2,3-Epoxypropyl Phenyl Ether: More Than Just Chemistry
A Look Back: Origins and Discovery
Chemistry weaves its way into nearly every modern convenience, and 2,3-Epoxypropyl Phenyl Ether stands as a quiet workhorse behind the scenes of industry and research. Its roots stretch back to the mid-20th century, a period known for chemical exploration that shaped the industrial landscape so deeply you still see its fingerprints everywhere, from plastics to advanced composites. This compound’s introduction coincided with rapid growth in organic synthesis and materials science, and chemists quickly caught on to its versatility. Researchers noticed how its reactive epoxide group could build complex molecules or link polymers with remarkable efficiency. It wasn’t a household name, but in labs and manufacturing lines, it found a dedicated following.
Understanding the Basics: What Is It?
2,3-Epoxypropyl Phenyl Ether sounds intimidating, yet its structure offers clues to its appeal. The molecule combines a phenyl group—a familiar player from benzene chemistry—linked through an ether bridge to a reactive epoxypropyl group. That small but mighty epoxide ring means trouble for sluggish molecules that need a boost: it’s a source of tension and activity. Anyone who’s handled such compounds quickly learns the importance of the oxygen atom and strained ring, which transform an inert group into a chemical wildcard. Most often, you’ll find this ether as a clear to slightly yellowish liquid, with a sharp, sometimes almost sweet odor. It doesn’t float atop water, so spills need careful cleaning to keep it contained.
Delving into Properties: Physical and Chemical Nature
Grab a datasheet and 2,3-Epoxypropyl Phenyl Ether shows up with a boiling point just above typical room temperature solvents. That low volatility demands good ventilation when using it at scale. Density lands close to water, making separation by gravity more challenging during processing or waste treatment. Chemically, the most eye-catching feature is the three-membered epoxide ring, which sits highly strained and ready for reaction. A small change in pH or the presence of a catalyst opens that ring wide, giving access to new chemical spaces. You often see this property exploited in synthetic chemistry when building networks of polymers or sticking new functional groups onto aromatic frameworks. Though stable enough for shipping, it still requires an eye for detail in handling: exposure to strong acids or bases shortens shelf life fast.
The Details: Technical Specs and Labeling Demands
Labels on bottles don’t just give a CAS number—they also spell out potential hazards, storage conditions, and purity grades. In my experience, labs working with 2,3-Epoxypropyl Phenyl Ether demand high purity to avoid knotted reaction products or unwanted color changes. Storage in tightly sealed containers, out of direct sunlight, preserves usability over months. Labels bear hazard pictograms and clear warnings: this stuff isn’t for unsupervised hands. Regulatory compliance involves both national chemical standards and international shipping rules. Safety data always stays close by, reminding even the seasoned chemists that a single careless contact leads to skin or eye irritation pretty fast.
Getting There: How It’s Made
Synthesis of 2,3-Epoxypropyl Phenyl Ether requires a careful balance of reagents and temperature. Most industrial routes begin with reaction of phenol with epichlorohydrin under basic conditions. Lab techs combine the ingredients slowly, keeping the mix cool, and separating the crude product with simple extraction techniques. Final purification often runs through vacuum distillation, stripping out unwanted byproducts and concentrating the active ether. No magic here: just solid technique and steady attention to detail. Process tweaks—swapping bases or solvents—let chemists dial in yields or control byproduct formation, but the fundamental process changes little because it works so reliably.
Reactivity and Modifications: Transforming the Molecule
That epoxide ring opens doors—literally and figuratively—because it welcomes attack from nucleophiles like amines, alcohols, or acids. Skilled synthetic chemists use this toolkit to graft new groups onto the backbone, forming anything from pharmaceuticals to tough resin networks. In polymer science, the ring-opening step tethers individual molecules together, building flexible chains or rigid networks. Reactive blending with other monomers, especially those with multiple active hydrogens, produces thermoset plastics or specialty coatings known for chemical resistance and mechanical stability. The epoxide group’s reactivity also means the product rarely stays in its original form unless that’s the design goal. Its ability to bridge disparate chemical spaces gives designers and builders—both in chemistry and industry—the flexibility to solve real-world problems.
What’s in a Name?
Don’t let a mouthful of syllables hide a familiar face: 2,3-Epoxypropyl Phenyl Ether carries a variety of trade and scientific names. "Phenyl glycidyl ether" pops up most often on labels, while chemists sometimes shorten it to PGE or reference it by its IUPAC title in papers. Synonyms allow quick matching to regulatory lists and safety data, important when tracking hazards or supply chains. Industry watches for name overlap—different sources or suppliers sometimes brand the same molecule under different terms, which can confuse those looking for compatibility data or specific technical attributes. The name game underscores a broader point in chemistry: clear communication keeps projects on track and accidents at bay.
Working Safely: Standards in Practice
Anyone who’s handled 2,3-Epoxypropyl Phenyl Ether up close knows why safety doesn’t end at gloves and goggles. Vapors can irritate airways, and spills on skin shouldn’t linger. Experienced operators use chemical fume hoods, ventilation, and splash-resistant clothing to cut the risk of allergic or toxic reactions. Eye wash stations and spill kits sit close at hand. Regulatory agencies assign hazard codes and exposure limits, and companies keep regular records of training and usage to satisfy audits and satisfy insurance. Those precautions aren’t just for show: documented cases include respiratory sensitization after repeated or intense exposure, reminding everyone in the lab to treat this compound with respect every time.
Where It Works: Real-World Uses
Few chemicals touch as many sectors as 2,3-Epoxypropyl Phenyl Ether. In coatings and adhesives, it acts as both hardener and performance boost. Materials scientists rely on it as a building block for specialty thermosets, including circuit boards and aerospace composites, where its ability to form durable, heat-resistant networks pushes performance higher. It finds utility in plastic modifiers, stabilizers for resins, and sometimes as a chemical intermediate in making pharmaceuticals or agricultural agents. Testing labs use it in analytical chemistry for derivatization, a trick that enables better detection of certain molecules in complex samples. Its presence travels quietly through the production lines of paints, plastics, sealants, and specialty items ranging from dental devices to anti-corrosive primers. Wherever resilience and chemical compatibility matter, its chemistry finds friends and followers.
Pushing the Limits: Research and Future Prospects
Research into 2,3-Epoxypropyl Phenyl Ether stretches far beyond classic industrial uses. Scientists look for ways to tweak its structure for new functions: introducing subtle changes to the phenyl ring, for example, fine-tunes its compatibility with modern polymers or imparts new optical properties for electronics. The drive for greener chemistry has sparked efforts to replace hazardous raw materials or streamline synthesis with less waste, and this molecule often features as a test case for safety and sustainability studies. Advances in polymer chemistry may open the way for high-performance, recyclable plastics that rely on its versatile chemistry. Toxicity research also shapes R&D, with teams tracking low-level exposure impacts and bioaccumulation risks. As environmental rules tighten, industry must meet new standards for purity and emissions, which pushes everyone involved to rethink traditional processing and waste handling.
What We Know—and What We’re Still Learning: Toxicity Research
Toxicologists and occupational health experts have explored this compound’s effects for decades, piecing together its safety profile one study at a time. Skin and eye irritation rank at the top of documented issues, though rare cases link chronic exposure to asthma or long-term respiratory trouble. Absorption through skin or inhalation sparks most alarms, especially in high-concentration or poorly ventilated work areas. The compound’s metabolic breakdown hasn’t revealed lasting damage at low doses, but new research continues to monitor long-term risks. Guidelines for permissible exposure keep shifting in step with fresh discoveries, keeping health and safety professionals on their toes. Diagnostic labs have developed better detection methods for workplace air and biological samples, tightening control and early intervention triggers. The growing focus on chemical allergies and late-onset effects gives health surveillance programs even greater importance.
Looking Ahead: The Next Chapter
The future of 2,3-Epoxypropyl Phenyl Ether rests on more than tradition and solid chemistry. Industrial users, researchers, and regulators all look for materials with lower environmental footprints and higher performance. As the push for sustainability grows, innovators will seek alternatives that match its chemical smarts without compromising health or environmental safety. Digital design tools and automation in synthesis promise cleaner manufacturing, cutting waste and boosting yields. The rise of advanced functional materials, like smart coatings and recyclable composites, invites continued research into both the molecule and greener cousins inspired by its structure. The ongoing dialogue between innovation and caution keeps the story moving forward, as users everywhere demand chemicals that serve a purpose without mortgaging tomorrow’s environment.
What is 2,3-Epoxypropyl Phenyl Ether used for?
The Role of 2,3-Epoxypropyl Phenyl Ether in Industry
2,3-Epoxypropyl phenyl ether might sound strange if you haven’t worked in a lab or in manufacturing. For people in the plastics, coatings, and adhesive businesses, though, this chemical shows up as a useful specialty material. I saw its effects firsthand during a summer I spent in a small plant outside Tulsa. Epoxy resins needed to meet toughness standards, so every batch included compounds like this ether to tune the structure for strength and stability.
Building Better Plastics and Resins
On the shop floor, plastic must deliver. It doesn’t get a free pass if it’s brittle after you shape it or if it crumbles under stress. 2,3-Epoxypropyl phenyl ether, with a structure featuring both an aromatic group (the phenyl) and an epoxide ring, gives formulators a tool to solve these practical demands. Drop a small percentage into an epoxy resin blend, watch as the final cure resists temperature shifts, and notice how it stands up against wear. It doesn’t just serve as a filler—it fine-tunes performance. Studies point out that resins with this ether show stronger chemical resistance, which makes a difference in settings like construction, automotive, and even electronics.
Adhesives That Stay Put
In adhesives, especially epoxies meant for metal or circuit boards, the recipe matters. My coworker once joked that half our jobs were troubleshooting glue. If your adhesive cracks too soon, moisture seeps through, and everything fails. Adding 2,3-epoxypropyl phenyl ether lets manufacturers achieve balance—flexible enough to handle vibration, sturdy enough to keep its grip during heat cycles. Since this compound reacts with curing agents, it helps form strong, cross-linked networks that resist peeling and chemical exposure. According to chemical suppliers and independent lab data, adhesives containing this ether outlast traditional formulas in damp or corrosive settings.
Protective Coatings for Harsh Environments
Industrial coatings often act as the thin barrier between metal and ruin. My uncle’s hardware store carried “marine-grade” paints that he swore by. The secret, in many cans, lies with additives like 2,3-epoxypropyl phenyl ether. This chemical gives the final coat improved grip on steel or aluminum, blocks water molecules, and stretches the time between expensive repaint jobs. For storage tanks on oilfields or shipping containers at ports, coatings built this way offer real savings. Some research even highlights its ability to block UV light and shield pipes from acids and other tough chemicals.
Safer Work with Smart Handling
While this chemical makes products stronger, it’s not something to take lightly. Epoxy compounds, if handled carelessly, can create health risks. I learned quickly to double up on gloves and keep the work bay ventilated. Responsible manufacturers train staff, use tight controls, and follow health agency guidelines with every batch. Modern data sheets spell out exposure limits and disposal rules, supporting safer workplaces. These investments bring better worker health and fewer long-term costs.
Innovation and Future Directions
Demand rises for better, longer-lasting materials, especially as industries chase performance with less waste. Chemists push hard to reduce hazardous byproducts and design eco-friendlier alternatives. Research now looks at plant-based starting materials, ways to recycle cured products, and milder curing agents.
In the end, 2,3-epoxypropyl phenyl ether anchors itself in real-world uses: stronger plastics, tougher adhesives, and coatings that don’t let down their users. Anyone who cares about the reliability of cars, bridges, or laptops stands to benefit when companies take care with their chemicals—and keep pushing for safer, smarter solutions.
What are the safety precautions when handling 2,3-Epoxypropyl Phenyl Ether?
Understanding the Risks Before Getting Started
2,3-Epoxypropyl phenyl ether crops up in labs and manufacturing because its reactive epoxy group opens doors for all sorts of chemistry. The perks come hand-in-hand with real risks. Chemicals packed with epoxide groups may cause skin burns, eye damage, and breathing trouble if mishandled. Knowing the dangers helps prevent the headaches that follow carelessness.
Personal Safety Means More Than Gloves
My days in the lab taught me one thing: Splatters happen, and they don’t wait for you to suit up. Before working with a substance like 2,3-epoxypropyl phenyl ether, wearing nitrile gloves, chemical splash goggles, and a lab coat forms the first line of defense. Toss in closed shoes to protect against drops. I’ve seen solvents blister skin through thin gloves. Thick, tested protection works better in the long run.
Sometimes, face shields come in handy if mixing or transferring large volumes. Respiratory protection earns a spot on the bench once the vapor levels tick up or spills go sideways, especially in cramped workspaces. Skin absorbs some epoxies quickly; keep barriers between chemical and body at all times.
Ventilation Turns the Tide
Chemical fumes linger. 2,3-Epoxypropyl phenyl ether isn’t perfume—its fumes irritate eyes and lungs. Good fume hoods, strong exhausts, or open windows make a real difference. I’d never pour or measure this in a closet-sized room. After feeling my nose and eyes burn just from a whiff of poorly vented chemicals, I respect fresh air.
Spill Readiness Saves Regret
Every bottle has a habit of tipping when you least expect it. Have absorbent pads ready near your workspace. Neutralizers need to match the chemical—never toss water on an epoxy spill without checking compatibility. Emergency eyewash stations and showers need to be primed and easy to reach. Once, a coworker panicked and scarred her arm after fumbling over the eyewash button; regular drills can save pain.
Labeling and Storage Make a Difference
Labels might feel boring, but the wrong bottle mix-up gets dangerous fast. Keep clear, permanent labels that spell out hazards. Tightly sealed containers go in cool, low-light cabinets, far from acids or peroxides. Back in college, someone left a container of reactive liquid on a sunny shelf—pressure built, the lid popped, and the cleanup took hours. Good storage prevents bad surprises.
Health Monitoring and Access to Data
Regular health checks for those working often with epoxides add a layer of protection. Knowing the signs of overexposure—red skin, sore throat, headaches—means help comes faster. Safety data sheets need to be within easy view, not buried in drawers.
Training and Team Communication Shape Results
The only thing worse than a surprise reaction is working next to someone who doesn’t know the hazards. Everyone in the lab or plant needs hands-on training with spill kits and emergency stops. During my time overseeing new interns, I found practice beats printed instructions; real-time drills boost memory.
Staying cautious with 2,3-epoxypropyl phenyl ether isn’t about paranoia. It revolves around good habits, solid teamwork, and knowing when to slow down. These steps protect health and keep the focus where it belongs—on discovery and results.
What is the chemical structure of 2,3-Epoxypropyl Phenyl Ether?
Chemical Structure and Down-to-Earth Practicality
Digging into the nuts and bolts of 2,3-Epoxypropyl Phenyl Ether reveals a molecule with real character. This compound pulls together a phenyl group and an epoxy ring through an oxygen bridge. Nothing too fancy – just a benzene ring (that phenyl group) with a dangling ether linkage, capped by a three-membered oxirane ring. In plain terms, 2,3-Epoxypropyl Phenyl Ether carries the formula C9H10O2. Imagine attaching a glycidyl group to a phenol: you’d land right here.
The chemical structure gives the molecule its punch. That little epoxy ring – three carbon atoms straining in a triangle – stores enough energy to power through all sorts of reactions. This “springiness” at the molecular level lets chemists lock this ether into new chains, cross-link it in resins, or graft it onto just about anything with a loose hydrogen atom. The phenyl half of the molecule keeps things reasonably stable, letting it play well with others, but not to the point of being boring in a reaction.
Epoxides in the Real World
In my work with resins, the practical importance of structure keeps surfacing. Plenty of manufacturers rely on epoxides like this one because that energetic ring forms bonds quickly under the right conditions. Once mixed with an amine hardener, 2,3-Epoxypropyl Phenyl Ether links up fast and strong.
And beyond adhesives and coatings, there’s a comfort in knowing exactly what you’re working with. The phenyl ether section resists many corrosive agents, so these compounds last in paints or composite materials. That’s why you’ll spot molecules built like this in everything from floor coatings to electronics encapsulation. Durability counts when protecting a circuit board or patching cracks in a bridge.
Concerns Around Health and Environment
No chemical comes risk-free, and my hands-on time with reactive epoxides left a mark. Skin sensitivity can flare up if you don't glove up. The oxirane ring isn’t too choosy about what it’ll react with, which includes some proteins in your skin. Long-term exposure potentially causes allergic reactions or respiratory issues. Many in the lab keep safety data sheets close, not collecting dust. In production, ventilation systems and personal protective gear matter as much as the chemical itself.
There’s also a conversation to have about safe disposal. Epoxides too often wind up as waste, and those energetic rings don’t just fade away. Breaking down these chemicals in wastewater costs time and money. So developers look to green chemistry to tame aggressive epoxides or create structures that degrade more gently. I know a team that switched to waterborne resins to avoid solvents, keeping volatile organic compounds off the shop floor—and out of the air.
Building Safer and Smarter Materials
Thinking forward, sustainable chemistry will likely steer the future of compounds like 2,3-Epoxypropyl Phenyl Ether. Scientists are working the problem from both ends: making the molecule less hazardous, and inventing catalysts that control exactly where and how the epoxy ring opens up. As advances keep rolling out, you can bet on more durable products with a lighter touch on health and the planet. Responsibility and chemical design shouldn’t pull in opposite directions.
How should 2,3-Epoxypropyl Phenyl Ether be stored?
Why Storage Conditions Shape Safety
Nobody forgets their first whiff of a strong chemical, that sting in the nose signaling danger. For folks handling 2,3-Epoxypropyl Phenyl Ether, safety routines around storage aren’t just a checkbox—they save skin, lungs, and sometimes lives. This compound, known in many labs for its usefulness and reactive nature, poses both health and environmental risks if left unchecked.
Over the years, visits to small shops and big manufacturers have taught me that safe storage begins the moment the drum or bottle arrives. The wrong temperature, careless placement, or a cracked seal can spell trouble. Regulations from OSHA and the European Chemicals Agency echo that real dangers grow fast with missed steps.
A Cool, Dry Place Isn’t Just a Saying
Chemicals like 2,3-Epoxypropyl Phenyl Ether don’t take kindly to heat or moisture. Rooms that keep to a steady, low temperature slow down the vaporization process. Hotter conditions let fumes collect, and sudden spills get worse. I’ve seen containers bulge in the summer heat: that’s pressure, and a leak waiting to happen.
Keeping out water and humidity matters for another reason. Moisture finds its way past the faintest crack or loose cap. Water can trigger reactions that make the ether more toxic or volatile. Dry storage blocks that trouble before it starts.
Tight Seals and Solid Shelving
One overlooked factor in storage accidents comes from old or damaged containers. I once saw an entire shelf cleared because one bottle wept from a worn-out lid. That kind of thing can mix with other stored solvents, creating a cocktail nobody asked for.
Shelving made from metal, with trays for catching spills, provides a basic safeguard. Storing containers at waist level or lower keeps them from crashing down. Government workplace safety guides stress regular checks on seals and labels. Painted reminders and checklist routines help, especially in busy workspaces where people grab and go.
Separation from Incompatibles
Mixing chemicals creates new chemicals, sometimes without warning. I recall a place where someone stored phenolic ethers with acids and learned the hard way about corrosive byproducts. Acid or oxidizer storage cabinets cut the risk of this. In college labs and big factories, rules call for physical separation—dedicated cabinets or rooms get the job done.
Even if local codes feel distant, the science stays the same. Keep ethers apart from strong bases, acids, and oxidizers. Modern safety data sheets back this up and provide mixing charts that guide layout decisions within a storeroom.
Frequent Monitoring—More Useful Than the Hardest Lock
Locks, alarms, and ventilation systems matter, but a checklist in the hands of a trained worker catches problems earlier. Scheduled inspections have helped our team spot sticky residue on caps or corroded drum rings before a mess spread. Good policies put responsibility in real hands, not just paperwork.
For anyone new to handling 2,3-Epoxypropyl Phenyl Ether, it pays off to start with a critical look at your space: temperature, airflow, labels, storage gear, and cleanup kits. Mistakes grow silent over time—the right setup and constant attention keep people and the planet safer.
What is the CAS number of 2,3-Epoxypropyl Phenyl Ether?
Getting Precise with Chemicals
Every chemical in the modern toolbox comes with an identity crisis. One day it’s got a long, tongue-twisting name; on another, it’s an obscure abbreviation. The CAS number solves that problem with hard numbers. For 2,3-Epoxypropyl phenyl ether, the number is 122-60-1.
That eight-digit CAS number makes life a whole lot easier for folks in the lab or on the plant floor. Anyone who’s cataloged raw materials or kept a chemical inventory has run face-first into messy synonyms and ambiguous names. A CAS number keeps everyone speaking the same language. It cuts through confusion—no matter if someone calls it glycidyl phenyl ether or some other alias, seeing 122-60-1 on a drum lets you know exactly what’s inside.
The Human Impact
Years of working with specialty chemicals taught me that details matter more than most people realize. A lot of mishaps start not with bad intentions but with small mix-ups—a misplaced label or a typo on an order sheet. More than once, I’ve seen downtime and wasted batches because someone grabbed the wrong grade. One wrong molecule, and reaction systems throw tantrums or safety margins evaporate. CAS numbers help close those loopholes. It’s not some simple bureaucratic obsession; it protects workers and bottom lines.
Take epoxy chemistry as an example. 2,3-Epoxypropyl phenyl ether—CAS 122-60-1—works as a reactive diluent, making bulky epoxies easier to pour or mix. If another similar-sounding chemical sneaks in, the formula can change: weaker bonds, unpredictable cure times, safety risks rising. Standardizing on CAS numbers helps limit those headaches.
Safety Beyond the Lab
Outside the lab, CAS numbers also matter for public health. Emergency responders rely on them during spill responses. EHS managers need to get hazard information right—if someone enters a regulatory report without a CAS number, they risk incomplete records or fines. The consequences aren’t just regulatory; my old colleague ended up with months of pump rebuilds after anonymous “epoxy additive” fouled up seals simply because its ID wasn’t crystal clear from the start.
Countries that track restricted or hazardous chemicals list regulated substances by CAS number. That precision streamlines export paperwork and helps avoid liabilities when products cross borders. This isn’t just red tape. In my experience, the right number means a shipment clears customs faster or a permit application avoids last-minute questions.
Better Practices Moving Forward
People trust labels. Companies owe it to their teams to move past generic names, especially for chemicals like 2,3-Epoxypropyl phenyl ether, which find their way into products from coatings to adhesives. Training staff on the value of CAS numbers and double-checking them on shipping documents can save money, time, and possibly lives.
Digitizing inventories and tying purchase orders to these numbers cuts down surprises. A good software system links every safety data sheet, order, and regulatory filing to the CAS number. In my shop, that reduced rework and tightened compliance. Investing a little effort upfront goes a long way to keeping operations clean and safe.
Facts prove it: 2,3-Epoxypropyl phenyl ether carries CAS 122-60-1. Building every chemical process around correct identification keeps risks low and everyone a bit safer at the end of the day.
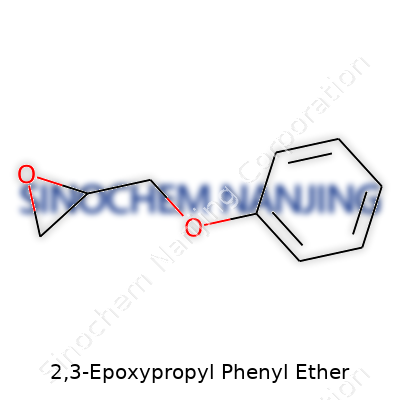
| Names | |
| Preferred IUPAC name | 2-(Phenoxymethyl)oxirane |
| Other names |
Glycidyl phenyl ether Phenyl glycidyl ether 1,2-Epoxy-3-phenoxypropane Phenyl oxirane 3-Phenoxy-1,2-epoxypropane PGE |
| Pronunciation | /ˈtuː θri ɪˈpɒksiˌproʊpɪl ˈfiːnɪl ˈiθər/ |
| Identifiers | |
| CAS Number | [122-60-1] |
| Beilstein Reference | 1208737 |
| ChEBI | CHEBI:38133 |
| ChEMBL | CHEMBL32128 |
| ChemSpider | 15499 |
| DrugBank | DB14019 |
| ECHA InfoCard | 03bca3f8-1b19-41c5-8fd1-4b52497eaf61 |
| EC Number | 204-998-1 |
| Gmelin Reference | 3931 |
| KEGG | C06506 |
| MeSH | D004946 |
| PubChem CID | 77191 |
| RTECS number | SL6475000 |
| UNII | 580C3J16QG |
| UN number | UN2319 |
| CompTox Dashboard (EPA) | DTXSID6020142 |
| Properties | |
| Chemical formula | C9H10O2 |
| Molar mass | C9H10O2: 150.17 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | aromatic |
| Density | 1.10 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.44 |
| Vapor pressure | 0.04 mmHg (25°C) |
| Acidity (pKa) | 15.59 |
| Basicity (pKb) | 13.86 |
| Magnetic susceptibility (χ) | -69.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.502 |
| Viscosity | 18.5 mPa·s (25 °C) |
| Dipole moment | 3.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -107.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4226.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H315, H319, H411 |
| Precautionary statements | P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2,3-Epoxypropyl Phenyl Ether NFPA 704: "2-2-1 |
| Flash point | > 110 °C |
| Autoignition temperature | 430°C |
| Lethal dose or concentration | LD50 oral rat 470 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1600 mg/kg |
| NIOSH | SN2985000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | IDLH: 250 ppm |
| Related compounds | |
| Related compounds |
Phenyl glycidyl ether Glycidol Epichlorohydrin Bisphenol A diglycidyl ether Allyl phenyl ether |

