2,3,4,6-Tetrachlorophenol: A Down-to-Earth Look at a Persistent Chemical
Historical Development
Back in the industrial boom of the mid-20th century, chemists started paying serious attention to chlorinated phenolics. They saw that adding chlorine atoms to phenol unlocked new possibilities for controlling fungi and bacteria in wood, textiles, and leather. By the late 1940s, 2,3,4,6-tetrachlorophenol was showing up in laboratories, soon making its way into factories. It didn’t take long for this compound to earn a place in the toolbox of wood preservers and pesticide formulators. As years passed, researchers realized these compounds, once prized for longevity, didn’t just wash away or disappear. Knowledge about the risks began accumulating, but production hummed along in many places, helped by the growing demand for more durable building materials and pest control methods.
Product Overview
The stuff shows up as an off-white to pale brown crystalline solid. Chemists identified it as an organochlorine compound with significant antifungal properties. Its molecular formula, C6H2Cl4O, makes it one of those molecules that walk the line between helping preserve wood and raising environmental headaches. Markets tend to favor this compound where high-resistance to decay, mildew, and insect attack is considered crucial. It’s one of the go-to chemicals for both technical applications and academic research dealing with organochlorine persistence.
Physical & Chemical Properties
2,3,4,6-Tetrachlorophenol stands out for its low solubility in water and strong solubility in organic solvents. The melting point runs about 71-74°C, and it decomposes at higher temperatures, releasing sharp, acrid fumes. Odor usually smells medicinal, with a bite that lingers in the nostrils. Chemically, the molecule resists breakdown in the environment, partly due to all those chlorine atoms crowding the ring structure. You won’t see it vaporize easily, but it does travel through soil into groundwater and sticks around in sediments for years. This quality shapes most worries about it, as slow breakdown creates opportunities for environmental buildup.
Technical Specifications & Labeling
Commercial lots typically arrive with a purity of at least 98%, stamped with hazard labels that warn about skin, eye, and respiratory irritation, and the potential for organ toxicity after long exposure. Standard labeling under GHS (Globally Harmonized System) flags flammability and environmental dangers, requiring chemical-resistant gloves, goggles, and thoughtful storage well away from acids, bases, and food supplies. Manufacturers must list batch numbers, CAS numbers (58-90-2 for this compound), and expiry dates on all bulk drums. Safety Data Sheets come packed with warnings, some based on past cases of accidental exposure and chronic workplace illnesses among wood treatment workers.
Preparation Method
The most common approach starts with phenol and teams it up with chlorine gas under iron(III) chloride or similar catalysts. The factory process happens in closed reactors, where temperature and pressure get tightly controlled to manage both product yield and the risk of runaway reactions. Anyone who’s spent time around chemical plants will recognize the intense smell that leaks out, even through fume scrubbers. After reaching completion, extraction and purification remove unreacted chlorine and by-products, leaving a pale crystalline residue behind. Waste streams typically undergo neutralization with caustic soda, but leftover brines and tars still test positive for stubborn chlorinated organics. It’s not a pretty business, and strict oversight makes a difference in keeping environmental contamination in check.
Chemical Reactions & Modifications
2,3,4,6-Tetrachlorophenol reacts both in lab glassware and on the larger scale. It forms esters and ethers with strong alkylating agents, and under controlled conditions, it can get reduced back to lower-chlorinated phenols. Alkaline hydrolysis breaks it down slowly, and mild oxidants have little effect, but with the right reductants, it can lose its chlorine substituents to produce diphenyl ethers and other specialty chemicals. This resistance to chemical change explains its longevity—and complicates cleanup after spills. Researchers who fiddle with the structure mostly aim to find derivatives that either linger less or pose smaller risks to the environment.
Synonyms & Product Names
People working with chemicals know this one may go by several names depending on where they buy it or what regulatory system applies. Sometimes it’s just called Tetrachlorophenol, but chemists might list it as Phenol, 2,3,4,6-tetrachloro-. Other naming conventions, like Dowicide 6 and Compound 3964, may turn up in old documents. The repeated use of “TCP” or “Tetrafos” crops up in plant maintenance and legacy product catalogs, making it tough for non-specialists to keep track of usage across borders or decades. This confusion often complicates identifying the stuff at old industrial sites or reviewing legacy product formulations.
Safety & Operational Standards
Working around 2,3,4,6-tetrachlorophenol means paying attention to site protocols. Plant managers lock down supplies in sealed, ventilated storage areas and post clear signage. Training focuses on preventing both inhalation and skin contact, and operating staff suit up with gloves, full-face shields, and chemical-resistant aprons. Emergency wash stations never sit far from treatment tanks. International rules set occupational exposure limits based on chronic toxicity research, usually capping airborne concentrations at low parts-per-million levels. Routine air and surface monitoring catch leaks before they reach dangerous concentrations. Long-term use in wood preservation once exposed workers to much greater doses, but greater awareness and regulation over the last three decades put an end to many careless practices.
Application Area
This compound built its reputation in wood treatment, safeguarding telephone poles, railroad ties, and building timbers from rot and termites. Paint manufacturers and leather tanners also relied on it to keep fungi and bacteria from chewing up natural materials. Localized use persists in some countries for non-food crop protection and algicidal applications in cooling towers and industrial water systems. Environmental persistence soon moved it out of household disinfectants and put it on the watchlist for water quality experts. Old utility poles and preserved timber structures still leak out 2,3,4,6-tetrachlorophenol decades after original treatment, showing just how slow this chemical leaves the environment.
Research & Development
Chemists working on safer wood preservatives now target molecules that break down faster and don’t stick around in the food chain. Research focuses on developing microbial agents and plant-derived compounds to give lasting protection with fewer side effects. Environmental scientists monitor legacy contamination in soils and waterways, running tests to measure breakdown rates and searching for ways to remove residues with bioremediation or advanced oxidation processes. Applied research sometimes looks at capturing industrial emissions right where the compound forms, using filters or adsorbents to trap or degrade chlorinated phenols before they escape outdoor air or water streams. This work demands reliable detection methods, leading to new sensors and lab techniques.
Toxicity Research
Toxicologists learned early on that 2,3,4,6-tetrachlorophenol gets absorbed through skin and lungs, accumulating in fat tissue and organs over time. Animal studies show effects ranging from liver and kidney damage to reproductive toxicity, especially at high doses or over long periods. The EPA and other authorities classify this compound as a probable human carcinogen. Waste handlers, chemical workers, and families living near old wood treatment plants carry higher health risks. Some communities near creek beds or factory sites still face long-term effects due to groundwater and soil contamination. Reviews of past workplace illnesses show symptoms linked not just to the phenol itself but to dioxin impurities that form during production or breakdown. These lessons shaped occupational safety rules and soil cleanup standards worldwide.
Future Prospects
The days of widespread 2,3,4,6-tetrachlorophenol use look numbered as both regulation and technology move toward alternatives. Demand for greener building materials, paired with stricter product approvals, already forced many larger manufacturers to pivot away from persistent organic pollutants. Still, the remediation industry faces the slow, expensive task of cleaning up old industrial sites and timber yards. Scientists push for better detection of minute residues and speedier tar and soil cleanup, while industry looks at using enclosed reactor technology to handle safer substitutes. Product bans and international agreements, especially under treaties like the Stockholm Convention, mean legacy stockpiles will need careful disposal and tracking. Communities and regulators can expect more discussion about health risks, long-term remediation, and the transition to safer chemicals as the story of 2,3,4,6-tetrachlorophenol draws closer to an endpoint.
What is 2,3,4,6-Tetrachlorophenol used for?
A Chemical With a Tough Reputation
Walk into any discussion about wood preservation or pesticide history and the name 2,3,4,6-Tetrachlorophenol pops up. This chemical stands out as one of those chlorinated phenols folks have counted on to keep certain fungi and pests at bay. It’s used in wood processing plants and in the agriculture sector, especially before more modern solutions took the spotlight. Back in the day, you’d find stacks of lumber treated with this greenish compound. Farmers and woodworkers trusted its ability to stave off decay, termites, and a long list of fungal invaders.
Why Some Industries Hold Onto It
Certain pests just don’t stop for less potent treatments. If you talk to someone who’s spent years working to preserve railway ties, power poles, or docks, they’ll probably bring up this compound and others in its family. Its power lies in its knack for sticking to wood, blocking out rot, and making it a tough meal for hungry insects. For a long time, its lower cost, wide availability, and simple application gave it an edge over alternatives, especially for industries that couldn’t afford to experiment with less-proven solutions.
Real Risks at Work and in the Environment
Stories from workers who handled this stuff regularly open your eyes to the risks. Skin rashes and complaints about headaches used to be common. Exposure happens through the skin, lungs, and sometimes in tainted drinking water in communities close to plants using these chemicals. Research has linked this compound, as well as its chemical relatives, to nervous system problems, liver damage, and even cancer. Reports from the U.S. Environmental Protection Agency and the World Health Organization lay out these health concerns without sugar-coating. Wildlife and water quality also pay a price. In places where spills or runoff hit streams and soil, the effects can last for years. Fish, amphibians, and even birds take a direct hit. Many countries responded to these risks with restrictions or outright bans except in cases where no safer substitute works as well.
Finding a Better Path Forward
Talking about solutions brings up bigger questions. Why do some companies keep using a chemical known for long-term harm? Sometimes, it’s money. Modern preservatives can cost more and need different equipment. Regulations rarely move as fast as research, so older chemicals stick around longer than they should. Whenever I’ve spoken to friends in the lumber trade, their main concern is safety—nobody wants to risk their health, but they also can’t afford to replace expensive inventories overnight. Change happens when regulators step up, researchers share safer alternatives, and customers demand better. Some substitutes already show promise, like borate-based treatments or heat-treated wood. The lumberyards and hardware stores choosing to phase out the old solutions deserve real credit.
Knowledge Leads to Better Choices
Community leaders and industry pros need straight answers about chemicals like 2,3,4,6-Tetrachlorophenol, not jargon or downplayed risks. More open sharing about what these substances do, who’s most at risk, and where contamination happens leads to smarter decisions. People working with treated wood benefit from protective gear and better training, but stronger, enforceable policies make the biggest difference. Families living near old treatment sites deserve cleanups and clear communication. Caring about these details protects health and helps everyone move toward safer, more sustainable practices.
Is 2,3,4,6-Tetrachlorophenol hazardous to health?
Getting Familiar With the Chemical
2,3,4,6-Tetrachlorophenol doesn’t pop up in dinner table conversations, but it lurks in a lot more places than most folks realize. Used as a disinfectant, wood preservative, and pesticide ingredient, it clings to soil, water, and dust. Decades ago, lumber yards and power plants leaned on it to fight mold, fungi, and pests. Even now, traces turn up in the environment—rivers, playground dirt, and even inside homes.
Risks in Real Life
My experience covering industrial pollution taught me to recognize the warning signs early. If a chemical has “chloro” in the name, pay attention. As with many chlorinated phenols, 2,3,4,6-Tetrachlorophenol sticks around for ages and doesn’t just break down quietly. The U.S. Environmental Protection Agency found it hangs out in soil and water—and makes its way into food chains. This stuff travels, even turning up in groundwater after a heavy rain or leaching out of old building materials.
Health Hazards
Direct contact triggers rashes, eye irritation, and headaches fast. If you get exposed over months or years, you’re looking at bigger problems. Scientists have pointed to liver and kidney issues, along with immune system impacts. Animal testing links it to organ damage and issues in the blood. The real gut punch comes from research showing it may trigger cancer after long-term exposure. In several studies, rats exposed to this chemical ended up with liver tumors. Based on that, health agencies in the U.S. and overseas flag it as a possible human carcinogen.
Workplace and Community Exposure
In many smaller towns, abandoned sawmills and factories left behind contaminated soil. Workers faced the highest risks—breathing sawdust, handling treated wood, or washing hands where dust settled meant direct contact day after day. People living near old dumps and woodyards tell stories of garden veggies coming up with brown spots and ground smelling odd after a rain. Even folks miles away noticed foul odors from contaminated water, which stuck around long after factories closed.
Why Action Matters
Ignoring chemicals like 2,3,4,6-Tetrachlorophenol comes with a cost. Kids playing in dirty yards, neighbors drinking well water from old industrial lots, and cleanup crews working without good gear—everyone pays the price. Regular blood tests and environmental monitoring help spot problems before they hit boiling point. Following best practices for removing or capping contaminated soil, using proper personal protective equipment, and enforcing strict limits for chemical use makes a big difference. Don’t let old habits set the stage for cancer clusters and mystery symptoms. The science doesn’t lie: this chemical causes harm, and prevention always beats treatment.
Practical Solutions To Reduce Risk
Communities dealing with historic pollution can team up with local universities to test soil and water. Homeowners should check if their properties sat on old industrial sites before letting kids dig in the dirt or plant food gardens. Simple fixes, like planting grass or mulching bare ground, reduce dust and cut exposure. At work, wearing gloves, using respirators, and keeping workspaces clean can stop most issues before they start. Better oversight from local and federal agencies keeps companies honest, especially around hazardous sites. No one wins by sweeping problems under the rug, so real progress begins with good information and stubborn vigilance.
What are the storage and handling precautions for 2,3,4,6-Tetrachlorophenol?
Respecting the Hazards in Everyday Practice
Anyone who has worked in a chemical lab or warehouse knows some substances create problems just by sitting there. 2,3,4,6-Tetrachlorophenol belongs squarely in that camp. It gives off a pungent odor that stings the nose and eyes, and skin contact can cause burns or rashes before you know it. I once saw a warehouse technician neglect gloves — it only took a day before blistering forced a trip to the clinic. Simple mistakes compound quickly around chemicals like this.
Physical Storage: Practical Choices Make All the Difference
Storing this compound poses real challenges. Heavy-duty containers, sealed tight with proper gaskets, stop leaks and block vapors. At an old facility, I helped switch standard plastic drums to lined metal containers after a spill corroded shelving. That swap saved a lot of headaches. Don’t let it sit near strong heat sources or in sunlight. In my experience, even a few hours of hot warehouse air can warp containers and release fumes.
It makes sense to chain containers or use containment pallets. A leaking drum on a cracked warehouse floor once sent cleaner running into a drain — we spent hours with local authorities tracking the mess. Strong, chemical-resistant floors and secondary catch basins cut this risk. These investments pay off, both for safety and peace of mind.
Labeling and Segregation
Communication stops accidents before they start. Large, bold hazard symbols and clear labels wake up anyone passing by. Segregating stockpiles, at least fifty feet from oxidizers, concentrated acids, or flammables, reduces unwanted reactions. I’ve walked through storerooms where chemicals crowded one another — one small splash, and suddenly alarms go off. Clear zones and regular checks prevent surprises.
Handling: Don’t Rush the Process
Protecting yourself starts with basic gear: heavy gloves, goggles, long sleeves, and a fitted mask. Once, I underestimated a fume — tight spaces without ventilation magnify the effect; even brief exposure triggered days of headaches and coughing. It pays to use fume hoods or work out in the open, with airflow directed away from you. Set up eyewash stations where people can reach them in seconds. Speed rarely beats caution. Filling containers too fast leads to dangerous spills, so measure and pour with a steady hand and full attention.
Training and Response Plans: Staying Ready for the Unexpected
I’ve seen quick thinking stop panic in its tracks. Training everyone who enters a store room, from janitors to technicians, closes weak points. Practicing spill drills every quarter, even if it seems tedious, prepares folks for real emergencies. Updated Material Safety Data Sheets posted nearby give everyone the facts they need in a hurry.
Disposal: Closing the Loop Safely
Getting rid of leftovers or contaminated items isn’t just paperwork. Many waste handlers won’t even touch drums without visible proof of containment and documentation. Once, a disposal company walked away because lids weren’t sealed and the forms were incomplete, leaving us in a bind. Triple-check closure, labeling, and logs before calling for pickup. Using licensed contractors and following local hazardous waste laws keeps you out of regulatory trouble — and out of the headlines.
Every Step Matters
Nothing beats taking the long view with 2,3,4,6-Tetrachlorophenol. Each detail — from keeping gear handy to double-checking containers before heading home — makes a difference. Ignoring little things only invites big problems later. Speaking from long hours around chemicals, treating each step with respect is the only real shortcut.
How should accidental exposure to 2,3,4,6-Tetrachlorophenol be treated?
Recognizing the Risks
2,3,4,6-Tetrachlorophenol is not something most folks come across every day, but it can show up in certain pesticides and wood preservatives. The tough thing about this chemical—like many chlorinated phenols—is the speed at which the body absorbs it, whether through skin, lungs, or gut. Exposure sometimes starts with a sharp, unpleasant odor or a burning feeling wherever it touched skin. Not everyone reads labels on the bottles or drums they find themselves handling, especially in high-pressure work environments. That’s how it happens: an accidental splash, spilled powder, or vapors in the air.
What to Do Right Away
No one benefits from hesitation after an exposure. Experiencing this firsthand while working at an industrial site taught me how panic only wastes precious seconds. Remove contaminated clothing as fast as possible; the chemical keeps soaking in otherwise. If it’s skin contact, rinse the area under running water—cold, not hot—because hot water could open skin pores, driving more toxin in. Take time. A quick rinse won’t cut it. Go for at least 15 minutes, scrubbing gently with unscented soap if available. Splashing water on your face isn’t enough; irrigate the eyes with clean water for a quarter hour if any gets in, even if they don’t sting at first. These are the same steps outlined by resources like the CDC and Poison Control.
Breathing Trouble
Being around the vapors, some people cough, some get dizzy. Air out the space fast—open doors and windows. If trouble breathing starts, move outside and get help. Don’t tough it out. Chlorophenol fumes attack the lungs, and shortness of breath hints at something more serious going on inside. Friends or coworkers nearby also need to avoid breathing the same air—secondary exposure spreads fast and matters just as much as the original accident.
Why Medical Attention Matters
Letting things slide is tempting. I’ve seen it: folks laugh it off, only to feel sick hours later. Chemicals like this don’t always do their worst right away. Swallowing even a small amount—intentional or not—can hit the liver, kidneys, and nervous system before lunch. Heading straight to the doctor or emergency room counts as common sense, not overreacting. Doctors can start treatments that move the toxin out of the body, maybe using activated charcoal or intravenous fluids. Blood tests help spot organ trouble before it’s too late. Without real treatment, complications can stack up: fever, sweating, low blood pressure, even seizures.
Prevention and Safer Practices
Accidental exposure often traces back to missing gloves, cracked goggles, or workspaces lacking clear labels and instructions. Taking five minutes at the start of a shift to check gear made all the difference for me and my crew. If a chemical smells bad or looks suspect, double up on protection. Keeping emergency showers, eyewash stations, and first aid kits in sight, not locked up in an office, means people actually use them. Ongoing training with real-life examples sticks much better than reading out loud from a manual. Telling stories from the field gets people’s attention in a way technical specs never do.
Community Education
Hazardous chemicals don’t stay on the job. Home workshops, storage sheds, even garden centers sometimes carry products with these ingredients. Schools and community groups can offer short info sessions about safe chemical handling. I’ve done one at a local library where we practiced what to do if something got in our eyes. Giving people steps to act quickly—removing clothes, rinsing properly, seeking help—means they stand a chance when things go wrong.
Big Picture
No one prepares for an accident by assuming it won’t happen. Acting fast, knowing the right steps, and respecting the danger of chemicals like 2,3,4,6-Tetrachlorophenol gives people and communities a much better shot at staying safe and healthy.
What are the environmental impacts of 2,3,4,6-Tetrachlorophenol?
Not Just Another Chemical—Real Worries with Real Consequences
Digging into the story of 2,3,4,6-Tetrachlorophenol means thinking about more than a line on a safety data sheet. This compound, known for its use in wood preservatives and pest control, isn’t just a leftover from an old job in the chemical plant. It leaves marks that show up far outside the lab—on soil, water, and health.
The Path It Takes—And Where It Ends Up
Chemicals like this one don't simply stay put after use. Small but steady leaks, spills, or careless disposal allow them to seep into soil and waterways. Once in the environment, it sticks around. Its chemical structure resists natural breakdown. Scientists tracking river and groundwater samples often bump into traces long after the original source dries up. In local communities, questions start to build: what’s in the well, and how far has it gone?
Life at Risk: Not Just Numbers or Lab Rats
People hear about toxicity charts and reference doses, but the impact lands in the lives of wildlife and communities. Fish and amphibians in streams pick up this persistent chemical. It disrupts how their bodies work, affecting reproduction and survival. If the food chain picks it up, birds and mammals start showing effects, too. I’ve seen local anglers get worried after reading warnings about eating their catch when chemicals like this get on the list.
Human Health Isn’t Just a Statistic
Some towns near contaminated sites start noticing odd health issues—long-term exposure can lead to skin problems, liver damage, even cancer. The International Agency for Research on Cancer categorizes it as possibly carcinogenic to humans. People start wondering if their tap water is part of the problem. Being honest, once a chemical like this shows up in local news, trust disappears fast.
The Buck Doesn’t Stop with Industry
It’s too easy to point blame at chemical factories and walk away. Strong regulatory action helps prevent new leaks and tighten up disposal. But old contamination sticks around. Clean-up projects cost money and time. It means digging out contaminated soil, specialized filters for groundwater, and regular checks. Relying on the idea that “dilution is the solution to pollution” doesn’t fly; the problem lingers unless someone digs in and solves it.
Choices and Solutions—Turning Worry into Action
A responsible approach needs eyes wide open to legacy pollution and future risks. That means enforcing strict controls on use, transparent tracking of where chemicals travel, and giving a voice to affected communities early. As a regular person, you watch for those “hazard: keep out” signs and wonder what else you’re not being told. Community involvement means more than public hearings; it’s about making information clear and accessible.
Safer substitutes for wood preservation, tighter container labeling, and tougher penalties for illegal dumping give real teeth to environmental protection. Companies need to remember: once these chemicals escape, they don’t just vanish—someone pays the cost, whether it’s a cleanup crew or the next generation fishing in once-safe waters.
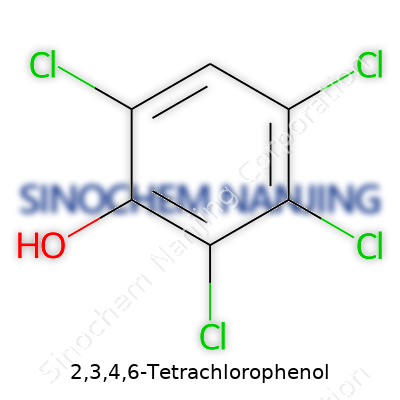
| Names | |
| Preferred IUPAC name | 2,3,4,6-Tetrachlorophenol |
| Other names |
Phenol, 2,3,4,6-tetrachloro- Tetrachlorophenol Dowicide 2S Nipacide TCP |
| Pronunciation | /ˌtɛtrəˌklɔːrəˈfiːnɒl/ |
| Identifiers | |
| CAS Number | 58-90-2 |
| 3D model (JSmol) | `3D structure; JSmol: C1=C(C(=C(C(=C1Cl)Cl)Cl)O)Cl` |
| Beilstein Reference | 1209283 |
| ChEBI | CHEBI:34660 |
| ChEMBL | CHEMBL1547 |
| ChemSpider | 7928 |
| DrugBank | DB11454 |
| ECHA InfoCard | 03b9527a-8995-4653-ab3d-c2cce5adaba0 |
| EC Number | 202-489-5 |
| Gmelin Reference | 120232 |
| KEGG | C02361 |
| MeSH | D013609 |
| PubChem CID | 6621 |
| RTECS number | SN1575000 |
| UNII | Z37H8Y08D8 |
| UN number | UN2468 |
| CompTox Dashboard (EPA) | DTXSID9020713 |
| Properties | |
| Chemical formula | C6H2Cl4O |
| Molar mass | 233.89 g/mol |
| Appearance | White to light tan crystalline solid |
| Odor | Phenolic odor |
| Density | 1.685 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 3.3 |
| Vapor pressure | 0.00016 mmHg at 25°C |
| Acidity (pKa) | 6.08 |
| Basicity (pKb) | 7.86 |
| Magnetic susceptibility (χ) | -0.00062 |
| Refractive index (nD) | 1.620 |
| Viscosity | 1.5 cP (50°C) |
| Dipole moment | 2.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 282.4 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -259.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1299.9 kJ/mol |
| Pharmacology | |
| ATC code | D08AE06 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes severe skin burns and eye damage, may cause respiratory irritation, toxic to aquatic life. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS05,GHS09 |
| Signal word | Warning |
| Hazard statements | H301, H315, H317, H319, H332, H334, H335, H341, H351, H373, H400, H410 |
| Precautionary statements | P260, P264, P270, P271, P273, P280, P301+P330+P331, P302+P352, P304+P340, P305+P351+P338, P310, P321, P330, P363 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 70 °C (closed cup) |
| Autoignition temperature | 150 °C (302 °F; 423 K) |
| Lethal dose or concentration | LD50 oral rat 700 mg/kg |
| LD50 (median dose) | 676 mg/kg (rat, oral) |
| NIOSH | PC8575000 |
| PEL (Permissible) | 1 mg/m3 |
| REL (Recommended) | REL (Recommended Exposure Limit) for 2,3,4,6-Tetrachlorophenol: "1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 10 mg/m³ |
| Related compounds | |
| Related compounds |
2,3,5,6-Tetrachlorophenol 2,4,5-Trichlorophenol 2,4,6-Trichlorophenol 2,3,4-Trichlorophenol Pentachlorophenol |

