Looking Closer at 2,4,5-Trichlorophenol: Past, Present, and the Road Ahead
Historical Development
2,4,5-Trichlorophenol grew out of a period when chemicals promised quick answers to tough problems. Scientists first made it back in the early 20th century. By the 1940s, big chemical companies used it to make herbicides and disinfectants in large batches. Memories of those early days are colored by its connection to 2,4,5-T, a main part of Agent Orange. History shows how enthusiasm for new chemistry sometimes outpaces understanding of long-term effects. Growing up near a paper mill, I saw this pattern: focus on progress, not always on consequences. Over decades, as communities near production plants grew concerned, regulators and researchers started paying close attention to risks linked with these compounds.
Product Overview
2,4,5-Trichlorophenol stands out as a chlorinated phenolic compound, widely recognized for its role in producing herbicides and wood preservatives. Its white crystalline appearance can mislead newcomers—something so unassuming packs a punch. Demand persists in specialized sectors, especially for crafting specialty chemicals, but broader market interest stays limited by environmental and health worries. Those who have worked with chlorophenols know they don’t just “go away.” Waste disposal and lingering residues become part of any serious industry conversation involving this compound.
Physical & Chemical Properties
The structure of 2,4,5-Trichlorophenol features three chlorine atoms arranged on a phenol ring, a design that gives it both its chemical stability and environmental persistence. You notice this compound holds up under a range of conditions—a property that appeals to industrial chemists but poses real headaches for environmental teams. Its melting point hovers around the 69-70°C mark, and in my experience, its faintly medicinal odor means you don't forget you're dealing with something that isn’t entirely benign. It's only sparingly soluble in water, but it dissolves easily in common organic solvents like ether or benzene, reflecting the practical challenges of both using and cleaning it up.
Technical Specifications & Labeling
Any experienced handler will note strict sourcing and labeling protocols. The label doesn’t just tell you purity or batch details; it reminds you about hazard classifications and environmental warnings. Packages carry the UN 2020 Dangerous Goods identification—something that signals potential for harm and a clear expectation for serious handling procedures. I recall early lab work with phenolics and the way even seasoned researchers never cut corners: all containers tightly sealed, clear hazard icons, and logs noting every gram used.
Preparation Method
Industrial routes for making 2,4,5-Trichlorophenol typically involve the chlorination of phenol under strict temperature and pressure controls. In older facilities, temperature spikes led to variable purity and, at times, uncontrolled side reactions. Chlorine gas requires meticulous safety protocols, and that's often the focus in chemical manufacturing stories—it's not just turning one chemical into another, but doing it with precision to limit unwanted byproducts, such as dioxins. Later improvements added better reactors and automated controls to reduce these risks; plant operators and nearby communities often pushed for these upgrades, knowing how much was at stake.
Chemical Reactions & Modifications
2,4,5-Trichlorophenol acts as a flexible intermediate, especially in the production of herbicides like 2,4,5-T. Nitration, alkylation, and etherification count among the modification techniques, providing raw materials for everything from pesticides to specialty disinfectants. The downside—every chemical tweak adds to the variety of breakdown products, some harmless, others persistent or toxic. Many of us who studied organic chemistry grew used to handling phenolic intermediates with gloves, fume hoods, and real caution. The risks aren’t theoretical; chronic exposure studies track the health of workers and populations near sites that rely on this chemistry.
Synonyms & Product Names
2,4,5-Trichlorophenol pops up in literature and labels under several names, such as “2,4,5-TCP” or “Phenol, 2,4,5-trichloro-.” Some decades-old trade names crop up in pesticide listings, especially from the mid-20th century. Anyone researching legacy pollution issues in North America or Europe eventually runs into these variants. This shifting terminology trips up environmental investigators who try to link chemical records to present-day soil or water analysis.
Safety & Operational Standards
Strict regulations cover every step of the 2,4,5-Trichlorophenol supply chain. Industrial plants, labs, and storage sites follow chemical hygiene plans that spell out handling, containment, spill response, and disposal. Workers wear full protection—gloves, goggles, and respirators. I’ve watched teams drill for spills and accidental exposure, knowing major fines or long-term health issues hang in the balance. Waste incineration or hazardous landfill options often weigh cost against compliance risk; underfunded or poorly regulated sites sometimes skip best practices, feeding the long-running debate about environmental justice and chemical safety.
Application Area
Major uses for 2,4,5-Trichlorophenol include the synthesis of herbicides, fungicides, and wood protection agents. Its use in wood preservation had a big impact on construction and agriculture, especially in regions battling rot or insect damage. Over time, regulatory bans in North America and Europe shifted much of this production offshore. Herbicide users and workers in related industries saw firsthand both the short-term benefits and the environmental aftereffects—a cautionary tale illustrating how cost savings in the short term can saddle communities with cleanup bills later.
Research & Development
Researchers focus on safer synthesis and effective degradation techniques. Universities and government labs chase ways to break down chlorinated phenols into less hazardous forms, using both bioremediation and chemical catalysts. I follow these efforts closely because they reflect a wider push to fix yesterday’s contamination with tomorrow’s innovation. Current studies favor engineered bacteria or fungi, hoping to break up leftover residues in soils and waterways. Results hold promise, but translating lab success to real environmental improvement takes time and ongoing funding.
Toxicity Research
Studies dating from the 1960s and 1970s first pointed to acute and chronic toxicity in animals. By the 1980s, mounting evidence linked repeated exposure to effects on the liver, kidney, and immune system. In the era before tight regulation, towns near production sites sometimes reported spikes in rare cancers or respiratory conditions. Modern long-term studies dig deeper into low-dose effects and accumulate data about persistence in ecosystems. Communities living with contamination work with independent researchers to get clear answers—reminding all of us that toxicity isn’t an abstract issue, but a deeply personal matter for many families.
Future Prospects
Many in the chemical and environmental fields agree the future for 2,4,5-Trichlorophenol lies in safe legacy management and the development of green chemistry alternatives. Industry won’t turn away from useful molecules, but ongoing restriction and improved safety tech will likely further limit use in developed economies. Decision-makers face hard choices—how to balance cleanup, worker protection, and access to useful materials. Technological advances, rising public awareness, and lawmaker attention suggest that safer alternatives and full-scale remediation are the best path forward. The long legacy of 2,4,5-Trichlorophenol offers lessons for students, policymakers, and anyone living near manufacturing corridors: progress demands caution, real transparency, and a firm commitment to learning from the past.
What is 2,4,5-Trichlorophenol used for?
Behind the Name: What Sets 2,4,5-Trichlorophenol Apart
2,4,5-Trichlorophenol is a mouthful, but its reputation goes beyond the technical name. Anyone who has walked into a chemical plant or studied the way industry shapes the world might recognize the sharp, distinct odor wafting from containers marked with a hazard label. This compound shows up in everyday objects more than most people realize, with its origins tracing back to the boom in synthetic organic chemistry. People built it for a reason: it gets the job done when it comes to crafting other chemicals.
How Industry Uses 2,4,5-Trichlorophenol
Manufacturers often reach for 2,4,5-Trichlorophenol as a building block. It plays a big part in making pesticides, herbicides, antiseptics, and preservatives. For decades, it helped create substances like the infamous Agent Orange, a herbicide with a dark chapter in history due to its dioxin contamination. Its ability to kill fungi and bacteria means it works as a preservative in wood processing and in some paint and leather treatments.
This isn’t a household chemical, and it’s not something you’ll find in a kitchen cabinet or under a bathroom sink. Still, its fingerprints are all over certain processes in paper manufacturing and industrial cleaning where microbial contamination can spoil products or gum up machinery. Some specialty laboratories use it for chemical synthesis, forming more complex molecules for pharmaceuticals and dyes.
Health and Environmental Concerns
Talk to anyone in the environmental field, and 2,4,5-Trichlorophenol sparks worry. The compound doesn’t break down easily, so it lingers in soil and water. Exposure poses health risks. According to the Environmental Protection Agency, it may damage the liver, kidneys, and nervous system. The manufacturing process can form stubborn impurities called dioxins, which stick around and build up in living tissue.
Growing up near a site where such chemicals once spilled, I learned early that what goes into rivers rarely just disappears. Local streams carried a mess of industrial leftovers, and experts spent years studying the effects on fish and plants. People in affected towns raise real concerns about the long-term impacts, especially since dioxins linked to this compound rate among the most hazardous substances known to science.
Facing Forward: Reducing Risks and Improving Safety
Balancing utility and safety isn’t easy. Industry has mostly switched away from using 2,4,5-Trichlorophenol in many applications, pressured by tighter regulations and public scrutiny. Some manufacturers now use alternative chemicals that break down faster or don’t generate the same toxic byproducts.
Strict handling practices and protective gear help reduce risks for workers exposed during industrial processing. Secure storage, containment, and cleanup protocols keep it from reaching wider communities. Regulatory authorities like the EPA monitor and restrict emissions, but monitoring remains a challenge, especially at older sites.
Addressing the legacy of contamination demands joint effort. Governments push for remediation, while advocacy groups keep a watchful eye on community health. As chemistry continues to develop, safer alternatives and better waste management offer hope for a cleaner future, yet the lessons from compounds like 2,4,5-Trichlorophenol linger in both soil and memory.
Is 2,4,5-Trichlorophenol hazardous to health?
Understanding 2,4,5-Trichlorophenol
2,4,5-Trichlorophenol, a chemical often used in disinfectants, wood preservatives, and as an intermediate for some herbicides, doesn’t turn up in everyday conversation. Still, it pops up in various products and industrial processes. The smell tends to stick—a bit like hospitals and labs—which brings its own brand of caution. I remember walking through old barns treated with chemicals like this, the lingering sharp odor settling in the air, making it hard to ignore the chemical presence long after use.
Health Concerns Linked to Exposure
Breathing in or touching 2,4,5-Trichlorophenol can lead to several short-term and long-term problems. Inhalation or skin contact can cause irritation, headaches, dizziness, and sweating. The less obvious threat comes from repeated or high exposure. According to the U.S. Environmental Protection Agency (EPA), this compound has been linked to effects on the liver, kidneys, and immune system in laboratory animals. The International Agency for Research on Cancer (IARC) places 2,4,5-Trichlorophenol among possible carcinogens to humans. That kind of classification doesn’t happen for no reason. Cases of contaminated sites with 2,4,5-Trichlorophenol often show up in news reports covering clusters of health complaints in nearby communities—nosebleeds, nausea, and in some places, higher than average rates of certain cancers.
Why the Chemical Lingers in the Environment
One thing that sets 2,4,5-Trichlorophenol apart: it doesn’t break down easily. I’ve seen sites where chemical spills happened decades ago, and tests still show high levels in the soil and groundwater. This stubbornness means hazards can stick around long after a factory shuts its doors. Runoff from contaminated sites can carry the chemical far beyond where it was originally used, spreading the risk to more people and wildlife. Fish and other aquatic life take up the chemical, making its way up the food chain, eventually reaching humans again.
Responses and Responsibility
Local and federal agencies do monitor for chemicals like 2,4,5-Trichlorophenol, but gaps remain. I’ve spoken with residents who feel left to fend for themselves, forced to do their own water or soil testing when official help falls short. Regulations exist, yet enforcement and cleanup lag behind, often held up by funding battles or debates over exactly how hazardous these chemicals are. Reporting and transparency from companies using or disposing of the compound could go further. Without strong oversight, unsafe disposal keeps happening—people on the ground notice the effects long before warnings come through official channels.
Looking Toward Safer Communities
Solutions mean more than just tighter rules—they call for investment in cleanup technology and better education for anyone living or working near places using persistent chemicals. Community pressure has moved mountains before; it takes making noise to get policymakers to pay attention. Support from scientists, doctors, and public health advocates helps put real data into the debate. Safer alternatives for industrial processes already exist, and switching over could cut risks fast. Even so, until sites contaminated with 2,4,5-Trichlorophenol get cleaned up and companies take responsibility for safer disposal, the risk to health remains real. Tackling this hazard can feel like pushing a boulder uphill, but the benefit to communities—and future generations—makes the effort worth it.
What are the storage requirements for 2,4,5-Trichlorophenol?
The Real Talk on a Hazardous Chemical
2,4,5-Trichlorophenol isn’t the sort of compound you stash on a shelf among cleaning supplies. Anyone who’s spent time around industrial chemicals knows this substance carries a serious punch. Workers see 2,4,5-Trichlorophenol pop up in wood preservatives, pesticides, or as a chemical intermediate, but the risks stick around well after the workday ends. Mishandling can trigger toxic fumes or fires, so storing this chemical securely keeps more than your regulatory record clean—it protects health and lives.
What Sort of Risks Are We Talking About?
Exposure to 2,4,5-Trichlorophenol irritates skin, eyes, and the respiratory system. Heating it releases noxious vapors, including dioxins, which bring environmental concerns and long-term health problems. In 2022, the CDC flagged skin contact and inhalation as common routes of exposure in accidental releases. Improperly sealed containers, or leaks in storage rooms, don’t just break the rules—they open the door to harm.
So, How Should It Be Stored?
The safest storage place offers two things: stability and containment. Decades working in facilities handling volatile chemicals show me it's usually the basics that matter most. Trichlorophenol belongs in tightly sealed containers built to handle corrosive substances. Glass or high-quality plastic usually gets the nod, always kept upright and clearly labeled to avoid any guessing games in an emergency.
Keep the storage environment cool and dry. Moisture not only invites corrosion; it can speed up chemical reactions, and that’s never good news. Rooms meant for hazardous chemicals often run with independent climate control and solid ventilation. A good airflow system helps whisk away fumes before they build up.
Anyone who’s seen chemical burns or respiratory shocks knows the value of separation. Keep 2,4,5-Trichlorophenol isolated from incompatible chemicals like oxidizing agents and strong acids. One misplaced drum or bottle can spark a reaction reaching far beyond one room.
No Substitute for Training and Vigilance
Protocols and checklists matter, but experience fills the gaps rules leave behind. I’ve watched teams with good safety habits spot small leaks early and prevent bigger messes. Mandatory safety signage and spill kits should never gather dust. Eye wash stations and showers serve workers best when everyone knows where to find them without thinking. Training everyone who steps near the stockroom—right down to the weekend cleaning crew—can make the difference.
Upgrading for Better Safety
Nobody wants to read about another workplace accident in the morning news. Automation can help, from real-time vapor detectors to automated shutdown codes on doors. In my experience, investing in safety—ventilated rooms, double-walled drums, sensor systems—pays off with peace of mind. Digitized inventory management tracks every container and reminds you to check shelf-life or storage conditions, catching mistakes before they turn into incidents.
Regulations drive safety improvements, but real safety culture springs from the floor, not the law books. Management buy-in, frequent drills, and respect for the chemicals in use define good practice. It’s a team effort: nobody operates in a vacuum, and shortcuts catch up eventually.
Learning from the Field
Disaster stories stick with me—corroded containers, forgotten stockpiles, rushed workers skipping labels. Each one brings a lesson: treat 2,4,5-Trichlorophenol with respect long before the inspectors arrive. Proper storage isn’t just about boxes and barrels—it’s about knowing your materials, having your team’s back, and taking pride in a workspace that sends everyone home safe.
How should 2,4,5-Trichlorophenol be disposed of safely?
Why the Way We Toss Chemicals Matters
2,4,5-Trichlorophenol comes up in plenty of settings, from industrial sites to research labs. Having worked in a chemistry lab and spent time around environmental teams, I know firsthand how tricky chemical disposal gets—especially with something toxic like this stuff. A bad spill or careless approach can turn a workplace upside down, not to mention what it can do to soil, water, or anyone close by. That greenish crystal can pose long-term health risks: researchers link it to organ damage, skin irritation, and even cancer for those exposed repeatedly. It can also persist in the environment for decades. Dumping it down the drain or tossing it in regular trash shouldn’t even cross the mind.
Where Mistakes Happen
I remember a colleague who once let a container of 2,4,5-Trichlorophenol sit on a shelf, forgotten and uncapped. The strong, bitter smell filled the lab, and nobody wanted to be the one to move it. That moment underscored how easily routine breaks down if there’s no culture of safety. The truth is, most people don’t toss out dangerous chemicals every day, so they take shortcuts or ignore the risks. If you don’t treat toxic waste with respect, accidents come faster than you’d think. The research backs this up: the United States Environmental Protection Agency (EPA) has published hundreds of reports on site contaminations caused by improper dumping of chlorinated phenols, with creek water showing residues in areas far from direct industrial use.
Safe Disposal Means Getting Experts Involved
No matter how small an amount, disposal shouldn’t be a do-it-yourself project. Staff should label every container clearly and double-check any storage instructions. In my experience, calling a licensed hazardous waste contractor makes a difference. Most reputable firms collect organochlorines like 2,4,5-Trichlorophenol and move them to facilities designed to neutralize or destroy them, usually by high-temperature incineration. This method effectively breaks down the toxic structure and prevents future contamination. Landfills don’t cut it—dumping this compound on ordinary ground leads to long-lasting harm, and even sealed drums can corrode and leak over time.
Keeping Track for Peace of Mind
Documentation isn’t just some bureaucratic step. Regulators want a clear paper trail for every gram that leaves a building. Having well-filled logs and signed manifests means nobody gets lost in the shuffle, and surprise visits from safety officials become far less stressful. Labs and factories must train workers to recognize hazardous labels, wear the right protective gear, and avoid any shortcuts, even during busy seasons. Eyes can sting and lungs can burn with a single mishap—a lesson too many learn only after the fact.
Building a Safer Routine
Solutions start with clear rules and real commitment. Managers shouldn’t wait for regulations to prompt change; they ought to make safe disposal part of regular schedules and reward good habits. Investing in better storage cabinets and clear signage reduces guesswork. Community coordination can help, too—sharing contacts for certified waste handlers prevents someone from going it alone and risking exposure. State and federal agencies offer plenty of up-to-date guidance on proper handling, so there’s no excuse for playing fast and loose. Protecting both human health and the planet starts with mindful, responsible action every single time hazardous waste comes up. Experts, planning, and education all fit together when handling something as serious as 2,4,5-Trichlorophenol.
What is the chemical structure of 2,4,5-Trichlorophenol?
Breaking Down the Basics
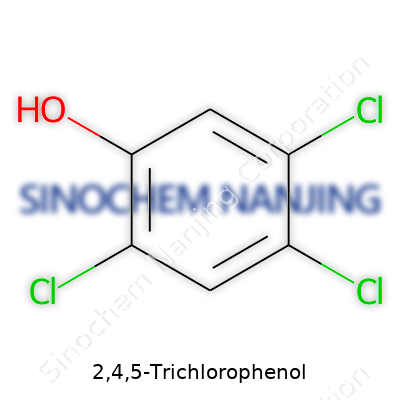
2,4,5-Trichlorophenol doesn’t typically make headlines, but it has a legacy that trails through many essential debates around chemistry, safety, and the environment. Its name gives away part of the structure: three chlorine atoms attached to a phenol molecule. To see its real impact, it's worth digging into the specifics of this compound’s chemical structure and how that shape ties into real-world concerns.
The Shape Behind the Name
On paper, a phenol molecule looks like a benzene ring, which has six carbon atoms connected in a hexagonal shape with alternating double bonds. In phenol, a hydroxyl (–OH) group attaches to one carbon. For 2,4,5-Trichlorophenol, three chlorine atoms replace three of the hydrogen atoms on the ring, specifically on carbons 2, 4, and 5. I once tried sketching this for a chemistry class: if you place the –OH group at the top (position 1), you count around the ring and plug in the chlorine atoms at the positions labeled 2, 4, and 5. This setup leaves a chemical that is both persistent and reactive in unique ways.
Environmental Footprint and Health Concerns
The trouble with 2,4,5-Trichlorophenol isn’t only what’s on paper or in a lab—its chlorine-loaded ring makes it quite stubborn in the environment. Chlorinated aromatic compounds, like this one, don’t break down easily. They can pass through water and soils for years. Decades ago, I talked to a farmer who worried about pesticide run-off near his property. This chemical and its cousins often enter the discussion since they're found as breakdown products in certain herbicides and wood preservatives. The pattern of chlorine atoms—set up by the molecule’s structure—makes it resistant to natural breakdown processes.
Research from the International Agency for Research on Cancer and EPA documents has highlighted ongoing health concerns. Exposure links to skin irritation, respiratory troubles, or more chronic risks, including possible carcinogenicity. Communities near manufacturing sites or timber industries have voiced their struggles with contaminated well water and persistent odors.
Ties to Larger Stories
2,4,5-Trichlorophenol isn’t an isolated story. Its structure shows up in the creation of other chemicals like herbicides. Notably, it played a key role in synthesizing 2,4,5-T, which mixed with 2,4-D to make Agent Orange. That connection gave me new perspective when looking at environmental policy—how even small tweaks to a molecule can ripple out and cause issues downstream from manufacturing to disposal.
What to Do Next
Looking for solutions pushes people toward chemistry that listens to both technical needs and community voices. Steps start with strong regulations—strict monitoring for waste and effluent from factories can catch leaks before they spiral. Old dump sites need cleanup crews trained both in chemical handling and public communication. Changing the way we develop new chemicals, focusing more on ingredients that nature can break down, matters in the long run. Universities, advocacy groups, and regulatory offices work best when they keep these lessons in mind—choosing structures that solve current problems without creating new ones.
Final Thoughts
The chemical structure of 2,4,5-Trichlorophenol tells a story that stretches far beyond the textbook. Each ring and bond on the molecule shapes how we need to respond—balancing innovation, safety, and responsibility. Paying attention to these details lays the groundwork for the next generation of safer, more sustainable chemistry.
| Names | |
| Preferred IUPAC name | 2,4,5-Trichlorophenol |
| Other names |
2,4,5-Trichlorophenol, pure Phenol, 2,4,5-trichloro- Dowicide 5 Dowcide 5 Penile 2,4,5-TCP |
| Pronunciation | /tuː, fɔːr, faɪv traɪˌklɔːrəˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 95-95-4 |
| Beilstein Reference | 1361637 |
| ChEBI | CHEBI:34607 |
| ChEMBL | CHEMBL1376 |
| ChemSpider | 10171 |
| DrugBank | DB07906 |
| ECHA InfoCard | 03bc7b7e-7682-4dc2-8cf5-dc076f3bf012 |
| EC Number | 3.1.1.63 |
| Gmelin Reference | 7786 |
| KEGG | C06516 |
| MeSH | D014250 |
| PubChem CID | 7282 |
| RTECS number | SN1575000 |
| UNII | N1P6M729M8 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID8020220 |
| Properties | |
| Chemical formula | C6H3Cl3O |
| Molar mass | 197.45 g/mol |
| Appearance | White to light tan crystalline solid |
| Odor | Phenolic odor |
| Density | 1.49 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 3.7 |
| Vapor pressure | 0.0015 mmHg (25°C) |
| Acidity (pKa) | 7.0 |
| Basicity (pKb) | 8.05 |
| Magnetic susceptibility (χ) | -73 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 2.15 cP (50°C) |
| Dipole moment | 2.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -276.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1425.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AE06 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, causes severe skin burns and eye damage, may cause respiratory irritation, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H315: Causes skin irritation. H318: Causes serious eye damage. H335: May cause respiratory irritation. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P311, P312, P330, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 113°C |
| Autoignition temperature | 235°C |
| Lethal dose or concentration | LD50 oral rat 325 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 325 mg/kg |
| NIOSH | SN15600 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 2,4,5-Trichlorophenol is 1 mg/m³ |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | TC: 20 ppm |
| Related compounds | |
| Related compounds |
2,4,6-Trichlorophenol 2,3,4-Trichlorophenol 3,4,5-Trichlorophenol 2,4-Dichlorophenol 2,5-Dichlorophenol 2,3,5-Trichlorophenol Pentachlorophenol 2,3,6-Trichlorophenol |

