2,3,4,7,8-Pentachlorodibenzofuran: A Closer Look
Historical Development
Before smartphones, right around the era when large industries began ramping up chemical production, 2,3,4,7,8-Pentachlorodibenzofuran (often called 2,3,4,7,8-PCDF) found its way into the environment. PCDFs didn't originate in labs with a plan for them. They turned up as byproducts when folks started manufacturing chlorinated chemicals like polychlorinated biphenyls (PCBs) and certain herbicides in the middle of the twentieth century. This compound’s history doesn’t just represent chemists pushing the envelope, but also mirrors the lack of foresight into long-term ecological effects. By the time regulators and scientists recognized the presence of PCDFs in emissions, waterways, and even food, the damage had already begun making itself seen in ecosystems and people’s health stories.
Product Overview
From a chemical standpoint, 2,3,4,7,8-PCDF shows up as a member of the wider family of polychlorinated dibenzofurans. No one sets out to manufacture it for a consumer product – its presence is almost always as an unwanted guest, a contaminant during the production of other substances. Because of this, industrial sites, especially those dealing with chlorine or burning waste, have turned up significant concentrations. This stuff isn’t something you’d keep on a shelf for direct use, yet its legacy stretches into soil, sediments, and even into living tissue throughout the food web.
Physical & Chemical Properties
2,3,4,7,8-PCDF takes the form of a colorless, odorless solid at room temperature, though few ever encounter it in pure form. Its chemical stability stands out — resisting breakdown from sunlight, heat, or microbial activity. A trait like this explains its ‘forever chemical’ nickname. Lipophilic by nature, it doesn’t slosh around in water easily, but instead clings to fats and organic material. This tendency to bioaccumulate helps explain why it turns up in the milk of mammals or the tissue of fish and why cleanup efforts lag far behind the pace at which 2,3,4,7,8-PCDF seeps into the world.
Technical Specifications & Labeling
Scientists break down PCDFs, including 2,3,4,7,8-PCDF, into congeners based on where the chlorine atoms sit on the dibenzofuran backbone. For regulatory or laboratory purposes, the compound often gets referenced by its CAS number (57117-31-4). Labeling in the lab always involves warning symbols for toxic and environmental hazards, plus instructions on containment. Technical bulletins highlight its melting point, solubility, and required handling precautions. Industrial stakeholders have had to learn a hard lesson: labeling this contaminant was too little, too late for many sites already damaged.
Preparation Method
2,3,4,7,8-PCDF never debuted through deliberate synthesis, but rather as a byproduct during combustion of materials containing chlorine compounds. Trash incinerators, metal processing, and especially the production of other chlorinated organics set the stage for its appearance. Every time these chemicals heat up or interact under poorly-controlled conditions, molecules break and reform, building complexity until out comes 2,3,4,7,8-PCDF among other PCDFs. It’s less about a recipe and more about unfortunate side effects of chasing other industrial goals.
Chemical Reactions & Modifications
Once let loose in the wild, 2,3,4,7,8-PCDF resists most of nature’s regular tricks for breaking down foreign substances. High-temperature incineration, above typical waste-handling levels, stands as one of the few reliable ways to destroy it. Advanced oxidation can pull a little weight in cleanup, breaking chemical bonds and stripping away chlorines in controlled lab settings, but on a practical scale the chemical shrugs off most efforts. Many researchers aim for techniques to reduce toxicity through dechlorination, though success rarely translates from test tubes to contaminated earth.
Synonyms & Product Names
In scientific circles, 2,3,4,7,8-PCDF isn’t just a mouthful, it’s also known as pentachloro-dibenzofuran among other names. The “five-chloro” designation highlights its key difference from similar polychlorinated dibenzofurans. Lab reports and toxicology papers tend to fall back on the abbreviation or the CAS number, simplifying communication but hiding the weight such names carry for communities experiencing exposure.
Safety & Operational Standards
Anyone handling or studying 2,3,4,7,8-PCDF quickly finds themselves immersed in a world of strict rules. Laboratory staff wear protective suits, gloves, and breathing filters. Facilities must run closed systems to limit emissions, and environmental sites nearby run air and water monitoring programs year-round. Local, national, and international regulations now target PCDFs for elimination, recognizing that even a small spill can linger for decades. Safe handling grows out of bitter experience, notably the Yusho and Yucheng food contamination incidents in Japan and Taiwan, where exposure left thousands with chronic health effects.
Application Area
This compound never gets a starring role in any planned manufacturing process. Its only real “application” comes as a tracer in environmental contamination studies or in toxicity testing. Scientists track its movement and breakdown, providing evidence for lawsuits and clean-up orders. Rather than fueling innovation, 2,3,4,7,8-PCDF’s legacy has been to freeze industrial expansion until plants prove they can avoid releasing dioxins and furans as byproducts.
Research & Development
Over the last decades, researchers have poured resources into understanding both where PCDFs like 2,3,4,7,8-PCDF turn up and how they act in living systems. Many studies dig into the compound’s ability to bind to proteins and disrupt hormonal systems, focusing on both persistence and toxic mechanisms. Modern monitoring depends on gas chromatography and mass spectrometry, providing ever tighter detection limits. Some labs look for genetically engineered microbes or chemical treatments that could one day offer a scalable clean-up option for contaminated soil and water.
Toxicity Research
If you spend time in public health, you can’t avoid the shadow of PCDFs. Studies repeatedly find a link between 2,3,4,7,8-PCDF exposure and disrupted immune systems, hormonal imbalance, and heightened cancer risk. Animals, especially those higher up the food chain, accumulate PCDFs in fat, passing residues to offspring through birth and breast milk. Human data, though harder to collect for ethical reasons, consistently point to similar effects, particularly when exposure happens young or drags on for years. Governments keep chasing lower limits because even in trace amounts, the risk lingers.
Future Prospects
Looking down the line, the world needs practical solutions for persistent organic pollutants like 2,3,4,7,8-PCDF. Industry has already slashed most deliberate sources, but legacy contamination sticks around, and accidental releases still happen. Stronger regulations, better filtration systems, and real investments in cleanup research offer a way forward. Community involvement has grown alongside technical fixes – locals demand transparency and participate in watchdog roles that keep polluters honest. As more is learned about PCDF’s lasting footprint on health and ecosystems, the hope rides on both policy innovation and new science to tip the balance from risk toward recovery.
What is 2,3,4,7,8-Pentachlorodibenzofuran used for?
Digging Into Its Purpose
People often assume every chemical exists because someone wanted it around. That is simply not true with 2,3,4,7,8-pentachlorodibenzofuran. This molecule belongs to a nasty family called dioxins and furans. These compounds show up not as intended products but as dirty byproducts—unwanted guests after certain industrial and chemical reactions. Factories dealing with chlorine, paper bleaching, incineration of waste, and old-school pesticide production end up with it. Nobody sets out to make pentachlorodibenzofuran because it's dangerous, costly to clean, and barely useful.
No Real Use, Big Consequences
In my experience working with environmental professionals, I’ve never seen anyone handle 2,3,4,7,8-pentachlorodibenzofuran with the goal of actually using it. Scientists and regulators usually focus on tracking and removing it. This stuff resists breaking down naturally and sneaks into soil, water, air, and food. It acts like many persistent organic pollutants: small amounts linger for years, then build up in the bodies of fish, birds, and even us.
Back in the 1980s, governments around the world started noticing these molecules piling up because of industrial activities. Scientists linked them to a range of health issues—cancer, liver damage, immune system problems, reproductive harm. Here’s where the facts come in: even tiny doses carry risk. The World Health Organization and the Environmental Protection Agency both highlight the dangers, stressing that levels need to be kept as close to zero as possible.
Why Stay Alert?
Pentachlorodibenzofuran does not power factories, fuel cars, or improve crops. Its only “role” comes from attention by public health agencies and environmental watchdogs. Researchers test soil, water, and food for traces. Factories that could create dioxins get hit with expensive pollution controls. This response ramps up fuel, energy, and waste costs—everyone pays in some way for the hidden legacy of these chemicals. Farmers have learned the hard way that once this stuff hits a field or river, it sticks around for decades.
For people living near older industrial sites or waste incinerators, monitoring makes a difference. A study out of Japan linked historical furan releases to changes in local birth rates and immune system issues in children. These health patterns push communities to demand testing, cleanup, and strict laws on waste burning and chemical handling.
Chasing Solutions
Nobody wins when furans pile up in the environment, so stopping them at the source stands as the key move. Using less chlorine in industrial processes, adopting closed-loop systems, and replacing out-of-date incinerators cuts these emissions. Some companies handle toxic residue with costly filters and incinerators set at higher temperatures, which destroy most furans. Regulators demand reports and run tests on food and air. Community activists push for real transparency. These layers of pressure and oversight help keep pentachlorodibenzofuran out of our bodies, though the fight never really ends.
Living through multiple chemical scares in my region, I’ve seen firsthand how clear rules, public involvement, and constant vigilance create safer neighborhoods. Facing up to ugly byproducts like pentachlorodibenzofuran means choosing health and transparency over short-term cost savings.
What are the health risks associated with exposure to 2,3,4,7,8-Pentachlorodibenzofuran?
A Close Look at a Hidden Toxic Threat
Toxins like 2,3,4,7,8-Pentachlorodibenzofuran aren’t household names, but their effects seep into communities and linger in bodies for years. This chemical belongs to a family of dioxin-like compounds notorious for showing up after trash incineration, pesticide manufacturing, or from the breakdown of industrial chemicals. Once these compounds escape into the air, they settle into water, soil, and food supplies. People breathe them in or eat plants and animal products contaminated by them.
The Evidence on Health Risks
Broad scientific research links dioxin-like compounds—including this specific pentachlorodibenzofuran—to harsh health problems. Exposure doesn’t just poke at one organ or mess with one system. Instead, it can disrupt immune response, weaken the reproductive system, and fuel cancers. Studies in both animals and humans have found immune suppression, which leaves individuals defenseless against infections. Endocrine disruption can fumble with hormonal balance, causing developmental delays for children and even fertility issues for adults. The International Agency for Research on Cancer dug into the evidence and regards chemicals in this family as known human carcinogens—plain and simple.
I’ve met farmers, welders, and trash sorters who describe odd rashes and odd aches after spending years in contaminated sites. Not everyone knows to blame what they can’t see or smell, but research matches their stories. Chloracne—a painful skin condition—shows up in folks with elevated dioxin body burden. There’s also proof that these chemicals alter the way the liver processes other substances, raising risks of long-term disease.
Long-Term Exposure Feeds Bigger Problems
What worries doctors and scientists is the persistence of pentachlorodibenzofurans. Unlike toxins that pass quickly, these build up in fat tissue and resist breakdown. They flow from mother to child through breast milk, linking generations to past pollution. Children face higher danger from exposure, not just because of their size but the fast pace of their development. A damaged immune or nervous system early in life makes it tough to recover fully.
Communities near hazardous waste sites often see more birth defects and cancer cases. The reality is that people with fewer resources shoulder greater exposure. Many don’t receive regular health screenings or detailed answers about their risks.
Pushing Toward Solutions
Cutting exposure starts with cutting release. Cleaner landfill practices and strict incinerator technology matter. Governments worldwide already limit how much of these toxins factories can spew, but enforcement doesn’t always keep pace with economic pressures. People can call for tighter monitoring by supporting local environmental watchdog groups and pushing industry leaders to set better standards.
Testing food and water for pentachlorodibenzofurans isn’t just a job for scientists; consumers, farmers, and public officials all play a part. Investing in robust monitoring, especially in communities near chemical plants or dumps, keeps contamination events from slipping under the radar. People who feel they might have been exposed should ask health providers about dioxin testing and not shy away from raising concerns, even if they feel ignored at first. Transparent records of what’s lurking in air and water will never solve every problem, but they give people the footing to defend their own health.
How should 2,3,4,7,8-Pentachlorodibenzofuran be stored and handled safely?
Recognizing the Risks
2,3,4,7,8-Pentachlorodibenzofuran packs a punch as one of those chemicals most folks have never heard of but should take seriously. This compound, a byproduct from manufacturing processes and waste incineration, ranks high on the danger scale. Research draws strong lines between this group of chemicals and cancer, immune system trouble, skin problems like chloracne, and hormone disruption. Workers and scientists who deal with these substances don’t take them lightly, not because of reputation, but because experience and science say that the tiniest amount drifting in the air or settling on the skin can cause health issues over time.
Safe Storage: More Than Just a Locked Drawer
There’s no shortcut here: lock up 2,3,4,7,8-Pentachlorodibenzofuran in sturdy, corrosion-resistant containers with airtight seals. Polyethylene or polytetrafluoroethylene containers offer chemical resistance, blocking leaks and keeping the compound from escaping. Treat this like handling a potent poison, not an everyday supply. You won’t find this sitting on the shelf next to the soap—access should stay limited to folks who understand the risks. I remember working in a university lab where a separate, vented chemical cabinet kept these high-risk compounds from mixing with others and turning a mistake into a nightmare.
Labeling never feels exciting, but with chemicals like pentachlorodibenzofuran, it’s not negotiable. Clear, strong labels with warnings stand as the only chance for someone new or tired at the end of a shift to recognize the hazard. Storage areas need reliability and clear ventilation—no one wants to breathe these vapors and later find their mask wasn’t up to the job. Every bit of dust or vapor floating from careless storage becomes a problem for someone down the hall or years down the road.
Handling: Suits, Gloves, and Good Habits
It doesn’t matter if you work in an academic, industrial, or environmental cleanup setting; every hand that touches this compound needs glove protection. Chemical-resistant gloves, goggles snug against the face, and a fitted respirator all play their part. Chemical splash suits might feel bulky, but the barrier between skin and this compound turns out to be much thinner than most people guess. Simple soap and water won’t cut it if there’s a spill on your arm.
Ventilation systems—a real investment—matter more than any single bottle. Local exhausts or fume hoods pull away contaminated air before it starts drifting. Filters that trap particulates and vapors send a clear message: people’s health comes before cutting a corner.
Waste and Accidental Release: Cleaning Up, Not Covering Up
No one has a perfect record for spills or mistakes. The real test happens in what comes next. Absorbent pads, safety containers, and emergency procedures give everyone a fighting chance. I’ve watched trained techs in action, scooping up powder, securing it in sealed drums, and calling hazardous waste disposal—never tossing it in the trash, no matter the deadline.
Routine checks of storage areas and safety equipment keep the workplace safer than any fancy policy statement. Reading about contamination cases in the news or medical journals, it’s easy to see that disasters start small—unmarked vials, forgotten spills, absentminded disposal. The fix rarely arrives from outside legislation or new technology. It happens when workers double-check each other, management pays for better extraction vents, and everyone treats safety data as more than paper pushing.
Keeping Responsibility Local
Protecting health, air, and water sources from this compound rests with every individual on site. Training and good habits add up to collective experience. Mistakes here don’t get second chances. Respect for the risks doesn’t mean living in fear; it looks like solid teamwork, active vigilance, and a chain of custody tighter than a family reunion.
Is 2,3,4,7,8-Pentachlorodibenzofuran regulated or restricted by law?
Toxic Legacy of Dioxins and Furans
2,3,4,7,8-Pentachlorodibenzofuran belongs to a family of chemicals called dioxins and furans. These names trigger concern in most folks who follow environmental health. Dioxins and furans form during burning processes, including waste incineration, pesticide manufacturing, chlorine bleaching in paper mills, or any place organic material meets chlorine at high heat. Grocery store food, the water tap, and backyard gardens all see the trace fingerprints of these molecules. The real worry lies with long-lasting effects, not a brief encounter.
Where Regulation Stands Today
Governments, both local and global, have stared down dioxins and furans for years. The big global treaty, called the Stockholm Convention on Persistent Organic Pollutants, names many variants of polychlorinated dibenzofurans, including the infamous 2,3,4,7,8 isomer, as substances nations must try to eliminate or strictly limit. The United States Environmental Protection Agency includes this compound within the group of dioxin-like substances it regularly assesses and restricts. The European Union takes a tough stance too. Both agencies tie regulations to emission limits, cleanup standards, and safety checks in everything from landfill leachate to milk production.
Dioxins and furans do not have easy clearance from the body. They settle. They bind to fatty tissues, build up over generations, and resist sunlight, bacteria, or basic weathering. The science has shown convincing links to cancers, immune suppression, hormonal problems, and developmental delays in children. Once these poisons settle into soil or water, anyone living nearby joins the circle of impact. Policy has stepped in for that reason.
Facing the Realities of Enforcement and Oversight
Laws work best when industries comply and authorities follow up. Not every incinerator runs at high enough temperature or perfect efficiency to destroy furans like 2,3,4,7,8-PCDF. Some slip through. Open burning of trash or poor practices in factories punch loopholes in even the strongest rules. Neighborhoods sitting near old landfills or former industrial sites often feel the brunt of past neglect, despite current legal controls.
Yet, every regulation with bite also depends on proper detection. Most average citizens have no practical way to measure the dioxin or furan content in local air, water, or soil. Specialized labs handle this work. They use methods like HRGC/HRMS (high-resolution gas chromatography/mass spectrometry), which require funding and trained staff. Those budgets do not stretch as far in some towns or developing countries. That leads to uneven protection.
Common Sense and Next Steps
Community members can make a difference. Organizing for air and water monitoring around high-risk industries can catch problems early. Backyard burning bans matter; these small fires add up. Supporting local and national environmental groups who keep eyes on old dump sites pays off too. Farmworkers, landfill workers, and people living along rivers polluted by industrial runoff face the most risk. Public pressure gets local officials moving when they worry about the next news report or voter backlash.
Safer substitutes in manufacturing, stronger waste handling rules, and better technology in existing plants all lower the odds of exposure. Sharing scientific results in plain language and bringing those results to city hall or the statehouse wins better funding for testing and cleanup. Every step toward less dioxin and furan in the dirt and dust puts fewer families in harm's way, and that is worth fighting for.
What are the environmental impacts of 2,3,4,7,8-Pentachlorodibenzofuran?
Deep Trouble from an Unwanted Guest
2,3,4,7,8-Pentachlorodibenzofuran doesn’t roll off the tongue. Most folks never hear of it unless trouble stirs. This chemical belongs to the family of “furans” – pollutants that sneak out during some industrial activities, especially where chlorine mixes with high heat, like in the pulp and paper sector or waste incineration. It did not come from a lab where somebody designed it with grace or safety in mind. It drifts into existence more by accident than intent.
It Sticks Around and Travels Far
After release, it clings to soil, hangs in the air, and latches onto plants and animals. Nothing about its presence feels temporary. Even decades after leaving a factory, traces show up miles away. Rain and wind do the heavy lifting, carrying it into rivers and lakes or dropping it on fields. Life in water picks it up, and soon it settles into the fat of fish and the eggs of birds. I’ve watched fish advisories go up beside lakes that once fed local communities, all because of these stubborn chemicals.
The Problem Grows in Living Things
We can’t see the trouble with the naked eye, but scientists check animal samples, and the numbers don’t lie. Low levels still pack a punch. Birds nesting in contaminated places lay fewer eggs, and young often hatch with twisted beaks or weak limbs. For mammals, including people, health risks pile up. 2,3,4,7,8-Pentachlorodibenzofuran slips into the food chain and doesn’t leave easily. It stores in bodies for years, raising cancer risks, hurting immune systems, and even changing how hormones work.
Communities Hit Hardest
Locals living close to big industries or waste dumps know this story better than anyone. Folks from river-based towns tell how they turned cautious about eating the catch of the day. Some families saw more illness over the years. In my own region, debates around cleanup drag on because money and responsibility get tossed around between companies and government, while the chemical keeps leaching into the ground and water.
Better Ways Forward
People do not have to stand by and watch things get worse. Some towns tighten controls on what goes into the incinerator. Cleaner techniques in the pulp and paper industry drop dioxin and furan leakage sharply. Laws got teeth, but strict monitoring saves more trouble than just buckets of paperwork. Several groups push for point-source cleanups, digging out the worst hot spots and restoring wetlands. Change takes time—and funding—but the right science and shared pressure from concerned communities help force the shift.
Knowledge stays important, too. Open records, local air and water testing, and honest talk keep people engaged in decisions that shape their land’s future. This problem does not go away overnight, but careful steps can heal ecosystems. We learn from what did not work before. Simple transparency and investment in safer technologies matter just as much as enforcement. The world needs less of this toxic guest if future generations hope for cleaner water, healthier wildlife, and food they trust.
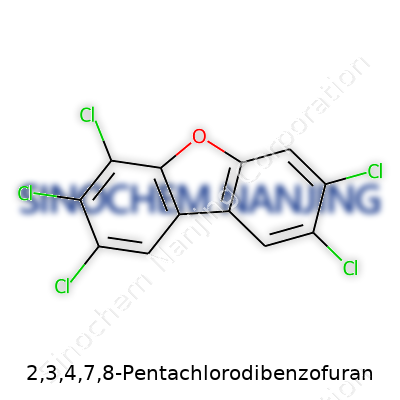
| Names | |
| Preferred IUPAC name | 2,3,4,7,8-Pentachlorodibenzofuran |
| Other names |
PCDF-110 2,3,4,7,8-PeCDF |
| Pronunciation | /ˌpɛn.təˌklaɪ.rəʊ.daɪˌbɛn.zəˈfjʊə.rən/ |
| Identifiers | |
| CAS Number | 57117-31-4 |
| Beilstein Reference | 1858737 |
| ChEBI | CHEBI:82110 |
| ChEMBL | CHEMBL4300260 |
| ChemSpider | 123154 |
| DrugBank | DB13363 |
| ECHA InfoCard | 08a985dc-563a-4d71-8d3b-9c8e24699715 |
| EC Number | 'EC 206-902-6' |
| Gmelin Reference | 120066 |
| KEGG | C14412 |
| MeSH | D016623 |
| PubChem CID | 9778 |
| RTECS number | GZ2250000 |
| UNII | UX15A8H72T |
| UN number | UN2718 |
| CompTox Dashboard (EPA) | DTXSID2020597 |
| Properties | |
| Chemical formula | C12H3Cl5O |
| Molar mass | 356.324 g/mol |
| Appearance | white solid |
| Odor | Odorless |
| Density | 1.741 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.94 |
| Vapor pressure | 1.2E-6 mm Hg at 25°C |
| Acidity (pKa) | 2.50 |
| Magnetic susceptibility (χ) | -0.00022 |
| Refractive index (nD) | 1.644 |
| Viscosity | Viscous liquid |
| Dipole moment | 4.79 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -60.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5720.7 kJ/mol |
| Pharmacology | |
| ATC code | Dioxins, including 2,3,4,7,8-Pentachlorodibenzofuran, do not have an ATC code. |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes damage to organs. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H310 + H330: Fatal if swallowed, in contact with skin or if inhaled. H351: Suspected of causing cancer. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P264, P270, P273, P280, P284, P301+P310, P302+P352, P304+P340, P308+P311, P310, P320, P330, P363, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-4-0- (Health-4, Flammability-1, Instability-0, Special-) |
| Flash point | 132 °C |
| Autoignition temperature | > 662 °F (350 °C) |
| Lethal dose or concentration | LD50 (rat, oral) 25 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral >5 mg/kg |
| NIOSH | PC8225000 |
| PEL (Permissible) | 0.001 mg/m3 |
| REL (Recommended) | 0.001 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Dibenzofuran Polychlorinated dibenzofurans 2,3,7,8-Tetrachlorodibenzofuran 2,3,4,7,8-Pentachlorodibenzodioxin |

