2,4,5-Trichloroaniline: A Chemical’s Story from Laboratory Curiosity to Cautionary Tale
Historical Development and Product Overview
Science books mention 2,4,5-Trichloroaniline as a fairly old discovery, rooted in the chemical boom of the early twentieth century. Chemists in that era developed a hunger for new molecules and, with it, an interest in anilines—a family with deep ties to dyes, pharmaceuticals, and later, pesticides. The journey of 2,4,5-Trichloroaniline isn’t just about clever lab work; it's a reflection of society’s shifting relationship with synthetic chemicals. Its synthesis grew out of the need for building blocks in manufacturing, especially as demand for chlorinated aromatic compounds spread around the globe. Over time, this chemical found a place in research benches as well as industrial vats, though today’s lens tilts toward careful handling and respect for what such compounds can do.
Physical and Chemical Properties Worth Noting
Let’s look at what puts this chemical on the map. 2,4,5-Trichloroaniline appears as a solid, usually colorless or faintly colored, resistant to quick breakdown thanks to those chlorine atoms. Its molecular formula, C6H4Cl3N, packs in three chlorines that affect its reactivity and shape its interactions with other substances. Its melting point sits well above room temperature, so it hangs together in solid form under most conditions you’d find in a regular lab. The dense arrangement of atoms gives it a higher molecular weight than simple aniline, and those extra chlorines help determine where—and how—it reacts, making it tougher for nature to digest compared to less bulky cousins.
Technical Specifications & Labeling: Not Just Details
Regulations worldwide demand accuracy in labeling for 2,4,5-Trichloroaniline. Chemical suppliers must follow standard nomenclature, and labels flag important items like purity, physical state, and potential hazards. Those who work with it can’t ignore warnings about protective gear, ventilation, and proper storage. Beyond safety, technical notes also flag its solubility, compatibility with solvents, and guidelines for disposal. Small print on labels becomes a first defense against hazards: not something to gloss over. Purity levels usually run high when it’s sold for synthetic or analytical uses, ensuring predictable outcomes in research and industry.
Preparation Method: No Mystery, But No Shortcuts
The preparation of 2,4,5-Trichloroaniline taps established organic chemistry, relying on selective chlorination of aniline or reduction of appropriately chlorinated nitrobenzenes. In practice, this means handling aggressive reagents and tight controls over temperature and byproducts. For those who have spent time at a lab bench, it’s clear that producing this compound isn’t just about following a recipe—every misstep raises the risk of side products or waste that complicates cleanup. Efficiency matters, as does minimizing exposure, so newer methods sometimes aim for milder conditions or greener reagents when possible.
Chemical Reactions & Modifications: Opportunity and Risk
Reactivity of the 2,4,5-Trichloroaniline core unlocks routes to further synthesis, allowing chemists to tack on different functional groups or swap out its chlorines. Nitration, sulfonation, and various coupling reactions open up new paths for dye, pigment, and pharmaceutical development. But reactive intermediates and the potential for toxic byproducts make this chemistry a double-edged sword. The power to modify aniline derivatives comes with responsibility. Mishandling can create persistent environmental contaminants or compounds with far greater toxicity than the starting material, a lesson history refuses to let us forget.
Synonyms & Product Names: Language and Regulation
Even a seasoned chemist must juggle the many names this compound collects: synonyms like 2,4,5-Trichlorobenzenamine, sometimes shortened to “TCA” in technical circles. Regulatory agencies and international safety codes assign numbers and codes, so what goes by one name in a catalog might appear under another in legal texts. Researchers and handlers must stay sharp to avoid confusion, especially when regulatory controls and restrictions change between regions. This web of names and codes isn’t just an annoyance—it often separates safe practice from dangerous neglect.
Safety & Operational Standards: No Room for Complacency
Working with 2,4,5-Trichloroaniline demands commitment to safety, both personal and environmental. In the lab, full PPE—gloves, goggles, sometimes respirators—are non-negotiable. Schools and work sites house it in locked storage, far from food, flame, and the casual visitor. Waste streams receive careful monitoring to prevent release into soil or water systems. Government regulations in places like the EU, US, and Japan demand clear protocols for spill management and exposure. I’ve seen the consequences when corners get cut: mishaps leading to skin burns, persistent odors, and headaches that last longer than the workday. Vigilance isn’t a choice; it’s the only way.
Application Area: Industry’s Double-Edged Tool
Historically, this compound played key roles in the making of pesticides, herbicides, dyes, and specialty chemicals. Notably, it once figured in the synthesis of compounds like 2,4,5-T, a defoliant infamous for both its effectiveness and its links to contamination. The presence of 2,4,5-Trichloroaniline in supply chains sparked debate and investigation after environmental and health crises in the latter half of the twentieth century. Today, most reputable manufacturers and academic researchers approach it with a mix of necessity and caution, aware that its benefits—controllable nitrogen source, reactive aromatic scaffold—come with steep risks.
Research & Development: Pursuing Progress, Not Just Product
Modern research involving 2,4,5-Trichloroaniline centers on safer syntheses, degradation methods, and detection techniques. Environmental chemists seek better catalysts to break it down in polluted water or contaminated land. Analytical chemists refine study of trace residues in food, water, and soil, equipping regulators and the public with clearer data on true exposure. In industry, R&D labs hunt for substitutes with lower risk of persistence or toxicity, sometimes drawing inspiration from nature or advanced materials. Yet, because it remains a useful marker for historical contamination, its study also illuminates broader lessons about how society handles persistent and bioaccumulative chemicals.
Toxicity Research: Hard-Learned Lessons
Toxicologists continue to dissect the dangers of 2,4,5-Trichloroaniline. Animal models highlight chronic exposure risks: liver and kidney effects, neurotoxicity, changes in blood chemistry, and strong warnings about reproductive or developmental harm. Unlike some old industrial chemicals, this one never masqueraded as safe. Dust or vapors cause irritation and harm on contact, and long-term contact leaves a footprint in the body’s tissues. Communities dealing with contamination highlight the gap between laboratory data and real-life consequences. These voices remind scientists, regulators, and industry workers that test tubes do not capture the full story.
Future Prospects: Respect Over Convenience
Looking ahead, the future of 2,4,5-Trichloroaniline probably belongs less to everyday commerce and more to controlled scientific investigation. Regulatory pressures and safer alternatives push industry away from reliance on aromatic chloroanilines, and stricter bans loom on the horizon in many regions. Remediation technologies, such as advanced oxidation or bioremediation, will grow in importance for sites already polluted. So long as researchers treat this compound with open eyes and strict controls, its study can highlight gaps in our approach to chemical stewardship. Lessons learned from its legacy shape a modern path for synthetic chemicals: openness, vigilance, and an insistence on health above quick gains.
What are the main uses of 2,4,5-Trichloroaniline?
Understanding 2,4,5-Trichloroaniline
Stepping into a chemistry lab always gave me a sense of urgency and responsibility. Some substances carry weight beyond their chemical formula. 2,4,5-Trichloroaniline is one of those. From years of reading material safety data sheets, I’ve learned to trace its path across industries and research labs, and it keeps popping up for practical reasons, not just as a footnote in textbooks.
Dyes and Pigments Production
Many of my science textbooks glow with bold colors. Few people stop to think about the origins of these dyes. 2,4,5-Trichloroaniline forms the backbone in the synthesis of several specialty dyes, thanks to its stability and chemical structure. Manufacturing companies use it as an intermediate for azo dyes, delivering strong colors that stick well to fabrics and papers. In some ceramics and plastics, you’ll also run into pigments built upon the backbone of this compound. Its molecular shape supports bright, lasting color.
Pharmaceutical Raw Material
Drug development is not a straight road. It’s filled with dead ends, side trails, surprising finds. I remember working alongside a chemist who searched for ever-more effective ways to construct complex molecules. 2,4,5-Trichloroaniline stepped in as a reliable building block for several medicinal compounds—often in pain relievers and antifungal drugs. It shows up in the early steps where molecular structures get pieced together, forming a platform researchers can modify. This flexibility gives pharmaceutical labs new tools in the race to fight disease.
Agrochemical Intermediate
Farmers need less guesswork and better yields. Science tries to deliver both. Here’s where 2,4,5-Trichloroaniline gets plenty of attention, especially in creating herbicides. Its role as an intermediate in synthesizing crop protection chemicals helps keep weeds from choking out harvests. History shows that chemicals based on this compound led to big jumps in food production. These solutions walk a fine line, since they also come with concerns on safety and environmental impact.
Concerns and Solutions
Continued use pushes questions into the open. I’ve stood beside enough environmental advocates to know the stories on both sides. Prolonged exposure or mishandling can put workers and the environment at risk. 2,4,5-Trichloroaniline breaks down slowly, and accidental spills create challenges for water and soil. Smart innovators now focus on containment, proper training, safer handling and robust protective equipment. In my experience, stricter regulations and worker education protect people and places, even in fast-paced manufacturing sites.
Breaking the cycle of chemical dependency doesn’t mean moving backward. Sustainable chemistry stands as a goal, not a buzzword. Replacing hazardous substances without losing effectiveness asks for fresh thinking, so researchers search for greener synthesis routes, seek out alternatives with fewer risks, and improve waste treatment systems to keep communities safe. My own job felt the pressure to search for not just quicker results, but cleaner processes.
Looking Ahead
2,4,5-Trichloroaniline keeps its place in some important fields, from dyes to farmland to the pharmacy shelf. Like many chemicals, its impact depends on the care behind its use. Choices made in the lab and the factory floor shape its legacy out in the world.
Is 2,4,5-Trichloroaniline hazardous to health?
What is 2,4,5-Trichloroaniline Used For?
Everyday life brushes up against specialty chemicals more than most people realize. 2,4,5-Trichloroaniline, a chemical not usually found under the kitchen sink, ends up in the supply chain of dyes, pigments, and pharmaceuticals. Some factories use it to manufacture pesticides. Most folks never see this substance directly, but its fingerprints are all over the modern chemical industry.
Health Concerns and Exposure Risks
Anyone who’s ever spent time in a lab knows the sharp smell of certain chemicals, making you question if your goggles fit tight enough. Chlorinated anilines like this one raise eyebrows because of their toxicity. Direct exposure—breathing in dust or letting it touch your skin—can give you headaches, worry your liver, and, in high enough doses, trigger more severe reactions. Studies on similar chemicals point toward symptoms like drowsiness, weakness, and, if unlucky, persistent skin irritation.
Workers in factories that make or handle this compound carry the greatest risk. Regulators such as OSHA in the United States keep a close watch because a slip in safety can mean someone gets seriously hurt. Proper gloves, masks, and strong ventilation do a lot to keep levels tolerable, but not every workplace follows the rules perfectly. Chronic exposure over years invites long-term health problems, although exact numbers for 2,4,5-Trichloroaniline remain in need of more research.
Environmental Pathways Bring Indirect Consequences
People far from production lines can still cross paths with this chemical without realizing. If a plant spills waste or dumps it improperly, it seeps into soil and water, sometimes traveling farther than expected. Fish and wildlife can suffer. There’s evidence from related chlorinated compounds that aquatic organisms don’t handle these chemicals well, leading to death or disrupted development. Water downstream from poor disposal practices can drag these compounds back into human water supplies.
What Science Says About Its Hazards
Data collected by public health authorities shows that substances related to 2,4,5-Trichloroaniline can cause cancer in animals, though the link in people remains under study. The chemical structure fits patterns seen in other hazardous anilines, which push toxicologists to stay cautious. Manufacturers, as a result, must file special paperwork and track how much they keep on-site. Not every risk appears overnight, but some compounds build up in the environment or living tissues more than anyone intended.
Reducing Harm and Making Smart Choices
I’ve seen firsthand how workers’ confidence grows when companies cut dust, invest in extraction hoods, and train people on handling risky chemicals. Eye-wash stations and emergency showers stop becoming rusty ornaments and turn into lifelines. Some factories track worker blood and urine for early signs of exposure, catching small problems before they escalate into unmet medical needs. Cleaner manufacturing techniques and strict transport rules shrink opportunity for waste to wind up where it doesn’t belong.
We all expect industries to put safety ahead of shortcuts. Regulatory agencies need sharp teeth and public pressure helps keep them honest. Responsible chemical use includes clear labeling, tough leak monitoring, and a business culture that doesn’t treat human health like an afterthought. Chemicals with a track record of harm should get extra scrutiny, and switching to safer alternatives where possible puts fewer people in harm's way. If you work around such substances or live near an industrial site, staying informed and speaking up gets results.
What safety precautions should be taken when handling 2,4,5-Trichloroaniline?
Why Handling This Chemical Isn’t Like Handling Table Salt
2,4,5-Trichloroaniline belongs to a class of compounds that never really make headlines for the right reasons. Its chalky, pale appearance belies the hazards it carries. Years spent in lab environments taught me that you only have to get careless with compounds like this once, and your body may never forgive you.
Know the Enemy: What Science Says
Research shows this compound can irritate skin and eyes, and inhaling its dust triggers coughing, shortness of breath, and sometimes worse. The compound’s ties to producing herbicides and dyes add another layer of risk, since related chemicals have links to dioxins, which science has connected to serious health outcomes.
Colleagues have shared stories about missed warning signs. Nausea, headaches, rashes — these are not inconveniences to brush off. The compound acts silently at times, and symptoms might pop up only hours after a mistake. Giving this chemical respect means fewer regrets later on.
Lessons from the Lab Floor
No one working with this stuff skips protective equipment. Nitrile gloves — not latex, since chlorinated organics can eat through latex. Splash-proof goggles, lab coats, and a good pair of closed shoes come before even opening the container. That humility in gearing up saves misery.
One old mentor stuck a sign over the chemical lockers: “Trust your senses, trust your habits.” He kept an eyewash station ready and inspected it regularly. The fume hood fan ran before the bottle left the shelf. Smelling even a hint of chemical usually meant, clean up and step away.
Good Habits Cut Down on Risk
Working next to a spill kit, not across the room from it, means help is always close. Clean benches, tidy spaces — they’re not just for show. Spilled powder or vapor can slip by unnoticed if the space looks chaotic. Leaving bench clutter behind keeps contamination to a minimum.
If I could tell anyone one thing: never work alone. The chemical doesn’t care how experienced anyone is. Accidents demand backup, and the buddy system never gets old. Colleagues who ignore training often regret it. Regular training—running through what-if scenarios—keeps reactions sharp even on slow days.
Smart Storage and Waste Handling
Store this compound in a dry, locked cabinet, away from strong acids and oxidizers. Moisture finds its way into weak containers and degrades chemicals, leading to leaks. Read the Safety Data Sheet — every new batch, every time — because suppliers can change processes without warning.
Waste from 2,4,5-Trichloroaniline doesn’t belong in the sink or regular trash. Certified hazardous waste containers and proper labeling keep everyone along the chain out of harm’s way. Rushing through disposal shortens careers and harms the environment far beyond the lab.
None of This Is Optional
There’s no shortcut. Anyone handling this chemical holds responsibility for themselves and everyone else in the area. Learning from those who came before, respecting the warnings, and taking the extra minute saves lives and keeps work possible. That’s not just expert advice—it’s lived truth.
How should 2,4,5-Trichloroaniline be stored?
Understanding the Hazards
Years back, I spent time in a lab where 2,4,5-Trichloroaniline made a regular appearance. The first lesson was straight: any chemical with ‘trichloro’ in its name does not belong anywhere near sloppy storage. Spills and accidents invite both toxic exposure and persistent cleanup nightmares. The facts speak plainly—this is a solid with a fair vapor pressure, and both skin and inhalation exposure can bring health problems like respiratory irritation, nausea, or worse. The risk grows if it’s not sealed tight or winds up close to acids or oxidizers that trigger dangerous reactions.
Keep It Cool, Dark, and Dry
Most of the trouble with 2,4,5-Trichloroaniline starts with poor storage. I learned quickly that leaving powders near windows or on benchtops just asks for clumping, decomposition, or accidental mixing. Every credible safety resource, from OSHA to the National Institute for Occupational Safety and Health, flags the basics: a tight-sealing container, tucked away in a dry place, blocks moisture, and staves off any dose of heat or sunlight that might speed up chemical breakdown. More than once I saw even experienced chemists shrug at ‘just a few degrees warmer’—yet small boosts in temperature accelerate how quick this stuff turns unstable, and nobody wants that risk in their workspace.
No Shortcuts With Containers
I’ve seen folks store hazardous solids in flimsy plastic or screw-cap glass jars. The safer bet always came from thick, high-density polyethylene or amber glass bottles with gasketed lids. Some might think labeling goes without saying, but even a single mislabeled container sparked confusion in our lab and could have ended worse. Clear hazard labels, date of reception, and batch numbers help anyone on duty identify risks immediately. No one wants to guess what white powder lives inside an anonymous jar.
Segregation Prevents Mishaps
Anyone storing chemicals long enough will witness a close call or two. Once a cleaning crew stacked oxidizers next to organics, and it nearly blew a shelf. 2,4,5-Trichloroaniline stores safest far from acids, oxidizers, and reactive metals. It fits best on secondary containment trays—leakproof, chemical-resistant platforms that catch trouble before it spreads. In more modern labs, ventilated chemical cabinets pull away fumes before they drift outside. These setups, mandated by many safety codes, give added peace of mind.
Solutions for Safer Practice
Hazard doesn’t end once the jar gets shut. Long years in chemistry taught me that handling counts for little if disposal and accident response plans don’t match the risks. Clear protocols ensure spills get cleaned quickly and accidents don’t escalate. Ongoing training—refresher sessions every few months—keeps staff sharp on risks and emergency contacts. Even staff outside the lab benefit when supply rooms set clear color-coded areas.
For places with tight budgets, investing in basic fireproof storage cabinets and secondary containers pays for itself by preventing accidents. Chemical management software minimizes confusion with up-to-date access logs and automatic reminders for expiring stocks. If there’s a standout lesson, it’s this: chemical safety takes real commitment, attention to detail, and a willingness to review habits regularly. With that approach, 2,4,5-Trichloroaniline sits safely on the shelf—out of sight and out of mind until the next research project calls for it.
What is the chemical structure and molecular formula of 2,4,5-Trichloroaniline?
The Shape of the Molecule
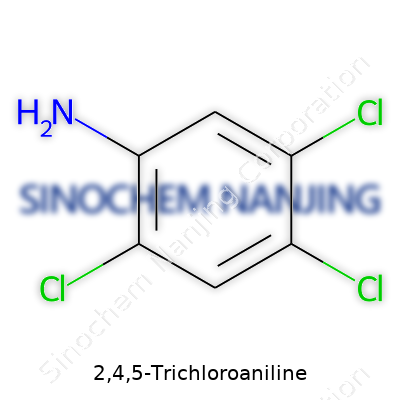
Chemistry comes alive by making sense of the molecules that shape our world. Take 2,4,5-Trichloroaniline. You won't see its name in every chemistry textbook, but it pops up plenty in the real world, especially for those who spend time in industrial labs or toxicology research. Its chemical structure is simple but effective: a six-carbon benzene ring stacked with one amino group and three chlorine atoms. These chlorines attach to the ring at the 2nd, 4th, and 5th carbons. The amino group (-NH2) sits on the first carbon. Visualizing this doesn’t need a textbook diagram—just think of a standard benzene ring, swap some hydrogens for three chlorines, and stick an amine group on the front.
Molecular Formula and Anatomy
If you've used molecular formulas to order chemicals or check safety sheets, the shorthand jumps out: C6H4Cl3NH2. To break it down, the skeleton of six carbons holds together a classic aromatic ring, familiar from compounds like aniline. In this one, three positions carry chlorine atoms, making the ring heavier and less reactive than basic aniline. The full formula—C6H4Cl3N—reflects all these modifications. This extra bulk from chlorine can matter a lot in the chemical’s behavior, from how it dissolves to its toxicity and the way it gets processed in the environment and human body.
The Significance in the World
2,4,5-Trichloroaniline is not just another compound on a database. It’s significant in labs focused on agricultural chemistry since it serves as a parent material or intermediate for making certain herbicides, especially older ones like 2,4,5-T. Some legacy uses of those herbicides still have ripple effects. Runoff and residues often end up in places they shouldn't, including farmlands and waterways. In my time working around agricultural testing, traces of this chemical have been fingerprinted in soil samples, even decades after application. For the communities that live near these fields, these molecules are more than a drawing—they connect to real concerns about water quality and soil health.
Risks and Safety
Handling chemicals, especially ones with a handful of chlorines, always brings safety into the conversation. Chlorinated anilines like 2,4,5-Trichloroaniline aren’t usually something you find outside professional settings for good reason. Direct exposure or accidental inhalation can irritate the skin, eyes, and respiratory system. Long-term exposure raises more serious flags, especially given the way similar compounds have shown up in studies of chronic toxicity and environmental buildup. Researchers and industry workers regularly suit up in protective gear—gloves, goggles, sealed hoods—to avoid direct contact. Laboratories often use fume hoods for a reason; a whiff of something off can signal trouble.
Searching for Better Solutions
In recent years, there’s been a push to move away from heavy reliance on these kinds of chemicals, both to lower occupational hazards and reduce long-lasting residues in the environment. Green chemistry offers some hope: chemists have designed alternative pathways that skip the need for such highly chlorinated aromatics when creating modern herbicides or dyes. As someone who’s spent time both in university labs and consulting with farmers, I’ve seen the value in educating end-users about the persistence of such chemicals in soil and waterways.
Understanding the molecular structure of compounds like 2,4,5-Trichloroaniline isn’t only useful for those in white coats. Everyday people who care about food safety, clean water, and responsible land management all benefit when companies and governments push for alternatives that don’t load our environment with difficult-to-remove toxins. It starts, as always, with knowing what you’re dealing with—right down to the last atom.
| Names | |
| Preferred IUPAC name | 2,4,5-Trichlorobenzenamine |
| Other names |
2,4,5-Trichlorobenzenamine 2,4,5-Trichloro-1-aminobenzene |
| Pronunciation | /ˌtraɪˌklɔːrəʊˈænɪliːn/ |
| Identifiers | |
| CAS Number | 95-82-9 |
| Beilstein Reference | 1461582 |
| ChEBI | CHEBI:38624 |
| ChEMBL | CHEMBL18773 |
| ChemSpider | 16302 |
| DrugBank | DB13756 |
| ECHA InfoCard | 03d1ef2a-7d7e-47be-8047-d31f5cfb91b7 |
| EC Number | 220-865-6 |
| Gmelin Reference | 60612 |
| KEGG | C06513 |
| MeSH | D014275 |
| PubChem CID | 9166 |
| RTECS number | GY9625000 |
| UNII | 8NRQ6F0G6W |
| UN number | UN2018 |
| CompTox Dashboard (EPA) | DTXSID6020670 |
| Properties | |
| Chemical formula | C6H4Cl3N |
| Molar mass | 197.45 g/mol |
| Appearance | White to gray crystalline solid |
| Odor | Odorless |
| Density | 1.64 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 2.98 |
| Vapor pressure | 1.74E-4 mmHg at 25°C |
| Acidity (pKa) | 3.68 |
| Basicity (pKb) | pKb = 9.5 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.661 |
| Viscosity | 204.3 cP (25 °C) |
| Dipole moment | 3.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -81.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2324 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS05, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H410 |
| Precautionary statements | P261, P264, P270, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P311, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2,4,5-Trichloroaniline: "2-2-0 |
| Flash point | 142°C |
| Autoignition temperature | 220 °C |
| Lethal dose or concentration | LD50 (oral, rat): 1300 mg/kg |
| LD50 (median dose) | Oral rat LD50: 2400 mg/kg |
| NIOSH | SN1820000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Aniline 2-Chloroaniline 4-Chloroaniline 2,4-Dichloroaniline 2,6-Dichloroaniline 2,3,4,5,6-Pentachloroaniline 2,4,5-Trichlorophenol 2,4,6-Trichloroaniline |

