2,3-Dimethylheptane: A Closer Look at a Lesser-Known Hydrocarbon
Historical Development
Chemistry classes often focus on familiar hydrocarbons like octane or benzene, but 2,3-Dimethylheptane sits quietly among its isomeric cousins, tracing its history through the story of organic chemistry's evolution. Early chemists isolated straight-chain alkanes from crude oil, refining separation techniques just as railroads and engines demanded fuel. Over the twentieth century, improved fractionation and analytical methods, such as gas chromatography and mass spectrometry, made it possible to pinpoint branched alkanes in petroleum streams. Scientific curiosity and practical demand led researchers to catalog compounds like 2,3-Dimethylheptane, not just for completeness, but because minor changes in structure—like adding a methyl group—can nudge volatility, combustibility, and other key traits. Surplus from cracking reactions and detailed mapping of petroleum products pushed even the less prominent alkanes, like this one, into the light, making their presence known in discussions about fuel quality and refining yield.
Product Overview
2,3-Dimethylheptane is an alkane hydrocarbon with nine carbons, dressed up by two methyl groups on the second and third carbons. This molecule rarely gains star status in textbooks or industry brochures, but its significance lies in its fingerprint in gasoline and as a reference standard in analytical chemistry. The molecule doesn’t draw attention for its unusual reactivity or unique creative uses, but its branching pattern tunes properties like boiling point and combustibility, which trickle down into performance discussions in fuel science. Think of it as a familiar sideline player—never the focus but impossible to ignore in the balance of refinery output.
Physical & Chemical Properties
With the formula C9H20, 2,3-Dimethylheptane falls solidly into the group of saturated hydrocarbons. It's colorless, generally liquid at room temperature, and possesses a characteristic faint odor—facts that won’t surprise anyone who’s splashed gasoline on their hands at a filling station. It boils at a slightly lower temperature than its straight-chained relative, nonane, thanks to its branching. Density drops a bit too, a result that lines up with the branching found in other alkanes. Chemically, this compound trades in the stability and predictability of alkanes—unreactive unless faced with hot flames or strong oxidizers, which suits its presence in fuels well. You won’t see it dissolving metals or picking up stray electrons the way aromatics or unsaturated hydrocarbons can.
Technical Specifications & Labeling
Most laboratories label 2,3-Dimethylheptane using the CAS number 3074-69-7, with C9H20 often listed for clarity. Purity typically lands above 98% for any meaningful research application, since contaminants skew test results. Chemists like seeing distillation range and density laid out plainly; they know even small deviations speak volumes about quality. Transportation rules assign it into the flammable liquid category, and labels warn plainly against heat, open flame, and poor ventilation. Industry trusts standard labeling from organizations like OSHA and GHS, and the Material Safety Data Sheets for alkanes uniformly stress fire risks and the need for solid ventilation.
Preparation Method
Small quantities of 2,3-Dimethylheptane can be coaxed out of petroleum distillates, especially in specialized research-grade settings. The real workhorse, though, comes from synthetic organic chemistry through alkylation. Starting with heptane, chemists use Friedel-Crafts reactions, carefully adding carbon fragments in the right locations, often guided by catalysts like aluminum chloride in hydrocarbon solvent. Controlling where methyl groups land is the main challenge—the chemistry rarely yields just one isomer without cleanup steps. Years spent leaning over a fume hood or prepping columns have taught me that the purity of starting material and reaction conditions make or break these syntheses, especially when demand focuses on a handful of grams, not kiloliters.
Chemical Reactions & Modifications
Alkanes like 2,3-Dimethylheptane behave with a certain stubbornness; their C–C and C–H bonds hang tight, resisting reaction except under forceful conditions. Combustion sits most prominently among its possible fates—exposure to oxygen and a spark, and the molecule breaks down into carbon dioxide and water, releasing heat. Halogenation, especially with chlorine under UV light, replaces hydrogens step-wise, creating useful intermediates for further chemistry. More elaborate techniques involve cracking, in which reactors split alkanes into smaller hydrocarbons, important in fuel technology. Isomerization plays a commercial role too, as refineries shuffle methyl groups to improve octane ratings for fuel blends. My own benchwork with alkanes always came back to their tendency to resist change unless pushed, a humbling lesson for anyone expecting fireworks.
Synonyms & Product Names
Most chemists refer to this compound as 2,3-Dimethylheptane, though occasionally you’ll see prefixes like “iso” sneak in, or numeric descriptors such as 2,3-DMH. The IUPAC convention dominates in academic circles to cut down on confusion, and commercial catalogs might list it under nonane isomers. Regardless of the label, the molecular structure remains the same, and the challenge is always ensuring what’s in the bottle matches the name stamped outside.
Safety & Operational Standards
Handling 2,3-Dimethylheptane doesn’t deviate far from established practices for other alkanes, though that predictability often lulls operators into complacency. Fire risk tops the list—rich vapor mixed with air and a stray spark, and anyone in the room learns respect in a hurry. Effective ventilation and spark-free controls get drilled into anyone working around volatile organics. Personal protective equipment rarely strays from the standard nitrile gloves, goggles, and flame-resistant coats, but consistent training remains the biggest barrier to avoiding accidents. If a spill happens, chemical-resistance and readiness matter more than heroic cleanup efforts. In my time supervising undergraduate labs, it was clear—repetitive reminders and visible signage always pay off more than fancy safety gadgets.
Application Area
This molecule falls into the vast pool of hydrocarbons churning through global refineries, shaping the final blend of gasoline and specialized solvents. Its branching structure nudges fuel properties enough to matter for refining operations, especially where high-octane demands push companies to optimize every variable. Research labs use 2,3-Dimethylheptane as a standard for chromatography—a reference point to calibrate instruments or untangle complex mixtures extracted from crude oil or environmental samples. Though not typically bottled or branded for direct commercial use, trace amounts contribute to the complexity and performance of transportation fuels, especially as chemists look for alternatives to aromatics and oxygenates.
Research & Development
Analytical chemists and fuel scientists dig through the properties of hydrocarbon isomers, aiming to predict how subtle structure changes affect performance. In this space, 2,3-Dimethylheptane helps sharpen fuel octane models, advancing beyond broad-strokes characterizations toward real predictive power. Instrument manufacturers lean on hydrocarbons like this to validate detection limits or retention times, using them as test cases for new tools and protocols. A key part of modern R&D involves refining catalysts to direct synthesis and isomerization, chasing greener routes and tighter selectivity. In the push for cleaner fuels, every component’s fate through an engine or atmosphere gets scrutinized, and even compounds in the chemical “background” become interesting data points.
Toxicity Research
Decades of toxicology research on alkanes show a consistent pattern: high molecular-weight, saturated hydrocarbons tend to cause limited acute toxicity, but vapor inhalation and skin contact lead to irritation or central nervous system effects if controls break down. Chronic exposure, though less studied for minor isomers like 2,3-Dimethylheptane, generally leads investigators to focus on respiratory and dermal routes. Animal studies with related branched alkanes point to a low risk of bioaccumulation and moderate biological impact, but gaps remain—especially with long-term or environmental exposure. Regulatory agencies lean on analogs to fill data gaps, a common approach in chemical risk assessment, but constant vigilance keeps inching science forward, highlighting the need for direct testing as new applications emerge.
Future Prospects
As fuel science drives toward efficiency and lower emissions, the tuning of hydrocarbon blends gains new urgency. Structural isomers like 2,3-Dimethylheptane step into the spotlight, not for dramatic properties but for their impact hidden in baseline performance metrics. Researchers digging into catalytic isomerization and predictive modeling look further afield for the next leap in synthetic routes, including routes incorporating renewable feedstocks or biocatalysts. Environmental scrutiny on volatile organics and the move away from hazardous oxygenates mean each component, including the minor players, ends up under the microscope. Continued advances in separation technology and environmental monitoring ensure this molecule, though rarely the hero, will remain part of the evolving conversation guiding energy, safety, and sustainability choices. Experience has shown that ignoring the “background” rarely pays, and knowledge of lesser-known compounds often sets experts apart from generalists in the field.
What is the chemical formula of 2,3-Dimethylheptane?
A Close Look at 2,3-Dimethylheptane
Everyday chemistry shapes things we take for granted, from the smell of gasoline to the plastics lining our gear. 2,3-Dimethylheptane doesn’t just exist as an abstract scientific curiosity; it shows up wherever hydrocarbons play a role. This molecule is part of the alkane family, which fills gas tanks everywhere. So, understanding the chemical formula of a molecule like this isn’t just about memorizing symbols but about understanding how atoms link up in real life. The chemical formula of 2,3-Dimethylheptane is C9H20.
Getting from Structure to Formula
Textbooks say every name in organic chemistry counts as a full set of instructions. Break down the name “2,3-dimethylheptane” and it starts to take shape in the imagination. The backbone, “heptane,” hints at a chain of seven carbon atoms. The “dimethyl” part means two extra methyl (CH3) groups branch off. These aren’t just tacked on at random; position numbers matter. Here, one methyl sticks off carbon number two, another off carbon three.
I remember learning how much difference just one branch can make. For example, n-heptane (plain heptane) burns differently from its dimethyl cousin because those branches change the way molecules bump into each other. The formula C9H20 tells us there are nine carbon atoms and twenty hydrogen atoms. It matches the structure: seven in the main chain plus two from the methyl groups. Hydrogen atoms fill up every spare bonding spot, making the molecule stable.
Why Accurate Formulas Matter
Anyone who’s accidentally used the wrong chemical at work knows getting the formula right makes a difference. Mixing up two hydrocarbons that sound similar can have real consequences. For instance, research into fuel performance depends on a deep understanding of molecular structure. 2,3-Dimethylheptane burns with a particular octane rating, changing how engines perform. Petroleum chemists rely on exact formulas to refine fuels or create synthetic lubricants that protect engines under heat and pressure.
Even outside the lab, these fine distinctions ripple through the supply chain. Shipping regulations use formulas to classify materials for safety. Industries map out recycling processes based on hydrocarbon categories. Getting the formula wrong could mean violating environmental standards and increasing health risks. Misinformation about a molecule’s makeup can lead to costly or dangerous errors.
Solutions for Avoiding Confusion
So many names and codes crowd the shelves of chemical catalogs. Clarity matters—for students, workers, and regulators. A simple solution: label containers clearly with both the structure and the full chemical formula. Standardized digital tools help catch errors before they reach the lab or factory floor. Companies can set up quick-training sessions to teach staff how organic names link to formulas. More collaboration across industries and academic circles also helps share best practices and avoid mix-ups.
In my work, asking a colleague to double-check before moving a drum of hydrocarbons paid off more than once. Instead of focusing just on memorization, I push for a hands-on approach—drawing out molecules, following each branch, and matching names to formulas. It’s a habit that builds lasting understanding and keeps mistakes in check, no matter what hydrocarbon crosses your path.
What are the main uses of 2,3-Dimethylheptane?
Where You’ll Find 2,3-Dimethylheptane in Use
2,3-Dimethylheptane doesn’t turn many heads on its own. At its core, it’s one of the many branched alkanes that show up in crude oil and refined fuels. This molecule doesn’t fuel your car directly — not in isolation, anyway — but it helps shape the mix you pour into your tank. Petrol and diesel fuels don’t flow from the ground perfectly tailored for engines, and a lot of work goes into tweaking octane ratings and combustion characteristics. That’s where molecules like 2,3-Dimethylheptane fit in.
The most direct use falls within fuel chemistry. Engineers and chemists rely on it for reference when fine-tuning how fuels burn in real engines. Its place as a model compound means it pops up in octane rating calculations, especially in research settings. These ratings decide if a car knocks and pings, or runs smooth at high combustion pressures. Blending scientists use it in mixtures that mimic real-life gasoline, so they can push engines harder without sacrificing reliability. In these labs, accuracy matters – wrong ratios mean wasted money and less efficient engines.
Building a Better Understanding of Fuels
Fuels don't just “get made”; they’re engineered. Every refinery is a giant chemistry set working to turn raw oil into targeted products. 2,3-Dimethylheptane serves as a reference point for how branched hydrocarbons behave compared to straighter chains. Researchers load up gas chromatographs and spectrometers with it to map out what’s in a sample and what tweaks will yield the cleanest burn or the best energy content.
Refinery operators might adjust processes based on how test batches rich in 2,3-Dimethylheptane react under different conditions. Oil companies also need to know how each hydrocarbon affects emissions, especially under tightening environmental rules. Cleaner, more efficient engines aren’t possible without knowing what every molecule will do once ignited. Every bit of data helps in creating fuels with fewer particulates and less carbon monoxide.
Studying Environmental and Health Impacts
Beyond fuel, 2,3-Dimethylheptane shows up in research on air quality and public health. Since small hydrocarbons turn into volatile organic compounds (VOCs), cities track compounds like this to measure air pollution. Scientists might measure it after car exhaust tests or while analyzing chemical spills. This links directly to how policies around urban pollution come about—numbers drive regulations, and accurate data can support stricter air quality standards.
Regulatory bodies like the US EPA keep a tight eye on VOCs due to their role in forming ozone and smog. Labs turn to 2,3-Dimethylheptane when calibrating machines to see if pollution abatement efforts are actually working. Lower readings mean cleaner air for everyone walking city streets or living near industrial sites.
Opportunities for Cleaner Industry
There’s a growing push for greener chemistry in refining and manufacturing. Teaching the next generation of chemical engineers often starts with basic hydrocarbons, including 2,3-Dimethylheptane. Through education and hands-on work, students run experiments that simulate what real refineries do and propose alternative pathways with less pollution. Cleaner gasoline and understanding every ingredient’s effect become possible once the science is in hand.
In practice, I’ve seen how accurate fuel characterization saves energy and cuts costs for large transport fleets. Knowing the role of minor components like 2,3-Dimethylheptane shapes not only what goes into a fuel truck, but how engines are designed for tomorrow. Embracing thorough research on basic chemicals pays off through better fuel mixtures, cleaner engines, and improved oversight of emissions.
Is 2,3-Dimethylheptane hazardous or toxic?
Understanding 2,3-Dimethylheptane in Everyday Context
If you’ve spent time around gasoline or worked in labs, chances are you’ve come across hydrocarbons like 2,3-dimethylheptane. This clear liquid belongs to the family of alkanes and often pops up during conversations about fuel components. Most people wouldn’t spot it in daily life, though it makes its way into the fuels that power our cars and trucks.
What Makes a Chemical Hazardous?
People sometimes hear the word “chemical” and brace for bad news. It’s smart to check a substance’s safety profile. With 2,3-dimethylheptane, there are a couple of ways to approach the topic. I always look at how likely someone is to touch, breathe, or swallow the stuff outside a controlled setting. The U.S. Occupational Safety and Health Administration (OSHA) and the National Institute for Occupational Safety and Health (NIOSH) do not specifically list 2,3-dimethylheptane, but guidance for similar alkanes gives us some answers.
Physical Risks Before Chemical Toxicity
The most immediate risk from 2,3-dimethylheptane comes from its flammability. I still remember the first time I handled a container of hydrocarbon in a lab—there was no mistaking the smell or the safety protocols. This compound catches fire quickly, which matters most in places storing large quantities. Industry storage uses thick signage and strict safety standards for a reason. You don’t want vapors accumulating in a closed room. Inhaling the fumes, especially in a poorly ventilated space, can make someone dizzy or give them a headache, just like breathing gasoline vapors does.
Is It Toxic to People?
Talking about toxicity gets a little technical, but there’s value in sharing what long-term field experience reveals. Regular exposure to most alkanes, including 2,3-dimethylheptane, tends to irritate eyes, nose, and skin. Splash enough of it on your hands and you’ll notice your skin drying out fast. Some studies on related alkanes show repeated skin contact triggers dermatitis. Swallowing it has serious consequences like aspiration into the lungs, which can lead to chemical pneumonia—never something to risk. I’ve seen accidental exposures in industrial settings handled by thorough rinsing and medical checks.
Environmental Fate and Hazards
Concerns stretch past human health. Dropping or spilling this chemical on the ground won’t make it stick around for years, but it does pose a threat to aquatic life if it slides into waterways. Like other light hydrocarbons, it floats and spreads, creating a film on water that blocks out oxygen. Fish, insects, plants—they all struggle when their habitat gets coated with oily residue. That’s why spill response teams work fast to contain and clean up leaks.
How to Reduce Risks
Having the right safety systems in place matters most. Good ventilation, regular monitoring for leaks, and safety data sheet training all make a big difference. Rescue gear and first aid supplies belong close at hand in areas managing such chemicals. When in doubt, personal protective equipment like gloves, goggles, and splash aprons offer an added layer I always reach for. In my experience, keeping a tidy workspace and knowing what to do in an emergency beats any fancy technology. There are no shortcuts, just practical habits and an honest respect for the stuff you’re working with.
The Bottom Line from Experience
Day-to-day contact with 2,3-dimethylheptane’s pretty rare if you’re not working with fuels or chemicals. For folks in those fields, the biggest worries revolve around fire hazards, short-term symptoms from inhaling vapors, and skin irritation. It doesn’t build up in people or animals the way heavy metals do, but it’s never wise to treat any flammable hydrocarbon casually. If you respect the risks and follow safe handling practices, it shouldn’t cause trouble. There’s peace of mind in routine safety checks, and a reminder that working thoughtfully puts control back in your hands.
What is the boiling point of 2,3-Dimethylheptane?
Understanding What the Boiling Point Really Tells Us
People often treat boiling points like tiny factoids you need for a quiz or to fill an empty spot in a textbook. For 2,3-dimethylheptane, it’s about 146°C (294.8°F). But it’s worth looking closer. Boiling points shape how chemicals get used, transported, and stored. If you deal with fuels or solvents, the boiling point gives you a quick sense of volatility, risk, and the practical side of handling a substance.
Those of us who've worked with chemicals know boiling points are more than lab trivia. I spent years in a shop where the wrong assumption about volatility led to ruined products or, worse, safety scares. Even a small change in a molecule, like methyl groups in 2,3-dimethylheptane instead of straight heptane, nudges the boiling point lower because branching stops molecules from stacking tightly. That little detail ends up making all the difference in a refinery or chemical plant.
Why Care About the Details of a Hydrocarbon?
It’s easy to lump hydrocarbons together and assume you’re only splitting hairs. Yet a refined fuel blend might change performance or environmental impact based on subtle differences. 2,3-dimethylheptane isn’t just another isomer; its boiling point pins its place in petroleum distillation columns and separates it out from other fractions. Engineers count on this property to make sure fuel grades stay within spec, especially with regulations tightening every year. That’s not paperwork—if a fuel doesn’t evaporate as expected, engines might misbehave or pumps could run dry.
Consulting published resources, you can confirm that value: handbooks like the CRC Handbook of Chemistry and Physics list the boiling point of 2,3-dimethylheptane around 146°C. Research articles back up the number, checked using distillation and precise temperature monitoring. Not all sources match, which reminds us to double-check figures instead of copying from one place to another. Consistent data keeps industries running smoothly.
Boiling Point Knowledge in Everyday Safety and Practice
Forget chemistry class for a moment. Think about anyone loading, storing, or mixing chemicals in real life. The boiling point acts as a line between safe liquid and unpredictable vapor. I’ve seen folks underestimate this: storage rooms got too warm, vapors built up, alarms rang, and cleanup wasn’t pretty. If more people respected the boiling points printed on safety sheets, fewer folks would wind up breathing in something they shouldn’t or risking a flash fire.
Following the numbers doesn't mean memorizing trivia. Keeping the right data in mind—especially for less famous compounds like 2,3-dimethylheptane—keeps everyday operations smooth and safe. Companies can work with suppliers to make sure labels aren’t wrong, and staff learns that boiling points aren’t just for chemists. Sharing clear, accurate information through training and updated safety practices protects jobs, facilities, and the environment.
Seeking Improvement in Chemical Handling
Industries that deal with hydrocarbons can take simple steps: use reliable sources, invest in temperature controls, and make sure every hand in the process knows the basic properties, not just the headline hazards. Regulators have a part to play by standardizing how data gets reported and audited, so the value for something like 2,3-dimethylheptane means the same thing in every lab or tank farm. In my experience, enough attention on details like boiling points stops problems before they start. That’s a habit worth passing down.
How should 2,3-Dimethylheptane be stored and handled?
Recognizing the Real-World Risks
2,3-Dimethylheptane shows up in labs, refineries, and chemical storage yards because it gets used in fuel research and as a reference compound. Beneath the technical jargon, this is a flammable liquid—clear, volatile, and capable of creating trouble if treated lightly. With any hydrocarbon like this, fire risk lingers over every decision. Open flames, heat sources, or careless smoking turn spills into blazes in seconds.
Building Safe Storage from the Ground Up
People sometimes tuck away chemicals wherever there’s free space. Bad idea with compounds like 2,3-Dimethylheptane. Flammables demand rooms with tough construction—no open woodwork or aging extension cords. Use cabinets or dedicated lockers built for hazardous liquids, preferably with self-closing doors and solid secondary containment trays. Metal safety cans work much better than repurposed plastic jugs. Large drums belong on bunded pallets or inside properly vented areas because vapor builds up fast in enclosed spots.
Temperature matters just as much as the container. Keep storage away from boilers, direct sunlight, and heat vents. Vapors will creep into the air if the place warms up. Good practice involves steady room temp, low humidity, and away from oxidizers—don’t stack paint thinners or acids shoulder to shoulder with hydrocarbons.
Personal Protection and Practical Know-How
A splash or big whiff of 2,3-Dimethylheptane won’t feel like a typical trip to the gas pump. In my experience, you never stop at just goggles and gloves—nitrile beats latex, and ignited vapor won’t show mercy for a rolled-up shirt sleeve. Lab coats without open cuffs and closed-toe shoes save skin from spills and flying shrapnel if a fire starts. Respiratory protection isn’t just a formality; disposable masks won’t stop fumes. Use proper cartridge respirators in poorly ventilated rooms, because this chemical’s vapor can build up quickly.
Ventilation and Fire Control: The Non-Negotiables
Fresh air makes the difference between a near-miss and a disaster. If you can smell the chemical, it’s in the air—vent hoods, extraction fans, and windows aren’t just there for comfort, they matter for survival. Storage areas with strong airflow reduce buildup and keep vapor concentrations below flammable limits, which protects both staff and visitors. Dusty old fire extinguishers give a false sense of security. Use powder or CO₂ models, not water—hydrocarbon fires only grow with water’s help. I make a habit of checking extinguisher tags every season, because they won’t work if forgotten and expired.
Packing, Labeling, and Moving without Regret
Old stories come to mind about messy, unlabeled bottles creating chaos during emergencies. Clear, tough labels displaying contents, date received, and relevant hazards prevent confusion and save time for everyone. When it’s time to move the liquid—even down the hall—grab a chemical-rated handcart, carry small loads, and announce your route in busy labs. Spills from overfilled or unsealed cans can drill the lesson home the hard way.
Regular training keeps memory sharp. Annual safety drills and refreshers run through practical responses: eye wash stations, fire alarm pulls, and emergency shutoffs. No one remembers long policy binders in a panic, but hands-on walk-throughs stick for years.
Responsible Disposal
Pouring leftover solvents down the drain isn’t an option. Hydrocarbons poison waterways and trigger environmental violations. Collect spent or dirty 2,3-Dimethylheptane in labeled waste cans and send them to licensed handlers—one accident in the waste stream ends careers and hurts communities.
Working with this chemical teaches care, respect, and a kind of vigilance that comes with handling things that work for or against you. Safe habits, not just checklists, let people go home healthy after long days with sharp, risky liquids.
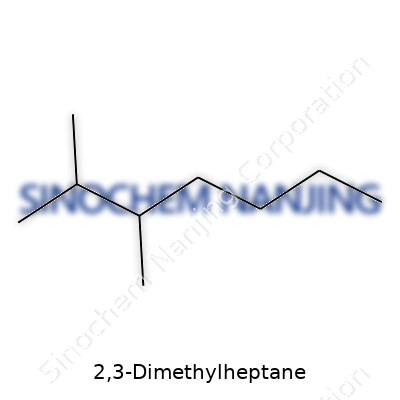
| Names | |
| Preferred IUPAC name | 2,3-Dimethylheptane |
| Other names |
Ethylisohexane Isooctane n-Octane, 2,3-dimethyl- |
| Pronunciation | /ˌtuː θri daɪˈmɛθɪlˈhɛpteɪn/ |
| Identifiers | |
| CAS Number | 584-95-2 |
| Beilstein Reference | 1701828 |
| ChEBI | CHEBI:89341 |
| ChEMBL | CHEMBL17262 |
| ChemSpider | 122152 |
| DrugBank | DB14274 |
| ECHA InfoCard | ECHA InfoCard: 100.117.436 |
| EC Number | 203-699-2 |
| Gmelin Reference | 55552 |
| KEGG | C08352 |
| MeSH | D004563 |
| PubChem CID | 11508 |
| RTECS number | MI7700000 |
| UNII | K3CB329P0K |
| UN number | UN2331 |
| CompTox Dashboard (EPA) | DJ32UWF3J7 |
| Properties | |
| Chemical formula | C9H20 |
| Molar mass | 128.26 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.718 g/mL at 25 °C |
| Solubility in water | Insoluble in water |
| log P | 4.4 |
| Vapor pressure | 0.511 mmHg (at 25 °C) |
| Acidity (pKa) | pKa < 0 |
| Magnetic susceptibility (χ) | -75.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.399 |
| Viscosity | 0.675 mPa·s (at 20 °C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 398.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -242.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4810.1 kJ/mol |
| Pharmacology | |
| ATC code | '2,3-Dimethylheptane' does not have an ATC code. |
| Hazards | |
| GHS labelling | 2,3-Dimethylheptane is not classified as a hazardous substance or mixture according to the Globally Harmonized System (GHS). |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P304+P340, P312, P331, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 71 °C (closed cup) |
| Autoignition temperature | 210 °C (410 °F; 483 K) |
| Explosive limits | 0.9–6.0% |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| NIOSH | RN0162000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of 2,3-Dimethylheptane is: "NA ppm |
| IDLH (Immediate danger) | IDLH: 900 ppm |
| Related compounds | |
| Related compounds |
Nonane 2-Methylheptane 3-Methylheptane 2,2-Dimethylheptane 2,4-Dimethylheptane Isooctane Heptane |

