic auxins like 2,4,5-T. By the 1960s, the compound got swept up in policies that pushed efficiency at almost any cost, a mindset very much of its era. Government-backed field experiments and industrial-scale production took off, but little thought went into the environmental outfall or the long-term risks to those who manufactured and applied these chemicals. Its notoriety hit a peak during the Vietnam War, through its use in Agent Orange, a legacy that still shadows discussion of this compound.
Product Overview
2,4,5-T, in practical terms, hit the market promising farmers and land managers the dream of tidy, productive fields. The chemical, formulated most often as a white crystalline powder, made easy work of brush and weeds competing with crops like wheat, soybeans, and corn. Large timber companies sprayed it across forests to clear undergrowth. Municipalities kept train tracks and highways weed-free using the same product. At the time, the run-off effects into rivers, bioaccumulation, and risks to non-target plants and animals rarely got more than a nod, a trend that seems starkly out-of-step compared to today’s risk management culture.
Physical and Chemical Properties
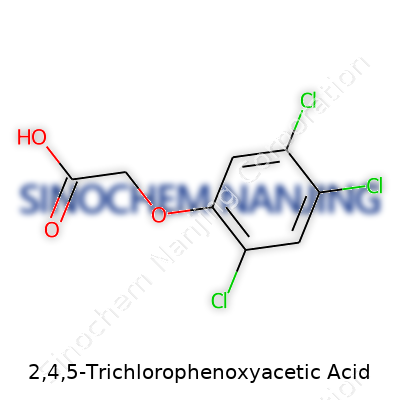
2,4,5-T’s chemical formula—C8H5Cl3O3—tells a story of three chlorine atoms tightly coupled to a phenoxyacetic structure. This configuration allows the molecule to mimic plant growth hormones, which tricks unwanted broadleaf plants into uncontrolled growth and, eventually, death. The compound, under standard conditions, appears as a white, odorless powder with limited solubility in water but better solvency in organic solvents. These physical traits made tank mixing and aerial spraying simple, but the limited water solubility made it stubbornly persistent in some soils and sediments, posing clean-up challenges decades after its last legal applications.
Technical Specifications and Labeling
Labels for 2,4,5-T during its heyday rarely included much beyond the standard cautionary statements and suggested application rates. Over time, pressure from toxicologists and activists forced manufacturers to include stronger warnings, especially after contaminant problems with dioxin by-products became public. It became clear that the technical details behind purity and allowable co-contaminant levels mattered far more than most regulators had appreciated. While today's environmental labeling rules would never allow the old practice of glossing over ancillary risks, the lag in updating labels back then contributed to a painful learning curve for everyone in the supply chain.
Preparation Method
Chemical producers synthesized 2,4,5-T by reacting 2,4,5-trichlorophenol with chloroacetic acid. Simple enough in theory, but cracks soon appeared when poorly controlled processes left behind 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD), a profoundly toxic by-product. Factory workers near contaminated sites and towns downstream from production plants learned the hard way that cheap shortcuts could haunt whole communities. Uncontrolled waste discharge and inadequate workplace protections put the real human cost of mass chemical synthesis in sharp relief. The long, slow grind of regulatory oversight caught up eventually, but not before irreversible mistakes.
Chemical Reactions and Modifications
Chemists explored modifications to 2,4,5-T, hoping to boost weed control or manage volatility. Formulation additives like surfactants helped spread the herbicide across plant surfaces more efficiently, but tweaks to the core molecule rarely solved the biggest headaches. Many of these experiments ended up abandoned, with researchers recognizing the risk of even more toxic by-products or persistent residues. The reluctance to repeat earlier oversights has since guided stricter lab protocols, shifting the culture toward proof of safety as a precondition for market entry rather than an afterthought.
Synonyms and Product Names
The chemical’s mouthful name stuck, but the market long favored shorthand. Labels touting “2,4,5-T” or less commonly, trade names like Brush Killer and Woodland Herbicide, lined co-op shelves. For insiders, the catch-all “phenoxy herbicide” summed up a class that included 2,4-D, which escaped much of the late-century stigma. Today, almost any mention of 2,4,5-T reads as a warning, and with dioxin’s reputation intertwined, most countries treat the brand and the class with extra scrutiny.
Safety and Operational Standards
Industrial safety around 2,4,5-T trailed far behind its widespread use. Workers mixed and sprayed the chemical wearing little more than coveralls, with few enforced measures to prevent inhalation or skin contact. Large-scale releases of TCDD during production harmed entire towns, as seen in Seveso, Italy, and in U.S. communities close to manufacturing sites. Later, worker safety standards—ventilation controls, chemical-resistant gloves and suits, regular blood dioxin checks—raised the bar, but only after tragic failures. Many lessons from that era shaped today’s regulatory framework, where thorough environmental impact assessments and occupational exposure limits form the backbone of chemical oversight. From a personal perspective, talking with retired field workers makes that lost vigilance hit home.
Application Area
Farming landscapes across much of the Northern Hemisphere once bore the mark of 2,4,5-T—rows of crops with edges burnt clean of broadleaf weeds, woodlands thinned to accelerate timber stands, and rights-of-way kept passable for utilities and road crews. The post-war drive to mechanize and increase yields left few corners of the world untouched by synthetic herbicides. At its peak, nearly every farm supplier carried drums of 2,4,5-T alongside other so-called miracle aids. With rising environmental awareness came bans and strict restrictions, shifting pest management toward integrated tactics and less toxic alternatives.
Research and Development
Research around 2,4,5-T, even after large-scale bans, keeps offering case studies on long-term contamination, toxicology, and chemical fate. Soil scientists still map dioxin residues decades after last application. Medical researchers document higher rates of some diseases among people who lived or worked near contaminated sites. The scale of field studies into safer weed management methods now reflects the determination to avoid repeating the mistakes that marked the 2,4,5-T era. Many public funding agencies now prioritize projects on remediation over chemical shortcuts.
Toxicity Research
The toxic legacy of 2,4,5-T revolves more around its chemical hitchhiker, TCDD, than the parent molecule itself. Toxicologists studying TCDD found links to cancers, birth defects, and disruptions across almost every natural biological process. Large cohort studies of Vietnamese populations, U.S. veterans, and factory workers strengthened these links over decades, driving both policy change and public mistrust of all phenoxy herbicides. Many of those affected live with chronic illnesses, while broad environmental surveys keep adding grim detail to what dioxin exposure can do at the genetic and cellular levels. The experience underscores how regulatory shortcuts in one era can impose suffering on generations that follow.
Future Prospects
The world has little appetite for another round of 2,4,5-T. Legal use now exists only in a few countries, mainly for regulated research or controlled clean-up trials. The chemical’s story has shaped a more transparent culture around chemical approval, where public health and environmental legacy arguments outweigh the promise of short-term gains. Modern researchers chase biopesticides and precision weed management—systems that draw on biology, not brute chemistry, for solutions. As a cautionary example, 2,4,5-T sits in the background every time a new molecule seeks approval. Regulatory authorities demand unparalleled evidence of safety from lab bench to farm field, and community involvement in risk discussions is non-negotiable. The industry’s institutional memory of this compound keeps pushing the field toward innovations with fewer hidden costs, and I find myself hoping that future generations will look back at cases like this as the benchmarks they never again want to meet.
What is 2,4,5-Trichlorophenoxyacetic Acid used for?
Looking Back at the Role of 2,4,5-Trichlorophenoxyacetic Acid
Most people outside agriculture or environmental science circles don’t talk much about 2,4,5-Trichlorophenoxyacetic Acid, usually called 2,4,5-T. For many decades, this synthetic compound played a role in how farmers and governments dealt with weeds and unwanted vegetation. People developed 2,4,5-T as a herbicide, and it quickly found its way into both commercial agriculture and large-scale land management, especially in the middle of the twentieth century.
Herbicide and Its Legacy
Farmers turned to 2,4,5-T because it tackled broadleaf weeds that sapped energy and nutrients from crops. Alongside its cousin 2,4-D, this compound made up the core ingredient in the defoliant mix called Agent Orange. During the Vietnam War, the U.S. military sprayed Agent Orange over jungles to remove enemy cover. This turned out to have consequences that keep scientists, environmentalists, and policymakers talking to this day.
As someone who’s spent time in rural communities where pesticide use isn’t just a side story but part of daily life, I’ve seen the push and pull between the desire to control weeds and the need to safeguard human health and ecosystems. What made 2,4,5-T’s story so fraught came down to more than just killing weeds: the byproducts. In particular, it often carried a contaminant, dioxin, which showed up at troubling levels.
Health and Environmental Concerns
Studies in the 1970s revealed a strong link between dioxin exposure and health problems. Researchers observed elevated birth defects, cancers, and other illnesses—especially among those exposed during the Vietnam conflict and in communities close to manufacturing sites. Dioxins linger in the environment and accumulate in the food chain. In my own research, I’ve come across soil reports and fish sampling data that trace dioxin residues decades after spraying stopped. This chemical’s stubborn presence helps explain the deep concern surrounding 2,4,5-T’s history.
In 1985, after mounting scientific evidence and increasing public outcry, regulators pulled 2,4,5-T from the market in the United States and many other countries followed. For people living near contaminated sites, the end of production didn’t erase the legacy right away. Cleaning up affected areas takes money, time, and political will. Residents, especially those who rely on fishing, hunting, or farming for their livelihoods, have spent years pressing for answers and action.
Lessons for Modern Agriculture and Policy
Modern farming keeps hunting for new solutions to weed control. Safer alternatives have replaced 2,4,5-T, but the story highlights how chemical shortcuts sometimes come back to haunt communities. Regulatory oversight has improved with lessons from this era, and today’s chemicals often face higher hurdles before approval. At the same time, people debate the right balance—fighting weeds while also considering long-term health and soil quality.
Farmers, scientists, and local governments need open lines of communication. Ongoing research into safe weed management, combined with transparency about the risks, helps everyone make better choices. Experience with 2,4,5-T shows why asking questions before large-scale deployment of any new chemical really matters. Future generations deserve fields and waterways that don’t carry invisible scars from the shortcuts of the past.
Is 2,4,5-Trichlorophenoxyacetic Acid hazardous to human health?
The History Behind the Controversy
Many people have heard of Agent Orange—the defoliant used in the Vietnam War. One of its main ingredients was 2,4,5-Trichlorophenoxyacetic acid, or 2,4,5-T for short. My own familiarity with stories from Vietnam veterans began long before I dove into the research; the lingering health complaints from those soldiers made me wonder about chemicals like this.
How Exposure Happens
Farmers and groundskeepers sprayed 2,4,5-T to kill weeds across fields and forests for decades. In the process, they often inhaled drift or got the chemical on their skin. Even without set out to research, family stories paint a picture—dad’s hands, cracked from work, absorbing things you can’t see, lingering scents in the barn. Households near sprayed fields sometimes detected a chemical odor, raising concerns over what crept into local water or food supplies. No one who grew up surrounded by farmland shrugs off the idea that what goes on crops sooner or later enters wells or kitchen tables.
Health Risks and What Science Shows
2,4,5-T on its own poses some risks, but the real trouble comes from dioxins—a group of toxic byproducts made during its production, with TCDD being the most infamous. People exposed to dioxins face higher risks of cancer, birth defects, liver issues, and hormone problems. The U.S. Environmental Protection Agency classifies TCDD as a human carcinogen. Studies of populations with heavy contact (like factory workers or communities near chemical plants) tracked increased rates of cancer and developmental problems.
The fate of a chemical in the environment matters too. 2,4,5-T breaks down more slowly in some soils. It can wash into streams, linger in sediment, and build up in food chains. I have seen community gardens tested for contaminants decades after old herbicide use, hoping the soil tells a clean story. People drinking from rural water wells have asked, “Are these safe?” because nothing dissipates overnight.
Regulation and Bans
In the U.S., regulators decided that the risk stacked too high against the benefits. Commercial use of 2,4,5-T left the market during the late 1970s and early 1980s, and most countries agreed, following similar bans. Old stores of the chemical sit locked away in environmental containment sites, underlining the consensus: it’s not safe to keep it in active use.
Lessons for Today
The story of 2,4,5-T highlights a lesson about trusting technology before understanding long-term effects. Blanket trust in labels and assurances from manufacturers or regulatory agencies does not always protect communities. Transparency around chemical ingredients and production methods needs more emphasis than ever. Real-world experience from people working with or living near agricultural chemicals trumps sterile test results alone.
Promoting better training for anyone handling pesticides, widening access to protective equipment, and increasing investment in chemical safety research yield practical steps forward. Respect for landscape and human health should guide every decision about what gets sprayed, where, and how often. Supporting farmers in moving to safer alternatives and backing research into non-chemical weed control moves the needle further away from old hazards. As someone who has watched neighbors fight for clean wells and local governments wrangle with cleanup bills, I see the ripple effects long after the last barrel leaves the field.
How should 2,4,5-Trichlorophenoxyacetic Acid be stored and handled?
Understanding the Risks
A lot has been said about the dangers of herbicides, but 2,4,5-Trichlorophenoxyacetic Acid (2,4,5-T) carries a reputation that stands out, not just for its weed-killing power but also for the risks in its use. Manufacturers once mixed it into products that went everywhere from large farms to suburban lawns, but its dark history includes connections to dioxin contamination and serious health stories. Anyone with experience in ag chemicals knows small mistakes can turn into big problems fast.
No Room for Lax Protocols
You can't cut corners on chemical hygiene. Storage starts with a basic rule: keep the area cool, well-ventilated, and locked up tight. Fumes from 2,4,5-T pack a punch, especially if they creep out in a closed room. Tucking containers away from sunlight and anything that throws heat helps cut the risk of the compound breaking down and releasing its nastier byproducts. All containers should carry labels right on the side where you can read them at a glance, even if you're in a hurry. In my own work, broken or faded labels have caused more headaches than anything, usually leading to a waste of time double-checking inventory or, worse, confusion during clean-up.
Don't Treat It Like Just Another Jug
One of the lessons learned in jobs around barns and garages with chemicals: not all plastic bottles offer the same protection. You want original, tightly sealed containers, stacked on spill pallets that catch leaks. Many of us have seen what happens when someone tries transferring herbicides or pesticides to second-hand jugs. Not only does that make it easy to lose track of what’s inside, but it also increases the odds of a spill if the container fails or reacts with the chemical. Clean storage areas keep dust, dirt, and water from getting into the mix, and it’s smart to keep an updated log for what comes in and goes out.
Hands, Eyes, and Lungs Deserve Real Protection
Handling calls for simple, sturdy habits. Gloves made of chemical-resistant material, long sleeves, and goggles—this isn’t overkill; it’s just practical. If you’ve ever gotten even a whiff of spilled herbicide, you know the respiratory irritation is real. Working outdoors or with exhaust fans cuts down on exposure. Immediate and thorough handwashing stops contamination from spreading to door handles, steering wheels, or kitchen countertops. Old-timers always remind anyone new that nothing good comes from rushing or skipping steps with substances like 2,4,5-T.
Responding If Something Goes Wrong
Spills end up being the moment of truth. Absorbent materials like specialized pillows or sand take care of small leaks, but larger spills call for trained teams and plenty of personal protection. Proper disposal matters just as much as storage—nobody wants groundwater taking a hit because someone poured leftovers down a drain. Following local hazardous waste guidelines not only avoids fines but actually protects neighbors and animals who don’t get a say in what ends up in the soil.
Learning and Improvement
Every community that’s faced legacy chemical issues picked up wisdom the hard way. Regular training for anyone coming into contact with 2,4,5-T, from warehouse workers to farmhands, builds a safer culture. Keeping material safety data sheets handy and reviewing incident reports keeps everyone on their toes. Progress comes from treating safety rules as common sense, not as red tape.
What are the environmental impacts of 2,4,5-Trichlorophenoxyacetic Acid?
A Closer Look at 2,4,5-T
2,4,5-Trichlorophenoxyacetic acid, or 2,4,5-T, holds a heavy history. Used for decades as a herbicide, its most infamous role came during the Vietnam War as a major component in Agent Orange. These days, the impact of 2,4,5-T lingers in both soil and public debate. I remember reading farmer stories from rural America, folks who used this chemical on crops and now face soil contamination years later. Concern rarely came from those who bottled or sprayed it, but the evidence started growing stronger as research poured in about its toxic byproducts.
Soil and Water Contamination
Runoff after herbicide application leads straight to the heart of environmental damage. 2,4,5-T binds to soil, but not always tightly enough. Heavy rain pushes it deeper or washes it into waterways. The process doesn’t stop there. Broken-down forms of this chemical retain toxicity, mixing with groundwater and, in some cases, winding up in drinking supplies. One infamous byproduct, dioxin (TCDD), sports a toxicity level that causes fear even among seasoned toxicologists. I recall one scientist putting it simply: a local river system can carry the legacy of 2,4,5-T for decades, impacting fish, plants, birds, and anyone relying on that water for crops or cattle.
Impact on Wildlife
Animals don’t operate on schedules or steer clear of treated fields. They graze and nest wherever they find resources. Exposure to 2,4,5-T and its byproducts affects animal hormones, immune systems, and can cause birth defects. Studies following bird populations near treated fields noticed higher egg mortality and issues in chick development. Stories from biologists stuck with me: frogs with limb deformities, declining pollinator numbers, and foxes showing signs of ill health after denning near contaminated sites. Each example adds weight to the problem and gives a human face to scientific statistics.
Human Health Risks
Dioxins in 2,4,5-T lead to health concerns far beyond what most people predicted in the early years of herbicide use. Exposure links to increased cancer risk, reproductive problems, and immune system disruption. Findings from epidemiological studies accuse dioxin of acting even at very low doses. Plenty of Vietnam veterans, as well as farmers and chemical workers, suffered rashes, liver damage, and severe nerve disorders. During community health meetings in rural towns, parents worry about legacy contamination in old farmland. Stories from Vietnam still haunt generations, proving that contamination doesn’t follow a simple timetable or respect borders.
Stopping Further Harm and Moving Forward
Regulators in many countries banned 2,4,5-T, but soil and water cleaning continues. Solutions take time, money, and commitment. Soil washing and chemical treatments work but do not fit small-scale farms or tight budgets. Local food movements focus on organic growing and soil health, setting examples for responsible practices. People living near past contamination sites organize to test water and advocate for government intervention. Honest labeling, consumer education, and holding chemical companies accountable drive progress. I’ve seen neighborhood groups come together, pushing for community gardens and schoolyard plantings as ways to rebuild trust in the land. Small changes stack up over years, and each clear water well and untainted harvest means another family breathes easier.
Are there any regulations or restrictions on the use of 2,4,5-Trichlorophenoxyacetic Acid?
The Shadow Cast by One Herbicide
People often remember notorious chemicals for one reason—harm. 2,4,5-Trichlorophenoxyacetic acid, or 2,4,5-T, landed in the global spotlight for all the wrong ones. It showed up in fields, forests, and even war zones like Vietnam, always tagged alongside “Agent Orange,” that infamous defoliant. What shocked me, growing up in a rural area with relatives who farmed, was how lightly folks talked about these weedkillers. Few discussed contamination or health, just how well weeds disappeared.
Harm Isn't Just a Story—It Leaves Proof
Decades have rolled by, but the lessons of 2,4,5-T remain. Scientists documented links between 2,4,5-T and birth defects, liver problems, immune disorders, and higher cancer rates. Dioxin, the poisonous contaminant that clings to 2,4,5-T’s reputation, explains the panic. Studies dug up clusters of health issues from communities near factories, servicepeople exposed in Vietnam, and workers who handled the substance day in, day out. For me, stumbling on the EPA’s warning sheets for 2,4,5-T made clear society couldn't ignore the health stories pouring in from affected families worldwide.
Regulations Have Teeth—and For Good Reason
No agency wanted to carry the risk. The United States Environmental Protection Agency (EPA) banned almost all uses of 2,4,5-T by 1985. Europe set roadblocks earlier, taking a harder line after years of soil and food contamination. Most countries now count 2,4,5-T as restricted or outright banned. Australia’s official take mirrors that of the U.S.—the chemical cannot be used. Heath Canada followed suit, ditching any pretense of “safe use.” Commercial sale or application became a relic of the past, not just a tough regulation with loopholes to jump through. It reminds me of asbestos removal at my old high school. Once a clear link to harm surfaces, regulators don’t tiptoe anymore.
Lessons for Today’s Chemical Landscape
Looking back, the 2,4,5-T story offers practical reminders for anyone handling chemicals. Public records show trust in regulatory review matters, but blind optimism costs lives or leaves scars that don’t fade. Industry argued for “safe levels,” but evidence kept mounting, not shrinking. The world learned, yet these lessons need retelling—chemical companies must share full details from early trials, and governments can’t stall action just to save money or reputation. Studies ought to look at long-term impacts, not only at quick, visible problems.
Solutions Take Real Work—Not Just Bans
Stopping the sale of a dangerous chemical looks like progress, and it is, but the job doesn't end there. Old stockpiles still sit in some storage sheds or forgotten depots. Cleanup programs require real funding and public awareness. Sometimes, the hardest change is persuading people that “the way we’ve always done it” needs rethinking. Sustainable weed control, better hazard training, and more rigorous chemical review could protect land and health where my family and yours build their lives. The memory of chemicals like 2,4,5-T becomes more than a warning; it becomes a nudge to keep watching—even when the weeds are gone.
| Names | |
| Preferred IUPAC name | 2-(2,4,5-trichlorophenoxy)acetic acid |
| Other names |
2,4,5-T 2,4,5-Trichlorophenoxyethanoic acid 2,4,5-Trichlorophenoxy acid |
| Pronunciation | /ˌtraɪˌklɔːrəˈfiːnɒksiəˈsiːd/ |
| Identifiers | |
| CAS Number | 93-76-5 |
| Beilstein Reference | 1918814 |
| ChEBI | CHEBI:2797 |
| ChEMBL | CHEMBL1087 |
| ChemSpider | 2046 |
| DrugBank | DB04285 |
| ECHA InfoCard | 03e73a28-e0b1-4a09-aa92-4909ea8598b1 |
| EC Number | 3.1.1.37 |
| Gmelin Reference | 8838 |
| KEGG | C02518 |
| MeSH | D014286 |
| PubChem CID | 6579 |
| RTECS number | AG6825000 |
| UNII | 6Z8XW4GW6H |
| UN number | UN3092 |
| CompTox Dashboard (EPA) | DTXSID4020732 |
| Properties | |
| Chemical formula | C8H5Cl3O3 |
| Molar mass | 255.49 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 1.65 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.8 |
| Vapor pressure | 2.17 x 10^-7 mmHg (25°C) |
| Acidity (pKa) | 2.73 |
| Basicity (pKb) | pKb = 11.86 |
| Magnetic susceptibility (χ) | -64.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.603 |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -523.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2347.7 kJ/mol |
| Pharmacology | |
| ATC code | QH401A04 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H351, H400 |
| Precautionary statements | P261, P273, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-1 |
| Flash point | 146°C |
| Autoignition temperature | 230 °C |
| Lethal dose or concentration | Lethal dose or concentration: "Oral rat LD50: 389 mg/kg |
| LD50 (median dose) | LD50 (median dose): 389 mg/kg (oral, rat) |
| NIOSH | NA9100000 |
| PEL (Permissible) | 10 mg/m³ |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
2,4-Dichlorophenoxyacetic acid MCPA (2-methyl-4-chlorophenoxyacetic acid) 2,4,5-Trichlorophenol 2,4,5-T Agent Orange |

