Editorial Commentary: The Story and Future of 2,3,4-Trimethylpentane
The Road From History to Utility
In the world of hydrocarbons, 2,3,4-Trimethylpentane holds a spot that pulls together chemistry, industry, and environmental discussions. Years back, as automotive technology took big steps forward, fuel engineers started dissecting what made gasoline “work.” Fuel knocking—once a roadside frustration—sent generations of chemists hunting for solutions. The story of iso-octanes, including 2,3,4-Trimethylpentane, takes root here. As scientists puzzled their way through hydrocarbon structures, they didn’t just stumble onto new molecules—they began to shape the very standards that define modern gasoline. This compound steps into the spotlight alongside its close cousin, the much-heralded 2,2,4-Trimethylpentane (iso-octane), both playing their part in raising octane ratings, cutting down on knock, and keeping engines running smoother.
Getting To Know 2,3,4-Trimethylpentane
Take a look at the clear, colorless liquid sloshing in a flask, and you see 2,3,4-Trimethylpentane for what it is: a hydrocarbon with the formula C8H18. Its structure shows three methyl branches sticking off a pentane backbone, making it one of many possible isomers in the octane lineup. In practice, it boils at around 116°C, with a density a bit lower than water. Pour it out and you’re reminded it evaporates quickly, smells like gasoline, and doesn’t mix with water—hallmarks for this category of molecules. It’s not flashy, but there’s a reason chemists keep going back to branched alkanes like this one when working on fuel innovation or separation studies. It stands up well in engine conditions due to its low tendency to pre-ignite, which shows up as a higher octane number on engine tests.
Labeling and Technical Details Matter
Clarity matters more than ever as chemical handling grows in scale and complexity. Regulations in North America, Europe, and Asia expect clarity in labeling—not only the name “2,3,4-Trimethylpentane,” but also its common synonyms, such as “iso-octane isomer.” Product data should show purity levels, but the reality on the ground is tighter: Labs need results to two or three decimal places when mixing reference fuels for standard tests. Safety labels warn about flammability, the risk of inhalation, and storage rules. Any slip in technical handling can put health or operations at risk, so good standards are not just a paper exercise.
How Chemistry Brings It To Life
Chemists often start with cracking large petroleum fractions and then separate out desired isomers using distillation and selective reactions, often enhanced by acid catalysis or molecular sieves. In many refineries, branched alkanes are coaxed into existence through isomerization processes. Reliable access to pure 2,3,4-Trimethylpentane depends on fine-tuned methods that separate out its isomers, with research teams zeroing in on process controls and catalysts that squeeze out a few more liters per run. Tiny impurities, lurking in the parts-per-million range, can skew results in fuel tests or degrade readings in research settings, making purity not just a preference, but a necessity for many users.
Chemical Reactions and Molecular Tweaks
The backbone of this molecule—an alkane—means it’s not easily knocked around in most chemical reactions. It resists oxidation and doesn’t open itself up to the kinds of reactions seen with double bonds or functional groups. That said, under harsh conditions—think high temperatures, UV light, or strong oxidants—it can break down into smaller fragments or form by-products. This stability gives it value for research, as well as for gasoline blending, where predictability makes for safe, reproducible results. Chemical companies sometimes modify it using controlled reactions to probe combustion patterns or environmental breakdown, usually in the context of laboratory studies or engine tests.
Alternate Names Don’t Always Help
Trying to navigate among chemical synonyms can feel like wandering in a forest of names. Besides its IUPAC designation, 2,3,4-Trimethylpentane may be lumped with the generic “octane” family or spotted in literature as a “C8 iso-alkane.” Each name has its home—regulatory documents, industrial catalogs, or research reports—but mismatched terms open the door to costly mix-ups. Anyone working in regulatory, supply chain, or lab operations needs an unambiguous path from label to substance.
Meeting Safety and Operational Benchmarks
Working with volatile hydrocarbons brings its own set of operational rules. 2,3,4-Trimethylpentane, no different from its peers, calls for strict controls in storage and lab settings. Safety protocols require ventilated spaces, flame-resistant gear, and vigilant handling to minimize fire risk. Spills need quick clean-up using absorbent materials and disposal methods that keep groundwater safe. In the last 15 years, industry regulators have tightened standards for chemical storage and tracking, nudged along by a rise in environmental disclosure laws. As someone who’s spent hours in research labs, it’s clear that checklists and preparedness drills are not red tape—they’re what keep people safe when routine becomes unpredictable.
Where It Finds Its Purpose
Of all the potential application areas, the gasoline industry stands out—2,3,4-Trimethylpentane blends into reference fuels that calibrate octane ratings, keeping engines quiet and efficient. Its role in research labs deepens as scientists use it to dissect combustion chemistry and emissions. Students, teachers, and industry pros alike come back to this molecule when illustrating hydrocarbon structure, volatility, and separation science. Over time, environmental regulations have nudged its use away from bulk fuel blending, but it remains front-and-center for calibration work, standards enforcement, and carefully controlled R&D.
Research Keeps Digging Deeper
Chemical research never stands still, and neither does the study of 2,3,4-Trimethylpentane. Scientists hunt for its combustion by-products, chasing clues about unburned hydrocarbons that escape from tailpipes. Analytical chemists probe how trace impurities affect readings in sensitive detectors or engine tests, while process chemists keep scanning for better purification methods. This research fuels debates about auto efficiency, urban air quality, and the broader push toward alternative fuels. Universities pair undergraduate learning with hands-on work involving this molecule, giving students a primer in real-world analysis, instrument calibration, and environmental chemistry.
What We Know About Toxicity
Every hydrocarbon brings health concerns, and 2,3,4-Trimethylpentane is no exception. The most pressing issues pop up with inhalation—you breathe in too much vapor and symptoms from dizziness to nausea can set in. Fire risk takes center stage in large handling operations, which is why fire control systems and leak detection routines can’t be shrugged off. Current toxicology points to low skin absorption compared to aromatic hydrocarbons, yet repeated exposure still carries risk for headaches and drowsiness. Regulators track workplace exposure closely, and research keeps digging for effects on the liver, kidneys, and nervous system, mostly using rodent models or in vitro tests. Long-term studies probe environmental impact, including its fate in soils and water, which inches into bigger questions about ecosystem health.
The Road Ahead
Looking forward, the role of 2,3,4-Trimethylpentane seems set to shift. As electric vehicles bite deeper into market share and cities clamp down on emissions, its volume use in traditional fuels will keep declining. Yet, its value in chemical research, standard reference applications, and process technology sticks around. Process innovation may yet bring greener ways to make alkanes, borrowing from bio-based feedstocks or integrated refinery schemes designed to lower the carbon footprint. Universities and governments shuffle research priorities every few years, but one thing holds true: learning from established hydrocarbons like this one can teach us plenty as energy systems evolve. All the hours I’ve spent in chem labs, swapping out reference standards or talking to engineers, have taught me that molecules like 2,3,4-Trimethylpentane do more than just fill a fuel tank—they spark the questions that keep science, industry, and safety always moving ahead.
What are the main uses of 2,3,4-Trimethylpentane?
A Closer Look at a Key Ingredient in Gasoline
2,3,4-Trimethylpentane, most people don’t recognize the name, but drivers everywhere feel its impact every time they fill up. This hydrocarbon blends right into the heart of modern fuel. What makes it so important? You look at the quality of gasoline and you’ll almost always find 2,3,4-Trimethylpentane contributing to a higher octane rating.
Octane rating shapes how engines handle heat and pressure. Fuels with plenty of 2,3,4-Trimethylpentane resist knocking, which means engines run smoother and more efficiently instead of coughing and rattling under stress. Coming from someone who’s pushed a car too hard once or twice, nothing feels better than knowing your fuel won’t let you down in the thick of city traffic or highway driving.
Cleaner Performance, Fewer Problems
Most drivers care about lasting engines. Fuel that keeps combustion under control protects engine parts from wear and expensive repairs. With 2,3,4-Trimethylpentane in the mix, gasoline does a better job at running cleaner, which means fewer buildups inside the engine. Years ago, mechanics used to see a lot more gunk clogging up valves and pistons; improvements in gasoline chemistry, with 2,3,4-Trimethylpentane right in the mix, changed that story.
Supporting Lower Emissions
Emissions targets get tougher every year. Every drop of fuel that burns cleaner means less pollution for everyone to breathe. Gasoline that handles pressure well stops incomplete combustion, which is one major pathway for carbon monoxide and hydrocarbons to end up in air. I’ve seen city air quality improve step by step as refining and fuel standards pull together, and better hydrocarbons help lead the way.
Versatility Beyond the Pump
Most folks see 2,3,4-Trimethylpentane as a fuel ingredient, though its uses can spill past that. Sometimes it works as a calibration standard in laboratories, especially those tied to research on fuel quality or pollution. I once visited an environmental testing facility where precise blends mattered; using this hydrocarbon as a reference standard let labs check the accuracy of their instruments faster and with more confidence.
Supply and Production Challenges
Relying on hydrocarbons means coping with refining limits and the ups and downs of crude oil markets. As gas needs change, fuel suppliers must juggle cost, efficiency, and environmental concerns. Refiners push to squeeze more of these valuable molecules from oil while keeping waste and emissions down—no small feat with older plants. Researchers and engineers work on new catalysts and cracking techniques, looking to get every bit out of each barrel. Some companies explore renewable routes, drawing from bio-based feedstocks as a way to cut reliance on petroleum.
Solutions for the Road Ahead
With electric vehicles growing, the world keeps shifting. Still, for trucks, planes, and millions of cars, high-octane fuel remains essential. Keep investing in fuel chemistry, improve refinery tech, and support alternative sources—these steps shape a future where people get performance and cleaner air together. It’s not just about what goes into the tank, it’s about how smart choices ripple out through communities and the environment.
Is 2,3,4-Trimethylpentane hazardous to health?
Understanding 2,3,4-Trimethylpentane in Everyday Life
Plenty of folks haven’t heard of 2,3,4-Trimethylpentane, but most have been close enough to touch it. This organic compound works as an octane booster in gasoline, helping engines run smoother and with less knocking. You can’t spot it in a busy garage, yet it’s there every time the fuel tank gets filled. I spent a chunk of my early career working around cars, always thinking about fumes long before I ever worried about any technical-sounding chemical names.
Real Risks Lurk in Small Exposures
Whenever a chemical shows up in fuel, folks have good reason to wonder about health risks. 2,3,4-Trimethylpentane doesn’t announce itself with a strong odor or obvious signs. It slips into the environment thanks to engine use, fuel spills, and evaporating gas stations. The U.S. Environmental Protection Agency and organizations like the National Institute for Occupational Safety and Health have both flagged similar alkylated hydrocarbons as cause for concern, especially if someone works around gasoline day in and day out for years.
Breathing it in can leave a scratchy throat, dizziness, or headaches, based on what field mechanics and research say. Animal research shows that long-term or high-level exposure could cause liver or kidney trouble. The lungs and central nervous system also take a hit, which is no small matter if you stand near gasoline vapors. Those who pump fuel for hours at a time, or clean up spills, face the most risk. Folks filling up their car for a few minutes a week deal with a much lower threat, though closing the gas cap quickly always feels like a smart habit.
Living With Chemicals in Fuel
Continue to drive along any major highway, and you notice a constant stream of vehicles burning gasoline. 2,3,4-Trimethylpentane keeps engines humming efficiently but doesn’t disappear after use. It can end up in the soil, sometimes reaching groundwater. I remember reading studies from states with lots of roadside wells, showing trace hydrocarbons in tap water. The amounts typically fall well below safety limits, but it only takes a single spill to cause worry in communities depending on those wells.
Fumes can hang heavy in closed garages or repair shops. Folks without proper ventilation or protective gear get the worst of it. I learned fast to open doors and windows wide, and wear gloves or masks on days with lots of engine work. Not every home garage has a fancy fume system, but even simple steps can lower risks.
Managing Health Risks Going Forward
Oil companies and public agencies have made some progress by improving leak prevention, reformulating some fuels, and keeping better tabs on spills. Most city-dwellers won’t run into this chemical at dangerous levels, thanks to safety rules about gas station design and underground tank inspections. Jobs in auto repair or fuel transportation need stronger protections.
People can push for regular air checks and better training for anyone handling fuel. Agencies can support cleanup crews with equipment and clear protocols in case of leaks. Living with modern fuels means keeping old risks in check, and demanding greater transparency about what goes into every gallon.
Learning more about 2,3,4-Trimethylpentane helps workers and neighbors feel safe around the fuels that power everyday life. Common sense habits—good ventilation, fast refueling, and reporting leaks—can go a long way to curb health worries tied to this little-known but widely used octane booster.
What is the chemical formula of 2,3,4-Trimethylpentane?
The Structure of 2,3,4-Trimethylpentane
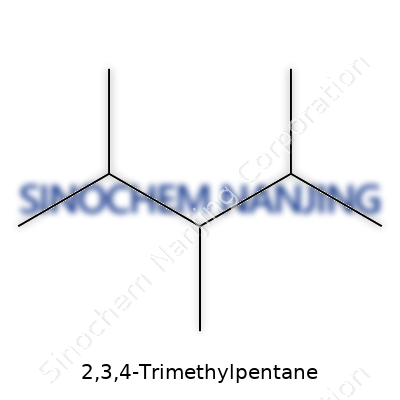
Looking at the name 2,3,4-Trimethylpentane, it sparks memories of organic chemistry classes where the smallest shift of a methyl group turned one molecule into another, and that change shaped things. Here, the backbone is pentane — a five-carbon chain. At positions 2, 3, and 4, methyl groups branch out, beefing up the molecule. Lay it out, count each carbon, and the chemical formula lines up as C8H18. No magic, just math: eight carbons, eighteen hydrogens.
Why the Structure Matters
Beyond memorizing formulas, it’s knowing what this structure means in real life. 2,3,4-Trimethylpentane is an isomer of octane. While in high school, most only hear about “octane” at the gas station. There’s more behind the pump. Isomers bear the same elements but differ by arrangement. The arrangement shapes properties like boiling point, volatility, and, for fuels, their knock resistance — what folks in the industry call octane rating. Refineries care because the right blend leads engines to run smoother, burn cleaner, and last longer. That matters for drivers and anyone breathing city air.
Facts Behind its Use
Think of all the different compounds mixed to make gasoline: some burn too quickly, others too slow. What stands out for 2,3,4-Trimethylpentane is how its structure stops engines from rattling — that annoying pinging or knocking under the hood. Chemists learned this after tirelessly measuring the knocking of pure samples, one by one. C8H18, especially in its isomeric forms, sets the benchmark in octane ratings. “Iso-octane” refers mostly to 2,2,4-Trimethylpentane, famous in fuel standards, but the 2,3,4 variant fits the conversation. Both share the C8H18 formula, just wired differently.
Digging Deeper: Importance for Everyday Life
Most people assume gasoline is just one thing. It’s a careful blend, crafted to avoid engine knocks and emissions. Without compounds like 2,3,4-Trimethylpentane, performance slips, pollution rises, and mileage suffers. Cars demand fuel that fits engines tuned for higher standards. Personal experience with older vehicles reveals the difference instantly — from rough idling to wear and tear. Formulas aren’t just technicalities; they’re the quiet force behind reliable commutes.
Looking at Solutions
Putting science and standards in the same seat opens up better answers for cleaner transportation. Relying on these high-octane isomers allows carmakers and fuel producers to phase out older, dirtier additives. Ethanol blends join the mix, but hydrocarbons like 2,3,4-Trimethylpentane remain crucial in balancing energy content and performance. The long-term path leads toward more sustainable options, but the chemistry learned from these formulas never fades. Every step toward synthetic fuels, bio-sourced blends, and emissions control leans on decades of formula-focused research. The answer to a question like “What’s the formula of 2,3,4-Trimethylpentane?” isn’t trivia. It’s part of the running engine in every car, every drive.
How should 2,3,4-Trimethylpentane be stored and handled?
Understanding the Substance in Everyday Terms
2,3,4-Trimethylpentane pops up most often in the fuel world, sliding in as an octane booster or blending component for gasoline. Behind the chemical name, you’re dealing with a colorless liquid that gives off volatile fumes. It pours easily, but don’t mistake its approachable look for something harmless. This material flashes at low temperatures, so even a small spill in the wrong spot creates plenty of hazard. The sweet smell can fool folks, but that doesn’t make it any less flammable—or less toxic when inhaled.
Common-Sense Storage
No one I know wants a fire in the garage, the lab, or the fuel depot. Keeping 2,3,4-Trimethylpentane in a cool spot, away from any open flames or sources of static, forms the right baseline. Forget cardboard or crumbling Tupperware; only steel or approved plastic containers with tightly fitted lids have any business here. These shouldn’t live in direct sunlight or next to oxidizers. Every person with a shed has learned that gasoline or similar chemicals don’t mix with careless stacking of paint cans or fertilizer bags. One bad lid or a tipped bottle and now you’re chasing fumes into every corner.
Good practice means labeling clearly so anyone—not just the chemist on duty—knows what waits in the container. I’ve found people rely on color codes and big, simple lettering, which helps during an emergency or after hours. Flammable-liquid cabinets or approved drums stay locked when not in use. That barrier pays off the minute a child or an untrained worker wanders by. Physical security isn’t just for big labs; every storage room matters.
Handling With Practical Precaution
Pumping or transferring Trimethylpentane calls for thoughtful prep. It evaporates quickly, so a breeze can send vapors across the room. Good airflow keeps you safer, but a spark near that vapor can turn routine handling into a nightmare. Even if you’re used to gasoline, don’t get cavalier. Splashing onto bare skin burns and dries out the area, and inhaling the vapor brings headaches that sometimes linger far longer than expected.
Gloves and safety goggles sound bothersome until you’ve tried washing a stinging solvent out of your eyes. During transfers, using proper funnels or pumps reduces the risk of splashing. I’ve seen too many workers shrug off these steps, thinking a quick pour won’t hurt, but all it takes is one accident to ruin that luck. If you spill even a little, absorbents and plenty of ventilation make cleanup less risky. Don’t grab the mop before checking the label on your absorbent—it should work specifically for hydrocarbons.
Why Training and Attention Protect People and Places
I’ve witnessed both well-run operations and jury-rigged setups. The difference shows in accidents—or the lack of them. Regular training helps workers recognize smells and symptoms, plus the signs of a poor seal or leaky valve. OSHA and fire codes call for refresher courses for good reason. People forget details; habits slip; a rushed job leads to mistakes. Clear processes and a willingness to call out shortcuts keep everyone honest.
Don’t rely on luck or the hope nobody notices a missing label or cracked nozzle. Keep material safety data sheets close by, so nobody guesses what to do in an emergency. Invest a little extra in fire extinguishers, grounding wires if you’re pumping, and warning signs. In homes, schools, or industrial sites, smart storage and responsible use guarantee both safety and regulatory compliance—and nobody faces a headline about preventable disasters.
What are the physical properties of 2,3,4-Trimethylpentane?
Getting to Know 2,3,4-Trimethylpentane
Beneath technical jargon and chemistry textbooks, 2,3,4-Trimethylpentane plays a quiet but important role in refining and fuels. People might know its more famous cousin, iso-octane, but this molecule also shows up in petrol mixtures, sneaking in with those other hydrocarbons that shape how engines run. It’s a clear, colorless liquid, straight out of a lab or a refinery tap — just the way most folks picture gasoline before any dyes hit it.
Physical Properties Shaping Its Story
The stuff flows like water when you pour it. Its density clocks in around 0.7 grams per cubic centimeter at room temperature, which means it floats on water and moves quickly if spilled. Holding it up to the light, you’ll see it’s not cloudy or thick—totally transparent, nothing hiding in there. Smelling it, there’s that familiar whiff: sharp, heavy, solvent-like, much like standing by a gas pump.
If you’re working in a garage or a research lab, knowing how this liquid behaves helps you avoid headaches. Its boiling point comes in close to 106°C. That tells you how quickly it evaporates and why it can vanish from an open dish long before you smell it too much. Its melting point drops down around -104°C, which keeps it flowing on the iciest days in storage. With a flash point near 18°C, it starts throwing off flammable vapors below room temperature. That keeps everyone cautious, because one spark or open flame in the wrong place could spell trouble.
Why These Properties Matter
Everyday people don’t ask about specific heat or vapor pressure, but in industrial circles, folks pay attention. With a vapor pressure over 30 mmHg at 20°C, this liquid makes itself known fast, evaporating from spills and open tanks. Workers don’t want those fumes hanging around too long; breathing them brings risk, not to mention fire hazards. Making a habit of wearing gloves and keeping spaces open to fresh air goes a long way.
Another thing: 2,3,4-Trimethylpentane barely mixes with water. Should it spill outdoors, it won’t dissolve and travel down the drain with the next rainstorm, but will float, making collection easier but also more urgent. Everyone remembers the drum-and-tarp tricks for scooping up light hydrocarbons in emergency drills.
Talking Reliability and Safety
The risk of using chemicals like this always circles back to how folks handle them. Factories and research centers tend to set up strict labeling and ventilation systems. Fire departments near heavy industry often train on how to control these flammable liquids, expecting the unexpected. People keep foam extinguishers handy, since water doesn’t do much good on hydrocarbon fires.
For anyone looking to improve on safety or environmental impact, tracking how hydrocarbons move through storage and distribution points always comes first. Catching leaks fast, switching to double-walled tanks, and always keeping flammable limits in sight—that’s where the smart money goes. It’s not the flashiest chemical, but treat it with care, and it pulls its weight.
| Names | |
| Preferred IUPAC name | 2,3,4-Trimethylpentane |
| Other names |
Isooctane 2,3,4-Trimethyloctane |
| Pronunciation | /ˈtuː θriː fɔːr traɪˈmɛθɪlˌpɛnteɪn/ |
| Identifiers | |
| CAS Number | 540-84-1 |
| Beilstein Reference | 1721198 |
| ChEBI | CHEBI:8937 |
| ChEMBL | CHEMBL43015 |
| ChemSpider | 11045 |
| DrugBank | DB01984 |
| ECHA InfoCard | 100.118.273 |
| EC Number | EC 211-309-7 |
| Gmelin Reference | 920 (Gmelin) |
| KEGG | C08381 |
| MeSH | D014259 |
| PubChem CID | 11638 |
| RTECS number | RY2175000 |
| UNII | UT0M64N33S |
| UN number | UN1262 |
| CompTox Dashboard (EPA) | DTXSID2020442 |
| Properties | |
| Chemical formula | C8H18 |
| Molar mass | 114.228 g/mol |
| Appearance | Colorless liquid |
| Odor | mild odor |
| Density | 0.718 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.80 |
| Vapor pressure | 1.65 kPa (at 20 °C) |
| Acidity (pKa) | 16.0 |
| Magnetic susceptibility (χ) | -7.2×10⁻⁶ |
| Refractive index (nD) | 1.389 |
| Viscosity | 0.696 cP (20°C) |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 359.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -232.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5198.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P261, P273, P301+P310, P331, P370+P378 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 51 °C (124 °F; 324 K) |
| Autoignition temperature | 415 °C (779 °F; 688 K) |
| Explosive limits | 1.0 - 6.0 % |
| Lethal dose or concentration | Lethal dose or concentration (LD50, Oral, Rat): 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 11 g/kg (rat, oral) |
| NIOSH | RN 564-02-3 |
| PEL (Permissible) | 1000 ppm |
| REL (Recommended) | 35 mg/m3 |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
2,2,3-Trimethylpentane 2,2,4-Trimethylpentane 2,3,3-Trimethylpentane 2,3,4-Trimethylhexane 2,3,4-Trimethylheptane |

