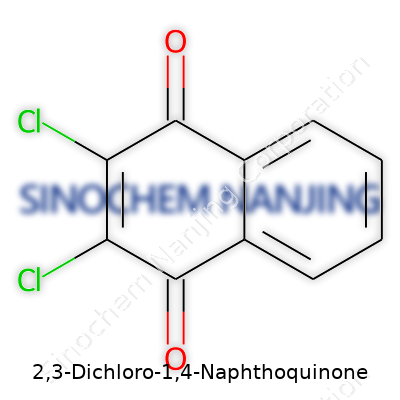
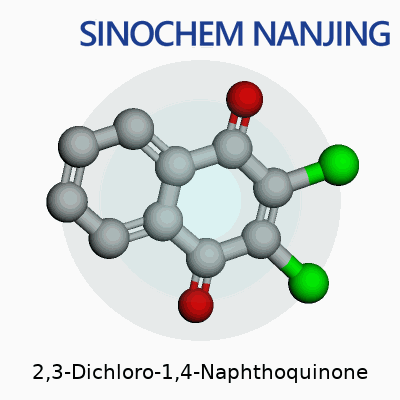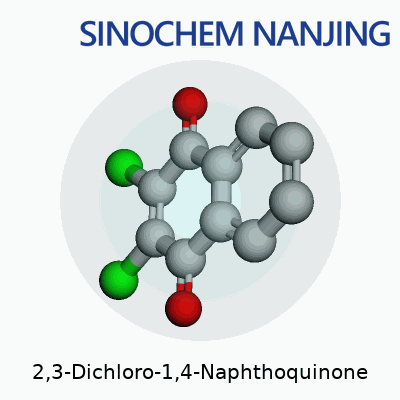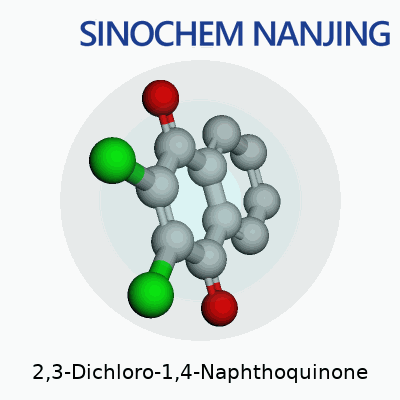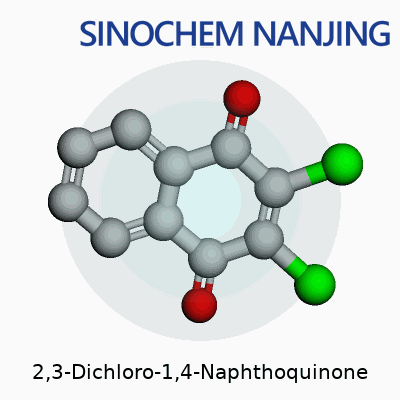
2,3-Dichloro-1,4-Naphthoquinone: Chemical in Context
Historical Development
Back in the days when chemical synthesis started to transform the fields of dye manufacturing and pharmaceuticals, folks learned that tweaking naphthoquinone molecules led to some real breakthroughs. Chemists noticed that putting chlorine in the right spots—specifically at positions 2 and 3 on 1,4-naphthoquinone—changed both the reactivity and stability of the compound. It wasn’t just idle curiosity. Early patents around the mid-20th century feature this molecule as part of larger batches of chlorinated quinones, giving a good glimpse into the practical drive behind its development. Today, people who dive into histories of organic synthesis run into this family of chemicals time and again, often tied to coloration chemistry and as building blocks for more complex molecules.
Product Overview
2,3-Dichloro-1,4-naphthoquinone doesn’t jump out in a crowded laboratory. It’s a yellowish solid, with a sharp—almost medicinal—odor that lingers longer than you’d expect. Most chemists bump into it as a reagent but the world outside lab benches hardly knows it by name. Still, its real value comes from the way it activates other molecules. Whether it’s used in the formation of dyes or as a precursor for advanced materials, manufacturers and researchers favor its versatility. You won’t see it on any supermarket shelf, but anyone synthesizing compounds in this structural family benefits from its availability and relatively stable shelf life.
Physical & Chemical Properties
As a crystalline powder, this compound stays stable at room temperature and doesn't require fancy conditions to keep from decomposing. Its melting point lands above 130°C—a classic mark for a tightly bound aromatic molecule. It dissolves in organic solvents like chloroform and dichloromethane, which come in handy during purification or downstream reactions. The two chloro groups make it more reactive toward nucleophiles, compared to the parent naphthoquinone, and this property opens further twists in syntheses. Inhalation or contact brings a biting odor that’s hard to forget, serving as a natural warning sign before things reach toxic levels. Experience teaches respect for chemicals that signal their danger upfront, and this one certainly fits that mold.
Technical Specifications & Labeling
Anybody serious about working with 2,3-dichloro-1,4-naphthoquinone starts by checking the label for purity, batch analysis, and expiration. Recognized suppliers present data on reaction residue, water content, and identifying marks from infrared or NMR spectrometry. Every bottle worth its salt lists a CAS number, which helps avoid nasty mix-ups with similar-sounding chemicals. Though it's tempting to gloss over paperwork, careful attention to labeling guards against impurities that can throw a wrench into sensitive synthesis projects. Real-world lab stories are littered with ruined experiments due to a stray contaminant. Lab protocols often require MSDS review before opening the container, for good reason.
Preparation Method
Most commercial batches of 2,3-dichloro-1,4-naphthoquinone stem from direct chlorination of 1,4-naphthoquinone. This synthesis usually relies on chlorinating agents like chlorine gas or other chlorides under controlled conditions. You can spot chemistry students learning about the exothermic bite of chlorination reactions the first time they run this prep. You need strong ventilation and a steady hand, since any shortcut risks both the product yield and personal safety. Old literature describes tweaks to minimize hazardous byproducts, but the bones of the process haven’t changed much in decades. More advanced work sometimes swaps these harsh conditions for milder, more selective approaches—saving resources and cutting down on waste.
Chemical Reactions & Modifications
Once you have this molecule, it doesn’t just sit on the shelf. Chemists love it because those two chlorine atoms call out for further substitution. Add a nucleophile and you get replacement reactions; add reducing agents and the quinone ring transforms, often producing unique hydroquinone derivatives. Pharmaceutical chemistry sometimes makes use of this compound as a stepping-stone to biologically active molecules, tweaking the core structure to tailor activity. In material science, researchers look at its ability to anchor onto surfaces or participate in oxidative coupling, spinning off lines of research involving smart coatings and sensors. The point isn’t the molecule itself—it’s the chemical agility it offers across so many different pathways.
Synonyms & Product Names
Different catalogs and chemistry texts refer to this compound under a pile of names, often reflecting differences in standards or local conventions. It pops up as Dichloronaphthoquinone, DCNQ, or sometimes just by shorthand ladder numbers. These aliases matter most for folks managing chemical inventories or scouring literature for cross-referenced studies. Common sense, built from years in the lab, says it pays to double-check identifiers before punching in an order or searching unique modifications. Name confusion has wrecked more than a few research budgets over the years.
Safety & Operational Standards
Safety isn’t an afterthought with 2,3-dichloro-1,4-naphthoquinone. Gloves, goggles, lab coats—the full kit—make a difference. The compound irritates skin and eyes and can hit respiratory systems if inhaled in too large a dose. Chemical splash goggles and a fume hood are standard practice, not overkill. Storage calls for sealed containers away from light and incompatible chemicals like strong bases or reducing agents. The lessons drilled in safety training come back in full force: check your fume hood, don’t pipette by mouth, and label everything. It’s remarkable how a small slip, such as open skin contact, can result in burns or long-term damage. Modern labs emphasize chemical hygiene, and cases involving chlorinated quinones are a textbook reason why.
Application Area
Industries tied to organic pigments, agrochemicals, and pharmaceuticals take a hard look at 2,3-dichloro-1,4-naphthoquinone. Dye manufacturers appreciate its ability to participate in synthesis routes that produce strong, stable colors, especially for fabrics that have to withstand sunlight and repeated washing. On the pharmaceutical side, it acts as a scaffold for molecules aimed at antibacterial or antifungal activity. Researchers in electronics poke at its redox properties, seeking ways to develop better semiconductors and sensors. The range of uses comes down to the flexibility of the quinone ring and the influence of those two chlorine atoms, which open countless doors for tailored functionality.
Research & Development
Every year, articles and patents showcase new applications and derivatives built on this chemical backbone. University research groups explore tweaks to the molecule, seeking greener synthesis methods that cut down hazardous byproducts. Work continues on using it as a precursor for complex heterocycles—potentially useful in treating emerging diseases or strengthening existing product lines. Collaboration between industry and academia thrives here, with real feedback about what works at the bench and what scales up safely. It’s not hype to say that small advances in how these core molecules are handled ripple out into major changes for entire markets. Keeping up with new literature sets a foundation for smarter, safer workflows.
Toxicity Research
There’s no denying that chlorinated naphthoquinones bring hazards to both users and the environment. Studies show that exposure beyond recommended limits causes skin, respiratory, and sometimes systemic toxicity. Regulatory agencies keep close tabs on workplace exposure, setting occupational levels and regularly updating guidance based on fresh toxicological data. In my experience, even seemingly minor exposures—like a drop on the skin or a whiff during a mishap—mean a trip to the eyewash or even a clinic. Mitigation strategies make a real difference. For waste handling, chemical deactivation and secondary containment stop runoff and accidental mixing. Honest discussion in the lab or workplace about the real risks, not just paper assessments, improves both safety and trust among staff.
Future Prospects
Advances in sustainable chemistry point toward more efficient and safer methods of production. Green chemistry tools like more selective catalysts or solvent-less syntheses promise to reduce the environmental footprint. Research into low-toxicity alternatives continues, but for many applications, there’s still no replacement for the versatility provided by this class of compounds. It’s up to chemists, both in industry and in research, to shape the next chapter—balancing performance goals with real concerns about health, safety, and environmental persistence. Transparent reporting, ongoing toxicological studies, and industry-wide sharing of best practices matter more now than ever. Whether we’re working in the lab, piloting new production lines, or teaching the next generation, learning from both past mistakes and fresh data is what keeps progress moving.
What is the chemical formula of 2,3-Dichloro-1,4-Naphthoquinone?
Understanding the Structure
In the world of chemistry, small changes make a big impact. Consider 2,3-Dichloro-1,4-Naphthoquinone. This isn’t a name that rolls off the tongue at a dinner party. Behind that technical label, though, there’s a clear chemical signature: C10H4Cl2O2. It represents a naphthoquinone backbone, rigged with two chlorine atoms at the second and third position, meaning the molecule holds onto its core but threads in new possibilities where the chlorines sit. The formula isn’t just an academic exercise, either. It matters for safety sheets, patents, and even for folks researching soil remediation or industrial dyes.
Real-Life Stakes Beyond the Classroom
Throughout college labs and later in industrial settings, knowing an exact formula helps avoid confusion. Mix-ups between isomers or other substituted naphthoquinones can mean deal-breaking safety hazards. I remember a story from my internship—someone misread a label in the lab. Instead of using a dichloro version, they used a dibromo version. What followed wasn’t exactly a disaster, but the product didn’t catalyze like it should. All because of two misplaced atoms—four letters in a formula that make the difference between a product working and a day ruined.
Why Clear Formulas Matter
People working in production or academic research both rely on formulas for more than paperwork. Regulatory agencies like OSHA and the EPA use these codes to guide best practices. The wrong compound could break a legal standard or pollute a waterway. C10H4Cl2O2 describes a specific chemical whose uses in dyes, intermediates, and sometimes electronics all depend on knowing those chlorine atoms are precisely where you expect.
Risks and Safety in Industry
These compounds can present risks beyond just the scientific world. Some chlorinated organics persist in the environment. They may bioaccumulate—ending up far from the site where they got loose. A plant operator who doesn’t double-check a formula might think they’re working with one thing, but only catch their mistake after something escapes containment. The formula isn’t just metadata; it’s a line of defense against error.
Supporting E-E-A-T: Skills and Trust
A researcher or technician gains trust by handling these details. Companies building transparent sourcing and data management need crystal clarity at every step, from purchase orders right through disposal. This means not just memorizing a formula, but treating those details as essential to process safety and product quality. Citations from bodies like PubChem and material safety data sheets all anchor back to having the right code. The reliability of this information forms the backbone of science-based industries.
Potential Solutions for Industry Challenges
Chemicals like 2,3-Dichloro-1,4-Naphthoquinone push companies to invest in sharper labeling, digital inventory, and even artificial intelligence checks on procurement systems. Training programs need to reflect modern needs: not just textbook recitation, but working under pressure, recognizing hazards, and clarifying documentation. Using QR codes and digital tracking, staff can pull up a compound’s formula and properties on a smartphone—no more squinting at a faded label, guessing between similar names.
Looking Ahead
Mistakes with chemical formulas can mean lost trust, sunk costs, and in the worst cases, hurt people or the planet. In my own work, keeping an eye on the details—especially the ones that look like mere routine—often reveals the biggest breakthroughs or the tightest saves. The formula for 2,3-Dichloro-1,4-Naphthoquinone is much more than a jumble of letters and numbers. It stands at the intersection of science, trade, and safety, shaping everything from new technology to environmental policy.
What are the primary applications of 2,3-Dichloro-1,4-Naphthoquinone?
Serious Chemistry in Action
Ask anyone who’s mixed chemicals for a living—few compounds offer as much range as 2,3-Dichloro-1,4-Naphthoquinone. You don’t see it on kitchen counters or hardware store shelves, but this molecule powers some core industries. Research labs often jump straight to the next hot material, but time and again, specialists reach for quinones when tough jobs demand tough chemistry.
Dyes and Pigments: Color the World
Textiles need stable color. Print inks fade if they’re not made with the right backbone. Makers count on naphthoquinone derivatives like this one to anchor dye and pigment structures. That’s not marketing fluff—decades of work show that it resists bleaching, heat, and most of what the weather throws at it. Manufacturers have found that this compound helps shape strong, vivid colors that don’t wash out or break down after a few laundries.
I remember working one summer in a textile testing lab. Samples that packed this molecule kept their reds, blues, and specialty shades even after hot water and bright sun. It shaped reliable color fastness at a time when brands wanted fewer returns from unhappy customers. Factories move fast. No one wants batches of shirts or drapes fading halfway through a season. This compound serves as the base for building those long-lasting shades that everyone counts on.
Medicinal Chemistry: Quiet Support Behind the Scenes
Medicines grow more complicated every year. Drug discovery teams appreciate a good building block when designing molecules that matter. Many antibiotics and antifungals trace their roots to naphthoquinone frameworks. You won’t see the compound itself in a pharmacy, but chemists leverage it for tailored compounds. Some studies highlight its ability to help construct new treatments, thanks to the chlorine atoms that open up reactive sites for fine-tuning.
Reliable source materials inspire innovation. In university research, grad students used this compound for a stepping stone to novel anti-cancer leads. Team meetings circled back to it because the core structure is sturdy—you can bolt on different chemical groups, explore unique activity, and keep iterating.
Agriculture and Industry: More Than Lab Curiosity
Fungicides and crop-protection agents pull from this naphthoquinone. Farmers want products that keep mold and blight at bay, especially in humid regions. The molecule’s reactivity lets industry chemists modify it quickly for new threats. Strong resistance to environmental breakdown makes it a backbone for products that don’t give up early in wet fields.
Beyond the farm, makers of specialty resins, adhesives, and plastics value this compound for its cross-linking potential. By promoting tighter bonds, it improves the lifespan and toughness of finished goods. Automotive and electronics applications depend on materials that won’t crack or corrode after one season.
Pushing for Smarter Solutions
Chemistry brings responsibility. While 2,3-Dichloro-1,4-Naphthoquinone builds useful stuff, it’s not risk-free. Handling calls for strong protocols in good ventilation, gloves, and responsible disposal. Reports point out the importance of keeping it out of waterways, because even trace residues disrupt aquatic life.
The global push for greener chemistry drives research toward safer derivatives. There’s momentum to reduce persistent chlorine atoms and switch to lighter-touch synthesis. Companies pursuing certifications like ISO 14001 examine the entire lifecycle of such ingredients, so the future for this compound involves both innovation and care.
Wrap-up
2,3-Dichloro-1,4-Naphthoquinone doesn’t grab headlines, but it fuels color, medicine, fields, and sturdy materials most people touch every day. Real expertise—tested in labs and factories—keeps finding ways to use this molecule where it matters, while making sure safety isn’t left behind.
What are the storage and handling precautions for 2,3-Dichloro-1,4-Naphthoquinone?
Why Safety Around Chemicals Matters
In any lab or industrial setting, a splash or spill isn’t just a hassle—it can change someone’s life. I've spent my share of hours in the lab, and every chemical takes on its own form of risk. With 2,3-Dichloro-1,4-Naphthoquinone, there’s no place for shortcuts. This compound carries health hazards that require respect and focus. The right choices in storage and handling can keep everyone safer, right down to the folks who clean up at the end of the day.
Fact: This Isn’t Your Everyday Household Cleaner
Let’s start with one raw fact: 2,3-Dichloro-1,4-Naphthoquinone doesn’t belong on open benches or next to food prep. The material safety data sheets don’t mince words. The chemical can irritate skin, eyes, and lungs. Eye contact may cause burns, and even a small inhalation can set off lasting respiratory trouble. A lot of us have seen quick mistakes lead to a race for the eyewash station. Containment, labeling, and personal gear make all the difference.
Protective Equipment Is a Non-Negotiable
In my own experience, skipping gloves or goggles has never been worth the gamble. Nitrile or neoprene gloves work best, and eye protection—goggles, not just safety glasses—should always go on before a container comes out. Lab coats, closed shoes, and, for certain tasks, respirators form the defensive line. Nobody needs a reminder after an accident, so make it a routine every single time.
Containment and Storage Choices
Don’t keep 2,3-Dichloro-1,4-Naphthoquinone anywhere humid or warm. The substance is more stable when it stays cool, dry, and shielded from sunlight. Glass containers with tightly fitting lids work best. I’ve lost count of times I've opened a cabinet and found poor labeling—every label should include the full name, date received, and hazard symbols. Sometimes it feels tedious, but it pays off if an emergency happens.
It’s easy to overlook secondary containment, but trays or dedicated bins spot small leaks before things spread. If you've got shelves, set chemicals like this one below shoulder height. No one wants to fumble with a slippery glass bottle over their head.
Prepping for Spills and the Unexpected
A lab without a spill kit is a risk nobody needs. For 2,3-Dichloro-1,4-Naphthoquinone, the right kit includes absorbent pads, inert powders, and sealable waste bags. Everyone who enters the work area should know the plan. Designated disposal containers keep hazardous waste out of the regular trash. I’ve seen people try to “just wash it down”—that doesn’t end well for anyone or the environment. Specialist chemical disposal services, or well-trained waste management staff, keep the risk contained.
Building Good Habits and Team Knowledge
Regulations like OSHA and local environmental rules aren’t just boxes to check. Training and drills sharpen everyone’s focus and speed. Consistent review of procedures helps the team stay ready, and open conversations after near-misses or mistakes build a stronger safety culture. I’ve learned more from coworker stories and shared lessons than from any manual.
A careful, consistent approach pays off. Chemical injuries and exposures usually trace back to lapses in storage, sloppy labeling, or ignoring routine gear. Keeping 2,3-Dichloro-1,4-Naphthoquinone secure and using the right equipment matters because everyone deserves to finish their day as healthy as they started.
Is 2,3-Dichloro-1,4-Naphthoquinone hazardous or toxic?
What’s the Real Risk?
2,3-Dichloro-1,4-naphthoquinone doesn’t sound like something you’d sprinkle on your breakfast. This is a chemical that shows up in the world of dyes, certain chemical synthesis, and research. Most folks will never handle it unless their work involves lab coats and goggles. But for those who do, the question isn’t just academic. Real risk comes into play.
Direct Contact and Health Effects
From personal experience in the chemistry lab, gloves and eye protection never seemed optional. Chemicals like this carry burn risk and can mess with your lungs or skin if spilled or inhaled. According to the European Chemicals Agency, skin contact can cause irritation, redness, or worse: damage that sticks around. The dust or vapor messes with breathing, and lab-based exposure brings headaches and dizziness. There are accounts in the literature warning about strong irritation and toxicity at both acute and chronic exposure. Few people outside chemical research circles know how serious these risks can get; extended exposure might even lead to organ issues.
Environmental Spill-Over
No one wants mystery chemicals in the water supply. Waste from factories, or accidental spills, turn into long-term headaches for communities. 2,3-Dichloro-1,4-naphthoquinone doesn’t just break down and vanish—some reports say it can stick around in soil and water. Persistent chemicals threaten fish, invertebrates, and even plants. We’ve all seen how one overlooked compound can ripple through whole ecosystems. Think back to public debates about pesticides or industrial waste; this situation carries echoes of those lessons.
Regulation and Worker Safety
Safety data sheets and regulatory agencies demand real attention to control. In my grad lab, the rules weren’t bureaucracy—they meant preventing chemical burns, rashes, or even fires. The International Agency for Research on Cancer keeps a long list of potentially cancer-causing agents, and while 2,3-Dichloro-1,4-naphthoquinone hasn’t been flagged as a known human carcinogen, it sits on the radar for further study. Occupational exposure limits can be tight. Employers and labs keep strict logs and waste protocols, not just to check off boxes but to avoid emergency room visits and legal fallout.
Transparency and Community Right-To-Know
Nobody deserves to be in the dark about what’s handled around them. I remember being part of a campus push for full disclosure on chemical inventories. Communities near chemical plants ask tough questions. If 2,3-Dichloro-1,4-naphthoquinone floats through the air or drains toward town, those neighbors deserve answers. Honesty here builds trust. Researchers and engineers won’t catch everything, but consistent reporting, safety drills, and community notification offer peace of mind.
Responsible Use and Alternatives
Some companies already look for safer alternatives or green chemistry approaches. Replacement, where possible, beats risky clean-ups later. Risk doesn’t vanish overnight, but ongoing research into substitutes marks a sign of progress. Those in the chemical field owe it to workers and neighbors to push for innovation that reduces these hazards.
Real-World Approach
No chemical works in isolation from the people and world around it. Rules, research, education, and respect for risk make all the difference. Chemicals like 2,3-Dichloro-1,4-naphthoquinone present hazards worth taking seriously—not just in fine print, but in every decision from lab bench to regulatory office. The goal isn’t to avoid all risk, but to weigh it honestly and act. That kind of responsibility keeps people safe.
What is the purity and available packaging size of 2,3-Dichloro-1,4-Naphthoquinone?
Understanding Purity
Chemicals like 2,3-Dichloro-1,4-Naphthoquinone invite plenty of questions from researchers, buyers, and engineers. Purity stays at the top of that list. In simple language, purity says how much of the bottle contains the real deal versus extras you didn’t sign up for. In my lab days, chasing after that “98%” stamp on a label wasn’t just about ticking a box on a form—it made downstream results more reliable. Labs expect most commercial batches of this compound to clock in at around 97% to 99% purity, sometimes nudging a bit higher for demanding applications, like analytical chemistry or synthesis of pharmaceutical intermediates.
I once worked with a supplier who offered a spec of 98% min. That number didn’t just look good; it led to smoother QC checks and fewer headaches during reactions, especially when side-products caused reactions to behave unpredictably. If you spot anything claiming 99%+ purity, you’re in premium territory—price follows, but so does performance.
Markets often offer technical grade, lab grade, and “high purity” options. High purity isn’t a luxury for chemical manufacturing; for pharma, those extra decimal points in purity can mean the difference between reprocessing a failed batch or getting to scale-up.
Packaging Sizes That Labs and Plants Actually Use
Small research setups hunt for packages starting at 5 grams. Scaling up, you stumble onto 25-gram bottles, then 100, then 250 grams. Industrial plants looking to churn out hundreds of kilos of intermediate or dye will want kilograms, sometimes arranged in 1 kg packs, 5 kg drums, and, for the big players, 25 kg fiber drums—resistant to moisture and usually fitted with liners for safety.
Packaging depends on storage needs, risk management, and, above all, safety regulation. At a bench scale, a 10-gram amber glass bottle protects the chemical from light and slows degradation. Larger operations move to robust HDPE drums or steel containers, so no one’s caught off-guard mid-project due to spillage or contamination. I’ve seen what cheap containers do—flaky seals, powder everywhere, waste, and extra cleaning. A good package is quiet insurance; you notice it most when it’s missing.
Why Purity and Package Size Should Matter
Getting shortchanged on purity translates to unpredictable chemistry and skewed analytics. That lands squarely in lost time, wasted money, and sometimes regulatory non-compliance. On the packaging side, mismatched sizes add hassle—either paying for what you never use, or scrambling for refills in the middle of a synthesis.
Knowledge here comes from hard-won experience. I learned to always ask for documentation: certificate of analysis, batch traceability, and shelf life details. Trusted suppliers don’t hesitate to send this over, backing up their purity claims. If they’re cagey or send generic specs, that’s a red flag right there.
Room for Improvement
To cut confusion, suppliers should give full transparency—batch numbers, expiry dates, impurity profiles, and clear packaging options. That makes researchers’ and manufacturers’ jobs easier. Labs can push for less-waste options: recyclable containers, right-sized packages, and expanded lot-tracing. Customers and suppliers can talk more to match supply with real-world demand, so nobody’s left storing drums they’ll never open—or dealing with a bottleneck due to under-ordering.
The chemistry field gets stronger when sourcing and documentation don’t add extra roadblocks. Purity and packaging may sound like background details, yet, in practice, they determine whether a project flows smoothly, meets its specs, and delivers safe results.
| Names | |
| Preferred IUPAC name | 2,3-dichloronaphthalene-1,4-dione |
| Other names |
2,3-Dichloronaphthalene-1,4-dione Dichloro Lawsone 2,3-Dichloro-1,4-naphthalenedione |
| Pronunciation | /tuː, θriː-daɪˈklɔːroʊ wʌn, fɔːr-næfˈθoʊkwɪˌnoʊn/ |
| Identifiers | |
| CAS Number | 84-47-9 |
| Beilstein Reference | 1859226 |
| ChEBI | CHEBI:81893 |
| ChEMBL | CHEMBL31922 |
| ChemSpider | 154256 |
| DrugBank | DB12936 |
| ECHA InfoCard | 03b601e5-44ba-491e-b8cd-767f5de1c822 |
| EC Number | 215-700-5 |
| Gmelin Reference | 35363 |
| KEGG | C10555 |
| MeSH | D017859 |
| PubChem CID | 69229 |
| RTECS number | QJ6300000 |
| UNII | 4Q696768WP |
| UN number | UN3341 |
| CompTox Dashboard (EPA) | DTXSID4059882 |
| Properties | |
| Chemical formula | C10H4Cl2O2 |
| Molar mass | 227.03 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.58 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.00018 mmHg (25 °C) |
| Acidity (pKa) | 3.9 |
| Magnetic susceptibility (χ) | -90.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.677 |
| Viscosity | 3.8 mPa·s (at 25 °C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -99.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -741.3 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P273, P280, P302+P352, P305+P351+P338, P332+P313, P337+P313, P362 |
| Flash point | Flash point: 138°C |
| Autoignition temperature | 190 °C |
| Lethal dose or concentration | LD50 oral rat 610 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 500 mg/kg |
| NIOSH | WX8575000 |
| PEL (Permissible) | 0.1 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |

