The Real Impact of 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate: Examining Roots, Realities, and Responsibilities
Historical Development: From Lab Curiosity to Field Application
Every molecule has a story, and 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate did not simply appear out of nowhere. Chemists and researchers, searching for effective solutions to mounting agricultural and industrial challenges, began synthesizing these sorts of carbamates decades ago, hoping to control pests more selectively and with less risk to non-target species. Higher crop yields and food security seemed within reach thanks to these advances in chemical technology. This compound, rooted in the broader family of carbamates that made headlines in the last century, became a staple in pest control strategies. Many industry veterans still recall the early skepticism: concerns over safety, soil longevity, and off-target toxicity. Today, the road from curiosity to relevance continues to provoke debate, especially as demands for sustainable practices grow louder from both the lab bench and the field.
Product Overview: Where Science Meets Practical Use
To those outside the sphere of chemical manufacturing, 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate often looks like just another technical name. In practice, this unique carbamate reflects a marriage of molecular innovation with day-to-day problems: pest resistance, unruly crop diseases, and the constant juggle between efficacy and environmental footprint. Not every molecule moves seamlessly into the hands of agriculturalists, but this one broke through, entering toolkits because of its targeted activity and reasonably predictable behavior. Here’s where honesty counts—every field pro I’ve spoken to weighs the promise of pest suppression against the hassle (and worry) of chemical spillover, weighing real risks and benefits through the lens of lived experience.
Physical and Chemical Properties: How Substance Shapes Application
Anyone who moves from bench-scale synthesis to field-scale application recognizes the gulf between theoretical knowledge and practical handling. 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate typically presents itself as a stable powder or crystalline solid, a trait not to be underestimated in busy operations where stability can mean everything for shelf life and transport. Its solubility profile impacts formulation: dissolve too quickly and you risk leaching, too slow and you frustrate the user hunting for fast results. The molecular structure drives how the compound interacts with living systems—a detail crop managers pay close attention to, particularly as they wrestle with time-sensitive missions like insect outbreak or disease flare-up. Too often, we overlook these quiet workhorses of chemical application: vapor pressure, hydrolysis potential, and pH-dependent stability always come up in field reports, especially in regions where sudden rainfall can turn a powder into runoff with little warning.
Technical Specifications and Labeling: The Devil in the Details
Guidance on use rarely wins beauty contests, but the right details shield growers, handlers, and the environment from needless risk. Labels run pages for a reason—every clause, pictogram, and cautionary statement reflects years of learning (and more than a few close calls). Specific instructions on dosage, dispersal methods, and personal protective gear stand as the first line of defense against misuse. Labeling continues to stir conversation in regulatory circles, too, as advocates push for plainer language, pictorial cues, and more comprehensive exposure data. My on-site observations echo this call; too many well-meaning professionals still find these documents dense and disjointed, urging industry and regulators to streamline without sacrificing substance.
Preparation Method: More Than Just a Lab Recipe
Synthesizing 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate doesn’t end with mixing a few reagents. Organic synthesis of this type usually starts from methylated benzofurans, progressing via carbamoylation using methyl isocyanate derivatives or equivalents. Each modification phase demands careful handling, as both precursors and byproducts can pose threats if not contained. Early process strategies often generated significant waste; more modern refinements aim to tighten yields, lower residual contaminants, and reclaim solvents for reuse. My interactions with process chemists spark constant reminders: efficiency improvements are not only about production cost—they echo downstream, affecting waste management and exposure risk for site workers and local communities alike.
Chemical Reactions and Modifications: Unlocking Value and Mitigating Risk
The structure of 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate opens the door to several functionalization strategies, offering a base for derivative creation tailored to niche markets. Hydrolysis remains a key vulnerability and must be closely watched, especially as real-world conditions rarely match the tidy confines of laboratory flasks. Certain substitutions can increase resistance to degradation, but every tweak ripples through the balance of toxicity and persistence. Some research circles favor such modifications to answer emerging pest challenges or regulatory crackdowns, but these explorations demand a deep dive into downstream ecological effects before any commercial rollout.
Synonyms and Product Names: Navigating a Sea of Labels
Chemicals rarely travel under a single flag. In supply catalogs, one might find 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate labeled under variant trade or systematic descriptors, typically shaped by patent filings or manufacturer branding. This fragmentation sows confusion: professionals must cross-reference databases, peer-reviewed studies, and local regulations to ensure apples-to-apples comparisons in safety evaluations, field trials, or residue studies. In my own auditing work, this patchwork of names and codes complicates efforts to harmonize data, particularly across borders where language and local practice diverge sharply.
Safety and Operational Standards: No Room for Shortcuts
Shortcuts in handling carbamates never pay off—this lesson gets hammered home everywhere from training classrooms to accident investigation reports. Contact, inhalation, or ingestion of this compound calls for robust response plans and up-to-date safety protocols. I’ve seen strong programs reduce incident rates dramatically, and these results usually trace back to leadership commitment, hands-on refresher training, and real-time risk communication on the factory floor. Chronic exposure risk, even below acute toxicity thresholds, underscores the vital role of routine medical screening for those regularly in contact. Operators insist on clear, accessible standards; outdated documentation or legacy infrastructure often trigger the sort of mishaps that media latch onto, eroding trust and stirring public backlash.
Application Area: Field Reality Over Textbook Ideal
2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate remains entrenched in agricultural pest management, drawing on a legacy of reliable action against target pests without the blanket impact of earlier organophosphates. Specialty crops gravitate toward such targeted solutions, valuing reduced residue and season-long control of resistant insect populations. Off-label uses inevitably crop up, sometimes born from necessity rather than ideal stewardship, particularly in regions squeezed by resource constraints. This on-the-ground reality reminds policy designers to temper expectations—uptake hinges on relevance to local practice, not abstract regulatory wish lists.
Research and Development: Real Progress Demands Rolling Up Sleeves
Innovation in the carbamate field faces a tough crowd: less tolerance for toxicity, stricter environmental endpoints, and more robust detection requirements. Research teams now aim for “greener” synthesis routes, drawing on renewable feedstocks or milder reaction conditions, but progress comes slower than press releases suggest. In my own experience, bridging the gap between promising lab results and scalable, cost-effective production often hits a plateau, mired in complexity or unforeseen side effects. Researchers continue mapping out metabolic pathways, environmental fate, and synergistic blends, hoping to anticipate problems before they hit headlines or disrupt markets.
Toxicity Research: Vigilance Above All
Carbamate toxicity tracking never fully ends. Early evidence established a profile of reversible cholinesterase inhibition, flagging acute exposure risks for humans and beneficial insects alike. Ongoing studies aim to sharpen our understanding of chronic, low-dose impacts—topics that rarely fit into sound bites: sublethal effects on pollinator navigation, accumulation in non-target vertebrates, or developmental quirks in aquatic life. Field practitioners wrestle daily with these realities, shifting application timing or method to minimize overlap with sensitive species. Regulators, to their credit, have toughened residue limits and environmental monitoring, responding to a populace less tolerant of pollution and ecological unknowns. Every year brings new data, but consensus remains elusive, especially as underlying mechanisms of toxicity reveal greater complexity.
Future Prospects: Responsibility Shapes the Road Ahead
As the public asks sharper questions about what enters their food, soil, and water, the future of 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate rides on more than technical performance. Thoughtful stewardship means keeping sight of risk, not just reward: rotating actives, limiting overreliance, and doubling down on education for users at every step of the supply chain. The chemical industry faces rising pressure to replace persistent synthetic agents with biodegradable or “soft” alternatives—work that demands not just new chemistry but partnerships across science, farming, and policy. If there’s a lesson to draw from past decades, it’s that sustainable solutions never arrive by accident. They emerge where transparent research, rigorous safety culture, and honest dialogue come standard, not as an afterthought. In the years ahead, compounds like this one may either adapt and thrive under stricter scrutiny or fade away in favor of approaches that build more trust among growers and the broader community relying on their harvests.
What is 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate used for?
Beyond the Chemical Name
Few people come across the name "2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate" outside of specialized fields. The compound falls within the family of carbamate pesticides, a class with a long history in modern agriculture. If you spend time around fruit orchards or commercial vegetable fields, trace amounts of these substances are usually in the air after pest control applications. In most cases, professionals use similar carbamate compounds to shield crops from destructive chewing and sucking insects. For example, carbamates like carbaryl and propoxur have made their mark since the 1950s for controlling aphids and beetles that love to feed on garden staples such as lettuce and tomatoes.
Widespread Use in Pest Control
Farmers often turn to this particular molecule as an active ingredient in insecticides. Its main role acts like an enforcer—paralyzing pests by interfering with their nervous systems. This capacity has fed global reliance on such options wherever pests threaten food security or public health. Municipal pest control uses similar compounds to knock back mosquito populations in problem summers, breaking cycles of disease like West Nile virus or dengue. Homeowners with lawns and gardens occasionally use products with carbamates to ward off grubs or weevils, though the licensing and access may differ from region to region.
Safety and Environmental Questions
Anyone who’s ever prepared to spray an insecticide, even on a backyard fruit tree, knows that safety comes first. Globally, agencies such as the EPA and EFSA take a hard look at these carbamate molecules before approving them for sale. Carbamates typically break down in soil faster than organophosphates, a different but older class of insecticide. This means they don’t linger as long in the environment—good for water supplies, better for soil microbes. Studies show residues can still pose a risk to aquatic life and pollinators, so responsible application matters a great deal. I always check for pollinator warnings and restrictions about using before blooming periods for crops like strawberries or clover, just to protect local bee populations. Strict label instructions and recommended waiting times form the core of safe and sustainable use.
Balancing Modern Needs, Protecting Health
My time volunteering at urban gardens gave me a practical reminder that pest problems look different depending on location, crop, and climate. Relying entirely on chemical control usually doesn’t last. Insects often adapt, losing sensitivity to old formulas and putting more pressure on farmers and gardeners to rotate active ingredients or try alternative approaches. This points to integrated pest management as a smart direction. Combining physical barriers, biological controls, crop rotation, and targeted use of chemicals such as 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate can hold pests in check with less risk to people or the food chain. Locally, farmer co-ops and extension offices keep everyone informed about the latest science on safety thresholds and emerging best practices.
Solutions for the Future
Ongoing research tackles the tough balance between effective pest control and long-term health of soil, water, and people. New detection technology lets labs trace smaller and smaller residues in crops, and that pressure for transparency reshapes how manufacturers design directions for use. Some regions, particularly with dense populations or sensitive habitats, now limit when, where, and how carbamate insecticides get sprayed. Consumer demand for organic produce also creates incentives to cut back or swap out older chemical solutions. There’s no simple end to the pest problem, but awareness, updated science, and shared community responsibility help move conversations away from quick fixes and toward sustainable stewardship.
Is 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate toxic to humans or animals?
What Are We Really Dealing With?
Most folks don’t recognize the tongue-twister name, but 2,3-Dihydro-2,2-Dimethylbenzofuran-7-Yl-N-Methylcarbamate pops up in the world of chemical pest control. This compound is a carbamate, a class widely used in insecticides. Similar molecules pop up in products aimed at getting rid of everything from mosquitos to crop-destroying beetles. I’ve spent time working on a farm, so chemicals like this, though tucked away on a shelf and printed in small font on a container, carry a heavy presence.
Health Risks in Plain View
The main worry isn’t how these names sound, but what happens when animals or humans cross paths with them. Carbamate insecticides, including this one, affect the nervous system. They block an enzyme, acetylcholinesterase, putting nerve endings on overdrive. Both in labs and in the field, toying with brain chemistry always brings trouble. Breathing it in, swallowing it, or getting it on your skin—each route opens the door to muscle twitches, blurry vision, sweating, vomiting, and even seizures if exposure gets high enough. During my time stacking crates in a storage shed, I saw more than one co-worker struggle with dizziness and headaches after working near freshly-treated crops—hard evidence that safety data sheets don’t always capture the day-to-day risk.
Animals don’t fare any better. Pets like dogs or cats, often curious in the wrong places, can run into trouble simply by licking treated lawns or eating poisoned rodents. Birds and bees face serious threats too; carbamates don’t stop for the “good” animals. There’s solid research on this: An article in Environmental Toxicology and Chemistry traced bee population drops to contact with carbamate insecticides.
Regulation and Reality
Agencies like the EPA do set limits. Official tolerances, maximum allowable residue limits, get released to reassure farmers and consumers. Anyone working in agriculture, though, knows that misuse—too much spray, not enough waiting after treatment—remains a problem. Rural clinics in farm country report cases of pesticide poisoning every harvest. A Centers for Disease Control study from 2020 pointed to over a thousand accidental pesticide exposure calls just in one year, many from communities living near farmland.
The Case for Safer Choices
Some growers switched to integrated pest management—mixing natural predators, crop rotation, and targeted chemical use. The result: fewer chemicals, less risk. I’ve visited operations relying on beneficial insects and careful monitoring; their produce passed the taste test, and the workers stayed healthy. This doesn’t fix every problem, but it shows one way forward that doesn’t pass all the risk to people and the environment.
Anyone with kids, pets, or a garden plot needs to keep chemicals in perspective. If something can knock out a beetle, it isn’t guaranteed harmless to a toddler or the family dog. Better labels, tighter controls, and safer alternatives offer a path that respects both our need to grow food and our responsibility not to gamble with public health. We all deserve to understand what’s in use around us and push for solutions where safety doesn't come last.
What are the storage and handling requirements for this product?
Why Storage and Handling Matter
Walking through the back rooms of various businesses, I’ve seen what happens when a product isn’t given the attention it deserves. From spoiled food in grocery stores to unstable chemicals in school labs, mistakes show up quickly—and fixing them can get expensive. Manufacturers put work into creating safe and effective goods. Taking shortcuts in storage and handling usually leads to waste or outright danger.
Temperature: Controlling the Environment
Temperature ranks high on the list of things people overlook. For some items, a few degrees makes all the difference. Vaccines demand refrigeration. Pharmaceuticals like insulin lose strength if they sit out too long, even at room temperature. On the other hand, canned food has a wide tolerance but loses taste and texture if stored near a heating vent. A simple thermometer helps a lot. Staff check it daily and record results—not just to follow rules, but to catch problems before they get bigger.
Humidity and Cleanliness: Fights Against Spoilage
Humidity sneaks up on you. High moisture in storerooms means mold, clumping, or corrosion—nobody wants that. Dry goods like flour and grains soak up humidity and lose shelf life. Electronics suffer from condensation. Keeping rooms dry means relying on dehumidifiers in certain climates, not just air conditioning. Floors and shelving should stay clear of spills. Keeping spaces free from dust isn’t just about appearance; many products carry labels that warn against cross-contamination, which can trigger recalls or health problems.
Packaging: First Line of Defense
Good packaging protects its contents from pests, sunlight, and accidental dropping. Bags with tears, boxes with crushed corners—both are red flags. I learned the hard way that rodents find even the tiniest opening. Inspecting packages before placing them on shelves pays off. For chemicals, manufacturers often print storage recommendations on the label itself, so it makes sense to keep those instructions visible, not hidden away. Some products call for tightly sealed containers to avoid leaks or evaporation—think solvents or strong cleaners.
Segregation: Playing It Safe
Stacking cartons to save space sounds tempting, yet safety takes a back seat. Not every product plays well with others. Put bleach beside vinegar, or acids near bases, and you risk a nasty reaction. I’ve seen warehouses use color-coded labels or signs for easy sorting. This helps staff remember not just what goes where, but why. Storing incompatible chemicals separately avoids both injury and costly cleanup.
Access and Security: Keeping Problems at Bay
Step into a storeroom without clear labeling and you know things can go wrong fast. Accidents spike when staff spend precious minutes searching for products. Organizing shelves by product group, using clear tags, and keeping up-to-date lists saves time and trouble. Some items—controlled substances, for instance—require secure locking and documentation with every use. Ignoring these steps invites theft or regulatory trouble. Regular checks by trained staff help catch mistakes before they harm someone or disrupt business.
Training: The Human Factor
No system runs itself. Training everyone to follow safe storage and handling keeps people safe and protects the bottom line. Hands-on demonstrations mean more than a binder of rules. Encouraging questions builds a culture of responsibility, turning storage into a team effort. Businesses that invest in practical training see fewer accidents and less waste—results you can measure in dollars and peace of mind.
What is the recommended dosage or application method?
Experience Teaches Precision
Sitting in a doctor’s office—or reading the back of a fertilizer bag—one question crops up more than any other: “How much should I use?” It sounds simple, but that number might change everything. I learned this the hard way with medication. Years ago, I tried to double up on cold medicine after a tough night. Headache, nausea, more misery followed. Labels matter, and the recommended dose exists for good reason.
The Science Behind Dosage
Pharmacologists and agronomists spend years studying how much of a product gets you the benefits without landing you in trouble. For medicines, too much puts organs at risk. The FDA reviews clinical trials that watch how bodies respond to different doses, weighing safety against results. In farming, the right amount of pesticide saves crops and avoids polluting streams. My neighbor once dumped extra weed killer hoping for a spotless yard. Fish in the creek didn’t survive. Responsible application keeps people and the planet out of trouble.
Rulebooks and Real Life
Professional groups—think American Medical Association for meds or EPA for lawn care chemicals—don’t pull numbers from thin air. The best results come from years of tests and thousands of volunteers. Reading the fine print isn’t a chore; it’s a roadmap. People sometimes skip instructions, believing a little extra won’t hurt or they want speedier results. It rarely works out. In my garden, skipping dilution steps left me with wilted plants and wasted money.
Reading Labels Pays Off
Every product from cough syrup to dog flea treatments lays out detailed instructions. For medicine, doctors calculate doses using age and weight, with children getting much less. Liquid or solid, tablet or cream, each format requires clear instructions. Chemists know absorption rates can change based on what’s in your stomach or the weather outside. Pesticides might need to be sprayed early in the morning before bees start their shift. Good outcomes depend on paying attention.
The Trust Factor
Trust runs both ways. Consumers trust their doctors, farmers trust the agronomist, and both trust the research and manufacturing process. When companies win regular inspections and list side effects, they show their work. I always feel better using brands that show certifications or explain their testing process. Reports from groups like WHO or reviews published in top journals build confidence. People want to know their decisions protect their family or their business.
Practical Ways Forward
Misunderstandings happen. Clearer labels can help. Asking your doctor to talk through a prescription—or reading support guides on a company’s website—saves time and worry. QR codes go straight to video demos now, breaking down safe use step by step. Neighbors often share what works; local experience sometimes fills gaps guidelines can’t cover. Investing a few minutes in learning proper application saves money and stress in the long run.
Moving Toward Safer Habits
Right dose, right time, right way. It sounds picky, but safe use protects families, communities, and the environment. The science is in the details, and respecting those details pays off. Whether it’s medicine in a cabinet or plant food, care today means fewer problems tomorrow.
Are there any safety precautions or protective equipment needed when using this chemical?
Working With Chemicals? Precaution Comes First
Spill a little baking soda, and you’ve got a minor mess and not much else. Grab a bottle from the chemical cabinet at work or in a school lab, and the stakes go higher. Folks don’t really appreciate the difference until skin starts itching, something fizzles, or you catch a weird smell in the air. I’ve sweated through plenty of science classes and warehouse shifts where people took shortcuts with protection, and almost every time, the problem came down to not taking safety rules seriously enough.
Goggles, Gloves, and Good Old-Fashioned Common Sense
The basic protective gear—goggles, gloves, aprons—doesn’t look fancy or futuristic. But each piece fills a job you can’t really fake. Goggles keep the splash out of your eyes. Gloves hold the harsh stuff away from your skin, blocking burns and dangerous absorption. An apron or lab coat stops chemicals from soaking into your clothes, so you don’t end up carrying the hazard home.
I once saw a guy shrug off gloves to speed through a batch of cleaning with bleach and ammonia. Minutes later, he struggled to breathe and had to bolt for fresh air, all because he didn’t respect what those chemicals could do. Accidents usually don’t wait around for you to gear up after something’s gone wrong. The right equipment needs to go on every single time.
Open Windows Aren’t Enough: Ventilation Saves Lives
Masks and fume hoods help too, especially with liquids that give off dangerous vapors. Relying just on open windows in most homemade setups doesn’t work. The heavy smell that stings your nose can mean toxic fumes are building up. Inhaling them for even a few minutes leaves you foggy-headed or, worse, with lasting lung problems. I’ve met workers who turned down respirators, hoping to “tough it out,” but nobody’s lungs hold up against repeat exposure to real chemical hazards.
The Key to Safety: Treat Every Chemical Like It’s the One That Can Hurt You
Not all chemicals wave red flags or strong odors. Clear liquids might be just water—or they could be a strong acid. Labels and safety sheets (SDS) look boring, but they tell you if you need thicker gloves or a face shield, or if you need to store something away from sunlight. Training helps, and I’ve realized those fifteen-minute videos before a shift actually stick with people. They remind everyone, even the old-timers who think they’ve seen it all, that shortcuts come back to bite.
Simple Fixes Make a Big Difference
Accidents drop when soap and water stay close by, and folks know where to find an eyewash station. Emergencies don’t wait for someone to hunt through a mess for help. Posting safety posters and checklists where everyone can see them, plus regular drills, turn safety into a habit instead of an afterthought. Companies that set real standards—and workers who push for better protection—see fewer injuries and less downtime.
Nobody’s invincible, and chemicals don’t give warnings before they do harm. Putting on goggles and gloves isn’t overkill—it’s how you make sure you’re around to share a coffee at the end of the workday.
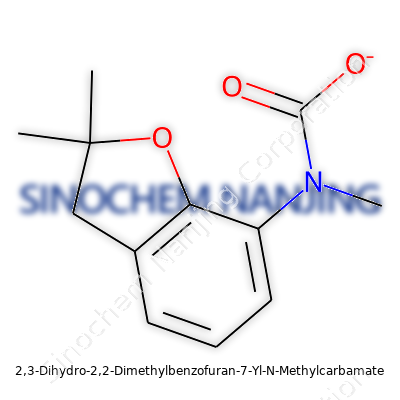
| Names | |
| Preferred IUPAC name | (2,2-dimethyl-3,4-dihydro-2H-1-benzofuran-7-yl) N-methylcarbamate |
| Other names |
Propoxur Baygon Blattanex Aprocarb Unden OMS 33 NPC 101 Prentox Vapiton Arprocarb |
| Pronunciation | /ˈdaɪ.haɪdroʊ ˈdaɪˈmɛθəl ˈbɛnzoʊfjʊˌræn ˈsɛvən aɪl ɛn ˈmiːθəlˈkɑːrbəˌmeɪt/ |
| Identifiers | |
| CAS Number | 17804-35-2 |
| 3D model (JSmol) | `3DModel/CTX (JSmol)` string for **2,3-Dihydro-2,2-dimethylbenzofuran-7-yl-N-methylcarbamate**: ``` CQOBQYXUVLIMQT ``` |
| Beilstein Reference | 2642213 |
| ChEBI | CHEBI:38864 |
| ChEMBL | CHEMBL2105931 |
| ChemSpider | 156146 |
| DrugBank | DB11438 |
| ECHA InfoCard | ECHA InfoCard: 100.102.791 |
| EC Number | 3.1.1.86 |
| Gmelin Reference | Gmelin 82832 |
| KEGG | C18433 |
| MeSH | D003883 |
| PubChem CID | 129849079 |
| RTECS number | GV8775000 |
| UNII | RY8281J8OJ |
| UN number | UN2588 |
| CompTox Dashboard (EPA) | DTXSID2022109 |
| Properties | |
| Chemical formula | C12H15NO3 |
| Molar mass | 209.25 g/mol |
| Appearance | white solid |
| Odor | Odorless |
| Density | 1.14 g/cm³ |
| Solubility in water | Insoluble in water |
| log P | 2.68 |
| Vapor pressure | 0.000049 mmHg at 25°C |
| Acidity (pKa) | 13.1 |
| Basicity (pKb) | 4.26 |
| Magnetic susceptibility (χ) | -54.72·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.573 |
| Dipole moment | 2.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 362.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -491.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6065.6 kJ/mol |
| Pharmacology | |
| ATC code | N05CM07 |
| Hazards | |
| Main hazards | Harmful if swallowed. Toxic if inhaled. Causes skin and eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H332, H400 |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P284, P301+P310, P302+P352, P304+P340, P304+P312, P305+P351+P338, P308+P311, P314, P320, P330, P363, P337+P313, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-1-0 |
| Flash point | 93°C |
| Autoignition temperature | 400 °C (752 °F; 673 K) |
| Lethal dose or concentration | LD50 oral rat 75 mg/kg |
| LD50 (median dose) | 7.5 mg/kg (rat, oral) |
| NIOSH | DN8225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Carbaryl Propoxur Bendiocarb Oxamyl Aldicarb Fenobucarb Carbofuran Methomyl Temephos |

