2,3-Dicyano-5,6-Dichlorohydroquinone: A Cornerstone in Chemical Synthesis and Research
Historical Development
Throughout the long path of synthetic organic chemistry, few building blocks attract as much focused attention as quinone derivatives. Chemists working in the 1970s began to play with halogenated hydroquinones and the idea of adding dicyano groups, aiming to make molecules that could serve as versatile intermediates. 2,3-Dicyano-5,6-dichlorohydroquinone soon emerged from this era's surge of curiosity and trial-and-error. Its development was spurred by the need for molecules with strong electron-withdrawing features. Academic labs, looking for easier routes to high-value aromatic compounds, saw its potential early on. Over time, technical literature built up around its promise, pushing this compound into specialized chemical catalogs and expanding its role both in academia and industry.
Product Overview
2,3-Dicyano-5,6-dichlorohydroquinone, sometimes dubbed DCDCHQ, shows up mostly as a pale solid in the lab, but its value is anything but plain. Researchers hold it up as a workhorse: rich in reactivity, flexible in use, and reliable as a core molecule that branches off into diverse chemical families. Its double cyano substitutions act like a magnet for further derivatization, giving synthesis chemists plenty to work with for tailor-made research adventures in condensed matter, advanced polymers, and pharmaceuticals. The pairing of chlorine atoms continues to draw in those interested in tuning electronic properties, a key factor in the push for more efficient organic electronics and catalysts.
Physical and Chemical Properties
Describing DCDCHQ starts by looking at its two key axes: the electron-deficient nature imparted by the cyano groups, and the stabilizing, but reactive, dichloro motif. It typically crystallizes with a light hue, signaling its purity in the hands of a careful synthesist. Its solubility splits the difference between polar aprotic solvents and the more forgiving solvents used in large-scale industrial batches. The cyano and chloro substitutions make it resistant to easy degradation, an attribute that showcases its practicality in arduous reaction conditions. Melting point records vary, but most standard sources agree that it doesn’t start to break down until reaching a respectable temperature threshold, reducing handling hazards under typical lab protocols.
Technical Specifications and Labeling
Most bottles of 2,3-dicyano-5,6-dichlorohydroquinone leave warehouse shelves with purity levels exceeding 98%. That margin matters for specialists in sophisticated electronic materials or drug precursor synthesis. Impurities, even below a percent, might skew research outcomes or trigger side reactions, breaking the hearts of graduate students everywhere. Accurate chemical labeling reflects not just molecular weight and batch codes, but also provides standardized nomenclature and common synonyms, an invaluable service for busy synthetic chemists juggling multiple reactants. Properly labeled containers also point out relevant safety standards, echoing lessons hard-won by those who came before.
Preparation Method
Synthesizing DCDCHQ involves a balancing act. Most routes center on a hydroquinone backbone, treated first with chlorinating agents like sulfuryl chloride, then carefully funneled through cyano group introduction—usually with copper cyanide or other reliable cyanation reagents. Chemists walk a narrow road: gentle enough to preserve the hydroquinone core; strong enough to ensure efficient substitution. Running these reactions under inert gas shields against unwanted oxidation or polymerization, persistent issues for any phenolic compound in reactive company. The process yields a recognizable product after standard workups, often recrystallized to ensure both structural integrity and analytical cleanliness.
Chemical Reactions and Modifications
The bread-and-butter of DCDCHQ’s value rests in its receptive sites for further transformations. Its electron-deficient core welcomes nucleophilic attack, giving a springboard for attaching amines or sulfur ligands. It also stands up to cross-coupling protocols, handing organometallic chemists a launching pad for drawing intricate patterns onto strained or fused ring systems. Electrochemical reduction opens even more avenues, as controlled reduction sometimes uncovers new potential for use in energy-storage materials. Replacing the cyano or chloro groups steps into the world of custom ligand manufacture, where subtle tweaks in ligand design change everything from metal binding to molecular recognition. The molecule isn’t just an endpoint; it’s a door to a dozen other rooms in the house of synthetic chemistry.
Synonyms and Product Names
Wandering through chemical catalogs, this compound goes by a small set of aliases, each reflecting some aspect of its structure: DCDCHQ, 2,3-dicyano-5,6-dichloro-p-hydroquinone, and sometimes even non-systematic names echoing the hydroquinone core or its dicyano heritage. Keeping the list short streamlines both procurement and regulatory paperwork, though a quick glance at a safety data sheet often reminds you which derivatives trade under which names. Precise naming prevents mix-ups—an important point, especially for researchers stacking up a dozen analogs for rapid screening.
Safety and Operational Standards
Lab veterans know the drill: gloves, goggles, and a working fume hood form the unspoken uniform for handling anything with cyano or chloro labels. DCDCHQ’s stability under mild conditions lulls some into a false sense of security, but dust inhalation and accidental skin contact remain genuine risks. Spreadsheets tracking exposure aren’t just for bureaucrats—those guidelines reflect a history of lessons learned, sometimes the hard way. Researchers keep material safety data sheets handy, looking up environmental handling and spill management, especially in teaching labs stocked with first-year chemists still gaining respect for reactive intermediates. Waste protocols for chlorinated organics usually grow stricter each passing year, shaped by environmental standards that try to balance research and stewardship.
Application Area
Modern industry and academia never stop digging for small compounds capable of big things. In electronics, some teams work DCDCHQ into the design of organic semiconductors, exploiting its fine-tuned electronic structure for improved charge transport and stability. Medicinal chemistry teams probe its aromatic core for tweaks leading to novel drug scaffolds, especially where electron-deficient aromatics turn out to block or activate target proteins. Material scientists aren’t shy with their curiosity, testing how its modifications perform under photo- or redox-active regimes for batteries and smart polymers. Broad applicability continues to prove out the value of focused, atomic-level tuning. Each field adapts its use, reflecting both advances in technique and shifting commercial priorities.
Research and Development
Research groups aiming for high-impact results often start by looking for molecules matching reliability with flexibility. DCDCHQ floats to the top because new derivatives branch out into unexplored chemical territories. PhD candidates and postdocs, busy building libraries for screening campaigns, keep returning to this compound because of its straightforward reactivity and consistent yields. Progressive research teams, especially those working at the interface of chemistry and materials science, include it in combinatorial arrays and customized ligands for organometallic catalysts. Reading through recent publications, it’s clear that DCDCHQ holds its own as a launching pad for chemical innovation.
Toxicity Research
Any molecule with multiple halogen and cyano groups draws oversight from toxicologists. Some findings indicate moderate acute toxicity by ingestion or inhalation, and animal studies flag the need for precaution. Long-term data gaps exist, largely because few end-user products deploy this molecule directly, but regulatory circles remind researchers about best practice in risk reduction. Preventive measures overrule any temptations to rush; a few minutes spent gloving up and checking vent fans beats days of uncertainty in the health clinic. Waste streams get treated as hazardous by default, more from prudence than documented environmental impact, as the track record on biodegradation and persistence still lags behind usage trends.
Future Prospects
Chemists always seek ways to wring more from versatile building blocks, and DCDCHQ hardly seems destined for retirement. The rise of green chemistry puts pressure on minimizing halogen content and waste, prompting teams to explore more efficient syntheses or safer derivatives. The electron-deficient core inspires those hunting for new organic conductors or redox-active agents, given ongoing pushes toward flexible electronics and next-level battery materials. Cross-disciplinary partnerships open up new perspectives, as synthetic teams start talking to computational modelers and device engineers. The push for sustainable chemistry might even see DCDCHQ serve as a development test case: balancing reactivity, safety, and performance in an industry hungry for smarter, cleaner molecules.
What is the chemical formula of 2,3-Dicyano-5,6-Dichlorohydroquinone?
An Up-Close Look at the Molecule
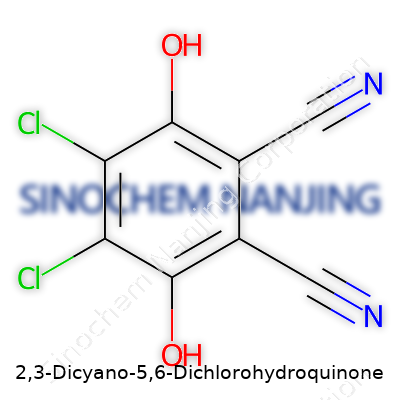
Scientists rely on chemical formulas to get straight to the core of what they’re handling. The formula for 2,3-Dicyano-5,6-dichlorohydroquinone is C8H2Cl2N2O2. Each letter and number peels back a layer of the compound, offering more than just a jumble of letters. The formula tells a real story about its chemical backbone—eight carbons, two hydrogens, two nitrogens, two chlorines, and two oxygens. No aspect is random.
Why Formulas Aren’t Just for Textbooks
I have seen students roll their eyes at complicated-sounding compounds, thinking this stuff can’t matter in the real world. Thing is, knowing a formula connects you to much more than academic trivia. For chemists and quality assurance folks, the difference between having a couple of chlorine atoms or a missing cyanide group could be a dealbreaker in making safe products. If you mix the wrong proportions or miss a detail in the structure, things can spiral—contamination, failed experiments, or worse, hazardous byproducts in manufacturing.
2,3-Dicyano-5,6-dichlorohydroquinone combines the core skeleton of hydroquinone, a chemical recognized for its role in photography and specialty manufacturing, with the potent additions of cyano and chloro groups. Think of those cyano groups—each one contains cyanide, which alters how the molecule behaves with its environment. The chloro groups aren’t just decoration either. Chlorine atoms shift how reactive and stable this compound stays under stress, a key fact in industrial settings.
Real-World Impacts: Health, Safety, and Production
The moment you hold a bottle of any chemical, you trust the formula stamped on its label. Lives and livelihoods depend on getting it right. This specific compound, C8H2Cl2N2O2, doesn’t just have academic roots. Manufacturers rely on precision. If you cut corners or misread the structure, you face lawsuits, recalls, and safety hazards. The formula doesn’t lie—if you’re working with dicyano and dichloro groups, you better fit these into your process control and worker safety plans. The Environmental Protection Agency and scientific journals document cases where improper handling of chlorinated and nitrated aromatics led to soil contamination, occupational exposures, and downstream pollution issues.
From my work in research, seeing people treat chemical names as just labels can cause lapses in judgment. Overconfidence and shortcuts open teams up to expensive and painful lessons. It’s not about fear-mongering, but about respect for the fact that chlorinated compounds can last for years in the environment and that cyanides can be lethal at low doses. Getting the formula right sets the groundwork for good science and for health and safety on the job.
What We Can Do To Stay Ahead
Knowing a formula isn’t enough; tracking how that structure behaves in different situations—under heat, in water, in waste disposal—sets apart a professional from someone just skimming the label. Data sheets, risk assessments, and trusted supply chains matter. Double-checking the chemical structure against reputable databases like PubChem or Sigma-Aldrich doesn’t just pad paperwork, but prevents avoidable mistakes. Teams should press for clarity: don’t settle for vague specs, push for certainty and traceability. Training newer chemists and plant workers to read formulas like C8H2Cl2N2O2 and really understand where each atom fits—that’s the foundation for clean labs, safe workplaces, and reliable results.
What are the main applications of 2,3-Dicyano-5,6-Dichlorohydroquinone?
Everyday Impact of a Little-Known Chemical
2,3-Dicyano-5,6-Dichlorohydroquinone isn’t a name that rolls off the tongue, and most folks outside lab work might not recognize it. Yet, the roles this chemical plays sneak quietly into products and processes that keep life moving, especially in science and industry. It brings value to analytical chemistry and electronics, sectors that shape today’s technology and health outlook.
Breathe Easy: Powering Chlorine Detection in Water
Clean water worries plenty of people and for good reason. Municipalities and labs depend on fast, reliable ways to measure chlorine levels. 2,3-Dicyano-5,6-Dichlorohydroquinone has carved out a niche as a key reagent in colorimetric chlorine tests. The principle is simple: the compound reacts with chlorine in water samples, changing color based on the amount of chlorine present.
This color change gives a straightforward signal. Workers can check chlorine levels on the spot and act fast if numbers drift out of range. Drinking water, swimming pools, even wastewater outflows benefit from this real-time insight. With water safety linked to health standards worldwide, a chemical that supports precise monitoring helps keep harmful microbes at bay and public trust intact.
Microelectronics: Playing a Quiet But Crucial Role
Electronics continues shrinking, packing thousands of functions into ever-smaller chips. The industry regularly seeks cleaner processes and sharper precision. 2,3-Dicyano-5,6-Dichlorohydroquinone functions as an oxidizing agent during some stages of semiconductor manufacturing. Its inclusion helps prepare ultra-clean surfaces and supports the fine-tuning of thin films.
Process engineers value predictability and repeatability. A chemical with a strong oxidizing punch and stable handling makes their lives a little easier when producing wafers or microchips. High yields and fewer defects mean more reliable tech in phones, cars, and home gadgets.
Laboratory Research: Opening Doors for Innovation
In academic and industrial R&D, scientists use 2,3-Dicyano-5,6-Dichlorohydroquinone as both a reactant and reference compound. Its structure allows researchers to explore new materials and chemical reactions. The world of organic synthesis often calls for tools that push boundaries—this compound fits that bill.
New medicines and materials sometimes trace their lineage to work involving this hydroquinone. Teams probing antioxidants, redox properties, or crystal engineering sift through options searching for compounds that improve performance or stability. Every new experiment might shape breakthroughs in drug design, specialty coatings, or analytical sensors.
Challenges and Next Steps
Despite its usefulness, using 2,3-Dicyano-5,6-Dichlorohydroquinone in the lab or plant brings up safety and waste questions. Strong chemicals with reactive groups can pose health risks if handled poorly. Proper training and strict protocols make a difference—gloves, ventilation, and real education around chemical risks help prevent trouble.
Waste management deserves real attention. Chemical residues should never reach water supplies or soil. Facilities that use this compound invest in treatment and disposal systems. Researchers keep hunting for greener alternatives or recycling angles to cut down on impact without sacrificing performance.
Looking Ahead
Chemicals like 2,3-Dicyano-5,6-Dichlorohydroquinone shape more parts of daily life than most people realize. The story of this molecule shows how careful science blends with smart oversight to make a difference, from water quality to cutting-edge tech. The search for efficiency, safety, and progress keeps this compound in demand—and keeps chemists reaching for better ways to use and manage it.
What are the storage conditions for 2,3-Dicyano-5,6-Dichlorohydroquinone?
Understanding Its Nature
2,3-Dicyano-5,6-dichlorohydroquinone belongs on the shelf with other specialty lab chemicals that people rarely see outside research or industrial settings. Folks who remember their days in the lab know that compounds with cyano and chloro groups often bring both reactivity and some real safety challenges. Two things stand out: this compound doesn’t play nice with heat or moisture, and any slip-up could mean wasted material or a safety scare.
Why Storage Details Matter
Improper storage brings risk, not just of spoiled chemicals, but of unexpected reactions, toxic fumes, or worse. I’ve seen the results of bottles stowed on the wrong shelf: clumps where there should be powder, stinging eyes, or in rare cases, alarms blaring. This isn’t a scare tactic, it’s reality in a working lab. Worse still, poor labeling and casual habits turn rare events into common ones. Whenever a new chemical arrives, people check the safety data sheet, but then habits slip, labels fade, and the dangers creep in.
What Good Storage Looks Like
The right approach for 2,3-dicyano-5,6-dichlorohydroquinone starts with a well-ventilated chemical storage cabinet. Keep it cool and dry. Heat accelerates decomposition and pushes volatility: even a few degrees can make a difference over time. Desiccators do a great job of defeating humidity, which is crucial because hydroquinones pick up water and degrade fast. Basic glassware with tight lids works, but a nitrogen-blanketed container cuts off oxygen, slowing down any unwanted reactions.
From the start, staff need to take another look at labels every few weeks—not just reading, but replacing the old ones if they’re fading or peeling. Outdated signage led to more than one accident in places I’ve worked. Some colleagues kept handwritten cards on every shelf, a habit worth borrowing. Good records beat perfect memory.
Personal Protective Gear and Facilities
Storage doesn’t end at the door. Chemical burns and inhalation trauma come from small mistakes—opening a jar without a mask, refilling a container in a crowded room. Safety glasses, fitted gloves, and long sleeves count as the bare minimum. A fume hood isn’t just for dramatic reactions; it’s a shield for anything that could aerosolize or fume. Relying on the nose to judge safety gives false confidence—by the time you smell it, you’ve probably inhaled too much.
Mitigating Spills and Leaks
Spills create real hazards. Walk into any shared lab, and you’ll spot the stain from last year’s cleanup: bleach didn’t do the trick, maybe because the staff missed the spill within seconds. Fast, proper cleanup means having absorbent pads, neutralizers, and bags nearby. In my experience, running for a mop wastes minutes that matter. Instead, teach everyone to grab a spill kit before they reach for the bottle.
Training and Audit Culture
Mistakes don’t just come from ignorance—they come from rushed work, skipped steps, or silent assumptions. Labs with regular training avoid more problems than those that trust everyone reads the binder. Spontaneous audits, not just annual reviews, work best. In my first research job, the best manager would spring surprise checks, not to catch errors, but to catch habits that slide. That habit stopped small problems before they became big ones.
Addressing Larger Challenges
It’s tempting to treat storage as a checklist item, but that attitude misses the bigger picture. Proper care for chemicals like 2,3-dicyano-5,6-dichlorohydroquinone protects health and the bottom line. Labs should invest in better containers, more frequent inventory checks, and strong training. These choices do more for safety and quality than any warning poster in the hallway.
Is 2,3-Dicyano-5,6-Dichlorohydroquinone hazardous or toxic?
Looking Past the Name: What We’re Dealing With
Long chemical names often make people tune out, but any compound used in industry or research can affect health or the environment. 2,3-Dicyano-5,6-Dichlorohydroquinone doesn’t roll off the tongue, but its impact deserves attention. Its structure contains two cyanide groups and two chlorine atoms. These are functional groups with a known history of problems. Cyanides and chlorinated compounds both raise red flags, not just in textbooks, but also in the real world where workers, communities, and the environment come in contact with them.
A Closer Look at What We Know
This compound hasn’t earned its own substantial chapter in the mass of chemical hazard data, yet its building blocks are familiar troublemakers. Cyanide groups, when released from a molecule, can disrupt cellular respiration. That’s not just a lab concern — acute cyanide poisoning has been documented for over a century, affecting tissues by starving them of oxygen. Inhalation or even skin contact with cyanides can present immediate risks. Chlorinated aromatics, on the other hand, show up again and again in discussions of environmental toxicity. Some persist in soil and water much longer than anyone would like, creating ongoing exposure and potential accumulation up the food chain.
Evidence, Not Guesswork
Scientific studies often prioritize widely-used industrial chemicals, so rare or novel substances sometimes fly under the radar. For university researchers or people in specialty chemicals, the absence of a detailed hazard report is an everyday frustration. But chemical law doesn’t work backwards; real harm has to happen, or a pattern has to show up, before tougher regulations get written. That means workers, labs, and local ecosystems could absorb the consequences first.
If we compare similar molecules, like other dicyanodichlorinated hydroquinones, toxic effects show up in both animal and cell culture studies. These can include organ damage, reproductive effects, and mutagenicity. Safe handling protocols come down to assuming the risk — not waiting for proof. The structure and documented breakdown products suggest possible formation of hydrogen cyanide gas during improper disposal or chemical reactions, an immediate inhalation hazard.
Doing Better: What Helps
People often only realize the dangers of chemicals when a mishap happens or symptoms build up in teams working with them. Simple moves such as requiring gloves, goggles, and fume hoods protect researchers and workers. Training matters even more. I have watched teams skip safety briefings, reasoning "It’s not one of the famous bad actors," until an incident proves them wrong. If you ask anyone who has had a close call, they’ll tell you that investing ten minutes in preparation beats spending weeks cleaning up after an accident.
Waste treatment must respect that both cyanide and chlorine demand special disposal steps to prevent release into the environment. On-site neutralization with proper reagents, not just washing down the sink, should be standard. Clear labeling and secure storage cut down on accidental exposure and mixing with other chemicals that might create even more toxic byproducts.
Building Community Awareness
Local community right-to-know programs work only if companies openly list chemicals present on site. Workers should have access to material safety data sheets and feel empowered to raise safety questions. Public health researchers who shine a light on "less famous" hazards help everyone avoid repeating avoidable mistakes.
Finding Answers Without the Wait
No law stops companies from proactively substituting safer alternatives once risks are suspected. Frontline workers, managers, and safety officers all play a role in asking, "Is there something less hazardous that gets the job done?" Industry change takes time but real progress happens when vigilance replaces complacency, one chemical and one workplace at a time.
What is the shelf life of 2,3-Dicyano-5,6-Dichlorohydroquinone?
Stability Stories from the Lab Bench
Many compounds look stable tucked away in a chemistry supply cabinet, but their stories grow complicated with time and the right conditions. Take 2,3-Dicyano-5,6-Dichlorohydroquinone, for instance. This crystalline substance, once prized by organic chemists for its role in electron-transfer, really only proves itself if its purity remains. No industry or laboratory wants to run reactions with degraded chemicals.
On paper, technical documentation often references two to three years of shelf life for this compound. There’s a catch, though. Those numbers only hold when tightly sealed containers avoid humidity, light and swings in temperature. Even with these precautions, slight decomposition can slip in unnoticed. If the lid doesn’t screw on tight, or some moisture sneaks in during transfers, batches might already go off-spec in less than a year.
Why Shelf Life Matters in Real Practice
Any experienced lab worker grows suspicious of “old” reagents. My own time in dusty research settings taught me that not all chemicals age gracefully. 2,3-Dicyano-5,6-Dichlorohydroquinone doesn’t announce its breakdown with color changes or strong odors. Instead, minute hydrolysis or slow oxidation nibbles away at effectiveness. Suddenly, yields in an oxidant-catalyzed reaction drop. The root cause isn’t a mystery: the compound, thought to be stable, faltered in storage.
Standard analytical checks—infrared spectra, thin-layer chromatography, and melting point—often tell the real story. Any deviation in the pattern suggests the bottle may only belong in the chemical waste drum. Relying on manufacturer-provided expiry can prove costly, particularly in high-precision synthesis where a failed experiment means lost days and wasted resources.
Facts from the Literature
As referenced in journals such as Journal of Organic Chemistry and chemical safety datasheets, 2,3-Dicyano-5,6-Dichlorohydroquinone stays stable in dark, cool, and dry places. Under best-case scenarios, one can expect two years or slightly more of reliable use. The presence of chlorines and cyano groups does offer a bit more resistance to aerial oxidation compared to regular hydroquinones, but laboratory mishaps remain common. Errant exposure to room air can start hydrolysis or reduce potency.
Practical Solutions for Storage and Testing
Reliability grows out of habits built around chemical storage. Use of amber glass bottles over clear jars already gives a few months’ advantage by blocking UV light. Pack desiccant pouches in storage cabinets, and keep chemicals away from sources of heat. Marking opening dates directly on reagent bottles beats relying on memory or vague records. If a chemical proves irreplaceable in a workflow, it pays to buy smaller amounts and stock more frequently, rather than risk loss from a bulk batch gone bad.
Regular scrutiny matters most. In research teams I’ve worked with, the best practice involved scheduled re-testing of sensitive chemicals before each use in important syntheses. Even a quick TLC check can catch a decline before the run goes awry. Companies and labs can build this mindset into their culture, training each new chemist to stay cautious.
Looking Forward
The importance of shelf life stretches beyond keeping chemicals on a shelf. It underpins every successful experiment and product. 2,3-Dicyano-5,6-Dichlorohydroquinone, like many specialty reagents, delivers on its promise only when everyone from procurement to final user respects its quirks and limits. Smart storage, routine checks and clear communication between teams beat any official expiry date printed on the label.
| Names | |
| Preferred IUPAC name | 2,3-dichloro-1,4-benzenedicarbonitrile |
| Other names |
2,3-Dicyano-5,6-dichloro-1,4-benzenediol NSC 110453 |
| Pronunciation | /ˈtuːˌθri daɪˈsaɪˌænoʊ ˌfaɪvˌsɪks daɪˈklɔːroʊ haɪˌdrɒk.siˈkwiː.noʊn/ |
| Identifiers | |
| CAS Number | 2454-89-5 |
| Beilstein Reference | 2226069 |
| ChEBI | CHEBI:132731 |
| ChEMBL | CHEMBL3291326 |
| ChemSpider | 171408 |
| DrugBank | DB08345 |
| ECHA InfoCard | ECHA InfoCard: 100.116.134 |
| EC Number | 207-079-2 |
| Gmelin Reference | 77763 |
| KEGG | C06602 |
| MeSH | D03.438.221.173.190.350 |
| PubChem CID | 16743 |
| RTECS number | KK1400000 |
| UNII | J977DOV5F7 |
| UN number | 3276 |
| CompTox Dashboard (EPA) | DJ79H9Y5BR |
| Properties | |
| Chemical formula | C8H2Cl2N2O2 |
| Molar mass | 286.03 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.72 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.41 |
| Vapor pressure | 1.42E-7 mmHg at 25°C |
| Acidity (pKa) | 1.21 |
| Basicity (pKb) | 1.54 |
| Magnetic susceptibility (χ) | -0.71e-6 cm³/mol |
| Refractive index (nD) | 1.682 |
| Viscosity | 1.24 cP (25°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -168 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P280, P301+P312, P302+P352, P305+P351+P338, P330, P337+P313, P362+P364, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 204.5 °C |
| Lethal dose or concentration | LD50 oral rat 320 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 5 gm/kg |
| NIOSH | KV7530000 |
| PEL (Permissible) | Not established |

