Reflections on 2,2,5-Trimethylhexane: An Under-Recognized Backbone of Modern Chemistry
Historical Development
Personal experience in industrial chemistry labs showed how specialty hydrocarbons, like 2,2,5-Trimethylhexane, rarely get the spotlight. Throughout the 20th century, demand for branched alkanes grew with the automotive boom and refining technology improvements. As I look back at old chemistry texts and research papers, it’s clear the development of 2,2,5-Trimethylhexane ran parallel to the need for high-octane fuels. Refineries pushed for new cracking methods, leading to intentional synthesis routes instead of only accepting this compound as a byproduct. Researchers in the oil industry documented improved anti-knock characteristics with structural isomers, including this one. This chemical found its way into fuel research at a time where chemists scrawled hand-drawn hydrocarbon chains on chalkboards, laying the groundwork for later advances in petroleum engineering.
Product Overview
2,2,5-Trimethylhexane belongs to the family of aliphatic hydrocarbons. Its structure is simple: a hexane backbone with methyl branches at two, two, and five positions. This branching defines its ability to act as a specialty fuel component. Application experience shows this molecule stands out in discussions about gasoline blending, where consistent combustion and vaporization play a huge role. Chemists working to tune vapor pressures or exploit specific boiling ranges see this chemical as an option for achieving targeted fuel properties. The world of fine chemicals and performance fluids often calls upon this hydrocarbon for its stability and energy density.
Physical & Chemical Properties
Handling 2,2,5-Trimethylhexane in the lab, you notice its clear, colorless appearance and faint gasoline-like smell. Its boiling point lands higher than some lighter branched isomers, offering stability while remaining liquid under common atmospheric conditions. Immiscibility in water doesn’t surprise anyone with familiarity in hydrocarbon chemistry. Compared to straight-chain or differently branched alkanes, its lower density and moderate vapor pressure make it suited for specialized roles where volatility matters—fuel labs and petroleum product testing come to mind. Its chemical inertness, courtesy of its saturated carbon skeleton, means it resists many reactions under standard conditions, though it won’t stand up long to halogenation, strong acids, or oxidizing environments.
Technical Specifications & Labeling
Labelling regulations carved out by REACH and the Globally Harmonized System mean clear, consistent chemical identification, both for users like myself and for institutions that track hazardous materials. The CAS number and molecular formula set the standard for procurement and proper classification. Various purity grades serve different needs: pure compounds enter research and standardized testing, while industrial blends go to performance fuel studies. These specifications range from typical purity sketches—95 percent and higher for analytical labs, less for bulk chemical processes. Reliable identification and traceability encourage safe, repeatable handling across labs and manufacturing facilities.
Preparation Method
Manufacturing 2,2,5-Trimethylhexane relies on well-established organic synthesis. From experience and reading refinery process manuals, it’s clear that isomerization and alkylation of lighter alkanes form the backbone of production. Commercial production can start with hexane or lighter feedstocks subjected to catalytic isomerization and methylation. Catalysts, often acidic zeolites or alumina-silica blends, direct specific branching patterns. Lab production typically involves controlled reactions monitored by GC-MS or similar spectroscopic methods, allowing chemists to verify the target structure and weed out byproducts. Good process control and careful catalyst management make for consistent yields, an ongoing challenge as demand and raw material quality fluctuate.
Chemical Reactions & Modifications
Despite being chemically stable in ambient conditions, 2,2,5-Trimethylhexane can undergo a variety of transformations in controlled settings. Chlorination and bromination come up sometimes in synthetic explorations, leading to halogenated intermediates with different reactivity. Oxidative cracking, which some chemical engineers still use today, turns it into valuable simpler molecules under harsh oxidation. By modifying conditions—temperature, pressure, the choice of catalyst—a single molecule opens doors to dozens of derivatives. These derivatives feed back into fields as diverse as fuel additives, research into combustion pathways, and in some cases, as intermediates for specialized polymers.
Synonyms & Product Names
Those who have spent time with chemical inventories know the headache of synonym confusion. 2,2,5-Trimethylhexane wears several hats depending on the supplier and the context—called sometimes iso-nonane or nonane isomer, though true nonane is straight-chained. The careful reading of chemical catalogs and material safety data sheets over the years taught me that relying solely on trade names or common descriptors can trip up even experienced chemists. Sticking to IUPAC nomenclature or the unique CAS identifier significantly cuts down on errors and makes communication between global teams a lot smoother.
Safety & Operational Standards
Like most hydrocarbons, 2,2,5-Trimethylhexane doesn’t play nice with open flames or poorly ventilated spaces. Anyone who’s seen accidents in poorly equipped workshops respects the flammability and vapor risks entailed. Good practice draws on long-standing OSHA and EU-OSHA guidelines: use chemical fume hoods, spark-resistant tools, and store in tightly sealed containers. Experience has shown that open transfer and careless cleanup lead to fire risks and inhalation hazards, especially with higher vapor pressure materials. Consistent, clear labeling, regular training, and thoughtful chemical storage—not just paperwork compliance—keep everyone safe and healthy in the lab and plant.
Application Area
Exposure to oil and gas research reveals 2,2,5-Trimethylhexane’s steady demand as a fuel component. Blending into test gasolines lets engineers benchmark octane ratings and combustion profiles, helping automakers and refiners minimize knocking and maximize efficiency. On the analytical front, this compound often finds use as a reference or model compound for hydrocarbon reaction studies. Sometimes, it serves as a calibration substance in analytical chemistry, valuable for gas chromatography and mass spectrometry work. Fine-tuning physical properties like volatility, lubricity, and energy content drives further usage in formulated fluids, specialized lubricants, and solvent compositions. Its role in simulating real-world mixtures for engine and equipment testing shouldn't be overlooked, proving just how interconnected chemical research and product engineering remain.
Research & Development
Development cycles in petrochemical companies and academic labs both rely on hydrocarbons like 2,2,5-Trimethylhexane to solve modern combustion and emissions puzzles. As governments push for cleaner fuels and lower emissions, chemists revisit even “mature” molecules to extract more performance and efficiency. Thoughtful work with branched-chain alkanes allows targeted studies to tease out unknown combustion intermediates or secondary pollution products, bridging the gap between bench and engine test cell. I remember research seminars where questions about molecular structure and combustion properties inspired lively debates and calls for better data on trace byproduct formation. In turn, these efforts push downstream into automotive and environmental policy discussions, proving fundamental chemistry remains tightly connected to the issues of our day.
Toxicity Research
Handling a range of hydrocarbons, you learn quickly to respect their biological activity. Toxicity research on 2,2,5-Trimethylhexane, much like related alkanes, shows it is no angel—high vapor concentrations bring dizziness or respiratory distress, and chronic exposures lead to headaches and other central nervous system effects. Environmental toxicology shows limited persistence due to volatility and photo-oxidation, but that doesn’t mean safe, unchecked releases. Long-term exposure risk remains a blind spot, especially for non-standard blends or uncontrolled industrial emissions. Regular monitoring, personal protective equipment, and rigorous adherence to established exposure limits form the only reliable shield against unforeseen health effects.
Future Prospects
Looking around at conferences and reading the latest journals, one sees how the future of 2,2,5-Trimethylhexane ties closely to future fuels and tighter emissions rules. No matter how far electrification advances, internal combustion engines and the chemical industry will need precise hydrocarbon building blocks for years. Research pushes further toward sustainable sourcing—bio-derived or waste-to-fuel pathways get more attention. Computational modeling and advanced sensors change how chemists and engineers measure combustion or emissions, raising demand for well-understood hydrocarbons like this one to serve as standards and test compounds. As chemical regulations, environmental scrutiny, and application technology grow in complexity, the demand for clear data and responsible use only increases. Synthetic chemists, chemical engineers, toxicologists, and environmental specialists will keep coming back to 2,2,5-Trimethylhexane to answer new questions, develop safer practices, and chart responsible growth for a changing industry.
What is 2,2,5-Trimethylhexane used for?
Fueling Every Mile: The Gasoline Connection
2,2,5-Trimethylhexane sounds like the kind of name you’d see in a chemistry book and quickly forget. Yet, this clear liquid matters every time you fill your tank. This hydrocarbon belongs to a class called isoparaffins, found right at the heart of high-octane gasoline. Engineers and chemists want fuel to burn cleanly and powerfully without knocking—an engine-damaging thump caused by uneven burning. Octane rating tells you how resistant a fuel is to knocking, and 2,2,5-Trimethylhexane pushes that number up.
Gasoline is more than just a mixture of random bits—its recipe gets tweaked to hit performance marks. Straight-chain hydrocarbons, like those once made from crude oil, can cause engines to sputter, especially under load and high compression. Isoparaffins like this one, with their branched structures, make the fuel more stable. High-performance engines, including those found in sports cars or aircraft, benefit the most because they squeeze every bit of energy out of each drop of fuel. The presence of compounds like 2,2,5-Trimethylhexane keeps these engines running smoothly.
Industry Leans on Chemistry
Refineries shape their outputs using procedures that have changed a lot over the years. Catalytic reforming stands out as a key step, where less-desirable hydrocarbons become more valuable ones. 2,2,5-Trimethylhexane does not occur naturally in significant amounts. Instead, it often comes from tweaks in refinery processes, converting straight chains to branched isomers. These chemical decisions have big effects downstream. Fuels blend together more efficiently, producing a final product that meets emission standards, gives good mileage, and leaves engines cleaner.
Air pollution and greenhouse gases shape the fuel industry more than ever. The EPA sets tough targets for tailpipe emissions, and car engines keep adapting. Compounds that let engines work without knocking help automakers meet those rules. Better combustion means less wasted fuel and lower emissions per mile. My own experience fiddling with cars taught me how much difference the right chemistry inside the fuel tank makes—a rough idle or a pinging noise can disappear just by switching to higher-octane gasoline.
Laboratory Use: Testing and Standards
Labs use 2,2,5-Trimethylhexane as a reference point for testing fuel properties. Testing agencies set global standards for diesel and gasoline quality, and this compound serves as a marker for certain characteristics, including octane behavior. Consistency matters, both for regulators and car owners, so using precise chemicals for tests helps keep those checks reliable. Universities and research facilities also draw on it to study combustion reactions and emissions profiles, seeking fuel advances that might lower pollution or boost efficiency.
Pathways Toward Cleaner Transportation
Since transportation accounts for nearly a third of U.S. greenhouse gas emissions, every tweak in fuel composition impacts big-picture goals. Some researchers are pursuing greener synthesis routes using renewables or bio-based feedstocks. One idea involves leveraging sunlight and catalysts to create branched alkanes like 2,2,5-Trimethylhexane directly from plant material instead of oil. Switching up how we source and manufacture fuel components might help cut the sector’s emissions.
If future vehicles rely more on electrification, the traditional role of hydrocarbons may shrink, but for now, millions of cars and trucks rely on this kind of careful chemistry to run better, cleaner, and longer.
What are the physical and chemical properties of 2,2,5-Trimethylhexane?
A Closer Look at Its Structure
No need to crack open a chemistry textbook to picture this molecule—it belongs to the branched hydrocarbon family. Structurally, 2,2,5-Trimethylhexane puts three methyl groups on a hexane backbone. That bulk gives it a different “feel” than plain old hexane. Scientists value this sort of branching, especially when they want to adjust performance in fuels and solvents.
Physical Properties Bring Distinct Advantages
Anyone who’s handled this compound in the lab can recognize its clear, colorless liquid nature. There’s barely a whiff—a mild hydrocarbon smell, and that’s about it. With a molecular weight of 142.28 g/mol, it’s heavier than straight-chain hexane.
Pour it into a beaker, and its density falls near 0.72 to 0.74 g/cm³ at room temperature. That means it floats on water. Its low boiling point (ranging from 130°C to 142°C) helps it vaporize easily under heat, much like gasoline. Pouring it into a closed container for storage can quickly build pressure. Its freezing point sits well below zero degrees Celsius, so the liquid stays fluid in most environments.
Its low viscosity lets it move freely—almost slippery. This trait makes it useful for blending into fuel or lubricants, where flow matters.
Chemical Behavior: Stability with a Twist
2,2,5-Trimethylhexane stays stable under normal lab conditions. It ignores most light, won’t rust metals, and resists breaking down if left alone. Branching in its structure helps keep it from “cracking” or reacting too easily, extending its shelf life. But toss in enough oxygen or heat, and combustion becomes a big risk. Hydrocarbons like this burn with a clean, blue-yellow flame—a familiar sight to anyone who’s worked with fuel samples.
Adding strong acids or oxidizers can provoke a reaction, but in practical use, there’s little incentive to do so. It won’t mix with water—like oil in a puddle, it beads up and separates. Still, it blends right in with other hydrocarbons, from alkanes to aromatic solvents, making it handy for fine-tuning fuel properties.
Where Facts Meet Application
This molecule often comes up in fuel research. Engine designers use branched alkanes like 2,2,5-Trimethylhexane to improve octane rating. A higher octane rating helps prevent "knocking," which damages engines over time. I remember early days in the lab, comparing straight and branched hydrocarbons—the difference in burning and the engine sound was always clear. The branched ones, including 2,2,5-Trimethylhexane, gave smoother, quieter runs.
Its clean-burning character reduces soot formation, pulling it ahead in the race to design efficient, cleaner fuels. This point ties directly into reducing local air pollution—a matter I’ve seen debated in city halls and environmental meetings alike.
Handling with Respect
Despite its usefulness, careless exposure spells trouble. Like many liquids found in the fuel and solvent world, inhaling vapors or letting it touch bare skin can cause irritation. Working with it meant gloves, ventilation, and storage in flame-proof cabinets. This isn’t overkill—it’s a matter of respecting potential hazards.
Disposal, too, needs careful steps. Unchecked dumping leads to groundwater concerns. I’ve watched enough clean-up crews spend hours tackling spills that could’ve been avoided with a bit of care up front. Local regulations set the standard for responsible disposal, and companies find it wise to follow them closely.
Raising the Bar for Cleaner Chemistry
The push for cleaner fuels keeps 2,2,5-Trimethylhexane in the spotlight. Adjusting structure at the molecular level can shift how fuels perform, reduce pollution, and improve public health. Its physical and chemical traits connect directly with the real world—less engine trouble, fewer emissions, lower risk to both people and the planet. That’s a lesson learned from the street to the lab and back again.
Is 2,2,5-Trimethylhexane hazardous or toxic?
What is 2,2,5-Trimethylhexane?
If you've ever worked around fuels or chemicals in a lab, 2,2,5-Trimethylhexane may have crossed your path. It's a branched alkane, showing up occasionally as a fuel additive or in certain research settings. Its structure puts it in the family of hydrocarbons found in gasoline. Outside specialized industries, most people barely give it a thought. Still, folks at refining sites and chemical plants encounter these substances every day.
Is 2,2,5-Trimethylhexane Toxic?
No red warnings stamp across most containers of this chemical, but that never means total safety. 2,2,5-Trimethylhexane sits among the broader group of alkanes, similar to the stuff powering cars. Most reports on alkanes suggest they hold low acute toxicity when inhaled or swallowed. Chronic effects show up more in high-exposure scenarios. Nausea, dizziness, or headaches sometimes hit after breathing strong vapors—something I've heard from technicians spending hours at a time fixing faulty pumps.
Peer-reviewed data on this specific alkane seems scarce compared with the steady stream on more common gasoline components like hexane or benzene. The primary risks seem rooted in its volatility. Vapors from large spills or poorly ventilated areas build up fast, displacing air and inviting lightheadedness or worse. Direct skin contact could cause mild irritation for the unlucky, but it's not the corrosive kind seen with acids.
Flammability Carries the Bigger Punch
Working with flammable liquids, especially those related to fossil fuels, always means respecting their tendency to ignite. 2,2,5-Trimethylhexane earns its place alongside other fuel components as a fire risk. In high school shops, teachers teach the lesson early: even a mild, sweet-smelling solvent can lay the groundwork for an explosion if safety slips. Open flames, sparks, or even static give enough ignition energy for a bad day at the plant.
Environmental Impact
Even though it breaks down in sunlight, letting 2,2,5-Trimethylhexane reach waterways raises environmental flags. Fish and aquatic life pick up toxins from similar hydrocarbons. The compound spreads quickly in surface water. Cleanup costs skyrocket if neglect meets a weak safety culture—a lesson learned the hard way in some refineries back in the 1980s.
Smarter Handling, Fewer Worries
Most hazards from these chemicals fade with good practices. Experienced workers lean into practical measures: solid ventilation, well-labeled containers, spark-free tools. Small changes, such as keeping material safety data sheets close and fresh air flowing, trim the number of people dealing with headaches and dizziness. Nobody, from new techs to long-timers, benefits from shortcuts. Regular training keeps the focus sharp and accidents rare.
Searching for Safer Substitutes
Big companies look for safer tech all the time. Newer additives cut emissions and lower toxicity. Regulators keep pressure up, spurred by real-world studies and community concerns. Every worker—chemist, operator, or engineer—wants to clock out healthy. That push for transparency means sharing reliable exposure studies and tracking near-miss incidents. Progress kicks in as safer alternatives hit the market, slowly edging out riskier compounds.
What Really Matters
Respecting chemicals has always boiled down to two things: straight facts and real-world vigilance. 2,2,5-Trimethylhexane doesn’t carry the same infamous reputation as benzene, but smart habits still belong in every workshop where it shows up. The best results come from good safety gear, honest conversation about hazards, and the kind of daily routines that keep folks safe from hidden risks.
How should 2,2,5-Trimethylhexane be stored and handled?
Understanding the Substance
2,2,5-Trimethylhexane counts as a specialized hydrocarbon, usually more familiar to folks working in chemical plants or research settings. This clear liquid gives off a smell reminiscent of gasoline. Most people never encounter it at home, but in a lab or industrial site, it becomes a regular companion. The similarities between this compound and fuels underline the real need for care every time it's stored or transferred.
Fire Risks Demand Respect
You take one look at the flash point and vapor pressure of 2,2,5-Trimethylhexane, and respect for fire safety jumps front and center. The vapor forms flammable mixtures with air. Any spark, cigarette, or static electricity has the chance to ignite these vapors. Anyone who’s ever watched an oil fire knows that fighting those flames takes more than water and wishful thinking. Recommended methods include keeping containers tightly closed and away from ignition sources. Grounding and bonding those containers cuts down on static build-up, which a lot of old-timers in the business learned the hard way after near misses.
Sensible Storage Choices Matter
Shoving a drum or glass bottle of 2,2,5-Trimethylhexane into a crowded chemicals cabinet can turn a clean workspace into an accident waiting to happen. A well-ventilated, cool storage area makes all the difference. Too much heat can encourage pressure build-up. Direct sunlight weakens storage containers and shortens shelf life. Proper signage reminds everybody about the hazards, and access remains limited to trained staff. Safety Data Sheets belong somewhere visible and easy to reach, not buried under a pile of paperwork.
PPE and Personal Awareness
Regular gloves and cheap goggles won’t cut it here. Nitrile gloves protect against skin exposure, and splash goggles shield the eyes. Chemical-resistant aprons get used more often than most people would imagine. Any seasoned lab worker can recall a time when good protection stopped a minor spill from turning into a trip to the emergency room. If skin contact happens, running water and plenty of soap come into play right away. Inhalation risk grows in poorly ventilated rooms, so working with proper fume hoods or local exhaust fans sets up an extra layer of safety.
Spills and Waste: No Room for Complacency
Ignoring a spill or tossing leftover solvent in regular trash guarantees trouble down the line. Spills call for absorbent mats, not paper towels. Any cleanup stays limited to people with training, and proper waste containers sit ready for used materials. Environmental responsibility calls for following all disposal regulations. No one wants their local water supply reflecting back the consequences of poor chemical stewardship.
Practical Training Trumps Paper Procedures
Reading procedures off a wall poster or safety manual doesn’t stick until you’ve run through a spill drill or worked shadow shifts with experienced staff. Guided practice gets people comfortable with grab-and-go actions during an emergency. Everyone benefits when questions about safety find answers right away. Strong safety cultures grow out of honest conversations and clear accountability, not just rules on paper.
Looking Forward
Companies can support safer workplaces by investing in regular training, reliable storage units, and updated personal protective equipment. Consulting up-to-date resources ensures workers base decisions on the most accurate information. By making safe storage and handling non-negotiable, future accidents and exposures drop even as productivity holds steady. Small actions day-by-day often prevent the next big story from making the headlines.
What is the molecular formula and structure of 2,2,5-Trimethylhexane?
Digging Into the Molecular Formula
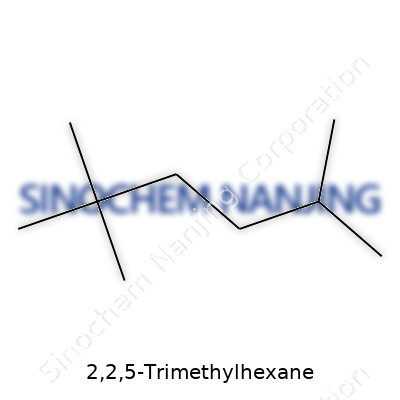
2,2,5-Trimethylhexane sports the molecular formula C9H20. Nine carbon atoms and twenty hydrogen atoms shape this saturated hydrocarbon. Take a moment to lay out the number of carbon and hydrogen atoms, and you get a sense of what places this compound squarely among the alkanes—a family of hydrocarbons known for their straightforward single bonds and chain-like structures. The structure's simplicity makes it a reliable building block in organic chemistry, especially for researchers mapping out synthetic pathways.
The Structure and Why It Matters
Let’s picture this: 2,2,5-Trimethylhexane features a six-carbon backbone. Out of this main chain, methyl groups branch off at carbons 2, 2, and 5. These branches give extra bulkiness to the molecule. In practical terms, 2,2,5-Trimethylhexane looks like a hexane molecule where someone has crowded extra methyl groups on specific carbons, changing its physical and chemical qualities.
This shape isn’t just chemistry trivia. The methyl groups on the same carbon—that’s two on the second carbon—force the molecule to take on a more compact, less linear form. You see direct impact on boiling point, melting point, and how the molecule dissolves. I remember back in an undergraduate organic chemistry lab, students often struggled to predict which alkanes would boil off first. The branching in 2,2,5-Trimethylhexane lowers its boiling point compared to its straight-chain siblings. These quirks make a real difference if you work in fuel design or chemical manufacturing, where volatility and combustion behavior matter.
Understanding Its Place in Chemistry
The way this molecule is constructed sheds light on bigger trends in organic chemistry. Alkanes with more branches tend to burn cleaner but with slightly lower energy release per molecule. In automotive contexts, branched alkanes like isooctane get highlighted as high-octane fuels. 2,2,5-Trimethylhexane shares several traits with isooctane, including a stable, branched profile. That puts it within reach for engine researchers who hunt for knock-resistant fuel components. If you’ve ever read about fuel efficiencies and cleaner burn in engines, you can thank these little branching tweaks for some of that progress.
Challenges and Future Opportunities
Producing pure 2,2,5-Trimethylhexane can be tricky, especially when separating it from close relatives in distillation. Tech advances in chromatography and synthesis have helped, although it still takes careful control and the right catalysts to get good yields. There’s clear value in developing greener, more precise synthesis methods so we minimize waste and energy use. Researchers have pushed for bio-based feedstocks in place of crude oil, which means compounds like 2,2,5-Trimethylhexane could one day come from renewable sources.
From a teaching standpoint, this molecule helps students learn about physical properties rooted in structure. The hands-on activities with model kits drive home how a small change—one extra methyl group, a twist in the chain—alters characteristics. Students remember that lesson the next time they look at more complex hydrocarbons or try to design molecules for a specific task.
Key Takeaways
Chemists and engineers working with hydrocarbons never ignore these details. The C9H20 formula and precise structure of 2,2,5-Trimethylhexane influence its use and production, making it relevant for anyone invested in fuels, materials, or fundamental organic chemistry.
| Names | |
| Preferred IUPAC name | 2,2,5-Trimethylhexane |
| Other names |
Isooctane Iso-Octane 2,2,5-Trimethyloctane |
| Pronunciation | /tuː tuː faɪv traɪˈmɛθɪlˈhɛkseɪn/ |
| Identifiers | |
| CAS Number | 590-76-1 |
| Beilstein Reference | 878121 |
| ChEBI | CHEBI:89398 |
| ChEMBL | CHEMBL2106627 |
| ChemSpider | 15366 |
| DrugBank | DB14056 |
| ECHA InfoCard | 03bbf914-1f50-4ca3-9a38-d378a9bc7387 |
| EC Number | 2087-67-6 |
| Gmelin Reference | 78021 |
| KEGG | C21130 |
| MeSH | D000438 |
| PubChem CID | 11762 |
| RTECS number | RN8225000 |
| UNII | F4T6P5V6JK |
| UN number | UN3295 |
| CompTox Dashboard (EPA) | DTXSID7040706 |
| Properties | |
| Chemical formula | C9H20 |
| Molar mass | 142.28 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.690 g/mL at 25 °C |
| Solubility in water | Insoluble in water |
| log P | 3.7 |
| Vapor pressure | 2.93 mmHg (25 °C) |
| Acidity (pKa) | pKa ≈ 50 |
| Basicity (pKb) | pKb: -1.53 |
| Magnetic susceptibility (χ) | -77.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.398 |
| Viscosity | 0.763 mPa·s (20 °C) |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 375.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -245.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –5229.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | '2,2,5-Trimethylhexane' does not have an ATC code. |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02 |
| Signal word | Warning |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P273, P301+P310, P303+P361+P353, P304+P340, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 55 °C (131 °F) |
| Autoignition temperature | 210 °C (410 °F; 483 K) |
| Explosive limits | 1–6.0% |
| Lethal dose or concentration | Lethal Dose (LD50, oral, rat): 5000 mg/kg |
| LD50 (median dose) | > 5000 mg/kg (rat, oral) |
| NIOSH | NA2977 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 120 ppm |
| IDLH (Immediate danger) | IDLH: 900 ppm |
| Related compounds | |
| Related compounds |
Isomeric octanes 2,2,4-Trimethylpentane Isooctane n-Octane |

