The Many Sides of 2,3-Xylenol: Looking Beyond the Chemical Formula
Historical Development
In the years since synthetic chemistry took off, aromatic compounds have played a big role in everything from dye manufacture to medicine. 2,3-Xylenol, tucked into the greater xylenol family, started showing up in research as scientists chased after chemicals they could modify and turn into something valuable. Early studies, driven by the demand for phenolic compounds in antiseptics and resins, flagged 2,3-xylenol as a promising candidate, partly thanks to its position as one of the less common isomers in the group. Access to more precise distillation and separation methods during the twentieth century pushed it from being just another byproduct into a compound with clearer industrial significance.
Product Overview
Ask a chemist what stands out about 2,3-xylenol, and you’ll probably get an earful about methyl groups and hydroxyl placement. Outside the lab, it doesn’t exactly show up on supermarket shelves, but it lurks behind the scenes in niche manufacturing and research. Workers in custom resin production, specialized solvents, and dye chemistry see its advantages every day. What often gets missed outside specialty circles is how much its odd combination of characteristics—both reactive and selective—opens doors for precision synthesis.
Physical & Chemical Properties
2,3-Xylenol is one of those organic molecules that stands out more in practice than on paper. The addition of methyl groups at the 2 and 3 positions creates a cocktail of reactivity and steric hindrance. Its melting point sits below room temperature, so it tends to appear as a liquid under ordinary conditions. Odor alone can make it tricky to handle day in, day out. In terms of solubility, it dissolves in organic solvents fairly well but acts stubborn around water. The aromatic ring grants some chemical stability but the hydroxyl group remains ripe for substitutions, esterifications, and coupling reactions.
Technical Specifications & Labeling
Labeling for 2,3-xylenol sticks to strict guidelines. Purity requirements typically specify anything above 98 percent for reagent grade, and all containers require clear hazard communication. Containers bear labels flagging both acute toxicity and flammability, and the documentation travels with every shipment. Anyone shipping or receiving this chemical learns to respect the potential risks; no one wants mystery chemicals in their inventory.
Preparation Method
Production for 2,3-xylenol usually starts with methylation of phenol or separation from crude xylenol mixtures. Alkylation with methanol or methyl chloride in the presence of acid catalysts offers higher selectivity and less environmental headache compared to older, dirtier routes. Some facilities pull it out from tar distillates, then lean on precise fractional distillation. As demand rose for specialized chemicals in the electronics and resins sectors, more refineries invested in better purification and recovery processes, emphasizing yield and consistency.
Chemical Reactions & Modifications
Many chemists see the hydroxyl and methyl combo as a starting point for elaborate synthesis schemes. The phenolic OH crowns the molecule as both a nucleophile and a leaving group, partnering nicely in etherification, ester formation, or azo coupling for dyes. Ring methylation supplies sites for further functionalization. Whether turning it into advanced polymer coatings, plasticizers, or more complex heterocycles, 2,3-xylenol keeps showing its value as a flexible substrate.
Synonyms & Product Names
On paper, chemists will call this molecule 2,3-dimethylphenol. Elsewhere, you hear names like 2,3-xylenol, meta-xylenol, or 2-hydroxy-meta-xylene. Trade literature sometimes muddles things, mixing up isomers or grouping all xylenols together—an oversight that can trigger confusion for buyers and lab workers alike. Accurate naming makes a difference, especially as regulatory agencies tighten requirements on chemical identification to prevent safety slip-ups.
Safety & Operational Standards
Anyone with experience handling phenolic chemicals knows both the risks and the right way forward. 2,3-Xylenol can irritate skin, eyes, and airways, and it packs some serious punch as a systemic toxin at higher exposure levels. Standard protocols call for gloves, goggles, and strong ventilation. Many companies install extra air filtering, especially since vapor exposure can creep up in warm workspaces. Training workers to spot leaks or spills, alongside regular checks on storage facilities, forms the backbone of responsible operations. Industry guidelines, based on real-world incidents and toxicological data, shape how every batch gets stored, labeled, and transported.
Application Area
While not as widely used as some of its isomer siblings, 2,3-xylenol carves out a niche in resin molds, high-performance adhesives, and certain photographic chemicals. Specialty dyes and pigment synthesis sometimes pivot on the subtle shift one methyl group can cause in end-product color or performance. In the pharmaceutical sector, its structure offers stepping stones toward more complex molecules. Electronics eventually joined the ranks, as demand for customized high-purity phenols expanded alongside new needs in coatings and resist materials.
Research & Development
Research groups look for ways to push aromatic chemistry forward through better catalysts, cleaner methylation agents, and safer phenol transformations. Universities and private labs test greener techniques for both making and breaking molecules like 2,3-xylenol. More recent studies explore replacing harsh solvents and optimizing yield while keeping hazardous waste low. The hunger for new plastics and next-gen electronic components keeps pushing for higher-purity, specialty phenols, extending 2,3-xylenol’s shelf life in R&D circles.
Toxicity Research
Toxicologists flag 2,3-xylenol as a compound that warrants close attention, especially inside industrial settings. Studies of animal exposure highlight central nervous system effects and metabolic stress at high doses. Its ease of skin penetration, typical of low-molecular-weight aromatic alcohols, means protection gear is more than just a box to check. Research looks at chronic low-level effects as well as acute poisoning, and regulators update recommendations as new findings stack up. Long-term environmental persistence doesn’t rival some heavier polyaromatics, but careful handling and clear disposal protocols matter either way.
Future Prospects
The drive toward sustainable chemistry puts molecules like 2,3-xylenol under the microscope. Regulations push for both greener synthesis and safer alternatives without losing industrial utility. As the landscape shifts, demand may grow for phenols tailored to high-spec applications, possibly expanding roles in electronics, medical coatings, or smart materials. Researchers hunt for ways to recycle or upcycle xylenolic residues, and green chemistry startups try swapping legacy feedstocks for biobased routes. In any scenario, as chemical manufacturing blends innovation with caution, 2,3-xylenol keeps drawing both opportunity and scrutiny.
What is 2,3-Xylenol used for?
An Industrial Fix for Modern Problems
2,3-Xylenol rarely gets the spotlight in most households, yet this little chemical keeps everyday life humming in the background. If you’ve ever picked up a can of disinfectant or a household cleaner and glanced at the ingredients, you probably noticed some obscure names. Chances are, 2,3-Xylenol helped build the chemical backbone of those products. Found in liquid and sometimes crystal form, it comes through petrochemical production—basically, reworking parts of crude oil or coal tar into building blocks for things we actually use.
Disinfectants and Cleaners Rely on It
One direct use sits in antiseptics and germ-killing solutions. At its core, 2,3-Xylenol disrupts bacteria and helps halt their growth. Hospital disinfectants need powerful ingredients that knock out pathogens before they can hitch a ride. My own years working in lab support drove home how much hospitals lean on tough cleaners to keep surfaces safe for anyone who steps through the door. 2,3-Xylenol plays a strong part in these cleaning agents—if it gets left out, infection risks start rising.
It doesn’t stop at hospitals. Commercial kitchen sanitizing sprays or the wipes in public restrooms lean on compounds like 2,3-Xylenol. Multiple studies, including those in the Journal of Hospital Infection, show that o-xylene derivatives add a punch to routine cleaning, reducing risk of bacteria sticking around between shifts.
A Middleman in More Complex Chemistry
Industry uses 2,3-Xylenol as a stepping stone, not just as a finished product. Companies blend or modify it to build dyes, resins, and herbicides that wind up in fabrics, plastics, and crop protection. This single molecule branches out into many, depending on what chemists transform it into next. From coloring synthetic textiles to making adhesives grip with more strength, 2,3-Xylenol supports more products than most realize.
Why Safe Handling Matters
With every useful property comes a demand for responsibility. Long ago, casual handling of aromatic compounds like 2,3-Xylenol led to health concerns. Exposure can irritate skin and lungs, and in factories, even minor spills can mean headaches for workers who don’t have the right protection. Regulations around the world now call for gloves, fume hoods, and storage guidelines to keep accidental poisonings at bay. The rise in regulatory oversight, from agencies like OSHA and equivalents in Europe and Asia, reflects growing awareness. As soon as these guidelines get strict, injury rates drop and workers feel safer clocking in every day.
Cleaner Alternatives and Green Chemistry
The reality for most industries: dropping hazardous chemicals cold turkey rarely pans out. Alternatives for 2,3-Xylenol in disinfectants exist, but performance or cost often lags behind. Some companies invest in bio-based phenols or plant-derived cleaners, though these hardly see the scale or reliability yet. A greater focus on better chemistry—from suppliers and manufacturers—could steer more businesses toward gentler options. R&D labs keep testing new variants that could slash toxicity without losing the effectiveness clean industries demand.
2,3-Xylenol supports public health, keeps production lines moving, and gives chemists a flexible tool. Understanding the balance between utility and safety lets us keep using this compound smartly without risking workers or the environment.
Is 2,3-Xylenol hazardous to health?
Everyday Chemicals and Real Risks
Many people do not think twice about the chemicals used in industrial processes and products all around them. One chemical that deserves attention is 2,3-Xylenol, known as a solvent and ingredient in disinfectants, dyes, resins, and pesticides. Working in environmental health taught me that not all chemicals share the same risk, but history gives us plenty of lessons about the need for careful handling of industrial ingredients.
What Science Tells Us about 2,3-Xylenol
2,3-Xylenol belongs to a group called methylphenols. These are cousins of phenol, a chemical with a strong track record for causing skin, lung, and eye damage when not handled properly. I remember flipping through the Material Safety Data Sheet and learning that exposure to 2,3-Xylenol through the skin or air can cause irritation. If someone were to breathe too much on the job, they could expect headaches, coughing, or even nervous system symptoms at higher exposures. Splashing the stuff on bare skin can mean redness, burns, or blisters in tough cases. Swallowing even small amounts leads to nausea, vomiting, and—in the worst cases—serious harm to organs like the liver or kidneys.
Lab studies back up these reports. The U.S. National Library of Medicine and the European Chemicals Agency both list 2,3-Xylenol as harmful when inhaled, swallowed, or absorbed. No one puts this chemical in household cleaners for a reason. Its safety margin sits pretty far from what you find in vinegar or lemon oil sprays. Research remains limited on long-term effects. What scientists have pieced together shows repeated exposure can build up over time, stressing out organs and raising cancer risks in animals.
Why We Should Pay Attention
Take a walk near an older industrial site, and you see signs for “Hazardous Waste,” “Authorized Personnel Only,” and the heavy presence of chemical storage drums. Working for a short time in waste management, I saw too many workers play down personal protective equipment. One careless accident with a substance such as xylenol can wipe out weeks of recovery time and run up thousands in health care bills. Strict labeling and safety controls exist for a reason; the stories of people burned, poisoned, or worse give context to dry hazard statements.
Skin exposure or breathing fumes is not just a workplace issue either. Improper disposal fouls up groundwater and soil, carrying risks long after the original barrels rusted out. Studies link methylphenols in tainted water to health effects in communities nearby. Children, pregnant women, and people with breathing troubles stand at the highest risk even at lower doses. This knowledge calls for real watchfulness — not only in the lab or factory but across the supply chain.
Better Solutions, Safer Choices
Eliminating health hazards from 2,3-Xylenol starts with practical changes. Safety training should stress real-life consequences, not just check a box on a form. Workers and supervisors need access to gloves, goggles, and fresh air systems. Downstream, companies that use such chemicals should tighten policies for storage, handling, and transport. Regular inspections, accident drills, and strong reporting make all the difference. On the environmental front, greener substitutes in production drop long-term risk. More research on non-toxic alternatives gives industries new choices, protecting workers and nearby families at the same time. Public demand for safer products can steer businesses away from hazardous ingredients. Lasting protection against chemicals like 2,3-Xylenol means putting health over shortcuts, from start to finish.
What is the chemical formula of 2,3-Xylenol?
The Heart of the Matter: 2,3-Xylenol’s Structure
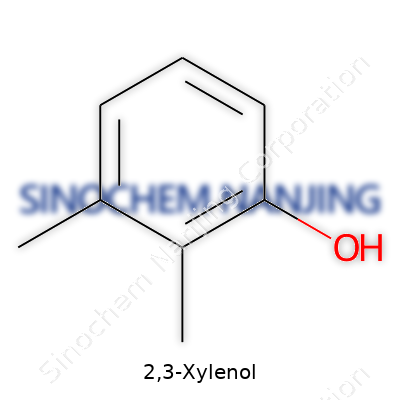
Curiosity never stops pulling me toward molecules and how small changes on a ring of atoms can create something brand new. Take 2,3-xylenol—not an everyday kitchen item, but a compound that deserves some attention. This molecule comes off as a slight twist in the xylene and cresol family. It has a benzene ring just like its chemical cousins. If you look at its formula, you see C8H10O. That means eight carbon atoms, ten hydrogens, and a single oxygen atom. Pretty neat and straightforward. But why does this arrangement matter?
The Details Behind the Numbers
Benzene, a staple foundation in chemistry, brings together six carbon atoms in a hexagon. Xylene shows up as benzene with two methyl groups (–CH3) attached somewhere on that ring. Now, toss in a hydroxyl group (–OH) for a phenol backbone. With 2,3-xylenol, the –OH hangs onto the first carbon, then methyl groups hook into the second and third carbons. This gives it its full name: 2,3-dimethylphenol. No need to recite molecular geometry—simple numbers tell the story. The chemical formula sticks with the basics: C8H10O.
Why the Structure Matters Beyond the Lab
In college labs, I remember discussions that circled around how small shifts on an aromatic ring brought wildly different smells and reactivity. 2,3-xylenol often finds itself in industrial scenes or research settings, serving as a building block for dyes, resins, or pharmaceuticals. Fiddling with those methyl groups, chemists can steer the direction of reactions for more specialized products. The structure controls how molecules react, how they get absorbed, or how they break down. Safety and handling always come up because phenols can irritate skin and lungs, so anyone working with it gets the proper gear and ventilation.
Connecting the Formula to Real Life Impact
Sitting behind these letters and numbers is a story of how basic chemistry fuels real change. Runoff containing phenol derivatives, if tossed carelessly, harms water sources, wildlife, and the communities nearby. Strict controls on discharge and production keep health risks down and groundwater cleaner. Even at home, many forget that certain degreasers or disinfectants contain related aromatic compounds—knowledge of their formula spells out the difference between safe use and unexpected exposure. Being aware of what’s in common cleaners or solvents starts with recognizing simple formulas like C8H10O.
Guiding Smarter Choices and Solutions
Over the years, I’ve seen both students and friends shy away from chemistry, worried by all the symbols. In truth, chemical formulas like the one for 2,3-xylenol are more than jargon; they unlock better decision making. When big factories or small businesses choose greener processes for making or disposing of phenolics, everyone benefits. Researchers keep developing alternatives with less toxicity or better biodegradability. The next generation of chemists need access to solid data, hands-on training, and open information on molecular identities and risks. Getting the right facts helps everyone—from the person studying molecules on paper to the communities living next door to production sites.
How should 2,3-Xylenol be stored?
Getting to Know the Risks
I’ve been around warehouses where chemicals share space with forklifts, empty drums, and coffee cups. Stuff like 2,3-Xylenol always stands out on the shelf, partly because the label looks intimidating, but mostly because it deserves real respect. This is a substance with both flammable and toxic sides — storing it haphazardly doesn’t cut it. The chemical’s vapors can irritate the eyes, skin, and respiratory system. Over time, neglecting storage can lead to fires, accidents, or slow health effects for those nearby.
Cool, Dry, and Away from Trouble
Heat is an agitator. From experience, chemicals in metal drums next to heaters or in sunlight always spell trouble. High temperatures can turn 2,3-Xylenol vapors from trace to choking—pressure builds, containers fail, and the results get ugly fast. A climate-controlled storage area with real ventilation keeps things safe. Humidity also plays its part, since dampness can corrode containers and raise the risk of leaks even before anyone notices.
Choosing the Right Containers
Polyethylene and metal drums with tight sealing tops usually work for 2,3-Xylenol. Reusing old containers seems thrifty until you meet the aftermath of a mix-up—two substances reacting or a slow leak that no one sees until they can smell it. Clearly labeled, undamaged containers cut out guesswork and panic when someone needs to move or inspect them.
Storing Far from Sparks and Flames
Nothing feels as nerve-racking as seeing a welding torch or a space heater too close to flammable stock. Flashpoints matter—2,3-Xylenol catches fire at a much lower temperature than a lot of newcomers guess. The fire triangle is simple: you give heat, oxygen, and fuel a chance to meet, and disaster follows. Dedicating a separate area for flammable materials, installing spark-proof lighting and equipment, and outright banning smoking keeps things under control far more than rules tacked to a bulletin board.
Ventilation Cuts Risk
Stale air makes accidents worse. A real ventilation system, not just a cracked window or a wall fan, draws vapors out and gives workers a fighting chance if something spills. Air movement also reduces long-term buildup that eats at containers and creeps into shipping zones. That’s not a luxury; it's basic safety for everyone who works anywhere near the place.
Emergency Gear Makes a Difference
Spills and splashes are not “ifs”—they’re “whens.” Having absorbent materials, chemical-resistant gloves, and goggles near storage areas leaves less room for panic. You end up with splashes that get cleaned and reported, not infections that send people home sick. Eyewash stations and safety showers close by can mean the difference between a bad day and a medical emergency.
Keeping Eyes Open
Routine checks turn out to be worth more than any sign posted on a wall. Leaks, rust, and missing labels show up often enough to remind anyone that complacency breeds accidents. Walking the aisles, looking for bulging drums or loose caps, lets any worker speak up before anyone pays the price. A real training session—where folks know the “why” behind the safety steps—changes the stakes completely.
Why It All Stays Important
Some substances only stay safe under the right conditions. 2,3-Xylenol is one of those. Years spent in and out of storerooms have made it clear: safe storage isn't about red tape, it's about people. Each choice counts. The mix of good storage practices, real training, and usable gear makes all the difference between a near-miss and a crisis. That keeps everyone going home safe at the end of the day.
What are the safety precautions when handling 2,3-Xylenol?
Understanding What You’re Working With
2,3-Xylenol comes from a group known as dimethylphenols. It often finds its way into research, chemical manufacturing, and even as a raw material for other products. Most people outside lab circles don’t cross paths with it, but for those who do, awareness is the first step toward staying safe. My own time in labs has shown me how substances that look harmless end up causing all sorts of problems without the right mindset and habits. 2,3-Xylenol can irritate eyes, skin, and lungs. Breathe it in too much and nausea or headaches can follow. Just a couple of spills taught me that gloves and goggles are never optional with this stuff.
Protecting Yourself Physically
Good safety gear puts a solid barrier between you and anything that could go wrong. Lab coats keep skin safe from splashes. Nitrile or neoprene gloves make sure direct contact won’t happen. Goggles protect eyes from even the smallest drops. If fumes become a concern, a face shield and a respirator support clear breathing. I learned early that latex gloves break down with some solvents and swapping them for nitrile kept me out of trouble. It’s easy to skip a step, especially when rushed, but cutting corners always comes back to bite.
Respecting Ventilation and Storage Rules
It isn’t just about wearing the right gear; the room itself matters a lot. 2,3-Xylenol fumes hang around, especially in warm or unventilated spaces. Fume hoods and exhaust fans work hard to clear the air. My old lab always blocked off a corner for working with volatile organics, well away from communal spaces and with solid airflow. Chemical cabinets, clearly marked and away from acids or oxidizers, keep stray reactions out of the picture. Never store it near food or drinks; accidents happen easier than people think.
Responding to Spills and Exposure
Spills can turn a good day into panic fast. For small accidents, absorbent pads or granules work best. Afterward, the affected area needs a good cleaning with soap and water. Bigger spills demand help—get folks out of the room, hit the emergency exhaust, and call a supervisor. Wash affected skin with water for at least 15 minutes if there’s contact. If someone breathes in too much, moving to fresh air comes first, and if symptoms linger, a trip to health services is a no-brainer. Safety showers and eyewash stations should always be nearby and checked often for function. These steps might sound obvious, but muscle memory kicks in during stress, and training becomes life-saving.
Building a Safer Culture
Rules and training shape safer habits. Every workplace should regularly talk through material safety data sheets (MSDS) and drill for the unlikely events, not just the routine ones. A strong safety culture doesn’t come from paperwork alone. It shows through in constant reminders, clear labeling, and people watching out for each other. In my experience, teams that talk openly about what went wrong after incidents see fewer repeats. It’s better to have an honest debrief than sweep mistakes under the rug.
Staying Ahead with Knowledge
Regulatory agencies such as OSHA set helpful standards, pushing labs toward better protective practices. Following their guidelines and keeping up with new research reduces accidents in subtle but important ways. As chemical handling continues to change with new technology and materials, everyone working with substances like 2,3-Xylenol benefits from stronger awareness and cooperation. It isn’t about checking boxes—it’s about making it home in one piece at the end of the shift.
| Names | |
| Preferred IUPAC name | 2,3-dimethylphenol |
| Other names |
2,3-Dimethylphenol |
| Pronunciation | /tuː θriː ˈzaɪliːnɒl/ |
| Identifiers | |
| CAS Number | 526-75-0 |
| Beilstein Reference | Beilstein Reference 2041484 |
| ChEBI | CHEBI:27736 |
| ChEMBL | CHEMBL14258 |
| ChemSpider | 14208 |
| DrugBank | DB03324 |
| ECHA InfoCard | 03b3ba1a-7feb-4d89-ad4a-2e2b5e7669bf |
| EC Number | 200-578-6 |
| Gmelin Reference | 50235 |
| KEGG | C01434 |
| MeSH | D015575 |
| PubChem CID | 6949 |
| RTECS number | ZE2450000 |
| UNII | 66QX09DD3X |
| UN number | UN2224 |
| Properties | |
| Chemical formula | C8H10O |
| Molar mass | 122.16 g/mol |
| Appearance | Colorless to pale yellow liquid with phenolic odor |
| Odor | Phenolic odor |
| Density | 0.958 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.38 |
| Vapor pressure | 0.17 mmHg (25°C) |
| Acidity (pKa) | 10.31 |
| Basicity (pKb) | 10.13 |
| Magnetic susceptibility (χ) | -64.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.535 |
| Viscosity | 3.93 mPa·s (25°C) |
| Dipole moment | 1.18 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 125.1 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -53.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3627.0 kJ/mol |
| Pharmacology | |
| ATC code | D02AE02 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Hazard statements: H226, H302, H312, H332, H318, H315, H335 |
| Precautionary statements | P210, P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,3-Xylenol: 2-3-0 |
| Flash point | 79 °C (174 °F; 352 K) |
| Autoignition temperature | 530 °C |
| Explosive limits | 1.3–7% |
| Lethal dose or concentration | LD50 oral rat 1210 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1210 mg/kg (oral, rat) |
| NIOSH | UY3500000 |
| PEL (Permissible) | PEL: 5 ppm (skin) |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Phenol Cresols 3,4-Xylenol 2,5-Xylenol 2,6-Xylenol |

