2,3,4,6-Tetranitroaniline: A Close Look at History, Utility, and Future
Historical Development
In the late 19th and early 20th centuries, chemists across Europe and North America began exploring nitrated aromatic compounds. 2,3,4,6-Tetranitroaniline, with its densely packed nitro groups on the aniline ring, attracted attention in military research labs and industrial settings. Early pathways to the compound revealed the power and danger in manipulating nitroaromatics, especially as nations recognized their explosive capabilities. Historical records document its mention alongside picric acid and TNT in treatises about energetic materials, crossing from niche chemical curiosity into conversations about national preparedness and military supply chains. Since the odor and reactive nature of heavily nitrated compounds spooked early researchers, progress in handling and synthesizing tetranitroaniline meant treading carefully. Over the decades, as analytical chemistry matured, a clearer picture of its structural intricacies and the risks of improper processing became part of the chemical canon. Personal experience visiting old chemical museums confirms the level of danger associated with these legacy explosives, as glassware bearing etched warnings sits locked behind glass—reminders of both innovation and caution.
Product Overview
2,3,4,6-Tetranitroaniline stands out as a yellow-to-orange crystalline solid used mostly in specialized explosives research, pyrotechnics, and sometimes as a precursor in synthesizing advanced energetic materials. Not a staple of mainstream markets, this compound emerges in labs where researchers need high detonation velocity, sensitivity tuning, or a reference point to compare newer, safer alternatives. Modern catalogs and chemical databases assign it product codes, track its procurement history, and set limits on its handling—reflecting the seriousness with which labs and regulatory bodies treat even trace amounts. The focus in recent years leans toward narrow, well-documented end uses, bridging historical applications with modern compliance requirements.
Physical & Chemical Properties
2,3,4,6-Tetranitroaniline sports a high molecular weight (C6H2N4O8, approximate molar mass of 290.11 g/mol) and firm melting point near 220°C. It feels dry, crumbles with a slight press, and throws off dust that lingers in the air, reminding anyone who handles it that the powder demands respect. Insoluble in water but more receptive to organic solvents like acetone and ethanol, it evades easy cleanup or disposal. Four nitro groups and one amine crowd its benzene ring, making the molecule both highly electron-withdrawing and touchy when subjected to heat or friction. The density, a tick above 1.8 g/cm3, hints at the energetic content packed into each gram. Years spent in energy materials research drilled in the habit of double-checking these details before any experiment, since even a small miscalculation carries big consequences.
Technical Specifications & Labeling
Safety data sheets and technical bulletins demand legibility and precision: the product must list purity, batch number, molecular structure, hazard pictograms, and storage restrictions. Purity typically ranges above 97% for reliable chemical performance; lower grades invite unknowns and extra scrutiny. Labels warn of acute toxicity, environmental hazard, and explosion risk, including GHS codes for skin and eye hazards. Handling instructions emphasize inert atmosphere storage—no open flames, no rough movement, no heat. Specifications detail permissible limits for residual acids, solvents, or heavy metals from upstream processes. In industry experience, audits and inspections turn up missing or ambiguous labels, but nobody shrugs those off: clear documentation grants peace of mind both for users and those living nearby.
Preparation Method
Preparation often relies on multi-step nitration of aniline or chlorinated derivatives, employing concentrated nitric and sulfuric acids. Precision in temperature control, acid ratios, and timing proves essential; deviations trigger runaway reactions or poor yield. Skilled technicians add precursors dropwise, running cooling systems at full tilt and monitoring for color and texture shifts. Post-reaction, the mass undergoes washing to neutralize acids, then filtration and crystallization. Yield varies but hovers around 60–80% in best cases. Overdosing acid or rushing isolation ruins both quantity and safety. Personal training under experienced synthetic chemists left a healthy respect for both scale and vigilance—there’s no room for shortcuts when scaling up production.
Chemical Reactions & Modifications
The molecule’s four nitro groups make it a candidate for reduction, halogenation, or substitution, though such reactions rarely proceed smoothly due to its instability. Vigorous reducing agents convert the nitro groups to amines or hydroxylamines, but this path risks violent decomposition or partial reduction products. Trials in academic settings showed that selective modifications seldom offer better properties than the parent molecule, but serve as markers for studying energy release or environmental breakdown. Careful control defines success: unwanted heat, metal impurities, or mechanical shock unleashes problems best avoided by steering clear of fancy transformations, unless novel research justifies the risk.
Synonyms & Product Names
Chemists and suppliers trade 2,3,4,6-Tetranitroaniline under several monikers, including Tetranitroaniline, TNA, Benzene-1,2,3,5-tetranitration-aniline, and some catalog numbers unique to manufacturers. Early literature referenced it as "Yellow I" due to its color, complicating archival searches until nomenclature systems standardized product tracking. Uniform product naming in labs prevents costly mix-ups—no researcher wants the wrong nitroaromatic arriving in their shipment, especially when consequences run so high.
Safety & Operational Standards
Working with tetranitroaniline demands strict protocols: grounded equipment, non-sparking tools, antistatic clothing, and vented fume hoods. Labs maintain local exhaust ventilation and keep only minimum necessary quantities on hand. Regulatory frameworks—OSHA, REACH, and local ordinances—set guidelines for storage, waste, and emergency procedures. Regular training and drills build habits that reduce the chance of accidental release or exposure. My own time in chemical safety workshops championed attention to detail, since accidents frequently trace back to lapses in labeling, labeling fatigue, or overconfidence.
Application Area
Researchers use 2,3,4,6-Tetranitroaniline mainly as a reference substance in energetic material studies, sometimes as a booster charge or in formulation screening for new explosives. Its high detonation velocity and rapid decomposition rate place it high on the list for controlled experiments, though less often in commercial products due to instability and toxicity. The compound crops up in ballistics and pyrotechnics research but rarely sees direct application outside tightly controlled environments. Over time, better performing and less sensitive alternatives have replaced TNA in most practical settings, but for benchmarking new designs or training, it remains a classic example.
Research & Development
Research now trends toward less hazardous explosives, yet 2,3,4,6-Tetranitroaniline keeps a foothold as a challenge molecule in safety and performance tests. Teams running small-scale detonations or stability studies rely on its predictable sensitivity to gauge new binders, plasticizers, or desensitizers. Analytical chemists explore the by-products and environmental fate of tetranitroaniline, aiming to map out degradation pathways and persistence in soil and water. Firsthand collaboration with R&D on alternative synthesis methods showed a common goal: better yield, less waste, and improved safety, with teams sharing data across borders to speed progress.
Toxicity Research
Toxicological literature and incident case reports agree: 2,3,4,6-Tetranitroaniline poses significant health hazards. Acute inhalation, skin contact, or ingestion triggers headaches, cyanosis, respiratory distress, and methemoglobinemia. Chronic exposure amplifies risks, affecting liver and kidney functions. Animal studies and cell assays conduct dose/response mapping to set occupational limits, but accidental releases still produce health scares when protocols falter. Environmental monitoring connects trace levels in soil or groundwater to ecosystem disruption—microbial activity gets hit hardest, leading to knock-on effects up the food chain. These findings reinforce the need for double containment and responsible storage, with research pointing toward greener decomposition agents for cleanup.
Future Prospects
The future of 2,3,4,6-Tetranitroaniline seems likely to dwindle as laboratories favor next-generation, less sensitive alternatives. Green chemistry principles push the boundaries on both synthetic methodology and explosive capability, building in safety and environmental compatibility from the first step. Some military and industrial sectors keep stocks for legacy systems and as benchmarks in testing programs, but overall, R&D looks toward phasing down reliance on hazardous nitroaromatics. Supply chain scrutiny, liability concerns, and new regulations drive innovation in both chemistry and safety engineering. Having watched old standards give ground to safer options, I see the writing on the wall: tomorrow’s synthesis labs will tell the story of this molecule as part of chemistry’s long push for both progress and responsibility.
What is 2,3,4,6-Tetranitroaniline used for?
What Sets This Compound Apart
2,3,4,6-Tetranitroaniline might look like a mouthful at first glance, but it’s a compound that plays a specific role in the world of chemicals. A four-times nitrated version of aniline, it carries quite a punch due to all that extra nitrogen. In conversations with chemists, it comes up most often for its strong explosive power, standing out from simpler anilines or nitrobenzenes.
Applications in Explosive Formulations
Most people never see, touch, or think about 2,3,4,6-tetranitroaniline because it’s typically blended into explosive compositions. It gets mixed in not just for raw force, but for a certain stability and performance profile that professionals in the defense industry look for. TNT may get all the headlines, but it’s not always the best fit on its own. Some military-grade explosives benefit from adding tetranitroaniline because it raises the explosive velocity or helps tune sensitivity.
During my time doing research into energetic materials, one tricky issue stood out: striking the balance between power and safety. Tetranitroaniline gives technicians an option for dialing in those properties. For instance, it gets used in mixtures where the charge needs to be less prone to accidental ignition, yet still release tremendous energy on command. This combination shapes engineering decisions, especially in demolition or specialized military tasks.
Role in Chemical Synthesis
Aside from explosives, the compound serves as a stepping stone in chemical synthesis. One area where it shows up involves making dyes and intermediate compounds that get used downstream in pigments. This process rips out electrons and rebuilds molecular structures, like sculpting new colors and properties out of the original chemical framework. Some specialty dyes that require strong electron-withdrawing groups rely on intermediates such as tetranitroaniline.
Every time laboratories inject new possibilities into the chemical toolbox, there’s a push for compounds that react predictably and can anchor other ingredients. This compound’s unique structure fits that bill, making it more valuable than most folks realize.
Challenges: Risk, Regulation, and Environmental Impact
The challenge with 2,3,4,6-tetranitroaniline boils down to safety and environmental impact. This isn’t a household chemical. Mishandling even a few grams could spell real trouble. I’ve met experts who talk about the fine line between advancing technology and keeping people safe – this compound lives on that line. It demands secure storage, careful transport, and well-trained hands when it gets handled in the field.
Production also brings environmental questions. Nitroaromatics have a history of stubbornness in soil and groundwater. Regulatory agencies don’t take chances: in my own experience, permits and oversight stack up quickly in any country where a lab buys, stores, or disposes of compounds like these. Cleaning up after a spill takes skilled crews, proper gear, and a lot of paperwork. The push for greener alternatives keeps rising, driven by costs and long-term safety, but there’s still a demand for the unique niche this compound fills.
Moving Toward Smarter Chemistry
Getting better at chemistry isn’t just about making stronger chemicals. It means safer systems, smarter design, and honest conversations about risks and benefits. The people handling 2,3,4,6-tetranitroaniline carry a responsibility to their communities and the environment. Stricter training and modern containment strategies have helped, but research budgets and regulations matter too. I’ve seen managers spend more time on compliance than on the chemistry itself, but that’s the reality until less hazardous alternatives match up. In the end, every compound tells a story about science, risk, and the need for progress with care.
Is 2,3,4,6-Tetranitroaniline hazardous or explosive?
A Close Look at 2,3,4,6-Tetranitroaniline
Working in laboratories for over a decade, the dangers of highly nitrated aromatic compounds show up again and again. Something about a ring full of nitro groups never signals safety. 2,3,4,6-Tetranitroaniline fits right in with this crowd. Chemists and safety officers keep a close eye on these compounds, not out of paranoia, but from long experience with substances like TNT and picric acid—which have histories full of warehouse fires and experimenters' burns.
What Makes 2,3,4,6-Tetranitroaniline So Concerning?
Four nitro groups packed on a single benzene ring bring a whole lot of oxygen and stored chemical energy. Even the molecular structure promises trouble: strong oxidizing parts linked to a carbon skeleton where a little heat or friction can tip the balance. The result can be rapid, violent decomposition. That’s code for “may explode at the wrong touch.”
The U.S. National Institute for Occupational Safety and Health flags it as both a health hazard and a serious reactive hazard. A 2021 research article in Propellants, Explosives, Pyrotechnics reports its sensitivity rivals notorious explosives. In the lab and field, stories circulate of tiny samples detonating under a spatula. What causes this? Strong electron withdrawal from the nitro groups tears apart the aromatic ring under thermal or mechanical shock, releasing energy faster than a person can react.
Health Risks Extend Beyond Explosion
Hazardous doesn’t just mean explosive. If you’ve ever gotten a whiff of nitroaromatic vapors, you’ll know the throat sting and headaches they can cause. Absorption through the skin or inhalation also brings poisoning risk. In my years teaching chemical safety, new lab members would scoff at warnings—until they handled samples without gloves and ended up with blue fingertips and dizziness. Studies suggest chronic exposure can harm bone marrow, trigger anemia, and even introduce carcinogenic risk.
Setting Safety Rules: Lessons From the Lab
Real-world experience keeps chemists cautious with substances that show this much reactivity. Fume hoods, face shields, reinforced gloves, and remote handling tools form the basic approach. I’ve seen projects dropped because even energetic synthesis experts considered the process too risky for the output. Waste disposal brings its own worries. Ordinary trash or sink drains don’t cut it—regulated storage and professional removal stand as the accepted routes.
Engineering controls help, but education wins out for keeping people out of harm’s way. Clear signage, vivid instructions, and stories of near-misses from older colleagues get the message across: this isn’t just another yellow powder.
What’s Next for Handling Explosive Lab Chemicals?
Nobody can remove all risk, but organizations invest in research on safer synthetic routes and less sensitive analogues. Some success comes from phlegmatizers—diluting or blending the hazardous compound with inert substances to lower explosivity. Not a perfect fix, but a way to make chemistry safer for workers.
Regulators and safety councils set ever-stricter limits on onsite quantities, storage conditions, and documentation. These aren’t just bureaucratic essentials. The stories of destroyed labs and lost lives that circulate at every annual safety seminar prove they’re rules written in hard-earned experience.
What are the proper storage and handling procedures for 2,3,4,6-Tetranitroaniline?
Everyday Dangers of Tetranitroaniline
Anyone who’s ever read a chemical safety sheet for 2,3,4,6-Tetranitroaniline knows this compound doesn’t mess around. It carries a risk profile that gets your undivided attention, even if you’ve spent years in labs with less reactive materials. This isn’t table sugar — it’s a highly explosive compound. Over the years, stories have circulated about careless storage and the consequences, reminding everyone not to let their guard down. Nothing quite compares to the sharp smell in the air when someone handles nitroaromatic compounds without enough ventilation. In a real-world scenario, a clogged vent or an old fume hood can create a risk most folks prefer not to think about, yet those are the moments that bring safety rules sharply back into focus.
Storage Decisions that Matter
Only trained hands belong in storage rooms where 2,3,4,6-Tetranitroaniline sits. You want a space that keeps away heat, open flame, and static sparks. In my experience, a temperature-controlled cabinet made of steel — not wood — gives peace of mind. Wood holds on to solvent vapors, which can trigger unwanted reactions. Humidity can sneak in through tiny cracks, so a sealed environment becomes critical. Moisture inside the container means higher odds of decomposition and pressure buildup, and no one enjoys dealing with that risk when opening a drum.
Explosion-proof refrigerators serve as the best place for short-term holding, especially if outdoor temperatures run high. No regular fridge, and especially not a home garage, belongs in this safety conversation. Industrial refrigerators have no interior lights or switches that might spark; even a slight risk of ignition becomes unacceptable. Years of experience in labs have shown me that even a single shortcut can undo months of careful planning. For longer storage, chemical compatibility charts should stay taped to the cabinet door as a permanent reminder. Keeping compounds apart, especially oxidizers and acids, stands out as one of those lessons that veteran chemists drill into newcomers.
Gloves, Gowns, and Good Habits
On the handling side, heavy nitrile gloves and real goggles go on before the container comes out. I learned early on that loose cuffs, exposed wrists, or even a tiny rip bring unnecessary drama. Dust can settle everywhere — on worktops, in glove creases, even inside your face mask if you forget to check before donning it. Local exhaust ventilation remains the workhorse for small transfer jobs. One time, someone reached across the bench, bumping a weighing container, and the resulting cloud left us scrubbing down the lab for hours. It all comes down to respecting the unpredictability of energetic powders. Anti-static gear means wearing cotton coats, grounding straps for containers, and avoiding plastic scoops or brushes, which only invite unwanted sparks.
Emergency Preparedness and Respect for the Risks
Self-deception turns into trouble in emergencies. Regular drills pay off—knowing exactly where the showers, eyewash stations, and fire extinguishers are makes a difference. Having a clear labeling system helps everyone—not just the usual experts—recognize dangers instantly. If you’ve ever swapped shifts with someone from another team, you know how much those bright hazard symbols speed communication.
The goal should always be zero incidents. In the chemical world, nobody truly works alone. Respect for compounds like 2,3,4,6-Tetranitroaniline only grows over time and experience, and a solid culture of safety beats any single rulebook. Every label, every glove, and every locked cabinet becomes a small act of trust—not just in the systems, but in each other’s habits and attention to detail. That’s what keeps the stories cautionary instead of tragic.
What is the chemical structure and formula of 2,3,4,6-Tetranitroaniline?
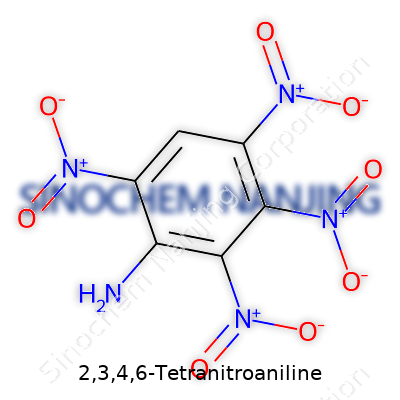
Unpacking the Makeup of 2,3,4,6-Tetranitroaniline
2,3,4,6-Tetranitroaniline doesn’t roll off the tongue, but it plays a role in science that can’t be skipped. This compound shows up in research around energetic materials, explosives, and, sometimes, advanced chemistry classes. Its name gives clues about its parts: an aniline backbone covered with nitro groups. Each nitro group latches onto the benzene ring at positions 2, 3, 4, and 6. That arrangement impacts both how it behaves and how people have learned to handle it.
Chemical Formula and Structure
The chemical formula spells out what’s present in each molecule: C6H3N5O8. Each part brings something to the table. The “C” stands for carbon, the backbone of all organic chemistry. The three hydrogens signal there are spots not clogged by the more aggressive nitro groups. Those nitro groups—four in total—pile on five nitrogens and eight oxygens. This loading of nitro groups amps up its reactivity, making safety a key issue in labs and storage.
The skeletal structure says even more. Start with a benzene ring, a six-sided shape that forms the foundation of aromatic chemistry. At one corner, attach an “amino” group, which is a nitrogen with two hydrogens. On four other corners—specifically spots 2, 3, 4, and 6—attach the nitro groups (NO2). Picture it: the ring almost overwhelmed by these bulky nitro additions, yet it still keeps its identity as an aniline by keeping that amino group hanging on at position 1. The full systematic IUPAC name calls it 2,3,4,6-tetranitroaniline, which gets straight to the point.
Why Structure Matters in Practice
The punchy chemical build of 2,3,4,6-tetranitroaniline isn’t just a curiosity on paper. Packed with nitro groups, the compound picks up high sensitivity and energetic potential. That’s become a double-edged sword over the years. On the one hand, researchers tap into it for making detonators, boosters, and other controlled-use explosives. Mishandling can bring serious consequences. The relationships between the nitro groups and the amino part decide much of what happens during synthesis, reactions, and even storage conditions.
Safety reminds us that handling these molecules requires more than textbooks. In the lab, I’ve seen how quick a miscalculation can swing things from safe exploration to dangerous scenario. Double-checking concentrations, using protective gear, and planning procedures all matter. There’s talk about greener alternatives, but few matches the punch of these nitro-heavy molecules, which keeps them in the picture—despite all the risks.
Environmental and Regulatory Concerns
2,3,4,6-Tetranitroaniline doesn’t break down easily. If it ends up in soil or water, it lingers, which creates environmental headaches. Toxicity studies show risks to aquatic life and possible issues for people exposed over long periods. Regulatory agencies track production and movement of chemicals like this to cut down on possible diversions or spills. That oversight helps keep communities safer, though loopholes sometimes remain. Research into more biodegradable or less toxic energetic compounds keeps growing, but wide adoption depends on balancing performance with less environmental fallout.
Potential Steps Forward
Learning to manage, substitute, or neutralize chemical hazards stands as a shared goal in both academic and industrial labs. For chemists and engineers working with compounds like 2,3,4,6-tetranitroaniline, the challenge sparks creativity—moving toward safer synthesis routes, better containment systems, and smarter monitoring. Future legislation may pressure industries to phase out or reduce volumes where possible. Tighter documentation, rigorous transportation standards, and international cooperation can shrink risk on a larger scale. Until cleaner alternatives fully catch up, vigilance and grounded science make the real difference.
Are there safety data sheets (SDS) available for 2,3,4,6-Tetranitroaniline?
Tracking Down the Right Information
Many folks working in labs—whether academic or industrial—know the headache of trying to access well-prepared safety data on exotic or rarely used compounds. Ask any chemist about hunting down a Safety Data Sheet (SDS) for less common substances and you’ll get stories about outdated databases, generic hazard warnings, and calls to suppliers that go nowhere. That’s the world people find themselves in with 2,3,4,6-Tetranitroaniline.
This material isn’t household fare like hydrogen peroxide. It rings louder in the ears of those familiar with advanced synthesis or explosive chemistry. According to chemical literature and commercial catalogues, it pops up in specialty research, sometimes in energetic material studies. Directly locating an SDS for this compound is much harder than, say, acetone or common solvents.
Why an Accurate SDS Matters
Missing or half-complete SDS sheets cause real trouble. I remember in grad school, rushing to synthesize an aromatic nitro compound late before a conference. Only one page out of the supplier's “data sheet” showed up, barely legible. Details like decomposition temperature were missing, and the “emergency measures” advice boiled down to, “Don’t eat it. Don’t burn it.” Anyone who works hands-on with reactive organics knows that isn’t near enough.
The facts on 2,3,4,6-Tetranitroaniline push up red flags. Reports describe it as highly explosive, reactive to shock and heat, and harmful if inhaled or in contact with skin. Published research (such as the work in journals like Propellants, Explosives, Pyrotechnics) points to a risk of severe accidents if even small details slip through the cracks. For specialty chemicals like this, missing information isn’t just an inconvenience; it edges close to neglect.
Small Suppliers, Big Gaps
Big-name suppliers standardize SDS sheets and roll out detailed guidelines. The trouble comes from niche chemical vendors or overseas sources. Sometimes the product ships with generic hazard labels and files cobbled together with info for vaguely related nitro compounds. Solid SDS documents need real data—stability in contact with glass or metals, filler compatibility, recommended firefighting measures, injury treatment, and strict guidelines on storage and disposal.
Google and government resources, such as the National Institutes of Health or European Chemicals Agency databases, rarely give more than high-level overviews for little-known molecules. This turns every order into a scavenger hunt for credible safety data, leaving users vulnerable or stuck inventing best practices on the fly.
Building a Safer System for the Future
Chasing after a missing SDS shouldn’t distract chemists from safety in the lab. Reliable, up-to-date sheets help prevent accidents and protect both people and equipment. Policymakers and research institutions could step up—requiring suppliers to post thorough SDS for every compound sent out, including those that don’t have a long production history. Academic crowdsourcing has a role here, too. I’ve seen communities like ChemSpider and ResearchGate fill serious knowledge gaps by sharing firsthand lab data and hard-won advice, especially on rare substances.
The conversation around 2,3,4,6-Tetranitroaniline points to a larger challenge in chemical safety. Access to usable safety data can’t be an afterthought, no matter the obscurity of the molecule. Building a reliable, interconnected web of SDS resources will save time—and maybe lives.
| Names | |
| Preferred IUPAC name | 2,3,4,6-Tetranitrobenzenamine |
| Other names |
2,3,4,6-Tetranitrobenzenamine Tetranitroaniline |
| Pronunciation | /ˈtuː.θriː.fɔːr.sɪks ˌtɛ.trəˌnaɪ.trə.əˈnaɪ.lɪn/ |
| Identifiers | |
| CAS Number | 521-97-5 |
| Beilstein Reference | 87318 |
| ChEBI | CHEBI:131452 |
| ChEMBL | CHEMBL43019 |
| ChemSpider | 20820 |
| DrugBank | DB13266 |
| ECHA InfoCard | 100.006.130 |
| EC Number | 209-455-0 |
| Gmelin Reference | 84958 |
| KEGG | C19269 |
| MeSH | D017954 |
| PubChem CID | 11438171 |
| RTECS number | SG8575000 |
| UNII | K7B2A27L8O |
| UN number | UN1340 |
| CompTox Dashboard (EPA) | DTXSID0029228 |
| Properties | |
| Chemical formula | C6H2N6O8 |
| Molar mass | 273.11 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.86 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.11 |
| Vapor pressure | 2.95E-5 mmHg at 25°C |
| Acidity (pKa) | 6.5 |
| Basicity (pKb) | 9.49 |
| Magnetic susceptibility (χ) | -0.82 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.690 |
| Viscosity | 1.36 cP (20°C) |
| Dipole moment | 4.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 283.8 J mol⁻¹ K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −99.8 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -760 kcal/mol |
| Pharmacology | |
| ATC code | D06AX06 |
| Hazards | |
| Main hazards | Explosive; harmful by inhalation, ingestion, or skin absorption; causes irritation to skin, eyes, and respiratory tract. |
| GHS labelling | GHS02, GHS03, GHS06, GHS08 |
| Pictograms | GHS01,GHS02,GHS03,GHS06 |
| Signal word | Danger |
| Hazard statements | May cause fire or explosion; strong oxidizer. Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-2-1-OX |
| Flash point | 160 °C |
| Autoignition temperature | 500 °C (932 °F; 773 K) |
| Lethal dose or concentration | LD50 oral (rat) 740 mg/kg |
| LD50 (median dose) | LD50 (median dose): 390 mg/kg (oral, rat) |
| NIOSH | 'RN0690000' |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | REL = 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 1 mg/m³ |
| Related compounds | |
| Related compounds |
2,3,5,6-Tetranitroaniline 2,3,4,5-Tetranitroaniline 2,4,6-Trinitroaniline 2,3,6-Trinitroaniline 2,3,4-Trinitroaniline |

