2,2-Dinitropropane: A Deep Dive into Its Story, Properties, and Impact
Historical Development
Anyone digging through the history of modern chemical industry will notice certain compounds pop up again and again, not because they draw headlines but because of the subtle weight they carry in technical circles. 2,2-Dinitropropane is one of those. Early research dates back to the expansion of nitro chemistry, right after the major European developments in the 19th century. By the time chemical synthesis matured into the twentieth century, scientists began looking further into nitrated hydrocarbons, hoping for material with sharper performance or cleaner reactivity. For decades, it existed quietly in the background as chemists tinkered with more complex molecular designs, but those early years of discovery set the stage for its later use. In labs and industrial settings, some noticed that compounds like 2,2-Dinitropropane offered both challenges and advantages, and that legacy continues in workbench conversations even today.
Product Overview
2,2-Dinitropropane, a compound with a straightforward skeletal structure and two bold nitro groups, stands out not through commercial fanfare, but from careful inclusion in niche applications. In the world of organic intermediates, fans of synthetic routes or energetic material know it as a tool for specific reactivity, or as a bench curiosity. Its understated profile means it doesn't show up on household chemical rosters or casual shopping lists. Although not mass-produced on the enormous scale of bulk solvents or fuels, it occupies an important spot for chemists seeking a controlled, reproducible source of nitrated propane derivatives.
Physical and Chemical Properties
2,2-Dinitropropane presents itself as a pale liquid, clear and convincing in purity, not relying on color to warn users about potency. A distinctive, piercing nitro odor signals caution, hinting at its energetic lineage. The compound boils at a temperature well above that of its simple propane parent, a direct consequence of its stacked nitro groups. Those same functional groups lend it a higher density and increased reactivity. A major point for anyone working with this compound involves its relative stability; compared to more sensitive nitroalkanes and nitroalkenes, 2,2-Dinitropropane won’t explode easily under mild conditions, though reckless handling flips the safety calculus quickly. It resists casual hydrolysis but surrenders its functional groups under strong basic or nucleophilic attack, opening pathways to more complex chemistries.
Technical Specifications & Labeling
Labeling legal requirements for compounds like 2,2-Dinitropropane have grown tighter throughout the years, shaped by safety concerns and regulatory shifts. Container labels flag it as both toxic and a potential fire hazard. Lots ship with clear hazard pictograms, precautionary statements, and essential use and disposal guidelines. Material safety documentation now rides alongside every legitimate sale, reflecting lessons learned from decades of chemical mishap. In practice, most facilities keep detailed logbooks for storage and handling, documenting batch origin, quality assessments, and personnel signatures. Only those with real chemical literacy get access, usually after a mountain of training and oversight.
Preparation Method
Production of 2,2-Dinitropropane doesn’t demand rare minerals or science fiction technology but does require careful control. The classic route starts with propane or a propanone precursor and deploys nitration conditions sturdy enough to bolt on two nitro groups at the right carbon. Industrial scale-up brought improvements in process containment, heat management, and waste neutralization. Laboratories focus less on speed and more on controlled yields and manageable scaling, often choosing batch over continuous processes. Concerns like off-gassing, containment breaches, or side product generation drive choices here, because mistakes cost both money and safety. After synthesis, purification becomes a must—any leftover acid or explosive impurity drastically raises storage risk. That purification demands patience, glassware, and a clock watcher’s discipline.
Chemical Reactions and Modifications
Any chemist who has handled 2,2-Dinitropropane recognizes a compound hungry for transformation. Both nitro groups open doors for reduction, substitution, and cyclization reactions. Under strong reducing conditions, the molecule surrenders its nitro groups to amines, or refashions them for use in heterocycle production. Its resonance and electron-withdrawing nature change how nearby atoms behave, which attracts synthetic researchers who enjoy tuning reactivity by leveraging these effects. In research, its predictable reactivity gives both comfort and creative possibilities when mapping out synthetic routes to more complex molecules, especially in pharmaceuticals or advanced polymers.
Synonyms and Product Names
No matter the setting, 2,2-Dinitropropane rarely parades under its full IUPAC title. Chemists in the thick of a project drop the tongue-twisting full name for quick alternatives such as “DNP” or the snappier “2,2-DNP.” These names carry through logbooks, labels, and email threads. It also shows up in some texts as “dimethylnitroethane” or “propanedinitro,” keeping things concise during a fast-moving discussion or a hasty handwritten note passed between benches. Rarely, obsolete synonyms materialize in archival research, which can trip up newcomers who haven’t memorized the jargon’s historical drift.
Safety and Operational Standards
In any serious facility, safety stands at the front of handling operations for chemicals like 2,2-Dinitropropane. Direct skin or eye contact demands immediate response, and well-trained staff know better than to breathe its vapors. Proper storage favors cool, dry places, away from shock or stray sparks, emphasizing the value of both chemical literacy and discipline. Regulations by authorities like OSHA, REACH, or local industrial codes set firm boundaries for use, disposal, and emergency response. No shortcuts get taken, because even minor mistakes with dinitro compounds can turn a routine operation into a headline-making mess. High-quality detection equipment, secondary containment, and frequent safety drills—none of these are optional for responsible operators. Anyone cavalier about safety doesn’t last long in these environments, and for good reason.
Application Area
2,2-Dinitropropane holds its own in a narrow but persistent constellation of uses. Synthetic chemistry claims the most interest, where it serves as a launching pad for more complex nitro derivatives, or as a controlled reactant in energetic materials research. Some applications exploit its relatively manageable stability and unusual electron dynamics as a way to push boundaries in advanced material design. In academia, its use as a study molecule—either for kinetic analysis or structure–reactivity investigations—remains steady, even as new compounds hit the scene. Despite its niche character, it has helped move forward technologies touching everything from specialty polymers to detonable mixtures used in science and engineering education.
Research and Development
Scientists drawn to 2,2-Dinitropropane tend to be those accustomed to navigating the line between theoretical promise and laboratory risk. Over the past generation, researchers have tried to stretch the molecule’s potential, both to eke out new reactivity and to probe safer handling practices. Efforts to tame and repurpose nitro compounds depend heavily on detailed mechanistic studies—how electrons move during a given reaction, which byproducts form, what conditions flip a reaction from safe to dangerous. Recent years have seen collaborations spanning between synthetic chemists, toxicologists, and safety engineers, who look for ways to use the molecule’s unique features without exposing workers or the environment to unnecessary danger. Some groups test modifications to the basic framework, swapping functional groups or molecular neighbors, hunting for productive novelties or safer substitutes.
Toxicity Research
Toxicity represents the heaviest shadow over the practical appeal of 2,2-Dinitropropane. Researchers, even those charmed by its synthetic range, cannot ignore its health risks. Uptake through skin, inhalation, or accidental ingestion puts people at risk for everything from headaches and nausea to more severe organ effects. Nitro compounds share a reputation for oxidative stress on cellular machinery, with dinitropropane drawing added scrutiny due to its molecular weight and volatility. Regulatory reviews draw on animal data, chemical analogs, and real-world accident investigations to set exposure limits and recommend robust engineering controls. Long-term epidemiological studies remain thin—this is no household chemical, after all—but the sector takes toxicity seriously, knowing that respect prevents tragedy.
Future Prospects
Looking to the future, the path for 2,2-Dinitropropane lies in refinement and caution. Creative synthetic scientists will keep turning to it as a dependable intermediate, especially if safer or more sustainable processes emerge. As regulatory scrutiny grows sharper and green chemistry picks up steam, there’s real pressure to balance utility with health and environmental safety. Alternatives may eventually eclipse it in some applications, but those looking for its special blend of reactivity, manageability, and niche performance seem unlikely to abandon it overnight. The molecule’s story offers a snapshot of the modern chemical industry: blending progress and precaution, technical know-how and human experience, in pursuit of better and safer science.
What is 2,2-Dinitropropane used for?
Background Behind Its Chemical Roots
2,2-Dinitropropane’s chemical composition makes it stand out in the world of organic compounds. The nitro groups attached to its molecular structure create a powerful punch most folks outside specialty labs never come across. This isn’t a household chemical; its nature means it belongs in properly equipped environments and managed by those with expertise.
Industry Applications: More Than Just a Niche Additive
Certain industrial players turn to 2,2-dinitropropane as a fuel additive. The explosive properties offer value to the formulations of dynamite and blasting agents. I’ve seen this firsthand walking the floors of chemical manufacturing sites. Safety protocols sit high on everyone’s checklist any time these compounds enter the mix. You don’t find it in ordinary gas blends at local stations. Instead, it gets reserved for special formulations where controlled energy release matters.
Another place this substance shows up is in the production of resins and adhesives. Neighboring compounds often struggle with stability when exposed to extreme temperatures. 2,2-Dinitropropane’s resilience in these cases can help manufacture specialty binders—think about adhesives for high-performance automotive or aerospace uses. These aren’t the simple glues in office drawers; they hold together things where lives often ride on the integrity of every bond.
Risks Tied to Storage and Handling
Strong chemicals carry strong risks. Nobody treats 2,2-dinitropropane lightly. Exposure can lead to serious health effects, ranging from dizziness and headaches to respiratory issues or even more dangerous fallout if handled carelessly. If a chemical plant stocks this on-site, strict safety rules follow: personal protective equipment, restricted access, proper ventilation. Even a small slip comes with consequences.
Leaning on my time in occupational health, I’ve watched the best programs drill safety procedures constantly. Chemical awareness saves lives. Having an action plan for leaks, spills, or accidental exposure is just good sense. When mixing or blending this with less reactive compounds, the crew always double-checks measures, knowing that even a tiny error carries greater risk than with standard industrial chemicals.
Why Public Knowledge Matters
It’s easy for folks to gloss over the presence of rare chemicals like this. Out of sight, out of mind—unless something goes wrong. Clear labeling, employee education, and proper disposal channels don’t just serve as red tape. These steps help keep surrounding neighborhoods safe, prevent disastrous leaks into water or soil, and sidestep potential legal trouble.
The regulatory environment takes these risks seriously. Laws from groups like OSHA and the EPA keep a close eye on how places use and store nitro-based chemicals. That tight oversight helps make sure companies keep their communities and workers out of harm’s way while pushing technology forward.
Room for Better Solutions
Chemists continue to search for safer alternatives. Sometimes, that means swapping to compounds that carry less environmental baggage or developing new methods for achieving the same results with smaller quantities. In the end, every advance offers a chance to reduce exposure risks and deliver safer processes for vital industries.
Is 2,2-Dinitropropane hazardous to health?
Getting Real About Chemical Safety
Chemicals with names like 2,2-dinitropropane often seem distant from daily life, yet their impact sneaks into more places than you might guess. This chemical, used in explosives research, laboratories, and certain chemical syntheses, carries health risks that can’t be ignored just because most people don’t knowingly come into contact with it. Real safety for workers and communities means digging into what these risks look like and how to lower them.
Health Hazards Worth Noticing
Small accidents with strong industrial compounds sometimes lead to big problems. 2,2-Dinitropropane, with its two nitro groups, stands out because it’s both flammable and toxic. Breathing in dust or vapor from this compound can irritate your airways, trigger headaches, and bring on nausea. Research and reports suggest that extended exposure harms organs—particularly the liver and kidneys. Some studies on animals point to possible damage to the nervous system as well.
If the stuff touches your skin or gets in your eyes, burning, itching, and redness can follow. Spills increase the chance for chemical burns. Over time, chronic low-level exposure has been linked with weakened immune response and liver stress. Not everyone will feel symptoms right away, but those who handle this compound in factories or labs learn fast that sloppiness leads to real pain.
Exposure Doesn’t Stay in the Factory
Handling or storing 2,2-dinitropropane brings risks not just to workers but also to neighbors. Fires or leaks can send hazardous fumes drifting beyond industrial fences. Improper storage sometimes leads to explosions. Reports from chemical plants show that lapses in vigilance—like missing ventilation or faulty handling protocols—can turn one bad day into a cleanup nightmare.
I’ve seen the anxious faces of coworkers following a small lab spill. Those moments drive home why training goes beyond paperwork. Keeping the risk theory alive means clear drills, good emergency gear, and accessible spill kits. Off-site, quick notification and honest communication let neighborhoods judge real risk and push for change when necessary.
Lessons from the History of Chemical Regulation
Stories from the past show how learning about chemical dangers lags far behind developing new compounds. The science community recognizes 2,2-dinitropropane’s hazards because accidents left marks on workers’ health decades ago. Older safety data sheets list targets like liver and kidney damage long before tough regulations caught up.
Tighter laws protect most people working with hazardous chemicals. OSHA and ACGIH, for example, set workplace exposure limits and recommend tight controls for nitro compounds. Still, enforcement isn’t perfect. Monitoring, regular testing, and keeping safety data visible matter much more than a binder on a shelf.
Lowering the Risks—Hands-On Solutions
Wearing gloves, goggles, and using good fume hoods aren’t optional around 2,2-dinitropropane. At work, people can ask for air testing and push for clear emergency steps. Disposal of waste matters—a few shortcuts lead to big fines and major health scares.
For people outside the plant, strong rules, clear labeling, and honest accident reports build trust. Supporting worker voice, whistleblower protections, and neighborhood watch groups does even more to keep corners from being cut.
Managing the risks linked to 2,2-dinitropropane keeps lives safer. Strict attention, honest sharing of hazards, and steady safety habits make the difference, both inside the lab and out in the community.
What is the molecular formula of 2,2-Dinitropropane?
Understanding 2,2-Dinitropropane
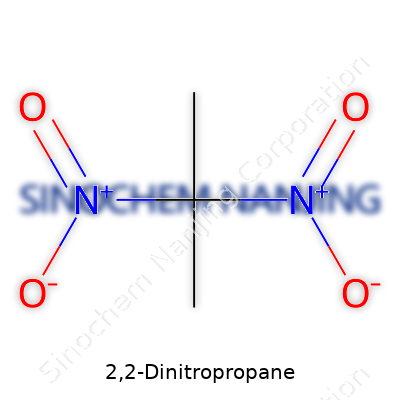
Chemistry teachings never really leave a person who’s spent a few afternoons in a lab, especially those moments puzzling over the structure of compounds like 2,2-dinitropropane. Stripping away the jargon, this substance brings together three carbons, six hydrogens, and four nitrogens, thanks to the two nitro groups attached at the second carbon on the propane chain. That leads directly to the molecular formula: C3H6N2O4.
Why Molecular Formulas Matter
It’s tempting to shrug off a formula as just numbers and letters on paper, but experience working with chemicals turns those symbols into much more—guides to safety, tools for efficiency, even clues about risk. Not knowing the formula of what you’re handling sets folks up for trouble, especially with energetic materials like dinitro compounds. That isn’t academic nitpicking; it plays into real-world consequences. The wrong mixture, an unexpected reaction—one small oversight costs lives and leaves a mess most clean-up crews would rather avoid.
Handling Risk Where it Counts
Having worked with a range of chemical stocks, the need for absolute clarity jumps out. 2,2-Dinitropropane, with its nitro groups, doesn’t just bring theoretical reactivity. Its formula communicates potential instability, signaling chemists not to treat the compound like a benign organic addition. Take a look at historical factory mishaps and the lesson sticks: precise molecular knowledge saves jobs, property, and sometimes the entire business.
The Path from Formula to Real-World Impacts
Beyond the lab, formulas like C3H6N2O4 feature in industries from explosives manufacture to specialty reagents. Workers get a crash course in why following chemical formulas to the letter keeps operations smooth. People who overlook those C’s, H’s, N’s, and O’s might find themselves staring down a barrel of government fines or, far worse, a hazardous event nobody wants to see replayed on the news.
Facts on Use, Safety, and Disposal
The facts line up: compounds with more nitro groups elevate reactivity scores. That places 2,2-dinitropropane in the same family as other sensitive materials, though it doesn’t commonly enter the public markets like some bigger explosives. Laboratory safety manuals and manufacturing standards reference its molecular signature, not just in research papers, but on every shipping label and mixing guideline.
The U.S. Environmental Protection Agency and OSHA highlight precise identification as a requirement, not a suggestion. This ensures tracing spills, understanding environmental impacts, and holding companies accountable all flow straight back to the correct formula. Without this, disaster management turns chaotic. Updates in regulatory filings almost always start with those same crucial formulas—because enforcement, intervention, and reporting all depend on having the right answer from the start.
Prioritizing Science-Based Training and Transparency
Having a deep respect for the basics—starting with the correct molecular formula—doesn’t just protect chemists or lab workers. It shields surrounding communities, sets a standard for global labs, and underpins rapid response in emergencies. Moving forward, investing in science-based training helps keep everyone safer and far better prepared to adapt to chemical incidents, large or small.
How should 2,2-Dinitropropane be stored?
Looking Beyond the Label: Real Risks in Storing Chemicals
Ask anyone who’s ever spent time in a warehouse or lab—2,2-Dinitropropane doesn’t advertise its risks. It lurks in metal drums, with a yellowish color and a faint odor, and the problems don’t shout. But 2,2-Dinitropropane brings dangers beyond a simple flammable liquid. I’ve learned from old hands and fire marshals alike: take shortcuts and the costs come fast. Explosions, fires, wrecked lungs—a carelessly stored few gallons give no second chances. Even small leaks build up, and those fumes reach far, outpacing your awareness. Life reminds you there’s a thin line between “routine” and “crisis.”
Temperature and Ventilation: Practical Steps Make the Difference
Small changes deliver real safety for this chemical. Forget about leaving containers in the sun or near steam pipes. I remember a colleague once stashed a can beside a boiler—ignorance almost cost him his job and the building. 2,2-Dinitropropane warms up easily, raising vapor pressure, pushing fumes into the air, and raising explosion risk. Refrigerated storage (2-8°C) gives a reasonable buffer for keeping both people and property safe. Ventilation isn’t just a checkbox; in stuffy spaces, vapors settle, and one spark can bring tragedy. I’ve seen how installing fans and keeping storerooms cool heads off disaster by keeping the air clear.
Separation: Where Small Details Save Lives
Mixing chemicals rarely ends well. Storing 2,2-Dinitropropane alongside acids, bases, or oxidizers practically invites a fire. A mishap in a neighboring warehouse—one shelf held a leaky oxidizer bottle next to a drum of nitro compound. Blew out half the wall before anyone grabbed the fire extinguisher. The lesson sticks. Keep it isolated. Sturdy cabinets, spark-proof latches, and warning labels leave less room for confusion or error. Never use metal shelving that creates sparks, and always ground metal containers to prevent static buildup.
Containers: Quality Over Convenience
Cheap, cracked plastic leads to spills, and rusty cans leak. Chemists and handlers always lean on containers certified for hazardous liquids—usually steel with tight-fitting, chemical-resistant seals. Regular checks for bulges, corrosion, or swelling spot early problems. Using secondary containment trays under containers catches drips and avoids a major mess. Sometimes it’s easy to want to “just stack it for a week.” But corrosion works quietly, and a tiny pinhole turns into a vapor fountain overnight.
Training and Emergency Planning: Prepared Teams Prevent Disasters
No shelf label or warning sticker replaces hands-on training. The best-run storerooms keep fire suppression, spill kits, and visible safety data sheets right where people can find them. I’ve seen panic turn routine incidents into chaos—never assume everyone knows what to do if something spills or if alarms blare. Regular drills and clear checklists create a culture where people spot mistakes before they grow.
Practical Solutions—Not Just Paperwork
2,2-Dinitropropane shows that safe storage draws on habits, not just policy. The facts speak for themselves: the Centers for Disease Control and Prevention documents explosion and fire risk at even moderate temperatures. No one forgets the lessons that follow an accident. Storage takes effort, but the payoff comes each time workers leave at the end of the day unscathed. It’s a matter of putting safety over speed, and precision over convenience.
What are the proper handling and disposal methods for 2,2-Dinitropropane?
Recognizing the Risks
2,2-Dinitropropane doesn’t have the same high profile as some other industrial chemicals, but that doesn’t mean it can be taken lightly. It acts as a powerful oxidizer, and direct contact can burn skin and eyes, irritate lungs, and trigger headaches. If it escapes into the environment, this chemical doesn’t just vanish—it can make water unsafe to drink and disrupt habitats for good. I remember a safety seminar from my lab days—nobody dared daydream when the instructor started passing around actual safety data sheets for chemicals like this. The reality of what one spill could cause made everyone sit up straighter.
Respecting the Rules
Local and federal laws have a lot to say about handling substances like 2,2-Dinitropropane. It’s easy to get impatient with regulations, yet they exist for good reason. The Environmental Protection Agency tracks hazardous waste for a reason: accidental fires, ground contamination, or just plain negligence can put whole communities in harm’s way.
From what I’ve seen, the best-run operations always plan ahead. Before anything arrives on site, they dial in procedures for storing chemicals like this in reliable, fire-resistant containers. Flammable liquids need distance from sources of heat, and storage areas need working exhausts, sensors, and emergency wash stations. These aren’t “just in case” features; they’ve become standard because one overlooked detail during handling can cause trouble that takes years to fix.
Smart Disposal Makes a Difference
Getting rid of old 2,2-Dinitropropane isn’t as simple as hosing it down the drain or tossing it in the regular trash. I’ve watched facilities bring in specialists for disposal jobs, and for good reason. They send all spent containers and residues to certified hazardous waste incinerators. Heat destroys what chemical treatments can’t. No one dumps this stuff in landfills—not if they want to avoid heavy fines or, worse, local groundwater issues that never quite go away.
Not enough people talk about the paperwork involved. Every batch that leaves a building comes with a batch of its own: manifests, shipping logs, chain-of-custody forms. Sloppy recordkeeping today means big questions tomorrow, and maybe a visit from regulators who want to see where those missing kilograms wound up. In my experience, there’s a sense of pride in ticking every box. Clean records mean people are less likely to cut corners with safety.
Taking Safety Culture Seriously
Hazardous waste handling only works with teamwork. Shortcuts rarely save time in the long run, and they almost always put health at risk. Good training pays off—nobody can put a price on eyes or lungs. Safety drills and clear signage set a baseline, but open conversations among staff make up the real backbone of a culture that looks out for each other.
Public data tells the story: where communities invest in updated training, double-check their storage protocols, and take disposal seriously, accidents drop dramatically. Better communication and up-to-date equipment give everyone a shot at going home safely. People trust companies that openly share how they protect workers and neighborhoods.
Building a Responsible Future
The world keeps using chemicals like 2,2-Dinitropropane in research and manufacturing, but we can choose how to handle the risks. Consistent education, clear rules, and investment in disposal make a real difference. It takes commitment at every stage—from delivery, to handling, to final disposal. The simple act of treating dangerous chemicals with respect can save lives and protect our environment for decades to come.
| Names | |
| Preferred IUPAC name | 2,2-dinitropropane |
| Other names |
1,1-Dinitroethane Dinitroisopropane |
| Pronunciation | /ˌtuː tuː daɪˌnaɪtroʊ ˈproʊpeɪn/ |
| Identifiers | |
| CAS Number | 594-61-6 |
| Beilstein Reference | 1906310 |
| ChEBI | CHEBI:28641 |
| ChEMBL | CHEMBL39937 |
| ChemSpider | 9983 |
| DrugBank | DB02141 |
| ECHA InfoCard | 17d1d9bb-8d33-4574-afb2-d3831e1e846a |
| EC Number | 209-795-7 |
| Gmelin Reference | 7417 |
| KEGG | C14145 |
| MeSH | D015244 |
| PubChem CID | 14031 |
| RTECS number | TZ5250000 |
| UNII | 1M73V14V3C |
| UN number | UN1289 |
| CompTox Dashboard (EPA) | DTXSID6074672 |
| Properties | |
| Chemical formula | C3H6N2O4 |
| Molar mass | 150.08 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.154 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 0.1 |
| Vapor pressure | 0.9 mmHg (20°C) |
| Acidity (pKa) | 12.3 |
| Basicity (pKb) | 11.19 |
| Magnetic susceptibility (χ) | −13.8 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.409 |
| Viscosity | 0.732 mPa·s (20 °C) |
| Dipole moment | 3.21 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 352.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -885.5 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Danger |
| Precautionary statements | Precautionary statements: P210, P261, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 2,2-Dinitropropane: 2-3-1OX |
| Flash point | 61 °C (142 °F; 334 K) |
| Autoignition temperature | 395 °C |
| Explosive limits | Explosive limits: 2.5–18.0% |
| Lethal dose or concentration | LD50 (oral, rat): 141 mg/kg |
| LD50 (median dose) | LD50 (median dose): 160 mg/kg (oral, rat) |
| NIOSH | TLV: NIOSH REL 0.1 ppm (0.37 mg/m3) TWA |
| PEL (Permissible) | PEL: 0.1 ppm (skin) |
| REL (Recommended) | 3 ppm |
| IDLH (Immediate danger) | IDLH: 75 ppm |
| Related compounds | |
| Related compounds |
Nitromethane Nitroethane 1,1-Dinitroethane 2-Nitropropane 1,2-Dinitropropane |

