2,4,4-Trimethyl-2-Pentene: A Commentary on Its Role, Risks, and Future Directions
Historical Development
Back in the early to mid-twentieth century, as chemists looked to the increasingly complex demands of industry and transportation, branched alkenes like 2,4,4-Trimethyl-2-Pentene carved out a role in shaping fuels and specialty chemicals. Gasoline quality kept grabbing headlines, and unsaturated hydrocarbons like this one offered solutions—especially when crude oil quality varied and refineries tried to hit tighter specifications. Discovery and early syntheses usually tracked with bigger efforts to control fuel octane or produce building blocks for specialty chemicals. As someone who has traced the pathway of hydrocarbon research, I’ve noticed that even small shifts in molecule structure opened whole new use cases in solvents, performance additives, or resin production. Knowledge trickled down from academic corners, where names like Ziegler, Natta, and the synthetic organic community developed approaches to tailor alkenes for downstream chemistries.
Product Overview
2,4,4-Trimethyl-2-Pentene stands out among branched pentenes. It lands right in the sweet spot for certain blending and reaction purposes—neither so volatile as to create handling nightmares, nor so inert as to limit process value. Folks in refining and petrochemicals know it as one of the main “trimethylpentene” isomers, important for tuning physical properties in specialty fuel fractions and synthesizing other organics. Its profile means it often serves as an intermediate—never hogging the spotlight itself but smoothing the way for higher-value products down the line.
Physical & Chemical Properties
This compound brings a predictable package: clear, colorless liquid at standard temperature and pressure, reeking of that faintly sweet, hydrocarbon scent typical of branched alkenes. Boiling point usually falls in the low to mid-100s Celsius, and as a non-polar molecule, solubility skews strongly toward organic media such as ether or hexane. One glance at its logarithmic partition coefficient shows significant hydrophobicity, placing it on the watch list for bioaccumulation studies—an area I follow closely given the real-world environmental stakes. Double bond location and branching together give it reactivity patterns that chemists leverage for further transformations, especially electrophilic additions or polymerizations when conditions allow.
Technical Specifications & Labeling
Labeling conventions orbit around its main IUPAC name, but the market sees synonyms like diisobutylene cropping up from time to time. Specifications get hammered out in context: purity targets above 98% remain typical for use as an intermediate, while trace impurity limits reflect the downstream process risks. I’ve read technical bulletins that stress the importance of moisture control, given that water reacts unfavorably under acid-catalyzed reaction settings. Labeling for shipping emphasizes flammability, with standard pictograms and clear hazard statements—there’s no room for error, as liquid hydrocarbon spills spell trouble for both plant safety and environmental compliance.
Preparation Method
The main industrial synthesis of 2,4,4-Trimethyl-2-Pentene takes root in acid-catalyzed alkylation. Reacting isobutene with isobutylene under carefully tuned acid catalysis leads to this pentene isomer as a principal product. Catalysts often skew toward strong mineral acids—think sulfuric or hydrofluoric—raising operational demands in plant equipment and safety protocols. If you walk through a production plant, the distinctive tang from the acid processes leaves no doubt about the chemical intensity underway. Yields and product distributions track with catalyst loading, temperature, and the presence of water or trace impurities. Downstream purification relies on fractional distillation, leveraging the boiling point spread between 2,4,4-Trimethyl-2-Pentene and its coproduced isomers or unreacted feed.
Chemical Reactions & Modifications
This compound’s double bond attracts attention from designers of synthetic pathways. 2,4,4-Trimethyl-2-Pentene reacts readily with halogens, hydrogen halides, and strong acids—classic Markovnikov and anti-Markovnikov orientations shape what new products appear. Hydrogenation transforms the alkene into the corresponding alkane, often under mild conditions with a palladium or platinum catalyst. I’ve watched polymer chemists use branched pentenes like this in oligomerization reactions, steering molecular weight and microstructure through catalyst tweaks. The down-the-line chemistry matters, since the physical and environmental fate of derivatives spins out into everything from gasolines to chemical feedstocks.
Synonyms & Product Names
For anyone navigating chemical catalogs, this compound turns up as 2,4,4-Trimethyl-2-Pentene, diisobutylene, or sometimes simply “DIB.” Disambiguating among commercial names demands close reading, since “diisobutylene” can cover a pair of structural isomers. Mislabeling here triggers ripple effects—wrong assumptions in stoichiometry, regulatory reporting, or even emergency response if accidents happen. The importance of settled nomenclature cannot be overstated, especially as companies globalize and standards get harmonized across legal frameworks.
Safety & Operational Standards
Anyone handling this liquid must take fire and explosion risks seriously—it flashes at low temperatures, forming flammable vapor-heavy air mixes at ambient conditions. Regulatory bodies put 2,4,4-Trimethyl-2-Pentene in the “flammable liquid” slot, right alongside structurally similar alkenes. From firsthand experience, I’ve seen what slip-ups in static discharge rules or open flames on production floors can do—flammable vapor concentrations build up fast. Inhalation doesn’t pose the same severe acute toxicity as benzene or toluene, but high exposures in confined spaces depress the central nervous system and displace oxygen. Skin contact tends to dry or irritate but rarely sensitizes. Inspections focus on spill trays, vapor detectors, and robust ventilation—not since foot-dragging on containment and detection has led to catastrophic losses in other hydrocarbon installations.
Application Area
2,4,4-Trimethyl-2-Pentene fits into several chemical supply chains. In refining circles, it lands as a blending stock dialed for higher octane, minimizing engine knock in spark ignition engines and matching fuel performance for modern emission standards. Outside fuels, derivatives end up in plasticizers, solvent systems, and resin precursors. Certain alkylation reactions rely on its double bond geometry, giving access to tertiary alkyl derivatives found in performance lubricants or polymer modifiers. Industry continues hunting for compounds that punch above their weight in terms of reactivity per unit cost, and branched pentenes often make the cut. As a side note, research labs keep this on hand for mechanistic studies and combinatorial synthesis sequences, especially in the pursuit of new functionalized organics.
Research & Development
Research cycles have focused on several fronts: new catalysts that up selectivity, process routes that reduce acid waste, and safer operational protocols topping the safety leaderboards. Academics and industrial researchers trade data on green alternatives—zeolites, ionic liquids, or supported Lewis acids that curb hazards without eating into throughput. Process engineers tout closed systems, in-line sensors, and predictive maintenance, all designed to keep risk down and productivity ticking over. In the last decade, computational studies pick up the slack, helping forecast what modifications to alkene structure change fuel or polymer performance. Innovation breeds optimism, but commercial roll-out lags behind when every new catalytic material invites long-scale production testing.
Toxicity Research
Even with low acute toxicity, 2,4,4-Trimethyl-2-Pentene demands vigilance. Its metabolic fate in mammals tracks with other similar alkenes, generally breaking down via oxidation and eventual excretion. Laboratory assays find no slam-dunk evidence of carcinogenicity, but the lack of long-term human epidemiology keeps risk assessors on alert. If improperly handled, vapor concentrations cause headaches, dizziness, and—at high doses—loss of coordination. Environmental health studies track how spilled hydrocarbons persist in soil, noting that branched structures degrade more slowly in cool, anaerobic zones. Experience shows early risk management—tight container closures, vapor recovery, and regular exposure monitoring—heads off the prospect of chronic exposures in workers or residents near production facilities.
Future Prospects
Looking ahead, 2,4,4-Trimethyl-2-Pentene straddles a line between legacy hydrocarbon chemistry and emerging demands for greener alternatives. Pressure mounts for sustainable feedstocks, cueing R&D attention toward bio-based routes, process intensification, or clever recycling schemes. Political and market winds push petrochemical players to rethink raw material sourcing, lifecycle risk, and emissions. New catalysis, continuous flow processes, and advanced monitoring architect a path forward. Stakeholders in both industry and academia find themselves at a crossroads: either double down on making these hydrocarbons cleaner and safer for people and ecosystems, or step aside as biobased chemistries gain traction. Choices made over the next few years set the tone for industrial chemicals as global society navigates toward a less fossil-dependent future.
What are the primary uses of 2,4,4-Trimethyl-2-Pentene?
What’s So Useful About 2,4,4-Trimethyl-2-Pentene?
Anyone walking into the world of chemical manufacturing will run into some strange names. 2,4,4-Trimethyl-2-pentene stands out as one of those tongue-twisters that actually pulls quite a bit of weight behind the scenes. This colorless liquid doesn’t usually make it into news headlines, but ask people who work with fuel additives, lubricants, and specialty chemicals—they know it as a workhorse.
The Octane Player in Gasoline Additives
Most folks just want their car to run well, but that smooth ride owes a lot to what happens at the molecular level. 2,4,4-Trimethyl-2-pentene steps up as a building block in the production of high-octane gasoline. The petroleum industry leans on this compound during the alkylation process, which means adding branches to molecules so the resulting fuel resists engine knocking and runs more efficiently. Increased octane means better performance and cleaner running engines.
The numbers back up this importance. According to research by the International Energy Agency, cleaner and more efficient gasoline cuts down on greenhouse gas emissions and air pollution, especially in urban areas where cars outnumber people. 2,4,4-Trimethyl-2-pentene doesn’t clean your air by itself, but it acts as a stepping stone in producing those cleaner fuels that make a difference every day.
Building Blocks for Synthetic Materials
If you’ve ever opened a bottle of motor oil or touched synthetic rubber grips, there’s a chance you’ve encountered something made using 2,4,4-trimethyl-2-pentene. The chemical industry loves compounds like this as starting points for synthesizing antioxidants and surfactants. These additives extend product life, improve stability, and shield engines and machinery from wear and tear.
Chemists often refer to 2,4,4-trimethyl-2-pentene as an “intermediate.” That means it helps synthesize other molecules that deliver real value, such as lubricant additives or detergents in household and industrial settings. From my own conversations with engineers in these industries, they point out that every percentage point in product stability or engine efficiency translates into millions saved and waste reduced.
Why Safety and Environmental Considerations Matter
2,4,4-Trimethyl-2-pentene isn’t a sleeper threat, but it does demand respect in handling. At production sites, teams rely on good ventilation and proper containment. The Occupational Safety and Health Administration and similar agencies offer clear guidelines. By respecting best practices, industry reduces spills, emissions, and worker exposure.
There’s an increasing push for greener chemistry, so companies now keep looking for ways to recycle or reuse pentenes and other similar hydrocarbons. Investing in closed-loop systems allows the industry to capture unused fractions and reduce the need for new raw materials. As far as sustainability goes, these changes will make a measurable difference, provided leadership pushes continuous improvement and knowledge-sharing between plants.
Looking Forward
As electric vehicles start to take over roads worldwide, compounds like 2,4,4-trimethyl-2-pentene won’t vanish overnight. Even outside of fuels, their role in making lubricants, plasticizers, and specialty chemicals remains solid. Anyone interested in chemistry’s real-world impact will find this compound quietly making everyday life possible—driving, maintaining, and protecting the machines and materials that keep modern society moving. Reaching for more sustainable production methods seems like the next smart step.
What is the chemical formula and molecular weight of 2,4,4-Trimethyl-2-Pentene?
Chemical Formula and Structure
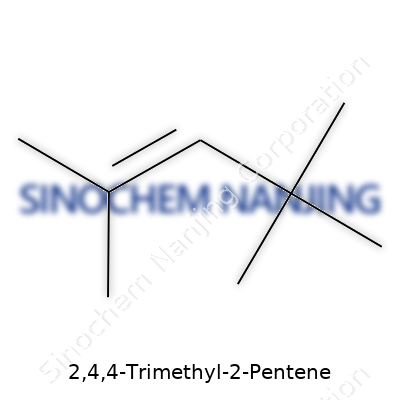
2,4,4-Trimethyl-2-pentene carries the chemical formula C8H16. The structure features an eight-carbon chain with three methyl groups snug on the backbone, and a double bond planted at the 2-position. Chemists look at this as a branched alkene, not just a mouthful for a name but a classic example of the variety found among hydrocarbons. Each molecule has sixteen hydrogens dangling off those carbons, so you get an isomer of octene, not far from cousins like 2,3,4-trimethylpentene in the lab.
Molecular Weight and Why It Matters
Flip open a handbook, and you’ll see the molecular weight of 2,4,4-Trimethyl-2-pentene is 112.21 g/mol. This matters whether you’re weighing it in a lab or figuring stoichiometric loads in industrial batches. I remember years ago, during an undergrad synthesis, just ignoring a decimal in a molar mass led to a scaled-up mess on the bench. Formula weight forms the backbone of safe, efficient chemical practice, and for scaling production, even tiny mistakes drive costs up or gum up the process.
Industrial Use and Significance
This alkene steps out of textbooks and into real-world processes. You’ll spot 2,4,4-Trimethyl-2-pentene in fuel blending. Its structure lends high octane, keeping engines from knocking and prolonging engine life, which isn’t just about nicer car rides—it ripples out to energy efficiency, maintenance savings, and emissions. Gasoline refineries have leaned on these branched alkenes since the days of leaded fuel. Refiners value its balance: not so volatile it evaporates away, not too heavy to drag performance down.
Safety and Handling in the Chemical Landscape
Lab veterans all have stories about why clear protocols matter. 2,4,4-Trimethyl-2-pentene flows as a clear, volatile, flammable liquid. Ignition points and vapor pressure stats dictate storage and transport. Workers depend on firm knowledge: no open flames, good ventilation, and tight seals on bottles. Recent years brought tighter regulations, driven by lessons from plant accidents and data on long-term exposure risks. Tight labeling, real-time monitoring, and easy-to-follow datasheets are now the norm in most chemical warehouses.
Ways to Support Sustainable Practices
Chemical firms can help by adopting greener synthesis routes and investing in closed-loop recycling for volatile organics like 2,4,4-Trimethyl-2-pentene. Instead of releasing these compounds after use, tech has caught up to reclaiming and purifying them, turning a waste stream into a feedstock. In the classroom and the lab, small changes stick: use spill mats, work with only what you need, and teach students why the basics—like knowing your chemical’s formula and mass—aren’t just for passing a quiz. They shape safety and sustainability down the line.
Staying Informed
Staying up to date with peer-reviewed journals, regulatory changes, and trade association bulletins keeps the work safer and smarter. I keep a bookmarked list of the CRC Handbook and regularly check the EPA’s database. Clear insight, never just blind adherence to procedure, builds trust: employees learn to spot risks, catch mislabeled bottles, and understand if new data means it’s time to swap in safer alternatives.
How should 2,4,4-Trimethyl-2-Pentene be stored and handled safely?
A Clear Look at Why Safety Matters
2,4,4-Trimethyl-2-pentene isn’t something you’d leave out on a lab bench or toss into a plastic jug in the garden shed. This stuff counts as a volatile organic compound, and if you’ve spent any time in a chemical storeroom or worked around bulk chemical drums, you understand those invisible vapors are a real worry. In my own lab days, I watched a seasoned chemist go through ritual-like steps just to move a can of this material. Flammable vapors escape fast. One spark means big trouble.
Practical Ways to Store It Safely
No one wants to pull up in the morning to a chemical fire. The storage spot should offer shade, good airflow, and a guarantee that the temperature won’t swing up and down like a yo-yo. Metal safety cans stand tough against leaky seals and don’t break down from exposure as soft plastics do. Dedicated flammable chemical cabinets—well-labeled, fitted with vents—keep this material from mixing with possible oxidizing agents. I’ve seen companies skimp and tuck containers behind old boxes; that shortcut shows up later as an insurance claim.
Regular folks may not realize just how much vapor this compound gives off at room temperature. That’s why a real fire risk exists, even in air-conditioned labs. Stats show industrial fires often happen because damp cardboard or ignored wood shelves pick up a trace odor and nobody takes action. Responsible storerooms swap out those absorbent materials for metal shelving, proper spill trays, and clear walkways. Out in the field, folks run regular checks for bulging cans. Corroded drums get replaced immediately. It costs less than post-incident cleanup or lawsuits.
Handling Mistakes You Can Avoid
Lab tradition runs deep: gloves (nitrile holds up), safety goggles, and no wide sleeves or strings that could catch a valve. Large volumes go into drum pumps—never hand pouring or jerry-rigged siphons. Stories circulate about some workers collapsing from fumes after cracking a vessel in a poorly ventilated room. In real terms, local exhaust ventilation and fume hoods don’t just keep the room comfortable—they stop someone from taking a snootful of unsafe vapors. I remember seeing well-labeled squeeze bottles set beside patch kits. One smart manager put color-coded tape on every line and container; nobody mixed up compounds after that simple step.
Regulations ask for proper signage, and with good reason. If a spill happens, fast eye washes and drench showers can spell the difference between a hospital visit and a wash-up at the sink. In busy industrial spaces, everyone benefits from spill kits, sand, or absorbents nearby. Training doesn’t stop with new hires; regular reviews catch gaps or bad habits before someone gets hurt.
Bigger Picture: Good Habits Keep Everyone Safe
Storing and handling 2,4,4-trimethyl-2-pentene takes more than reading a label. Choosing the right cabinet, using the best safety gear, and investing in regular training all help. According to agencies like OSHA and NFPA, fire risk goes down sharply as soon as staff learn the “why” behind each precaution. Chemistry isn’t just about formulas—it’s about respect for what every compound can do. Companies and schools, large or small, set a tone by following strong safety routines. Once these habits become part of the culture, the whole team looks out for one another, and risks shrink with each passing day.
What are the physical and chemical properties of 2,4,4-Trimethyl-2-Pentene?
What Sets 2,4,4-Trimethyl-2-Pentene Apart
Spend any time around a chemical lab or refinery, and you cross paths with countless compounds whose names barely fit on a label. 2,4,4-Trimethyl-2-pentene fits right in: the mere string of numbers and dashes can throw off the casual reader. Underneath the complicated name sits a clear, colorless liquid that brings something valuable to several corners of industry and research.
Physical Traits: Everyday Observations from the Lab Bench
2,4,4-Trimethyl-2-pentene doesn’t leave behind much for the senses. No striking color, just a transparent liquid. It’s got a distinct smell — not overpowering, but sharp and a bit sweet. Using it in my college lab, I always noticed it tended to evaporate faster than water. That volatility says a lot since its boiling point lands around 99 degrees Celsius. Stick it in the fridge, and it won’t freeze unless you drop the temperature below roughly -95 degrees Celsius.
The density of this compound usually sits a bit below water, at about 0.72 grams per cubic centimeter. That comes into play anywhere it mixes with other liquids, especially if separation by density is handy. Spills in the lab make clear the difficulty of mopping up such a light liquid—its movement across surfaces is quick and slippery.
Chemical Behavior: What Happens Under the Hood
2,4,4-Trimethyl-2-pentene stands as an alkene. That double bond gives it a place in many classic organic reactions. In real terms: add a strong acid, and it will want to grab a water molecule; bring in a bit of bromine, and the color fades as the reaction moves forward. The compound doesn’t mix with water, not even a little, but throw it in with most organic solvents — think acetone, ether, even benzene — and it blends in completely.
Combustion is another area that matters, especially given the fire risk around volatile organics. It burns easily, giving off heat, water vapor, and carbon dioxide — old friends for anyone who deals with hydrocarbon chemistry. Good ventilation in the workspace is important not only for safety but for controlling strong odors. Its vapors form flammable mixtures with air at room temperature, so strict controls and spark-free zones around storage or use areas keep everyone safe.
Why These Properties Matter in the Real World
Working in industrial settings, I’ve seen 2,4,4-trimethyl-2-pentene used as a building block for making other chemicals. It serves in the production of certain fuels and additives. Because it reacts predictably at the double bond, chemists rely on it for synthesizing more specialized compounds. The high volatility makes it valuable in processes where quick evaporation is an advantage, but it also means accidents can escalate quickly if basic precautions get skipped.
Balancing Opportunity and Risk
Many labs still use old glassware and open benches, but handling this compound demands up-to-date fume hoods and vapor suppression techniques. Proper labeling, secondary containment, and staff training make the difference between smooth operations and hazardous situations. Fire drills, clearly marked extinguishers, and an understanding of local chemical disposal laws all support safe, responsible use. From personal habit, I always double-check connections and gaskets before starting a reaction—anything to avoid releasing flammable vapors into the workspace.
The story with 2,4,4-trimethyl-2-pentene matches that of many modern chemicals: practical benefits paired with clear risks. Respect the properties, and they open doors in chemistry. Ignore them, and you pay the price quickly.
Is 2,4,4-Trimethyl-2-Pentene hazardous, and what precautions should be taken?
Understanding the Chemical
2,4,4-Trimethyl-2-pentene—sometimes popping up in industrial settings as a building block for other chemicals—doesn’t get much attention on the nightly news. In most cases, it lurks behind the scenes in oil refineries or as part of gasoline blends. But its low profile doesn’t make it harmless. This liquid evaporates easily, spreading its sharp, fuel-like odor. Exposure can sneak up during handling, transport, or any tank cleaning activities.
Where Hazards Show Up
I’ve seen plenty of folks shrug off clear liquids and odd smells in industrial plants, assuming “clear equals safe.” 2,4,4-Trimethyl-2-pentene doesn’t play by that rule. Even short contact can irritate the eyes, skin, and lungs. In tight spaces, its fumes push the air out—an oxygen squeeze nobody wants. Sore throats, headaches, and dizziness can follow a few unguarded sniffs. The bigger risk: in high concentrations, the vapor can even spark unconsciousness.
Breathing this chemical over weeks or months raises other issues. The lungs and skin become more sensitive, raising the chance of allergic reactions or long-term respiratory trouble. This isn’t just my experience—regulatory agencies issue warnings for a reason. The science ties repeated exposure to chronic problems. Then there’s the matter of flammability. Fumes from this chemical catch fire fast, even at lower temperatures, making workplace sparks or static big red flags.
Precautions—And What Actually Works
For anyone working around chemicals like this, the basics never go out of style. Good ventilation means less risk—nobody benefits from stagnant air when fumes can build. Simple fans often fall short; sometimes only a well-designed exhaust system gets the job done. I learned early to watch out for corners, pits, and other spaces where vapors pool. One slip or spark there, and things go downhill fast.
Protective gear should not be brushed aside. Gloves that resist chemicals, splash-proof goggles, and long sleeves help, but they only matter if worn every time—not just “when management’s looking.” Too many stories start with “I was just doing a quick transfer.” Skin contact leads to rashes or blisters, and removing contaminated clothing quickly is no joke. For anyone who works in teams, keeping a spill kit close by and knowing how to use it can keep a minor accident from blowing up.
Information and Training—A Missing Link
Plenty of workers see signs and labels but treat them as background noise. Real safety starts when people understand what chemicals like 2,4,4-Trimethyl-2-pentene can do, and how to actually respond when something goes wrong. Clear instruction—what to do if eyes burn, the right respirator to grab, how to secure storage drums—beats paperwork every time. Regular drills, honest talk about near-misses, and checks on PPE condition make a difference.
Better Work Practices—Reducing the Risks
It surprised me how much spills and leaks centered around simple slip-ups—a loose valve, a sloppy pour, ignoring a tiny crack in a hose. Routine inspections cut down on these risks more than any rulebook can promise. Choosing closed systems or transfer pumps over open buckets tightens safety. Fire extinguishers, grounding wires, and keeping spark-producing tools far from transfer work round out the plan.
The truth: 2,4,4-Trimethyl-2-pentene works best as a reminder that old-school vigilance still matters. The fixes aren’t flashy, but sticking to them means workers finish their shifts healthy. And no one at home has to wonder what’s waiting in the air or on the laundry.
| Names | |
| Preferred IUPAC name | 2,4,4-Trimethylpent-2-ene |
| Other names |
Diisobutylene Trimethylethylidenebutane 2,4,4-Trimethylpent-2-ene |
| Pronunciation | /tuː fɔːr fɔːr traɪˈmɛθɪl tuː ˈpɛntiːn/ |
| Identifiers | |
| CAS Number | 107-39-1 |
| 3D model (JSmol) | `C\C(=C(/C)\C)\C` |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:88399 |
| ChEMBL | CHEMBL153135 |
| ChemSpider | 13580 |
| DrugBank | DB14096 |
| ECHA InfoCard | 03a5c857-7265-4498-ae1a-9cdf48027c36 |
| EC Number | 203-492-7 |
| Gmelin Reference | 145894 |
| KEGG | C06534 |
| MeSH | D014240 |
| PubChem CID | 11227 |
| RTECS number | RZ1050000 |
| UNII | D89J30Y230 |
| UN number | UN2278 |
| CompTox Dashboard (EPA) | DTXSID7021734 |
| Properties | |
| Chemical formula | C8H16 |
| Molar mass | 98.19 g/mol |
| Appearance | Colorless liquid |
| Odor | mild odor |
| Density | 0.703 g/mL at 25 °C |
| Solubility in water | Insoluble in water |
| log P | 2.9 |
| Vapor pressure | 2.7 kPa (at 20°C) |
| Acidity (pKa) | 19.7 |
| Magnetic susceptibility (χ) | -8.74 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.417 |
| Viscosity | 0.674 mPa·s (20°C) |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 284.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –45.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4647.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P273, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 14 °C (57 °F; 287 K) (closed cup) |
| Autoignition temperature | 446 °C (835 °F; 719 K) |
| Explosive limits | Explosive limits: 1.0–6.0% |
| Lethal dose or concentration | Lethal dose or concentration of 2,4,4-Trimethyl-2-Pentene is: "LD50 (oral, rat): >5 g/kg |
| LD50 (median dose) | LD50 (median dose): oral, rat: 1700 mg/kg |
| NIOSH | SN9847000 |
| REL (Recommended) | REL: NIOSH considers this chemical to have no established REL. |
| IDLH (Immediate danger) | IDLH: 900 ppm |
| Related compounds | |
| Related compounds |
2,4,4-Trimethyl-1-pentene Isooctene 2,4,4-Trimethylpentane 2,3,4-Trimethyl-2-pentene 2,4,4-Trimethyl-3-pentene |

