Editorial Commentary: The Multifaceted World of 2,2'-Azobis(2,4-Dimethylpentanenitrile)
Tracing the Path to Innovation: A Look at the Historical Roots of AIBN Derivatives
Chemistry carries stories worth telling, especially in the field of organic initiators. The story of 2,2'-Azobis(2,4-Dimethylpentanenitrile), or ADVN, is tightly woven with a half-century of experimentation and creative research. After the mid-20th century boom in free-radical chemistry, industries chased after safer, more consistent ways to trigger polymerization. ADVN took the baton from older azo compounds, such as AIBN, but brought something fresh to the lineup—a better balance of stability and potency for processes demanding pinpoint thermal control. My own research in polymer science highlighted just how much manufacturers sought out reliability that didn’t risk runaway reactions or regulatory headaches. ADVN has quietly powered the growth of plastics and specialty polymers, showing up in plant logs alongside the more famous AIBN and V-65. This history is worth remembering, as every incremental improvement—higher decomposition temperatures, better product shelf life—built today’s chemical landscape.
Getting to Know the Compound: An Overview in Context
ADVN earns its place in industrial chemistry as a solid, free-radical initiator. Like its better-known cousin AIBN, it releases nitrogen gas and reactive free radicals as it breaks down. Where it stands apart is its higher decomposition temperature, giving process chemists a broader window for safe and controlled initiations. In plain language, this means you can design reactions that start and finish on your schedule, not the chemical’s. While ADVN isn’t as famous as AIBN, those in the business of controlled, high-precision polymerization know it can make the difference between a smooth-running operation and hours of troubleshooting. Its use ranges from bulk and solution polymerizations to applications involving specialty elastomers and thermoplastics.
Physical and Chemical Identity: What Matters Most Under the Microscope
Set a sample of 2,2'-Azobis(2,4-Dimethylpentanenitrile) under any decent lab light and you’ll see a colorless to pale yellow crystalline powder. It doesn’t make much of a fuss until you heat it. This compound holds together well at room temperature and doesn't explode into action the way some older initiators do at the first sign of warmth. Decomposition typically kicks in near 70°C to 80°C, making it workable for many late-stage industrial processes. Its modest molecular weight and limited solubility in water lend it flex in organic solvents—dichloromethane, acetone, even certain monomer mixtures—so it fits right in with the solvents process engineers already rely on.
Technical Specifications & Labeling: What Chemists Look For
Every bottle of ADVN worth trusting carries a label describing purity, typically hitting at least 98%, as well as guidelines for safe storage. Unlike AIBN, which releases isobutyronitrile, ADVN’s structure ensures a cleaner byproduct profile, often favored in specialty pharmaceutical or electronics work. Chemists keep an eye out for shelf life estimates, which can run a couple of years when stored cool and dry. Regulations force clear hazard labeling for explosivity, toxicity, and the need for gloves and splash protection. I’ve seen hazard data sheets grow thicker over the years, reflecting a real concern for worker safety, which is now baked into labeling and procurement paperwork.
Crafting the Compound: Preparation Methods Set the Standard
Synthesis demands skillful handling of highly energetic intermediates. Most ADVN production starts from nitrile precursors, forged into the final azo compound through a series of diazotization, coupling, and crystallization steps. Attention to process temperature and stoichiometry dominates because runaway side reactions waste product and create dangerous residues. The best routes yield a product that needs little post-synthesis purification, reducing organic solvent waste. Modern efforts emphasize greener methods—recycling reagents, capturing nitrogen off-gas, reusing wash solvents. These steps matter to anyone who has tried balancing product quality with the expectations of today’s environmental regulations. Facilities that handle these steps safely and cleanly earn trust, both from regulators and customers.
Reactivity and Modifications: The Chemistry Behind the Headlines
ADVN owes its value to a reliable, controllable decomposition path. On heating, it splits to form free radicals able to kick-start chain-growth polymerizations. It does not generate unpredictable byproducts under correctly managed industrial conditions—avoiding nasty surprises at the back end of a reactor. The process suits copolymerizations as well, allowing folks in specialty elastomer synthesis to tweak final properties just by adjusting the initiator load or target temperature. Researchers at university and in industry have also modified ADVN and similar molecules, attaching different substituents to push decomposition temperature up or down, tailoring the profile to suit specific up-and-coming polymer applications.
What’s in a Name: Synonyms and Alternate Product Labels
Chemical communication thrives on clarity. ADVN can show up in catalogs under names like 2,2'-Azobis(2,4-Dimethylvaleronitrile), ABVN, or CAS registry number 5108-72-3. Each synonym echoes a particular research tradition or industrial lineage. Purveyors may brand it around stability or performance, but chemists in the know read right through to the backbone of the molecule. As with any specialty chemical, double-checking synonyms before ordering prevents surprises at the receiving dock.
Staying Safe: Operational Standards That Matter
Nobody trusts blind luck in a modern chemistry plant, and that concern feels sharper with solid organic peroxides or azo initiators in play. Storage below 30°C, away from shock, friction, and open flame, sits at the core of safe operation. ADVN’s safety profile looks favorable compared to more volatile initiators, yet complacency has no place here. Regular training, spill protocols, fume extraction, and dust management combine to keep staff healthy. Emergency drills and close attention to shelf-life dates become a reflex. Industry has learned the hard way after a few notable incidents; workers now get the gear and the respect for risks that this chemistry demands.
Where ADVN Steps In: Application Across Industry
Ask any plant manager or R&D chemist what keeps their lines moving, and good initiators come up fast. ADVN helps drive polymerizations in both solution and emulsion setups, shining in styrene and acrylonitrile processes that run at higher temperatures. Its decomposition reliability means fewer batch failures and more consistent product properties, from high-impact plastics to weather-resistant coatings. Specialized fields such as medical device manufacturing or electronics often lean on initiators that play well with sensitive additives, where purity and predictable residue profiles count. ADVN supports those efforts, winning a spot on spec sheets and procurement orders by outperforming more temperamental alternatives.
Investigating the Unknown: Research and Development
Curiosity doesn’t rest in the modern lab. Academic and industrial teams push to wring more value from each molecule, studying ADVN derivatives for applications that demand even finer temperature control or faster radical release. Ongoing studies compare initiator blends, optimize reaction kinetics, and map out decomposition pathways under real-world processing conditions. The current push leans toward sustainable chemistry—less hazardous reagents, non-toxic byproducts, faster breakdown in waste streams. These goals align closely with the regulatory future chemical producers see coming fast. The work is never done, as new monomers and more demanding plastics emerge, pressing initiators like ADVN into new forms and custom blends.
Addressing the Shadows: Toxicity Research
Transparent discussion about toxicity forms the backbone of chemical trust. Recent years have brought sharper toxicology investigations, examining both direct and byproduct exposures. ADVN’s acute toxicity sits in the moderate range for compounds of its class, manageable with standard protective equipment and handling restrictions. Concerns focus on nitrile decomposition products and nitrogen off-gas, which calls for good ventilation and downstream carbon filtering. Chronic toxicity and environmental persistence often drive more questions from regulatory agencies, so long-term studies on soil and water fate remain an active area. Knowledge of these risks shapes how firms handle, transport, and dispose of ADVN, keeping communities and ecosystems safe.
Looking Forward: The Road Ahead for ADVN
Every day, chemists wake up looking for better, cleaner, safer answers. ADVN exemplifies the incremental, practical progress that industry prizes. The movement toward more sustainable, less hazardous chemistry will drive new ways to synthesize and deploy initiators, possibly phasing out older forms in favor of those with improved safety margins and smaller environmental footprints. As renewable feedstocks and green chemistry principles pick up steam, I expect ADVN analogues designed to break down readily in the environment or be produced with fewer hazardous intermediates. The future for this class of initiators looks promising so long as companies, regulators, and researchers remain honest about risks, pump resources into smarter R&D, and never cut corners on safety or transparency. Chemistry grows not just by invention but by clear-headed stewardship—ADVN’s evolving story serves as a reminder of that enduring lesson.
What is 2,2'-Azobis(2,4-Dimethylpentanenitrile) used for?
The Quiet Giant Behind Everyday Plastics
Stepping into any convenience store, the reality of modern plastics hits hard. Shelves sag from the weight of bottles, packaging, and containers that keep food fresh, drinks safe, and products a lot cheaper than glass or metal ever could. In this plastic universe, manufacturers lean on specialty chemicals to shape and toughen these products. One name that rarely gets screen time: 2,2'-Azobis(2,4-Dimethylpentanenitrile). It rolls off the tongue a bit rough, but its purpose is simple—helping plastics set, harden, and perform better.
How This Stuff Does Its Job
Makers of, say, PVC pipes or shoe soles often face a stubborn blockage: plastic resins don’t react fast enough, or at all, without help. 2,2'-Azobis(2,4-Dimethylpentanenitrile) acts like a kickstarter. Once heated, this compound breaks apart and throws off nitrogen gas—don’t worry, it’s the stuff that makes up most of the air we breathe. In the process, the compound churns out free radicals. These radicals get the main plastic-building reaction going and keep it running at an even pace. In plain language, this ingredient speeds up and keeps things moving in big polymer factories where even short stops cost real money.
Why Use This Instead of Others?
Chemical makers don’t pick ingredients just because they work. They check if the compound can last on the shelf, work at the right temperature, and keep things safe enough for humans. 2,2'-Azobis(2,4-Dimethylpentanenitrile) makes the cut for certain plastics because it works at a higher temperature than many alternatives. Factories cranking out foam insulation, wire coatings, or specialty gaskets like this kind of “radical initiator” because they can trust the reaction rate over a long run of production without surprises. That means fewer ruined batches, less waste, and more control over the final product’s toughness or flexibility.
Environmental and Safety Concerns
No one wants a chemical to boost profits but then cause headaches down the line. Safety teams in factories look at risk from both dust exposure and accidental decomposition. Like most azo compounds, 2,2'-Azobis(2,4-Dimethylpentanenitrile) needs cool, dry storage because it can kick off its reaction from heat or impact—not great near sparks or open flame. OSHA and European regulators keep an eye out and demand careful handling, meaning sealed containers, ventilation, and frequent checks for leaks.
Workers with chemical sensitivities might ask if any byproducts could cause harm. Nitrogen gas released during processing is generally safe, but some breakdowns spill out odors that would set off alarms in a food operation. These issues require smart engineering controls and plenty of on-the-job training. Many factories collect air samples and update safety data sheets regularly, so risks don’t get swept under the rug.
Room for Improvement
Making reliable, affordable plastics is tough without compounds like this one, but companies now juggle new demands: lighter environmental footprints, safer workers, and less chance of hazardous leftovers in products. Movers in the chemical industry are racing to come up with initiators that do the job just as well but stop short of causing long-term headaches for people or the planet. Only real investment and openness from big chemical companies will close that gap for good, making safer plastics—and, maybe, less of them—down the line.
What are the safety precautions for handling 2,2'-Azobis(2,4-Dimethylpentanenitrile)?
Why Strong Habits Matter in the Lab
Long days among chemicals teach people pretty quickly how much small mistakes can sting. 2,2'-Azobis(2,4-dimethylpentanenitrile) doesn’t give a second chance. It’s a radical initiator, used to spark reactions in polymers and plastics. It splits apart and releases nitrogen, letting chains snap together. Anyone using it faces certain risks—heat can start the reaction too early, dust can float into eyes or lungs, skin might itch for days after a splash. Precautions aren’t just checkboxes for inspectors; they’re a way to avoid all-nighters in the emergency room.
Respecting the Chemical: Don’t Cut Corners
Gloves don’t feel comfortable after twenty minutes, but nitrile or rubber gloves turn out to be crucial. Lab coats with long sleeves and close-toed shoes help keep splatters off skin, which can burn or scar for life. Eye protection sounds basic, till a single speck ruins your vision for good. I watched a colleague lose vision in one eye because safety glasses sat unused in the drawer—years later, he still talks about the cost of that decision.
Local exhaust fans do more than just clear the air. They yank vapors out before anyone breathes them in. Working under a fume hood, instead of at the open bench, cuts down on chronic coughs and headaches. That comes straight from long shifts in a plastics research lab where poor ventilation meant folks lost their sense of taste for days.
Storage: Out of Sight, Clearly Labeled
This compound can spark up with heat, friction, or static. Keep it cold—refrigerators between 2°C and 8°C work best. Keep it dry, away from sunlight, metal parts, or open wires. Someone in my old group once used a warm shelf because it looked convenient. Cleaning up took hours, and even though nobody got hurt, the smell stuck around. It’s not worth the time lost.
Label every container—big, bold markers help everyone stay aware. Segregate from acids, oxidizers, or anything that can trigger its breakdown. Seen too many close calls from folks storing everything in the same fridge; routine beats luck every time.
Spill Response and Waste Disposal
Small spills can turn dangerous fast. Scoop up powders with rubber spatulas, not hands. Dump everything—even cleaning rags—into approved waste cans, never down the sink. Waste workers deserve respect; watching someone fish a corroded tool out of the wrong bin burned this lesson in. Treat all waste as dangerous, even if the chemical smells sweet or looks harmless.
Always draft a step-by-step emergency plan. Medical attention can save skin and lungs if something splashes or gets inhaled. Nearest eyewash showers and first aid kits should sit within a five-second run, not in a closet across the hall. Within minutes, you can stop a small problem from turning critical. Practice drills mean less panic, more action.
Caring for People, Not Just Product
Following safety steps benefits everyone: chemists, janitors, emergency responders, people walking past the lab. Earning trust comes from getting home safe, day after day. Every well-fitting glove, every labeled bottle, every loud conversation about spilled nitrile means more people with all their fingers, all their sight, and a few more lessons learned the right way. Health isn’t an abstract value; it’s all about habits, lived out in real time, by people who want to stand tall and head home every night.
What is the chemical structure and formula of 2,2'-Azobis(2,4-Dimethylpentanenitrile)?
What Makes This Compound Unique
2,2'-Azobis(2,4-dimethylpentanenitrile) isn't a household name, but research labs and the polymer industry know it well. The reason? This compound plays a crucial role as a radical initiator, breaking its bonds under heat to kickstart polymerizations. These reactions shape the plastics used every day, from containers to automotive parts. So, understanding its structure lays the groundwork for using it safely and effectively.
Diving Into the Structure
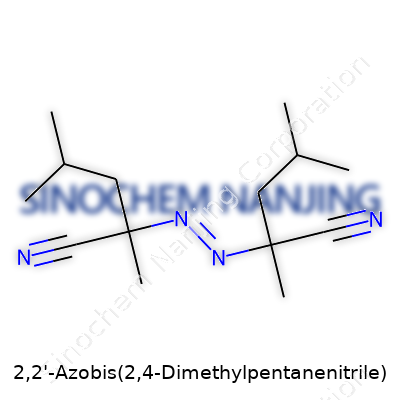
Looking at the chemical formula, it’s C14H24N4. That means each molecule stacks up 14 carbon atoms, 24 hydrogens, and 4 nitrogens. Its backbone features two identical arms, each stemming from the azobis (azo) bridge—a pair of nitrogen atoms joined by a double bond (–N=N–) right in the middle. Each nitrogen hooks onto a 2,4-dimethylpentanenitrile group, which brings in the branching. You get a zigzag of carbons, a couple of methyl (CH3) groups, and a nitrile (–C≡N) at the end of each arm.
If you draw it out, the center features the azo bridge:
Me2CH–CH2–C(CN)(CH3)–N=N–C(CN)(CH3)–CH2–CH(Me)2
People sometimes visualize it by grouping the repeating units, almost like a butterfly with two wings extending from a shared body.
Why Chemical Structure Matters
Structure serves as more than a formula on paper. It tells you real-world things—like how likely this compound will split under heat, whether it dissolves in a given solvent, and the safety challenges that come along. Those four nitrogen atoms, particularly in the azo linkage, carry the power to split and free up radicals, making this molecule suitable as a high-energy initiator in free-radical polymerizations. If the structure were less branched or lacked those nitrile groups, you’d watch reactivity and decomposition rates shift, sometimes drastically.
In practice, chemists who plan to use it need the structure at hand. Picture working in a lab and trying to predict the outcome of a polymerization. If you ignore molecular size or the types of side groups, yields will look erratic, results will prove impossible to replicate, and the tiniest oversight could trigger safety hazards. Case in point, the decomposition of this compound generates nitrogen gas and free radicals—a lucky break for polymerization, but a hazard if conditions aren’t controlled. A well-documented accident in an under-ventilated plant taught many that without careful control and a clear grasp of decomposition temperatures, the process could escalate into dangerous territory.
Turning Knowledge Into Safer Practice
Good handling practices start with the structure. Accurate identification lets chemists anticipate which conditions could push the material into rapid breakdown. Guidelines from agencies like the National Institute for Occupational Safety and Health (NIOSH) tie workplace exposure and storage rules straight back to how this compound behaves at the molecular level. Storage below 30°C and good ventilation are better than any one-size-fits-all approach to chemical safety.
In my work, I’ve seen how many mishaps stem from a gap in this knowledge. Check the structure, and it’s clear why 2,2'-Azobis(2,4-dimethylpentanenitrile) needs tailored procedures for weighing, mixing, and disposing of unused material. Integrating chemical understanding into safety routines isn’t just about ticking boxes; it builds a culture that stops incidents before they start.
How should 2,2'-Azobis(2,4-Dimethylpentanenitrile) be stored?
Recognizing the Risks in Everyday Practice
Storing 2,2'-Azobis(2,4-Dimethylpentanenitrile) isn't something to take lightly. Decades of experience in chemical handling have taught me to respect both the chemical and the lessons laid out in safety manuals. This compound falls into the category of organic peroxides, and its biggest reputation comes from how easily it can decompose or ignite under the wrong conditions. At manufacturing plants, labs, or even in transit, an inattentive moment can set off real hazards.
Controlling Temperature Isn’t Just a Guideline
Many have heard someone say, “Keep it cool.” Here, that advice means life or death for equipment and people. 2,2'-Azobis(2,4-Dimethylpentanenitrile) starts to break down rapidly as temperatures approach 50°C. Spiking temperatures not only spoil the material but send toxins airborne and could cause dangerous pressure buildup. Years working with such compounds taught me that refrigeration isn’t optional. Cold storage—ideally below 30°C—reduces the risk by slowing decomposition and keeping the work atmosphere safer. One summer, a broken air conditioner and forgotten box led to an emergency at a lab I visited, leaving a permanent impression on everyone involved.
Moisture and Light: Unexpected Enemies
Water and strong sunlight both trigger unwanted reactions. Many organic peroxides detest wet conditions. Storing them in a dry place is more than just good housekeeping; it can prevent runaway reactions that escalate quickly. I keep these containers far from chemicals like acids, bases, and anything that reacts with nitriles or peroxides. It only takes one sloppy shelf arrangement to cause a near-miss, or worse.
Container Choices Matter
Working closely with storage suppliers, I always choose airtight, light-resistant containers. Opaque plastics work well, but metal canisters sometimes wear faster, especially if leaks go unnoticed. Original packaging delivers more than convenience—it keeps experts’ engineering on your side. I trust those molded seals and chemical-resistant linings to keep oxygen and humidity at bay.
It’s Not Only About the Chemical, It’s About the People
In a busy storeroom, labels often fade and get ignored. Every time I train new staff, I remind them that proper labeling doesn’t just tick boxes. It tells anyone who walks past what’s inside, how to treat it, and what to do if something spills or catches fire. Readable hazard signs, up-to-date instructions, and regular safety drills build habits that save lives.
Ventilation Should Never Be Ignored
Once, an inadequate ventilation system in a chemical warehouse nearly caused disaster. For substances that release gases upon decomposition, fresh airflow isn’t a luxury. Any enclosure storing 2,2'-Azobis(2,4-Dimethylpentanenitrile) should have working air extraction and gas detectors if large amounts get stored. A faint sweet smell or haze means trouble. Staff need to know what that scent or haze means, and who to call.
Disposal and Access: Keep Tight Control
Access control stops unauthorized personnel from making costly mistakes. I’ve seen accidents arise from curious hands opening the wrong lid. A signed logbook, security tags, and restricted keys slow things down just enough to make people think. Expired or degraded material shouldn’t linger. Having a trained chemical waste handler dispose of it removes risk, prevents confusion later, and keeps inventories accurate.
Wrapping Up With Real-World Priorities
Following these storage principles draws from lessons learned on the job, industry consensus, and plenty of published case studies, including chemical safety bulletins. Regulatory agencies like OSHA and the European Chemicals Agency lay out practical checklists that parallel these experiences. Chemistry doesn’t offer mercy for shortcuts. In my own work, stubborn respect for guidelines paired with creative vigilance has kept both people and property out of harm’s way.
What are the hazards and first aid measures associated with 2,2'-Azobis(2,4-Dimethylpentanenitrile)?
Understanding the Hazards
Working around chemicals such as 2,2'-Azobis(2,4-Dimethylpentanenitrile) calls for a dose of practical awareness. This compound acts as a radical initiator—mainly in the plastics and rubber industries. Its reputation for instability, especially above room temperature, should raise eyebrows. The risk isn’t just something theoretical: if storage or handling gets sloppy, the material becomes twitchy and may decompose, giving off toxic gases or even causing fire or explosion.
I’ve seen a few chemical stockrooms where labels fade and the distinction between safe and risky gets blurry. In the case of this nitrile, heat and friction spell trouble. Even a minor slip, like resting a beaker on a sunny windowsill, could trigger a cascade. The decomposition process releases hydrogen cyanide and nitrogen oxides—neither kind to the lungs or nervous system.
Exposure Routes: Don’t Let Your Guard Down
A splash, a waft of dust, or even a soaked glove creates an exposure scenario. Chemicals like this one can irritate the skin, eyes, and respiratory tract. In worse scenarios, toxic effects travel beyond irritation. Hydrogen cyanide is notorious for choking off oxygen supply at the cellular level, and it acts fast.
Short cuts around protective clothing or eye shields tempt fate. I learned early in my lab days that the difference between a mild scare and a trip to the ER often boils down to double-checking gloves and goggles. This isn’t paranoia; the occupational risk for skin redness, rashes, or more severe poisoning remains very real.
First Aid Measures: Quick Thinking Counts
Things move fast after accidental exposure. If skin contact happens, rinsing the area with water for at least 15 minutes keeps damage in check. Removing contaminated clothing, jewelry, or shoes right away also keeps the chemical from soaking in further.
Eye exposure needs immediate and thorough flushing with a gentle stream of water, keeping eyelids open to wash out every trace. Medical attention comes next—no waiting until after lunch. Inhalation spills often happen where ventilation fans don’t do their job. Getting the person to fresh air tops the list; lay them down, keep them warm, and call for emergency medical help.
Swallowing this chemical turns the situation dire, so avoid inducing vomiting. Instead, rinse the mouth and get professional medical assistance fast. Sometimes adrenaline drives people to skip steps, but having written procedures posted where everyone can see makes the difference.
Taking Precautions
Handling this compound means respecting its low threshold for instability. Store it below the temperature recommended by the manufacturer, tightly sealed, away from heat or spark sources. Good air circulation is a must, especially in processing or weighing areas. Fact sheets and labels in plain language bring everyone on board. Regular safety training, not once a year but as a living routine, keeps protocols fresh.
Spill response kits need to be at arm’s reach, not buried in a manager’s office. Some companies go further by using color-coded containers and real-time temperature monitors, and that attention to detail pays off. Workers need access to fresh air, clean water, and protective gear. Eye-wash stations should stay uncluttered, checked regularly, and included in every workplace walkthrough.
Preventing Future Incidents
Hazardous chemicals test human habits as much as technical skill. For 2,2'-Azobis(2,4-Dimethylpentanenitrile), the stakes run high. Staying alert, reporting cracked container seals, and speaking up about equipment failures or unusual smells help build a safer environment. No shortcut or skipped checklist is worth a chemical burn or worse. Experience in the field hammers home a simple truth: consistent attention—paired with good habits and honest communication—forms the best defense against accidents.
| Names | |
| Preferred IUPAC name | 2,2'-azobis(2,4-dimethylpentanenitrile) |
| Other names |
Vazo 67 Azobis(2,4-dimethylvaleronitrile) ADVN |
| Pronunciation | /ˌtuːtuːˈeɪzoʊbɪsˌtuːˌfɔːrˌdaɪˈmɛθəlˌpɛnˈteɪn.nɪˌtraɪl/ |
| Identifiers | |
| CAS Number | '16952-62-8' |
| Beilstein Reference | 1858733 |
| ChEBI | CHEBI:87141 |
| ChEMBL | CHEMBL3180848 |
| ChemSpider | 16232 |
| DrugBank | DB14018 |
| ECHA InfoCard | ECHA InfoCard: 100.127.082 |
| EC Number | 229-713-7 |
| Gmelin Reference | 1348929 |
| KEGG | C19306 |
| MeSH | D017945 |
| PubChem CID | 12538 |
| RTECS number | RR1400000 |
| UNII | 02EUX33A3E |
| UN number | UN3242 |
| Properties | |
| Chemical formula | C14H24N4 |
| Molar mass | 266.403 g/mol |
| Appearance | White crystals |
| Odor | Odorless |
| Density | 0.965 g/cm³ |
| Solubility in water | Insoluble |
| log P | 1.22 |
| Vapor pressure | 0.34 mmHg (25 °C) |
| Magnetic susceptibility (χ) | -54.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4190 |
| Viscosity | 1.49 mPa·s (at 25 °C) |
| Dipole moment | 3.7 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 485.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -20.8 kcal/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –8409.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H241, H302, H312, H332 |
| Precautionary statements | P210, P220, P221, P234, P273, P280, P305+P351+P338, P306+P360, P310, P370+P378, P403+P235, P420 |
| NFPA 704 (fire diamond) | 1-3-3-❖ |
| Flash point | 37 °C (closed cup) |
| Autoignition temperature | 255 °C (DE) |
| Explosive limits | Explosive limits: 1.5-7% |
| Lethal dose or concentration | LD50 oral rat 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 449 mg/kg (rat, oral) |
| NIOSH | TTW66900 |
| PEL (Permissible) | Not established |
| REL (Recommended) | No REL established |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Azobisisobutyronitrile Azobisdimethylvaleronitrile Azobis(2-methylpropionitrile) |

