2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid: Depth, Development, and Future
Historical Development
The journey of 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid stretches back to the late twentieth century, tangled into the rapid development of cephalosporin antibiotics. Synthesizing this molecule wasn't an accident or mere chemical curiosity—it answered the call for new scaffolds that could dodge stubborn bacterial resistance. Scientists, facing the medical realities of penicillin and earlier cephalosporins losing ground to evolving bacteria, drilled down into the structure of thiazolyl acetic acids. By the 1980s, derivatives of this acid began shaping powerful third-generation cephalosporins. The molecule gave scientists a fresh backbone that resisted beta-lactamase enzymes, and its introduction marked progress in clinical pharmacology. Behind its discovery, research teams gathered in cramped labs and dealt with trial after trial—putting every outcome to the test, learning from failed attempts, and using bench chemistry as a key ally against infectious disease.
Product Overview
2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid now lives at the core of cephalosporin side-chain chemistry, playing a critical role in drugs that treat everything from pneumonia to meningitis. Pharmaceutical firms pursue this compound not for its standalone activity, but for its ability to boost the potency of antibiotics like cefotaxime and ceftriaxone. The substance usually appears as a powder, locked in industrial vials that travel through tightly regulated supply chains before ending up in pharmaceutical assembly lines. Given its value in helping build life-saving drugs, chemists and medical researchers have given it plenty of attention over the decades.
Physical & Chemical Properties
Picture a white or pale-yellow solid, with limited solubility in water but more flexibility when introduced to certain organic solvents. Its molecular formula, C6H7N3O3S2, shows a weight of around 233 g/mol. Handling the powder can sometimes release a faint sulfur-like odor. Its thiazole ring brings both stability and unique reactivity, especially in aqueous and mild acid or base conditions. The methoxyimino group stands out, providing resistance to hydrolysis and helping drugs built on this scaffold last longer in the bloodstream. Its melting point sits near 195-200°C, making storage conditions—dry, cool, protected from strong light—essential for any lab or plant that works with it.
Technical Specifications & Labeling
Producers mark each container by purity percentage—often above 98%—alongside batch numbers, manufacturing dates, and hazard classifications, in line with global shipping requirements and safety standards. Every label includes signal words recognized by regulatory bodies, such as “Warning” or “Caution.” Comprehensive certificates of analysis accompany shipments for audit trails and performance verification. Storage instructions reduce degradation risk, usually spelling out temperature limits, moisture sensitivity, and ventilation notes. Chemical suppliers use QR codes and barcodes to ease tracking for recalls, shelf-life management, or incident response.
Preparation Method
On the synthetic front, this compound grows out of union between thiazole derivatives and protected glycine analogues. The core step uses condensation of 2-amino-4-thiazolecarboxylic acid or its esters with methoxyiminoacetic acid, usually via activated intermediates. Skilled chemists gear up with precision glassware and powerful extraction solvents, adjusting pH and solvent ratios throughout each production cycle. Purification may involve repeated crystallizations or chromatography, with critical attention paid to removing organic impurities and residual solvents. Reproducible product quality rests on mastery of each stage—the acid chloride formation, the meticulous temperature controls, and careful handling of nitrosating agents.
Chemical Reactions & Modifications
The compound’s utility comes from its rich thiazole chemistry, opening the door to a wide variety of side-chain modifications. Functionalizing the amino or methoxyimino groups can yield novel beta-lactam derivatives. Medicinal chemists covet this flexibility, as tweaking just one functional group can sharply affect a cephalosporin's spectrum of activity, pharmacokinetics, or stability. For example, acylation reactions can introduce bulky protecting groups, enhancing pharmaceutical properties. Reductive amination or halogenation at specific sites also generates intermediates for related antibiotics. For those in medicinal labs, it’s a molecular playground—every shift in structure means a new shot at tackling pathogens the medical world worries about.
Synonyms & Product Names
This compound carries an armful of alternative names, as often happens among legacy chemical intermediates. Researchers in literature may spot it under designations like “Methoxyimino thiazolylacetic acid”, “AMTA”, or “MAA-thiazole.” You’ll find it referenced in product catalogs as “Thiazole glycine acid derivative” or by specialized numbers given by suppliers, such as “LT-41031.” Each name points back to the same structure, yet variations persist based on local naming preferences or patents. This web of synonyms means scientists and procurement teams stay vigilant, confirming exact CAS numbers—many rely on 10465-81-3—for every purchase or research project.
Safety & Operational Standards
From my years in chemical operations, handling this class of intermediates always called for strict safety rituals. Personal protective gear—goggles, nitrile gloves, lab coats—protects against dust inhalation or accidental skin contact, as thiazole derivatives can be irritating. Materials data sheets flag respiratory hazards and recommend using chemical fume hoods during synthesis or scaling-up processes. Waste management protocols ensure residues, solvents, and filters go into designated containers, handled by licensed chemical disposal experts. Emergency wash stations and eyewash units must stand ready, and every production batch gets logged for traceability, vital for regulatory compliance and workplace health. Facilities audit staff adherence, check safety equipment, and keep emergency contact numbers visible.
Application Area
Chemists and pharmaceutical manufacturers draw on this compound chiefly for cephalosporin synthesis, leveraging its structure to give rise to antibiotics with broad clinical reach. It’s hard to overlook how drugs built from this skeleton have reduced mortality rates from bacterial infections since the late twentieth century. Hospitals and outpatient clinics depend on extended-spectrum cephalosporins to manage serious infections, especially in immune-compromised or critically ill patients. Beyond antibiotics, researchers have tested its framework for agricultural fungicides and selective herbicides, though human health remains its top arena.
Research & Development
Academics and industry scientists continue exploring how modifications to the thiazole or methoxyimino groups influence pharmacological outcomes. Teams sift through structure-activity relationships, hoping small changes could yield agents with even lower resistance profiles or fewer side effects. Pharmaceutical R&D pours millions into high-throughput screening systems using libraries based on this acid. It fascinates many in medicinal chemistry, and more than a few doctoral theses have mapped new synthetic routes or biotransformation pathways using this compound as a springboard.
Toxicity Research
Toxicology screens show that, while this intermediate rarely enters clinical settings directly, it’s neither benign nor especially dangerous when proper protocols govern contact. Animal studies and cell-culture assays flag mild cytotoxicity at high concentrations; most occupational illness risks relate to mishandling or chronic exposure rather than acute toxicity. Regulators, such as those in the US and EU, mandate annual reviews on safety data sheets and workplace monitoring for facilities that handle the substance in bulk. In my own chemical lab years, any research involving this compound got reviewed by institutional biosafety and chemical safety boards, with mandatory environmental and health monitoring in place.
Future Prospects
Looking ahead, this compound’s story is not over. The rise of antibiotic resistance across the world keeps the pressure on medicinal chemists to innovate. Structure-guided design—especially AI-driven by newer computational tools—relies on scaffolds like this one as test beds for novel therapies. A growing movement in pharma pushes for greener synthesis methods, less solvent waste, and non-toxic byproducts. As regulatory agencies tighten controls and the public demands more sustainable manufacturing, producers experiment with enzymatic or biocatalytic preparation routes for thiazole containing building blocks. Research sponsorship and cross-institutional collaboration fuel ongoing effort, so it’s likely new uses or streamlined syntheses for this compound will emerge. As long as bacterial threats evolve, so will chemists’ efforts to outpace them, with acetic acid derivatives such as this one continuing to play a pivotal role in the race.
What is the chemical structure of 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid?
Getting to the Root of the Molecule
Every time I walk into a lab, glassware ready and notebook in hand, I can’t help but focus on structure first. 2-(2-Amino-4-thiazolyl)-2-(methoxyimino)acetic acid sits as a prime example, showing how chemical makeup steers function, especially in medicine. This compound features a core thiazole ring, amped up by both an amino and a methoxyimino group. The real eye-catcher: everything connects back to a simple acetic acid. The choices chemists make with every atom offer more than just academic satisfaction—they set the stage for real-world application.
Why Each Part Counts
The thiazole ring nails down stability and sharpens the molecule’s reactivity. Toss a nitrogen and a sulfur together: that combo creates binding sites that matter—for everything from enzyme inhibition to bacterial targeting. The amino group tucked onto the ring acts like a handle for hydrogen bonding. This isn’t just textbook theory; I’ve chatted with researchers who point to these interactions as the reason certain molecules knock out bacterial infections when others fall short.
The methoxyimino side earns its stripes by defending against destructive enzymes. Modern medicine needs structures that hang tough against beta-lactamases—the enzymes bacteria use to shred antibiotics. It’s more than a defensive feature; it’s a lifeline. I’ve watched as pharmaceuticals with clever side chains gave doctors another answer as old treatments started failing.
Acetic acid doesn’t get left behind. In organic synthesis labs, I saw young chemists turn to this group to tune solubility or bioavailability. A carboxylic acid hooks into targets like enzyme active sites or proteins. That means better absorption, maybe better outcomes for patients waiting for relief.
Choosing the Right Structure for Impact
Time and again, structure-activity relationships steer drug design. Scientists have tested hundreds of tweaks in the search for therapies that last longer in the body, punch harder against tough infections, or stay safer in the bloodstream. 2-(2-Amino-4-thiazolyl)-2-(methoxyimino)acetic acid lines up as a building block for cephalosporin antibiotics. These compounds brought major wins to hospitals where older drugs ran out of steam.
Still, no structure solves everything. As resistance rises, medicine needs chemists who can spot when clever functional groups start losing their punch. I’ve seen seasoned researchers push beyond the old recipes, drawing from decades of accumulated knowledge and embracing new synthetic routes—like blending thiazoles with unexpected side chains, hoping one will hit a bullseye against a superbug.
A Path Forward in Chemistry and Healthcare
Sharing knowledge across disciplines gives the best shot at new solutions. I’ve watched teams bring together synthetic chemists, biochemists, and clinicians just to dissect how the next structure could raise the bar. Reliable sources like peer-reviewed journals reinforce every lesson learned, sticking close to facts and avoiding hype.
Modern life keeps raising the stakes for antibiotic innovation. 2-(2-Amino-4-thiazolyl)-2-(methoxyimino)acetic acid, through its balanced but powerful structure, serves as proof that attention to each atom pays off. Focusing on function and creative design, while always demanding safety and evidence, puts the right options on the pharmacist’s shelf—and, ultimately, in patients’ hands.
What are the main applications or uses of 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid?
The Backbone of Modern Antibiotics
Walk into any pharmacy, dig through the names of the strongest antibiotics on the shelf, and you’ll land on a family called cephalosporins. Most people never wonder where these watchdogs for infections come from. They likely never read the fine print on a vial's label. But the medicine’s power, its ability to knock out deadly bacteria in hospital wards, comes from a compound known by chemists—2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid. Tough to say three times fast, but ask any pharmacist who has prepped antibiotics for an ICU, and odds are they recognize those building blocks.
Why should anyone care? For over three decades, these molecules have been the foundation for developing cephalosporin antibiotics that stubborn bacteria struggle to outwit. The presence of that methoxyimino group makes new drugs better at dodging bacterial enzymes that could otherwise chop antibiotics to pieces, making older medicines useless. I remember sitting through medical briefings on resistant bacteria, hearing stories of ICU patients turned around by nothing but modern cephalosporins powered by these sorts of compounds. Without them, our best tools would go dull fast.
Why Pharmaceutical Makers Keep Coming Back
Chemists recognized years ago that the thiazole ring and the extra amino chunk on this molecule made it a prime starting spot for safer, more advanced antibiotics. I’ve talked with a few pharmaceutical researchers who spend their careers in labs, building new drug candidates from this acid. They watch pathogens in petri dishes, adjusting the molecule just slightly to outmaneuver new forms of resistance. Every new cephalosporin in the hospital—think cefotaxime, ceftazidime, ceftriaxone—has roots in this structure.
This isn’t about minor tweaks that only matter on paper. Patients with neutropenia, newborns with sepsis, people with tough bone infections—these are the real winners when a scientist manages to build in extra staying power against bacterial threats. Beyond the hospital walls, clinics rely on oral forms for people fighting off pneumonia or complicated urinary tract infections at home. All of those therapies trace back, in part, to effective use of this one compound’s chemistry.
Factoring in Global Health and Access
Access remains uneven, especially in countries where storing and monitoring antibiotics is tough. Generic drug makers often rely on this acid because it’s proven, with a track record of producing stable, reliable antibiotics. There’s value in sticking with what works. As global health systems try to keep up with spreading resistance, protecting the brilliance of these compounds is a fight that doctors, drug makers, and patients all share.
Building the Next Generation
It’s tempting to think modern drug chemistry happens behind closed doors, far from reality. In truth, developing safer, more targeted antibiotics depends on the creative reuse of known cornerstones. My talks with infectious disease specialists always circle back: if you want antibiotics that last into the next decade, you go back to the kind of solid chemical frameworks that 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid provides. Protecting, refining, and distributing them responsibly demands solid science and honest access programs—not just quick profits or shortcuts.
Every life saved from serious infection in the hospital traces, in part, to this reliable building block, trusted and improved upon by chemists, manufacturers, and medical teams the world over.
What are the storage and handling recommendations for this compound?
Everyday Choices Make or Break Lab Safety
Ask anyone who’s spent long stretches in a university lab or an industrial plant — storage and handling decisions shape more than just the quality of an experiment. They set the tone for safety, efficiency, and often the health of everyone involved. Plenty of us have seen the impact of slicing corners. I still remember my first summer in a synthetic chemistry lab — a colleague left a bottle of acetone half-open, straight under the window in August, and the entire room reeked like nail polish remover by lunchtime. Nobody got hurt, but an oversight like that could have gone way worse.
Why the Details Matter
Every chemical brings its own risks and quirks to the table. Some are famous for picking up moisture. Others build pressure if left in a warm spot. Flammable solvents can ignite off a spark or careless static. Anyone dealing with compounds like these checks the Safety Data Sheet before even placing an order—regulators expect it, and so do smart workplaces.
Stable temperature, good ventilation, and the right containers aren’t just for peace of mind. Let’s take sodium metal: it stays calm in mineral oil, but a little humidity starts a dangerous reaction. Same goes for light-sensitive compounds you see in analytical labs—they need amber bottles, away from direct sunlight. Overlooking even simple storage advice leads to spoilage or, in a few bad cases, explosion or fire.
Organizing and Labeling Save More Than Time
Clear labeling and sensible shelf layouts mean that nobody has to guess. I’ve watched new staff shuffle through unmarked bottles, risking a fumbled pour or an accidental mix-up. Good habits start with readable labels: compound name, lot number, hazards, and arrival date. Some chemicals degrade fast, so rotating stock—using older supplies first—prevents wasted product and tricky disposal problems.
Personal Protective Gear: The Everyday Insurance
One thing that’s never outdated: gloves, goggles, and lab coats. Even “harmless” powders irritate skin or lungs after repeated exposure. I can’t count how many times a face shield saved me from a splash. Supervision also matters—new hires shadow veterans because mistakes happen quickest when people feel rushed or untrained.
Taking Maintenance Seriously
It’s easy to let equipment checks slide during busy weeks, but faulty seals or worn caps cause leaks that turn storage rooms into hazards. At my old job, a cracked beaker of concentrated acid sat at the back of a cabinet. The smell tipped us off, but only because someone checked the room during a routine sweep. Catching problems early stops bigger messes down the road.
Solutions Start With a Culture of Respect
Building a strong safety culture means talking openly about close calls and sharing lessons learned. Nobody operates in a vacuum. Training refreshers, regular audits, and a willingness to speak up if something feels off combine to prevent trouble. I’ve always appreciated workplaces that treat every bottle—common or rare—with that same respect.
In the end, the right storage and handling choices don’t just check off boxes for inspectors. They send everyone home at the end of the day, just as healthy as they arrived—and keep science moving forward without unnecessary setbacks.
What are the safety or hazard considerations when working with this product?
Why Safety Can't Slip Your Mind
Anyone who’s ever opened a bottle of cleaner, paint thinner, or fertilizer knows the sharp tang that climbs up your nose and brings tears to your eyes. I’ve spent a few weekends fixing up my place, and more than once I’ve felt that burn in my throat. This isn’t just discomfort—it signals risk. Working with chemicals means you trade home comforts for gloves, goggles, and lots of ventilation. Too many folks treat product labels like decoration, but they pack information that can spare you a trip to the emergency room.
Personal Responsibility Starts with Reading
I’ll admit, there’s been times I ignored the fine print on a label, thinking, “It can’t be that bad.” I learned the hard way after a kitchen scrub left my hands red and itchy for days. The product contained bleach and ammonia—it’s dangerous to mix those two, even in trace amounts. Breathing in the fumes triggered a coughing fit that sent me outside, gasping for air. That moment stuck with me. Every word on those labels carries weight: from “irritant” and “flammable” to “toxicity.” Ignoring these warnings isn’t just careless, it’s gambling with your health.
Common Hazards: What Can Go Wrong
Some products burn skin on contact, others can start a fire or explode if handled wrong. Solvents like acetone and toluene evaporate quickly, filling rooms with invisible vapors that dull your senses. Breathing this stuff in damages more than just your lungs—it can fog your memory, slow your reflexes, and over time, endanger your liver or kidneys. I’ve witnessed workers quiсkly dashing back into a closed room for “one quick thing,” only for dizziness to send them reeling. Even dust from mixing powdered substances can trigger asthma or worse. A spark near flammable liquids jumps straight to disaster, and news headlines filled with workshop fires all blur into the same root cause: neglecting safe practices.
Protecting Yourself and Others
In my years of helping neighbors with renovation projects, nothing slowed progress like a preventable accident. Splashback from a careless pour, a lid left open, or tossing used rags in a trash can grew riskier by the hour. I saw one friend suffer a bad chemical burn because gloves "felt clumsy.” Comfort is never worth the risk. Basic protections—good gloves, long sleeves, eye shields, solid shoes—matter far more than convenience. Open a window, run a fan, and if the instructions say “wear a mask,” don’t improvise with a t-shirt. Plenty of folks think chemical burns or lung injuries are a price baked into hard work, but real strength means refusing shortcuts.
Solutions That Work
Keeping chemicals locked away from kids or pets means storing them up high, tightly sealed, and far from food. Cleanups need proper disposal—never down the drain or into the trash without checking instructions. Used containers should find their way to hazardous waste sites, not the recycling bin. I make it a point to download SDS (Safety Data Sheets) directly from the manufacturer and keep printed copies close to my work area. If something spills, I don’t just grab some paper—kitty litter or special absorbents stop spread and soak up the mess safely. Sharing these habits with friends and family can mean the difference between a good story and a nightmare headline. Take the warnings seriously. Your skin, your lungs, and your peace of mind will thank you.
What is the purity and specification data for 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid?
What Purity Means in Real Science Work
I’ve worked with a fair share of drugs in different labs. Even small changes in raw material quality can throw months of research off course. Purity stands as the most critical factor because everything from side reactions in drug syntheses to potential impurities in the vial matters. When we ask about the purity of 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid, we aren’t just talking about a number. We push for nearly pharmaceutical grade—98% or higher—because contamination means wasted investment, unreliable results, and, later, patient risk if it moves along the pipeline. A batch at 95% might sound good for some chemicals, but in this case, it raises red flags for anyone working toward clinical or industrial uses.
Specification Data Matters for Safety and Progress
It always comes down to clear data: melting point, loss on drying, water content, residual solvents, appearance, assay by HPLC, and identification by NMR or IR. These numbers aren’t just technical filler. Take the melting point, which hovers around 194–198°C for a pure sample. Once you see a figure outside this range, it points to impurity or poor storage. Water can creep in easily. Too much, and you risk unexpected hydrolysis. Good suppliers keep water content below 1%. Loss on drying often gets limited to the same low value.
I’ve handled samples where missing identification tests turned up switched compounds in the bottle—months vanished in the blink of an eye. NMR and IR data must match published spectra. Residual solvents like DMF or methanol need more than casual checks. Even 0.1% DMF can block regulatory approval. Reputable suppliers trace every impurity to low parts-per-million levels—documenting each step. Anyone who skips those numbers exposes researchers, manufacturers, and eventually patients to serious unknowns.
Where Facts and Accountability Intersect
Years spent in regulatory-controlled environments have taught me inspections aren’t just about documentation. Labs need certificates of analysis for every batch. These certificates show not only raw numbers but who tested, when, and how. Regulatory agencies like the FDA have cracked down after finding inconsistencies in specification reporting. They don’t hand out warnings for nothing.
Transparency extends beyond the sheet in the lab drawer. By demanding suppliers provide results for heavy metals, particle size, and bioburden, buyers keep the whole chain honest. These checks also guard against cross-contamination and help ensure downstream synthesis doesn’t fail for unknown reasons. Every point of testing—chromatographic purity, enantiomeric excess, absence of byproducts—gets recorded not for tradition, but for safety, reproducibility, and intellectual honesty. I’ve seen the damage poor data inflicts: unreliable clinical results, lost grants, failed projects, and—far worse—patient risk.
Better Solutions Require Demanding Standards
Any real progress in chemical or pharmaceutical research comes from demanding open, audited records and third-party validation. There’s no shortcut. Working relationships with suppliers mean hands-on verification, not just trusting a COA. Joint audits, shared lab testing, and open corrective actions give everyone clearer certainty. A simple open-door policy—sharing data between partners, confirming analyses, and allowing inspections—translates into better science and safer outcomes. That’s what good science and good business both expect, and it’s what patients and researchers deserve.
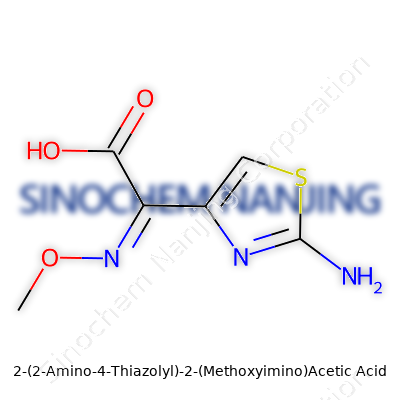
| Names | |
| Preferred IUPAC name | 2-[(2-amino-1,3-thiazol-4-yl)(methoxyimino)acetyl]oxyacetic acid |
| Other names |
2-(2-Amino-4-thiazolyl)-2-(methoxyimino)acetic acid Cefotaxime Impurity 13 Cefotaxime related compound K 2-Amino-4-thiazolyl methoxyimino acetic acid 2-(2-Amino-4-thiazolyl)-2-(methoxyimino)ethanoic acid |
| Pronunciation | /tuː-(tuː-əˈmiːnoʊ-fɔːr-θaɪˈæzoʊl-il)-tuː-(məˈθɒksiˌɪm-ˈaɪnoʊ)-əˈsiːtɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | [72630-70-5] |
| 3D model (JSmol) | `3D11AAMOJBGROG-UHFFFAOYSA-N` |
| Beilstein Reference | 1912140 |
| ChEBI | CHEBI:85178 |
| ChEMBL | CHEMBL1236 |
| ChemSpider | 21559686 |
| DrugBank | DB04560 |
| ECHA InfoCard | 03d5a894-d926-4172-8262-986b7f94aed1 |
| EC Number | EC 246-315-6 |
| Gmelin Reference | 87816 |
| KEGG | C14524 |
| MeSH | D000900 |
| PubChem CID | 72950 |
| RTECS number | AR8530000 |
| UNII | 92X6CT26OL |
| UN number | Not regulated |
| CompTox Dashboard (EPA) | DTXSID1039245 |
| Properties | |
| Chemical formula | C6H7N3O3S |
| Molar mass | 231.22 g/mol |
| Appearance | white to off-white solid |
| Odor | Odorless |
| Density | 0.52 g/cm³ |
| Solubility in water | soluble in water |
| log P | -1.33 |
| Vapor pressure | 3.46E-10 mmHg at 25°C |
| Acidity (pKa) | 2.0 |
| Basicity (pKb) | 7.2 |
| Magnetic susceptibility (χ) | -51.2×10^-6 cm³/mol |
| Refractive index (nD) | 1.730 |
| Dipole moment | 4.94 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 208.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -314.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -673.3 kJ/mol |
| Pharmacology | |
| ATC code | J01DD62 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P363, P337+P313, P362+P364, P501 |
| Lethal dose or concentration | LD50 oral rat 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Mouse (oral) 8250 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-(2-Amino-4-Thiazolyl)-2-(Methoxyimino)Acetic Acid: Not established |
| REL (Recommended) | Check Label at 2–8°C |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Cefotaxime Ceftriaxone Ceftazidime Cefepime Cefuroxime |

