2,3,5-Trichloropyridine: From Discovery to Future Prospects
Historical Development
Scientists set out searching for new chlorinated pyridines in the early twentieth century. They knew that small changes in molecular structure turn an average pyridine into a star player for industry and pharmaceuticals. 2,3,5-Trichloropyridine emerged out of that curiosity, first reported in organic chemistry literature in the mid-20th century, as researchers chased after more selective herbicides and unique building blocks for chemical synthesis. It didn’t take long for experts to spot this compound's advantages over its siblings—chlorine atoms on the pyridine ring push the properties of the molecule into new territory, making it interesting for chemical manufacturing as well as academic research. As demand for specialty chemicals ramped up through the decades, the processes for making 2,3,5-Trichloropyridine kept evolving, with environmental and safety pressures spurring continuous tweaks to both method and scale.
Product Overview
2,3,5-Trichloropyridine earned its keep thanks to three chlorine atoms perched on a six-membered nitrogen ring. That structure turned it into a highly sought-after intermediate for pharmaceuticals, agrochemicals, and other specialty compounds. You rarely find this substance as an end-use product on its own. Chemists pull it off the shelf mainly as a reactive building block. Its popularity grew because it unlocks synthetic routes to other complex molecules, especially for companies driving toward patentable drugs or new classes of crop-protection agents. In my own work with organic synthesis, I found its ease of handling compared to more volatile or reactive chlorinated aromatics particularly useful in scaling up reactions.
Physical & Chemical Properties
2,3,5-Trichloropyridine shows up as an off-white or slightly yellow crystalline solid, carrying a faint but distinct aromatic odor. It clocks a melting point around 52–56°C and a boiling point close to 247–249°C. With a molecular formula of C5H2Cl3N and a molecular weight near 200.44 g/mol, the density sits comfortably at roughly 1.5 g/cm³. It dissolves well in many organic solvents such as acetone, dichloromethane, and benzene, but don’t expect miracles with water—solubility there remains low. The chlorine atoms, anchored at the 2, 3, and 5 positions, pull electron density around the pyridine ring, which shifts its reactivity, making nucleophilic substitution a favored pathway. This chemical stability mixed with controlled reactivity becomes a large part of why manufacturers reach for it during process development.
Technical Specifications & Labeling
Suppliers put strict specs on 2,3,5-Trichloropyridine because any trace contaminants can torpedo downstream processes. Purity levels over 98% tend to be standard expectation, sometimes pushed to 99% for pharmaceutical grade batches. Common impurities might include lower-chlorinated pyridines, inorganic salts, or even starting materials that dodge complete conversion during synthesis. Tech sheets call out melting point, residual solvents, water content by Karl Fischer, and sometimes assay by high-performance liquid chromatography. Containers wear hazard symbols for acute toxicity and environmental risk, tied to global safety standards like GHS and REACH. Each label lists batch information and recommended protective measures, helping end-users trace back supply and keep labs in regulatory alignment.
Preparation Method
Production of 2,3,5-Trichloropyridine leans heavily on direct chlorination routes starting from parent pyridine, using chlorine gas under controlled temperature and pressure. Manufacturers typically use catalysts or tailored conditions to direct chlorination to the 2, 3, and 5 positions, suppressing formation of unwanted isomers. In my time on a process development team, we found batch-wise operations easier to control than continuous ones at the pilot scale because heat management around exothermic steps as well as quenching unreacted chlorine demanded close attention. Reaction mixtures get quenched, organic layers separated, and crude product purified through distillation or crystallization. Waste gas scrubbing and efficient ventilation take top priority, given the hazardous nature of chlorine both in raw materials and as byproduct.
Chemical Reactions & Modifications
What makes this compound special isn’t just its own chemistry but what you can do with it. Nucleophiles such as amines or thiols attack selectively at the positions activated by chlorine atoms, opening up routes to a host of new pyridine derivatives. Chemists often harness those substitutions in making intermediates for crop-protection agents, dyes, or pharmaceuticals. For example, a simple substitution with methylamine kicks off the synthesis of antiviral candidates or herbicide scaffolds. In a research setting, halogen-metal exchange reactions using Grignard or lithium reagents unlock further substitutions, letting scientists patch in new groups with surgical precision. Broad reactivity brings plenty of opportunity, but it demands respect for safety, as even minor slips during modification reactions can mean runaway exotherms or emission of hazardous vapors.
Synonyms & Product Names
Nobody likes confusion over the name of a chemical, especially with strict supply chain requirements. You’ll most commonly hear 2,3,5-Trichloropyridine, but catalogs and commercial shipments may list it as TCPY, Trichloropyridine, or sometimes by chemical registry identifiers like 13349-76-5. Language barriers can cause bumps when sourcing raw materials overseas—one reason most labs keep the CAS number close during procurement. A few companies sell it under proprietary labels, tacking on suffixes or trade names depending on target industry. No matter what company logos decorate the drum, the active ingredient’s chemical footprint stays unchanged.
Safety & Operational Standards
2,3,5-Trichloropyridine does not belong anywhere near casual handling. Exposure risks run from skin and eye irritation to more severe respiratory impacts. Gloves, goggles, and chemical hoods form the frontline barrier in any serious lab or manufacturing site. Spills get contained with inert absorbents, and any area touched by vapor or dust receives prompt clean-up. Regulatory bodies classify this chemical as hazardous to aquatic life, prompting strict protocols on storage, waste treatment, and transport. Regular audit trails and safety drills make sure everyone remembers the stakes—having witnessed one minor leak in a former role, I saw firsthand how fast a routine shift can spiral into an emergency without rigorous controls in place. Keeping safety data sheets up to date and retraining regularly earned their place in any operation working with trichloropyridines.
Application Area
Few products outside the pharmaceutical and agrochemical pipeline benefit from the flexible chemistry built on 2,3,5-Trichloropyridine. It anchors synthesis for fungicides, insecticides, and herbicides that ultimately boost crop yields across the agricultural sector. Drug discovery teams gravitate to this building block for its role in anti-infective and anti-inflammatory agent development. In a manufacturing setting, engineers use it to introduce specific functionalities along a synthetic route, confident that each substitution steps closer to the finished product with reduced byproduct headaches. The dye and pigment industry also picks it up when aiming for particular shades and stabilities in color formulations, showing the reach of a well-designed pyridine scaffold. Working alongside both biotech and chemical engineers, I saw how each team tuned their procedures around this compound’s unique toolbox of properties.
Research & Development
Academic and industrial labs haven’t stopped tinkering with how they use 2,3,5-Trichloropyridine. Green chemistry pushed teams to shorten synthetic steps, cut down on solvent usage, or swap hazardous reactants for safer alternatives. Catalysis research aims to dial in regioselectivity for new substitution patterns, often with the hope of granting faster market access for pharma or crop-protection products. I’ve read about groups adding flow chemistry methods or microwave-assisted synthesis to their toolkit, slashing reaction times or bumping yields using technology still catching up in the mainstream batch world. Data-driven process optimization—leaning on machine learning or automated reaction sampling—leads operations out of the era of trial and error. Patents keep rolling in covering new applications, and competition to drive down production costs hasn’t let up either.
Toxicity Research
Chlorinated pyridines—including 2,3,5-Trichloropyridine—don't get a free pass in toxicology. Animal studies report moderate to acute toxicity when inhaled or ingested. Bioaccumulation in aquatic systems comes up in environmental hazard reviews. Repeat exposure, particularly through inhalation, produces adverse effects on the respiratory system, and potentially the central nervous system. Ongoing research probes how breakdown products move in soil and water, so the industry can tighten up waste handling and effluent controls. Medical researchers also scrutinize how the compound interacts with liver enzymes and DNA, keeping safety barriers high. My own experience with hazardous chemicals persuaded me long ago to champion stricter risk assessments and shield on-site staff with high-grade personal protective equipment and medical surveillance whenever needed.
Future Prospects
Rising global demand for more effective and sustainable agrochemicals keeps 2,3,5-Trichloropyridine firmly on the radar of chemical manufacturers. More sustainable methods—like catalytic direct chlorination using greener oxidizing agents—aim to cut waste and energy use. In pharma, builders keep looking for ways to stitch together complex drug molecules faster, often capitalizing on the reactivity and selectivity built into chlorinated pyridine rings. Regulatory frameworks grow sharper each year, pushing for full life-cycle analysis and lower toxicity profiles, nudging producers toward cleaner, safer processes. Looking ahead, integration of recycling, closed-loop waste treatment, and real-time monitoring could set a new gold standard for handling compounds in this class. For young chemists and engineers, learning to work with these versatile intermediates safely opens doors to innovation across both science and industry.
What are the main applications of 2,3,5-Trichloropyridine?
The Quiet Player in Chemical Synthesis
Every now and then, a chemical compound works its way into several everyday products without anyone noticing. That's what happens with 2,3,5-Trichloropyridine. Many people outside the chemistry world hardly know it, yet it quietly helps produce things we all rely on. This compound isn't flashy, but it sure gets the job done.
Paving the Way for Pharmaceuticals
I once worked in a lab that developed molecules for new drugs. A team member showed me how 2,3,5-Trichloropyridine became a starter block for important ingredients in therapies. Scientists value its well-placed chlorine atoms, since these help craft complex molecules found in antihistamines, antiviral drugs, and some experimental cancer treatments. Its unique structure lets researchers add or swap out chemical groups easily, so they can build pharmaceuticals faster, spending less time wrestling with dead ends in synthesis.
Crop Protection and Modern Agriculture
My uncle runs a farm, and he always keeps an eye out for better solutions to deal with pests. Agrochemical companies push the boundaries here, and 2,3,5-Trichloropyridine drives that progress. It acts as a crucial link in the production of certain herbicides and insecticides. What stood out to me in conversations with agri-specialists was how targeting specific weeds or insects gets easier with compounds built on this chemical’s backbone. That helps farmers see less waste, improve yields, and use less active ingredient in the field—reducing some of the strain our planet faces from overused chemicals.
Dyes, Pigments, and Specialty Materials
Color matters. Factories making bright, lasting dyes often use 2,3,5-Trichloropyridine to anchor chlorine groups onto pigment molecules. If you’ve ever bought a piece of clothing and the color stuck around after months of washing, there’s a decent chance this chemistry played a role. In textile plants I’ve visited, operators count on chemicals like this to keep colors from fading, especially for tough workwear and outdoor fabrics.
The Challenge of Safe Handling and Environmental Concerns
Every chemical worth its salt brings some baggage. Friends working in environmental health stress getting safer handling and disposal protocols for 2,3,5-Trichloropyridine, since traces can persist in soil and water if not contained. Regulators and responsible manufacturers follow strict rules—constant air and water monitoring, closed systems for synthesis, and investment in better waste cleanup. Still, small producers sometimes cut corners, so clear guidance and stronger inspections always help push things in the right direction.
Real-World Opportunities and Alternatives
Companies around the world look for greener ways to work with halogenated pyridines. I’ve seen academic groups try to redesign certain processes to use less chlorine or find biodegradable substitutes, though progress feels slow at times. Open conversations between chemists, farmers, and policymakers build trust and focus attention on innovation. That’s how smarter materials and medicines grow out of something as unassuming as 2,3,5-Trichloropyridine.
What is the chemical formula and molecular weight of 2,3,5-Trichloropyridine?
Breaking Down the Chemistry
Chemistry classes typically throw around chemical formulas as mere codes. Really, these codes pack in the entire structure and reality of a substance. Take 2,3,5-Trichloropyridine. The chemical formula is C5H2Cl3N. Looking at that, you spot five carbon atoms, two hydrogens, three chlorines, and a nitrogen—no fillers, no hidden ingredients. The molecular weight, a number that doesn’t change, lands at 198.44 grams per mole. These figures seem trivial to folks outside the lab, but this core info keeps researchers and product developers from running into trouble.
Beyond the Numbers: Why the Formula Guides More Than Just Math
As someone who has spent hours puzzling through bottles of chemicals, I know how much trust lands on clear formula notation. Picture a manufacturer considering new pesticides or pharmaceuticals. Just a single extra atom in the formula leads to surprises—maybe toxicity where you don’t expect it, or a process failure. That is exactly why correct and consistent chemical notation does more than fill a spot in a spreadsheet. It lays out the rules for safety sheets, storage protocols, and shipping details. Every regulatory agency from OSHA to REACH works from these basics. If your numbers are off, you risk fines, health issues, or the infamous product recall. That reputation damage doesn’t wash away quickly.
Weight: Not Just a Stat
Molecular weight, which clocks in at 198.44 g/mol for 2,3,5-Trichloropyridine, doesn’t just end up in an equation. Chemists in the pharmaceutical and materials industries calculate dosages, mixing ratios, and handling limits. Miss the correct weight, and suddenly that drug batch sits out of range, or that polymer experiment breaks down. It’s easy to assume weighing precision belongs to science fiction labs, but teaching labs at universities and small startups work just as carefully. They don’t get the luxury of a do-over if storage tanks or mixing lines go awry. Not to mention, accurate weights allow for proper disposal—important for any industry with hazardous byproducts.
Real Risks of Getting It Wrong
In manufacturing, a slip in notation or calculation easily snowballs into larger safety issues. For instance, 2,3,5-Trichloropyridine, with its chlorine groups, usually signals greater reactivity and toxicity than a plain pyridine ring. Reading the formula keeps operators aware of what kind of gear and containment to prepare. Even a bench chemist with years in the field checks and double-checks formulae because regulatory inspections don’t let anyone off the hook for mistakes. Safety data sheets, emergency spill protocols, and labeling on shipping containers all track back to the right numbers and weights being there.
A Way Forward for Safer Handling
Industry still wrestles with mislabeling, old paperwork, or lazy shortcuts when updating chemical logs. Upgrading digital systems that tie raw data to shipping labels and production forms offers a clear chance to cut mistakes. Newer barcoding and lot-tracing help too, but nothing replaces training for the folks actually handling these materials. It’s not enough to dump the chemical in a database—the people managing it need to understand why each letter and number matters. Consistency saves time and money, but above all, it protects lives in the lab or on the shop floor.
What are the handling and storage precautions for 2,3,5-Trichloropyridine?
Choosing the Right Storage Conditions
2,3,5-Trichloropyridine isn’t your typical household chemical. Over time, I’ve seen how careless storage habits lead to real headaches—corrosion, leaks, and exposures that put health and the environment in harm’s way. This compound, with its low water solubility and resilience against light, still calls for respect at every step. Secure a cool, dry spot, well away from heat sources and out of direct sunlight. Good ventilation prevents vapor build-up, so air-circulating storage rooms do a better job than cramped cabinets. Forget about storing food or drink in the same area; contamination risks never pay off.
Dedicated chemical storage cabinets, built from resistant materials, help contain spills. We’re talking about containment trays or seamless shelving. If a drum leaks, catchment basins or spill pallets limit the escape. The fewer people with access, the better—only folks trained in hazardous materials should grab the keys to these cabinets.
Proper Handling Reduces Risks
Thick gloves, lab coats, eye protection, and chemical-resistant shoes become the unofficial uniform when working with 2,3,5-Trichloropyridine. Fumes from this compound may sting the nose and eyes even with a quick splash, so anyone pouring or measuring should use fume hoods or local exhaust systems. Wearing respiratory protection cuts down on accidental inhalation, and always, always wash up afterwards. Don’t trust bare hands, even for quick adjustments; skin absorption remains a real hazard.
Never pipette or siphon by mouth—a surprising number of accidents come from old habits. Label every container, no matter the size or temporary nature. In busy labs or warehouses, I’ve watched mix-ups happen far too fast. If storage containers aren’t compatible, the odds of corrosion or catastrophic leaks shoot up. Choose containers built for halogenated compounds to keep trouble at bay.
Minimizing Spills and Environmental Damage
Cleanups start before spills happen. Absorbents, neutralizers, and emergency showers need to live close by. Once, in a facility I worked at, a slow leak under a shelf turned into a slick—hazmat responders spent hours making the site safe again. Training teams on rapid response and keeping a clear line to emergency contacts turns confusion into quick control. Waste never belongs down the drain or in regular bins. Instead, dedicated disposal containers, collected by licensed handlers, keep harmful compounds out of landfills and waterways.
Putting Safety into Practice
Regular inspections spot leaks and aging containers before they become stories for the news. Some companies keep logs to track product age and condition; small investments like these save money and headaches. Technology helps: leak alarms, inventory systems, and access controls tighten up the process. But nothing replaces a culture of vigilance, where speaking up about slippery floors or failing seals is encouraged.
Strong rules backed by habits give 2,3,5-Trichloropyridine a place in industry without inviting disaster. Every worker, from new techs to senior chemists, learns the same steps. Simple diligence cages a dangerous chemical, saving both health and the company bottom line.
Is 2,3,5-Trichloropyridine hazardous or toxic?
Getting to Know the Chemical
2,3,5-Trichloropyridine shows up in the world of specialty chemicals and pharmaceuticals. Folks use it for synthesizing advanced molecules, not something you come across on a grocery store shelf. The compound carries a bitter, sharp odor. Anyone who's spent time in a chemical lab knows that sharp smell often comes with a warning—use care. Just because it helps in the lab doesn’t mean it’s harmless in daily life.
Human Health Concerns
Take spills and exposure seriously. Chemists working with this material wear gloves and goggles for good reason. According to PubChem and the European Chemicals Agency, extended or repeated exposure can irritate the skin, eyes, and respiratory system. Swallowing just a small amount might cause headaches, nausea, or abdominal pain. There isn't much long-term human data, but animal studies paint a worrying picture—liver problems and damage to organs started showing up in some lab results.
Many organic chlorinated compounds stubbornly cling to tissues in living things. Trichloropyridine might not act as rapidly as acutely toxic substances like cyanide, but building up over time can sneak up on people working with it day in, day out. Symptoms rarely flash up immediately, which makes things more dangerous if workers skip personal protective equipment or ventilation. Ignoring small amounts of exposure can add up, sometimes painfully.
Environmental Hazards
Chemicals like 2,3,5-Trichloropyridine rarely stay put. Factories using it risk leaking waste into water or soil, making a mess for local ecology. Studies show that related pyridine compounds stick around in groundwater and break down at a glacial pace. Fish and plants nearby take the brunt—routine wastewater checks often turn up harmful residues if companies slack on disposal routines.
The potential for bioaccumulation makes things hairy for local wildlife. A simple rainstorm after a spill sends toxins downstream, affecting more than just one corner of the factory. Every wildlife professional knows the headaches that come from persistent chemical pollution: fish kills, dead plant zones, even toxins in drinking water if municipal treatment misses these tricky molecules.
Solutions and Ways Forward
Smart facilities put safety up front. In my own work, running a well-ventilated hood with good personal protective gear always tops the protocol. Proper storage matters—lock up trichloropyridine in sturdy containers away from heat or open flames. Facilities that take health seriously post clear hazard labels, train employees on first aid for spills, and never cut corners on masks or gloves.
Disposal matters even more. Don’t pour waste down the drain, even if it seems diluted. Incinerate with licensed professionals or ship hazardous waste to certified treatment plants. Environmental regulations give a framework, but it’s up to company cultures to keep every shift safe. Authorities like OSHA and REACH don’t hand out warnings for fun. Years in industrial labs taught me that the “it’ll be fine” attitude leads to headaches—sometimes far worse.
Looking Ahead
Learning about the hazards connected with 2,3,5-Trichloropyridine should push companies and researchers to respect it. Worker safety programs, ongoing environmental testing, and respect for chemical risks set apart the best operations from the rest. Any progress against chemical exposure starts with direct action, clear rules, good training, and strong habits—habits that keep people healthy well after the workday ends.
What is the typical purity and packaging of 2,3,5-Trichloropyridine offered?
Packing Up Purity in the Chemical World
In every lab I’ve ever worked, purity drives everything. Chemists reach for reagents like 2,3,5-Trichloropyridine expecting tight specifications, because anything less just wastes time and twists results. Most manufacturers deliver this compound with purities above 98%, often tipping closer to 99%. They don’t do this out of courtesy. Years of feedback taught suppliers where corners lead to trouble—whether it's wasting catalysts, gumming up synthesis, or costing a chemist an entire week of planning.
Quality control teams lean on tools like gas chromatography and NMR to verify batches. When a drum or bottle shows up missing a COA or looking off-color, researchers notice. Purity differences show up right away: fine white to off-white powders tell buyers a supplier paid attention to every detail from the start of synthesis through final drying.
A Practical Look at Packaging
Nobody wants surprises when handling chemicals, especially something as persistent as 2,3,5-Trichloropyridine. Most suppliers choose to ship it in high-density polyethylene bottles, sealed tight and often double-bagged in foil. This keeps out water, oxygen, and dust in transit. I’ve seen what happens when those seals don’t hold up—clumping powders, odd smells, or worse, dangerous leaks. That’s why sturdy packaging matters.
For large volume buyers, steel drums with interior linings shield the contents from the world outside. Smaller amounts—250 grams to a kilo—usually land in rigid plastic jars. Labels should always shout out the batch number, weight, and hazard warnings in clear print. Missing information tells buyers to look elsewhere or test every detail before heading into synthesis. Poor labeling can turn a routine order into a guessing game.
Why Anyone Should Care About These Details
If impurities slip in unnoticed, projects veer off course. Research processes may stall over months chasing ghosts in a reaction flask. Purity isn’t just a number on a piece of paper; it’s a promise that experiments stand a solid chance of repeatable results. I saw a drug project cost an extra six months because analysts tracked back a problem to contaminated pyridine. Money lost, morale down, and trust chipped away all because one supplier dropped standards.
When packing lets moisture through or shipment boxes take too many knocks, an entire lot can become unusable. Chemicals like 2,3,5-Trichloropyridine don’t come cheap, and nobody enjoys tossing thousands of dollars in ruined material. Safe, reliable packing means researchers actually get what they ordered, ready for the next step.
Paths Toward Better Chemical Sourcing
Experienced buyers talk with their suppliers before any order is placed. They request certificates of analysis, sometimes a sample run, and always specifics about purity and packaging. Open communication pushes suppliers to maintain higher standards. Larger customers occasionally audit suppliers or ask them to sign quality assurance agreements. These steps reduce uncertainty and cut down on wasted effort.
Digital reporting, with QR codes printed on labels, now makes it easier to track lot histories and catch any recurring issues. It isn’t just about covering bases—tracking and vetting help everyone. Transparency reduces costly delays and bolsters safety across the board, something everyone in the field can appreciate.
Reliable purity and safe packaging in every shipment save labs time, resources, and frustration—it’s as simple as that.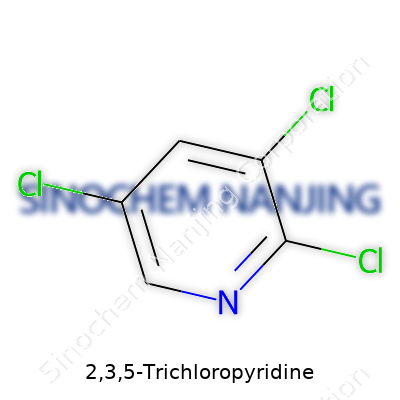
| Names | |
| Preferred IUPAC name | 2,3,5-Trichloropyridine |
| Other names |
2,3,5-Trichloropyridin 2,3,5-Trichloro-pyridine 2,3,5-Trichlorpyridin 2,3,5-Trichloorpyridine |
| Pronunciation | /ˌtuː,θriː,faɪvˌtraɪˌklɔːrəˈpɪrɪdiːn/ |
| Identifiers | |
| CAS Number | 24589-78-4 |
| Beilstein Reference | 120924 |
| ChEBI | CHEBI:86049 |
| ChEMBL | CHEMBL19393 |
| ChemSpider | 93387 |
| DrugBank | DB08335 |
| ECHA InfoCard | 03e1895e-b2a7-46db-8ef2-73a6e21d3ef4 |
| EC Number | 207-566-9 |
| Gmelin Reference | 8831 |
| KEGG | C14215 |
| MeSH | D015242 |
| PubChem CID | 69412 |
| RTECS number | UY9625000 |
| UNII | Q7XN4Q697M |
| UN number | 2810 |
| CompTox Dashboard (EPA) | DTXSID4058815 |
| Properties | |
| Chemical formula | C5H2Cl3N |
| Molar mass | 183.41 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Aromatic. |
| Density | 1.523 g/mL |
| Solubility in water | Slightly soluble |
| log P | 2.94 |
| Vapor pressure | 0.04 mmHg (25°C) |
| Acidity (pKa) | 3.72 |
| Basicity (pKb) | 6.24 |
| Magnetic susceptibility (χ) | -70.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.584 |
| Viscosity | 1.215 cP (20°C) |
| Dipole moment | 2.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –57.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1862 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P273, P301+P312, P305+P351+P338, P308+P313, P330, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-0-🛢️ |
| Flash point | 71 °C |
| Autoignition temperature | 530 °C |
| Lethal dose or concentration | LD50 oral rat 500 mg/kg |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (oral, rat) |
| NIOSH | SN2625000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | Not established |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
2,3,6-Trichloropyridine 2,3,4-Trichloropyridine 2,4,6-Trichloropyridine 2,3-Dichloropyridine 3,5-Dichloropyridine 2,5-Dichloropyridine 3-Chloropyridine 2-Chloropyridine |

