2,3,4-Trichloro-1-Butene: Chemical Progress, Risk, and Responsibility
From Lab Curiosity to Industrial Staple: The Historical Course
A few decades ago, 2,3,4-Trichloro-1-butene wasn’t much more than an academic project for organic chemists tinkering with halogenated alkenes. Chemistry in the twentieth century thrived on the ability to modify basic hydrocarbon backbones, and trichlorobutene proved attractive for this reason. The chemical world, eager for new intermediates in step-wise organic reactions, started exploring it for its potential in making pesticides, specialty polymers, and pharmaceuticals. Each new application created further demand for process improvement and stricter safety oversight, especially once early regulations on industrial organohalogens tightened. Trichloro-1-butene graduated from limited lab use to wider manufacturing in the 1980s, piggybacking on the pesticide boom and the rise of complex plastics.
Understanding What’s in the Bottle
With a sharp, pungent odor and a clear to pale yellow appearance, 2,3,4-Trichloro-1-butene looks innocent enough to the untrained eye. Those who’ve spent time around chemical storerooms, though, know that appearance means little. This molecule, with three chlorine atoms bonded to a four-carbon chain and a double bond sitting at the end, brings with it a potent mix of volatility and potential reactivity. Its boiling point sits higher than many simple alkenes, thanks to the halogens weighing it down, but it still gives off vapors easily and doesn’t need much provocation to take part in addition or substitution reactions. Handling it demands sturdy gloves, proper ventilation, and respect for both its physical potency and its habit of clinging to surfaces.
Label Clarity and Technical Details Matter
Years spent in production facilities taught me the sheer value of proper chemical labeling. One misplaced decimal on a material safety data sheet spells trouble for operators, lab techs, and even downstream clients. For substances like trichlorobutene, the tech specs do more than reference moisture content or purity; they usually note stabilization methods, recommended storage temperatures, and contamination limits. Overlooking these small details often leads to avoidable accidents, recalls, or environmental penalties. Standard labels provide the CAS number—871-18-7 in this case—alongside warnings for skin and respiratory exposure and notes about compatible storage materials.
Making 2,3,4-Trichloro-1-Butene: A Walkthrough
Synthesizing trichloro-1-butene usually starts from a simple base like butadiene, which undergoes direct chlorination under controlled temperature and illumination. In practice, the route often uses a halogen carrier to favor substitution at the 2,3,4 positions, maximizing yield and minimizing unwanted by-products. Tweaking reaction conditions—chlorine feed rates, pressure, solvent choice—can cut those by-products, but imperfect runs still create chlorinated waste that needs careful neutralization. Engineers working on these processes, myself included, get a firsthand look at how seemingly subtle tweaks can shave hours off production times or, just as easily, generate headaches with off-spec batches.
Reactions and Chemical Versatility
Few chemicals stay idle for long. Trichloro-1-butene is a responsive feedstock, taking part in nucleophilic substitutions, hydrolysis, and addition reactions. Chemists draw on its double bond and electron-withdrawing chloro groups for targeted synthesis, building everything from pest-killing agents to reactive intermediates in specialty polymers. I’ve watched as research teams chased more selective catalysts, intent on converting this molecule into new heterocycles or grafting it onto polymer backbones. The tweaks and tricks emerge from competitive pressure and the ever-present drive to wring higher value from each kilogram produced.
What’s in a Name?
Look through purchase orders or research papers and you’ll see synonyms like Trichlorobutene, 2,3,4-TCB, or even 2,3,4-Trichlorobut-1-ene. Mistakes happen easily when chemicals have unwieldy IUPAC names or similar-sounding derivatives, so the industry relies on consistent nomenclature and cross-referencing CAS numbers to ensure everyone handles the same substance. Experienced chemists double-check every name, especially during international transactions where regional vocabulary or translation issues have led to embarrassing—and occasionally hazardous—mismatches.
Working Safely: Standards and On-the-Ground Lessons
Textbook safety advice reads one way; actual practice can diverge. In the field, safe handling of trichloro-1-butene doesn’t just rely on material safety data sheets. Tight protocols mean regular air monitoring, personal protective equipment maintenance, and training workers to watch for subtle exposure symptoms before they escalate. Eye and respiratory irritation remain the most immediate risks, but chemical burns and chronic low-level toxicity call for equal attention. I’ve watched more than one veteran operator sidestep danger through muscle memory and constant vigilance—practices that should be standard, not exceptional.
Where It Goes: From Factory to Application
Much of the global output of 2,3,4-Trichloro-1-butene finds its way into agrochemical plants, where it forms a key precursor to selective pesticides and intermediate compounds. Some manufacturers also use it to functionalize polymer chains, particularly in making flame-retardant additives or modifying resin properties. Its ability to accept further substitution or addition reactions makes it valuable for custom syntheses in pharmaceutical labs or R&D environments hungry for exotic building blocks. Over time, newer, less hazardous alternatives have started to replace trichlorobutene in some applications, but the compound remains deeply embedded in legacy processes across multiple sectors.
Digging Deeper: Ongoing R&D and the Push for Safer Chemistry
Research on 2,3,4-Trichloro-1-butene pulls together academic curiosity and market demand. Green chemistry researchers seek cleaner synthesis routes, reduced energy consumption, and routes that cut chlorinated waste. Analytical chemists are mapping out subtle reaction pathways, hoping to predict impurity profiles and tighten downstream quality. The biggest push in recent years revolves around replacing this molecule with more benign alternatives where possible, designing catalysts to scavenge or neutralize unwanted by-products, and modeling long-term environmental behavior with more powerful simulation tools. These R&D efforts underscore a key truth: every “new and improved” molecule brings a fresh batch of unknowns to solve.
Toxicity Research and Environmental Scrutiny
Few people working on chemical plant floors forget the shadow cast by chlorinated hydrocarbons and their impact on health and environment. Rodent studies point to acute and chronic toxicity at relatively low doses, with findings flagging potential carcinogenic risks and disruption to respiratory or liver function. Local communities near production sites, faced with air and water quality concerns, have pushed regulatory authorities to clamp down on emissions and set strict discharge limits. Internal monitoring programs now run alongside independent audits, reflecting increased transparency and community oversight. Years of hands-on plant experience taught me that respecting these substances goes beyond compliance; genuine risk awareness and rapid transparency can make the difference between an incident contained and an emergency that lives on in local memory.
Looking Forward: Where Does 2,3,4-Trichloro-1-Butene Go from Here?
Innovation in industrial chemistry never moves in a straight line. Future demand for trichlorobutene will depend on evolving regulations, breakthroughs in less hazardous chemistry, and industry willingness to retrofit or reinvent entrenched processes. Some producers look to bio-based feedstocks or non-chlorinated alternatives as sustainability pressures mount. Others focus on closing the loop with higher recycling rates or real-time emissions capture technology. The core question rippling through boardrooms and lab benches alike is whether society can strike the right balance between progress and caution. Hard-earned experience shows that with persistent vigilance, open channels between industry and public, and a commitment to evidence-driven improvement, we can continue to benefit from chemistry’s advances without repeating the mistakes of the past.
What is the chemical formula of 2,3,4-Trichloro-1-Butene?
Breaking Down the Name
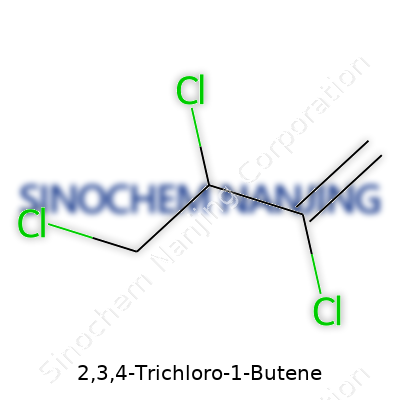
Seeing a name like 2,3,4-Trichloro-1-Butene makes a person wonder what’s really going on beneath those words. The trick lies in understanding each part. The backbone here is butene—a four-carbon chain with a double bond, straight and unpretentious. The “1-butene” signals the double bond starts at the first carbon. Add “2,3,4-trichloro,” and you’re putting three chlorine atoms on carbons 2, 3, and 4. The name points right to the structure, and I think it’s best just to lay it out.
Getting to the Chemical Formula
Start with butene: C4H8. That’s what pops into the mind of anyone who’s spent enough time staring at organic molecules. Substituting hydrogen atoms with chlorines on the second, third, and fourth carbon knocks off three hydrogens. Each swap gives a chlorine instead. So, C4H5Cl3 lines up as the right answer.
Why Chemical Naming Matters
It’s tempting to see these formulas as chemistry’s version of secret codes. I remember learning this system back in high school, where every line and number had meaning. The formula of 2,3,4-Trichloro-1-butene doesn’t just give a name; it tells chemists exactly what atoms sit where. Without a naming system like IUPAC’s, confusion runs wild—one misplaced chlorine and you’ve got something entirely different, with new hazards, uses, and reactions. Chlorinated compounds bring more risks, sometimes serious ones. Knowing a molecule’s formula can make the difference between safe handling and disaster.
Uses and Concerns
Compounds like this don’t just show up in dusty labs. Chlorinated butenes serve as building blocks in creating pesticides, pharmaceuticals, and specialty chemicals. Their reactivity makes them useful for adding other groups or building larger structures. But there’s a flip side. More chlorines mean a bigger environmental footprint and tricky handling. Chlorinated hydrocarbons have earned a notorious reputation for persistence in soil, air, and water. In cities and on farms, careless use or disposal causes health problems and fouls up ecosystems. Having worked at a chemical plant in my early days, I saw firsthand how strict regulations keep spills and leaks from turning into nightmares.
Safer Practices and Looking Ahead
Safety isn’t just a checklist; it’s a habit. Teams track chemical inventories, require training before anyone gets close to handling, and maintain gear in perfect shape. For 2,3,4-Trichloro-1-butene, this means careful storage, monitoring for leaks, and making sure nobody cuts corners. Smart companies move beyond old solvents and find alternatives to harsh chlorinated compounds, aiming to meet both business demands and community expectations. Researchers push for green chemistry, designing replacements that don’t stick around forever or build up in our bodies.
Understanding Brings Control
Turning down the fear means reading the fine print of chemicals. A formula like C4H5Cl3 might look intimidating, but with practice, those numbers and letters stop being a puzzle and start guiding decisions. Knowing what’s inside each bottle—by name, by formula, by nature—is what helps people work smarter, safer, and more responsibly. For all of us who’ve ever cracked open a chemistry set, that’s the everyday power of getting the formula right.
What are the main applications of 2,3,4-Trichloro-1-Butene?
Looking Closely at Its Role in Chemical Manufacturing
You won’t find 2,3,4-Trichloro-1-butene showing up in everyday household items, but it quietly supports a range of manufacturing processes. I’ve learned over the years that certain chemicals serve as building blocks, much like flour in bread-making—without them, a whole batch of materials and products would be impossible. This chlorinated butene fits right into that framework.
The most common application for this compound is as a starting material for other chemicals. It acts like a connector piece in a complicated machinery of chemical reactions, leading to products with a wide range of purposes. Manufacturers value it because the three chlorine atoms in the chain make it easy to alter, attach to, or convert into more valuable substances. In the chemical industry, flexibility like this means everything: you can develop pesticides, specialty polymers, or pharmaceuticals just by picking the right partners for your reaction.
The Pesticide Pathway
The agriculture industry relies on molecules engineered from building blocks like 2,3,4-Trichloro-1-butene for pest control. Farmers constantly struggle to keep crops safe from insects, weeds, and fungi. Overuse of outdated ingredients has led to pesticide resistance and environmental concerns. Here, this compound opens up the playbook for new pesticide chemistries with greater specificity, which helps with safer and more sustainable farming practices.
According to data from the Food and Agriculture Organization, upwards of 30% of food gets lost before reaching the consumer due to pest damage. So, more precise and effective pesticides—built off molecules like this—impact real-world food security. I have seen the shift among growers toward safer, modern solutions that reduce collateral harm to soils and pollinators, and chemicals with this type of backbone offer more options.
Industrial Synthesis and Specialty Plastics
The versatility of this molecule doesn’t stop at agriculture. Chemical plants use 2,3,4-Trichloro-1-butene for making specialty polymers, resins, and coatings. Chlorinated compounds show up again and again in fire-resistant cables, sealants, and high-performance materials. Wherever there’s demand for extra durability—think automotive parts that face extreme temperatures or electronics insulation—these advanced polymers prove their worth.
Exposing plastic materials to harsh environments can cause most types to crack or erode. By tweaking the chemistry and using intermediates from this family, manufacturers produce tougher formulas that last much longer. Extended durability reduces waste and manufacturing frequency, which fits into a larger push toward resource efficiency.
Health and Environmental Concerns
Chlorinated chemicals carry risk if not handled properly. The debate usually comes down to safety around workers, communities, and ecosystems. Mishandling causes everything from skin irritation to long-term environmental damage. Responsible use involves careful monitoring, closed-loop systems, and strong safety measures at production sites.
Safer alternatives get a lot of attention nowadays, but progress takes time. It’s possible to lower risk by enforcing stricter disposal protocols and pushing investment into green chemistry. Companies making regular use of this compound can work more closely with regulators and researchers to improve how the entire supply chain operates. My experience tells me that shared responsibility across industry, government, and local communities makes the biggest difference here.
What is the CAS number for 2,3,4-Trichloro-1-Butene?
The Importance of Chemical Identification
Behind every compound in a laboratory or factory, there’s a string of numbers known as the CAS number. For 2,3,4-Trichloro-1-Butene, this unique identifier is 41051-15-4. These numbers don’t just fill a box on a safety form. They simplify communication, reduce errors, and open access to data worldwide. If you’ve ever hunted for a chemical only to find ten names for the same thing, the value of a consistent identifier stands out fast.
Why Accuracy in Chemical Tracking Matters
Sitting at a lab bench or digging through regulatory paperwork, you see that names can be slippery. One country calls something by a brand name, another sticks to a formal IUPAC label, and someone else says “that chlorinated butene.” Researchers, regulators, and industry veterans all lean on the CAS system because it works across language, loyalty, and tradition. In large organizations, even a slight slip in chemical identity can halt production, increase hazards, or blow up a project timeline.
Real World Impact of CAS Consistency
Think about an environmental scientist investigating water samples near a chemical plant. Results come back showing traces of a chlorinated butene. Without a CAS number, nothing ties results to a catalog, a shipment log, or a safety data sheet with precision. Once you have 41051-15-4, you can link data from toxicity reports, cross-check with workplace exposure limits, and sort legal or environmental status down to the last detail. Misidentification costs money and, sometimes, health.
Transparency in Regulation and Trade
The world of chemicals isn’t just about beakers and gloves. International trade runs on documentation. Customs paperwork asks for the exact CAS number. Regulatory authorities check these numbers against restricted lists, enabling products to move or stop at borders. With 2,3,4-Trichloro-1-Butene’s number, compliance teams make informed decisions, avoid accidental law-breaking, and publish trustworthy records. Reliable identification keeps the supply chain dependable from warehouse to end-user.
How CAS Numbers Build Trust and Traceability
In my experience managing materials for both research and scaled-up manufacturing, I’ve seen suppliers and buyers argue over product identity because of missing or unclear labels. The introduction of a CAS number often settles the matter. Detailed records grow out of clarity: from raw materials intake, through processes, to waste disposal. 2,3,4-Trichloro-1-Butene isn’t just another name in a textbook—it arrives, is monitored, and is stored safely because that numeric code never gets confused with another substance. Anyone in labs, clinics, or regulatory offices can verify the identity quickly. Fewer mix-ups mean safer workplaces.
Building Good Practices for Chemical Safety
Great systems don’t hide mistakes—they prevent them. Part of that is using the right CAS number for every chemical, every time. For 2,3,4-Trichloro-1-Butene, the use of 41051-15-4 belongs on all shipping records, safety sheets, research notes, and regulatory filings. Code consistency doesn’t eliminate risk, but it does cut confusion. Most accidents in chemical environments trace back to poor record-keeping or mislabeling. Cascading errors slow down innovation, increase costs, and threaten both workers and the public.
Solutions and Steps Forward
Education works best over enforcement. Companies invest in staff training and checklists that emphasize reading and using CAS numbers every day. Label printers that auto-embed this identifier help, especially in busy warehouses and labs. Digital inventory systems flag inconsistencies before they slip downstream. Sharing best practices through professional groups and online forums raises the standard. With the right habits, even small teams keep track of complex inventories with confidence. Safety, compliance, and trust follow close behind.
Is 2,3,4-Trichloro-1-Butene hazardous to health or the environment?
A Chemical with Questions
2,3,4-Trichloro-1-butene doesn’t show up in popular news feeds or everyday conversations. This chlorinated butene mainly stays in industrial zones, where it gets used as an intermediate. But unfamiliar doesn’t mean harmless. Most chemicals that contain several chlorine atoms spark concern, and people who work with similar solvents and intermediates know the risks never drift far from reality.
What Happens to People Around It
Direct contact always worries workers. Based on data from the likes of the EPA and NIOSH, trichloro-alkenes have a reputation for irritating skin, stinging eyes, and bothering the respiratory system. Volatile organic compounds of this kind escape into the air easily. People breathing them in—especially in hot, enclosed spaces—can find themselves coughing, feeling dizzy, and struggling to focus. Any sort of spill can raise exposure through the skin or by inhalation.
I remember walking through a plant where just handling trichlorinated products left that recognizable chemical odor in my clothing. Safety rules existed for a reason: gloves, goggles, proper exhaust. Awareness slipped the moment someone forgot to check for leaks. Even after just a few hours, workers described short-term headaches, watery eyes, and mild nausea, but also a nagging worry—we didn’t know what exposure built up in people’s bodies over years.
Long-term studies remain thin. Animals forced to live with similar chlorinated butenes end up with signs of liver and kidney stress. Chlorinated hydrocarbons in general have been tied in other research to higher cancer risks with long enough exposure. Authorities list many of these chemicals as potential or suspected carcinogens, and no one wants to find out in hindsight that 2,3,4-trichloro-1-butene belongs in that category.
Impact on the World Outside the Lab
The story extends beyond workers. Disposing of chlorinated intermediates often leads to soil and water contamination. These compounds stick around. Microbes can’t easily break the carbon-chlorine bonds; water treatment plants struggle too. In many rivers near old chemical plants, persistent chlorinated chemicals remain trapped in sediment, slowly leaking out over decades. Wildlife—especially fish and those eating them—show high levels of these contaminants. This builds up in the food chain, reaching humans through fish or contaminated crops.
Research shows even low-level contamination disrupts growth and reproduction in aquatic life. That isn’t an abstract worry. If water around towns picks up enough chlorinated residues, the local catch becomes unusable, and people end up with health advisories.
Better Practices and Solutions
Minimizing hazards starts with substitution. Finding safer alternatives, particularly in open systems, always reduces risks. Closed-loop production lines, better protective gear, and stricter maintenance of ventilation systems make a practical difference. Regular air and water monitoring on-site catches leaks or spills before they spread. Waste treatment needs updating—using technologies that break down persistent chlorine bonds, like advanced oxidation or specialized incinerators, stops these chemicals from sneaking into landfills or waterways.
So, 2,3,4-Trichloro-1-butene may not have a high public profile, but it brings genuine risks. Living with such chemicals day-to-day means accepting responsibility for both worker health and environmental stewardship. Until science clears up the big questions, treating this compound with caution—at every stage—remains the safest approach.
What are the storage and handling requirements for 2,3,4-Trichloro-1-Butene?
Everyday Responsibility in Chemical Storage
Working around chemicals like 2,3,4-Trichloro-1-Butene reminds me how easy it is to take shortcuts—until something goes wrong. This compound can irritate the skin and eyes or trigger breathing problems if vapors escape. Many treat these risks lightly, but it’s the basics that keep a workplace safe.
Keeping It Contained
Take storage. Using a tightly sealed container made from compatible materials avoids leaks. A steel drum or high-density polyethylene container prevents corrosion or reaction. I’ve seen cheap plastic jugs split and turn into a nightmare. A dedicated chemical store, cool and dry, with good airflow cuts down on dangerous vapor buildup. It’s tempting to pack extra stock in a crowded closet, but crowding creates more hazards and makes leaks harder to spot.
Storing this chemical means marking every drum, even if everyone thinks they know what’s inside. Proper labels help emergency crews know what they’re up against, should something spill or catch fire. Regulations like OSHA and GHS don’t just exist for big companies; even small labs have to follow labeling and inventory rules. A shelf collapse or mixed-up container might sound rare, but these simple steps stop small problems from turning into disasters.
Handling Isn't a Shortcut Game
Personal protective equipment isn’t an afterthought. At my old plant, a manager had to go home after an accidental splash—chemical burns make for a rough day. The right choice often includes chemical-resistant gloves, goggles, and an apron. A chemical fume hood or good local ventilation means no one breathes in vapors by mistake. A cheap dust mask won’t cut it, either. Keeping a safety shower and eyewash close by isn’t just a best practice—it’s common sense.
It helps to treat every transfer, mixing, or disposal step as a risk. Drip pans and spill kits let workers catch problems right away. Regular equipment inspections spot weak points in seals and valves, catching leaks before they turn into emergencies. Skipping these steps just piles up trouble for someone else down the line.
Why It’s Worth the Effort
There’s real value in proper record-keeping. Writing down storage amounts and dates creates a roadmap during audits or cleanups. Safety Data Sheets (SDS) should never live in a dusty binder—everyone who handles the material ought to know where to find them and what’s inside.
Training is the last—yet most powerful—defense. Watching new hires look over the rules with wide eyes always reminds me: repeated refreshers keep risk top-of-mind. Whether you’re the old hand or just starting out, a confident team that knows spill and fire drill routines inside out is everyone’s best safeguard.
Looking Ahead
Change starts with culture. Safe storage and handling don’t just follow policy; they become part of the workplace DNA. Some folks see these routines as boring, but the facts show otherwise. In 2020, the U.S. Chemical Safety Board found most incidents tied back to skipped protocol—something preventable. If everyone makes smart storage and handling a habit, worry about 2,3,4-Trichloro-1-Butene fades into the background, leaving safety front and center.
| Names | |
| Preferred IUPAC name | (E)-2,3,4-Trichlorobut-1-ene |
| Other names |
Tris(trichlorobutenyl)chloromethane 1-Butene, 2,3,4-trichloro- 2,3,4-Trichlorobut-1-ene |
| Pronunciation | /ˌtuː,θriː,ˈfɔːr-traɪˈklɔːroʊ-wʌn-bjuːˈtiːn/ |
| Identifiers | |
| CAS Number | 18980-55-5 |
| Beilstein Reference | 1718738 |
| ChEBI | CHEBI:87341 |
| ChEMBL | CHEMBL261185 |
| ChemSpider | 10221260 |
| DrugBank | DB02308 |
| ECHA InfoCard | ECHA InfoCard: 100.014.324 |
| EC Number | 203-964-0 |
| Gmelin Reference | 8894 |
| KEGG | C19228 |
| MeSH | D017869 |
| PubChem CID | 79017 |
| RTECS number | EZ3850000 |
| UNII | QVL6K98891 |
| UN number | UN2418 |
| CompTox Dashboard (EPA) | DTXSID40882431 |
| Properties | |
| Chemical formula | C4H5Cl3 |
| Molar mass | 159.42 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | sweet |
| Density | 1.38 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 2.98 |
| Vapor pressure | 1.23 mmHg (25°C) |
| Acidity (pKa) | 14.41 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.5060 |
| Viscosity | 2.821 mPa·s (25°C) |
| Dipole moment | 2.19 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 354.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -96.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1876.9 kJ/mol |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P303+P361+P353, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0-▢ |
| Flash point | 54°C |
| Autoignition temperature | 435°C |
| Lethal dose or concentration | LD50 (oral, rat): > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 660 mg/kg (oral, rat) |
| NIOSH | LS-1076 |
| PEL (Permissible) | PEL: 1 ppm (Skin) |
| REL (Recommended) | 0.1 ppm (0.59 mg/m³) |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Chloroprene 1,2,3-Trichloropropane 1,2-Dichloro-3-butene 4-Chloro-1-butene cis-1,3-Dichloropropene |

