2,2,4-Trimethylhexane: More Than Just a Hydrocarbon
Looking Back: The Evolution of 2,2,4-Trimethylhexane
Walking through the history of chemical development, few people outside of chemistry circles stop to think about compounds like 2,2,4-Trimethylhexane. This molecule, a branched alkane in the octane family, quietly gained a place in motor fuel innovation decades ago. Researchers chasing better engine performance noticed that straight-chain hydrocarbons caused knock in early gasoline engines. Branching the carbon skeleton—enter 2,2,4-Trimethylhexane—increased octane ratings, reducing knock and pushing engines to run smoother and stronger. This compound started as a curiosity in organic synthesis, then made a name as petroleum refining improved and the automotive world asked for fuels that kept up with the promise of speed and power.
What Sets 2,2,4-Trimethylhexane Apart
This alkane doesn’t carry the fame of “octane” gasoline, but it shares a central feature: it's highly branched. 2,2,4-Trimethylhexane’s structure—eight carbons in total with three methyl groups strategically attached—sets it apart from straight octane and delivers better resistance to engine knock. This resistance does more than just let cars go faster; it extends engine life and cuts back on those damaging pressure surges. The product finds its way into test blends and high-performance fuel formulations because its chemical backbone mirrors the practical demands of real-world driving. Calling it just another hydrocarbon undercuts what it actually brings to the world of fuels and solvents.
Behind the Numbers: Properties and Specifications
Lay the compound down next to others in the gasoline spectrum and a few details jump out. It's a colorless liquid at room temperature, with a boiling range close to typical gasoline fractions. Chemically, it's inert—meaning it won’t react much outside its family—but that’s a double-edged sword. Good stability keeps storage straightforward, but environmental persistence raises eyebrows. Its density, vapor pressure, and flash point line up with typical hydrocarbon fuels, which means standard safety protocols for flammable liquids make sense here. Industry uses specifications like purity (often above 97% for lab grade), but most folks thinking about blending or research focus more on octane numbers, volatility, and combustion behavior than on a list of decimals.
Technical Labels and Real-World Tagging
On the technical front, 2,2,4-Trimethylhexane comes labeled according to chemical catalogs—as C9H20, with the CAS number 1678-24-0 ranking as its unique tag in regulatory databases. Chemists sometimes bump into its synonyms in literature: Isooctane, although more strictly reserved for the 2,2,4-trimethylpentane isomer, or simply “trimethylhexane” in older texts. These names hint at overlapping usage, sometimes muddying the waters for those hunting for specific properties. In fuel research, the shorthand often drops the numbers, but the real chemical world insists on accuracy—2,2,4- on hexane, not pentane.
From Crude to Compound: Manufacturing Challenges
Making 2,2,4-Trimethylhexane isn’t for the home chemist. Industrially, the process uses alkylation—reacting isobutene with 2,3-dimethylbutane in catalytic conditions. The reaction throws off plenty of byproducts, and the art of refining involves careful separation using distillation. Despite decades of trial and error, improvements in yield and purity remain critical topics in petrochemical research. The steps consume energy, and catalysts wear out, generating both cost and waste. Plant operators balance throughput and selectivity, faced with real consequences in both economics and environmental effects. The roots of its production tie back to the golden age of petroleum chemistry, but today’s refiners live in a world where margins are tight and scrutiny runs high.
Chemistry in Action: Reactions and Tweaks
Most uses rest on the backbone of this molecule staying put—its stability counts as a selling point when looking for low-reactivity blending agents. Still, under the right heat or with a spark of catalysis, 2,2,4-Trimethylhexane breaks down, isomerizes, or even cracks to smaller fragments. In the lab, researchers use high temperatures to study its pyrolysis products, mapping out potential environmental fates or checking fuel residues for signs of incomplete combustion. While not typically the starting point for more complex syntheses, its structure finds relevance in mechanistic studies where researchers want to watch molecular branching play out in real-time.
In the Field: Where 2,2,4-Trimethylhexane Matters
Everyday drivers probably never hear the name, but their cars run better partly because of testing with compounds like this. Refineries use 2,2,4-Trimethylhexane as a benchmark in fuel testing, checking against octane standards to guarantee engines get the right mix. Its unique support for anti-knock performance feeds innovation in racing fuel and aviation gasoline, where a small improvement can make a real difference. Beyond fuels, the solvent properties sometimes attract researchers who want a non-reactive, low-polarity liquid for chemical or industrial testing. For years, analytical labs measured the effects of additives using blends containing this molecule, helping shape how new fuel components stand up under stress.
Safety and Industry Operations: The Human Factor
Flammable liquids demand care. Like many hydrocarbons, vapors from 2,2,4-Trimethylhexane can ignite with just a spark. Industrial protocols focus on proper ventilation, grounding, and containment. Workers need training to handle spills safely and use personal protective equipment without cutting corners. Regulatory groups classify it with other gasoline-range hydrocarbons, so guidance from OSHA, NIOSH, and similar bodies makes a difference every single day in handling. Ignoring vapor exposure or the risks of long-term inhalation doesn’t pay off. Experience on the loading dock or in the sample room teaches respect that textbooks can’t deliver.
Pushing the Envelope: Research and Innovation
Analytical chemists use 2,2,4-Trimethylhexane to dig into combustion science, looking for ways to wring more efficiency from gasoline while slashing emissions. Universities run tests with it to model how new engine designs cope with real-world fuels. This compound pops up in academic journals, serving as a simple but robust model for branched paraffins. Research on advanced catalysts ties directly to the economics and sustainability of making high-octane components; as refining transitions to renewable inputs, synthesizing molecules like this from biofeedstocks marks a new frontier. Engineers explore how these hydrocarbons interact with alternative fuels, hybrid blends, and even next-generation lubricants.
Toxicity, Health, and Environmental Concerns
Health studies remind us that “simple” hydrocarbons can bring complicated risks. Inhalation of concentrated vapors can lead to dizziness, headaches, or—in rare cases of heavy exposure—more severe nervous system effects. Researchers map environmental fates, tracing how such compounds break down in water or soil. Persistence and potential for bioaccumulation raise issues for regulators. Studies show most of these branched alkanes volatilize quickly, lowering direct waterborne risk, but their presence in urban air continues to get attention. Workers exposed over long periods face added risks, and regulators weigh new findings when crafting exposure limits. Public trust in petrochemical safety ties directly to candor about these risks and visible enforcement of limits at every step.
Looking Ahead: What’s Next for 2,2,4-Trimethylhexane?
Every year brings sharper scrutiny to what goes in the tank and out the tailpipe. As electric vehicles carve a bigger place in transport, chemical innovators still look for better liquid fuels—especially for aviation and emergency services where batteries lag behind. 2,2,4-Trimethylhexane stands as both a benchmark and a target for synthetic biology, catalysis, and green chemistry. Researchers gear up to synthesize these molecules without fossil carbon, using engineered microbes or renewable feedstocks. The road forward depends on practical breakthroughs. Real progress means not just meeting old standards for octane, but doing it cleaner and smarter. Lessons learned from this one molecule shape the way chemists tackle the transition to cleaner energy, confronting the tension between efficient fuels and responsible stewardship head-on. Whether it starts in a refinery, a bioreactor, or a laboratory bench, the future of molecules like 2,2,4-Trimethylhexane reflects a world balancing performance, safety, and sustainability—one innovation at a time.
What is 2,2,4-Trimethylhexane used for?
The Role of 2,2,4-Trimethylhexane in Fuel Blending
2,2,4-Trimethylhexane, a branched-chain hydrocarbon, frequently finds purpose in the fuel world. Walk into a lab or refinery and you’ll see folks blending compounds like these to reach a specific octane rating in gasoline. Without enough branching in hydrocarbons, engines can rattle and knock, leading to rough rides and potential engine damage. 2,2,4-Trimethylhexane, with its structure, resists knocking better than many straight-chained alternatives. So, it supports safer and more efficient engines.
Through my own time working with fuel additives, engineers always chase a balance: making sure a vehicle runs smoothly with as little emissions as possible. Here, 2,2,4-Trimethylhexane matters because it lets refiners reach governmental standards for emissions without sacrificing performance. It’s not the only component in gasoline, but its contribution shows up every time a car starts quickly or climbs a steep grade without hesitation.
Supporting Research and Test Standards
Researchers use pure samples of 2,2,4-Trimethylhexane to benchmark instruments or test theories about combustion. It’s a compound straight out of an organic chemistry kit, stable and easy for chemists to measure. In standardized engine tests, controlling for one variable at a time builds trust in the results, and this compound’s predictability helps. I remember long mornings in a test facility swapping test fuels, and traceable consistency kept our measurements honest.
The National Institute of Standards and Technology (NIST) and similar organizations catalog compounds like 2,2,4-Trimethylhexane for this reason. Analytical labs count on it as a reliable choice when calibrating gas chromatographs, providing reference points that keep industry results valid.
Environmental and Human Health Angle
Gasoline blending doesn’t just focus on making engines run better. Choices line up with how toxic a compound might be or how much ozone it creates in tailpipe emissions. 2,2,4-Trimethylhexane, grouped under the C9 alkanes, doesn’t have the heavy risk profile of lead additives from decades ago, but safety precautions hold true. I’ve seen more conversations in recent years about personal protective equipment, proper ventilation, and disposal of hydrocarbon mixtures—no one wants old mistakes repeating themselves.
The Environmental Protection Agency (EPA) tracks compounds like this, watching for their environmental persistence. While hexanes break down more quickly in sun and air than heavier compounds, accidental releases or poor handling can still threaten water and soil quality. Industrial users need training and compliance programs to limit risks, and regular audits from regulators keep everyone aware of responsibilities. Simple, clear hazard labeling goes a long way to keeping people safe in labs and on the shop floor.
Finding Alternatives and Future Solutions
Push for alternative fuels grows each year, partly from the need to reduce petroleum reliance, partly from climate change urgency. 2,2,4-Trimethylhexane still plays a key role in existing gasoline, but researchers look for bio-based substitutes or drop-in molecules that would cut emission footprints while offering the same anti-knock benefits. Pilot projects sometimes use isooctane from green sources, but the jump from small batches to full-scale isn’t easy.
Leaving behind heavy technical jargon, the future feels like one of blending tradition with innovation. Careful handling, solid training, and smart chemistry ensure that compounds like 2,2,4-Trimethylhexane serve well today, while new approaches work steadily in the wings.
What is the chemical formula of 2,2,4-Trimethylhexane?
A Closer Look at a Simple Hydrocarbon
Anyone who’s ever filled up at a gas station has come close to 2,2,4-Trimethylhexane without even noticing it. This compound doesn’t make headlines like carbon dioxide or methane, but it plays a steady role in day-to-day life. If you look at its chemical structure, 2,2,4-Trimethylhexane carries the formula C9H20. That’s nine carbon atoms and twenty hydrogens. This formula points to a saturated hydrocarbon—an alkane, nothing fancy or reactive, but something that keeps industries rolling.
Why This Formula Matters Beyond the Lab
There was a moment, back in my high school chemistry class, when we talked about octane numbers and the knocks you hear in gasoline engines. Turns out, 2,2,4-Trimethylhexane has a hand in keeping engines running smoothly because everything about its molecular structure supports high-octane ratings. Higher octane means less knocking, greater efficiency, and fewer repairs. On road trips, the fact that this nine-carbon molecule is in the tank lets the car handle more without shaking itself part.
Looking at the facts, C9H20 might sound like just another chemical string, but its importance takes shape when considering its practical role. High-octane fuels owe a lot to molecules like this one. The world relies on this chemical for better internal combustion, and by extension, less wear on vehicles and lower maintenance costs for everyone.
Supporting the Big Picture: Science and Society
What always strikes me is how a chemical formula stands for more than atoms; it links to decisions in business, regulation, and the environment. C9H20 features in big studies about alternative fuels, because researchers keep searching for ways to meet high-octane needs without putting more stress on the planet. Gasoline blends change over the years as carmakers bump up engine pressure and governments set cleaner standards. Knowing what goes into those blends gives regular people more power to follow debates about energy and pollution.
Producers build on this knowledge to create fuels that serve both drivers and the environment. Efforts keep moving forward—some companies look at biology for new sources, hoping to match the performance of 2,2,4-Trimethylhexane with plant-based versions. Universities run projects on breaking the dependency on crude oil and using science to make old molecules like this one in modern, sustainable ways.
Finding Opportunities for Change
We all know gasoline carries big environmental baggage. The trick is not just sticking to old methods but experimenting with the formulas behind the scenes. If alternatives can mimic C9H20 without harmful by-products, progress feels real. That means supporting research, pushing for transparent labeling, and holding companies to clear, science-driven standards.
2,2,4-Trimethylhexane’s formula marks a starting point rather than a finish line. By understanding what makes it valuable—and where it falls short—scientists, regulators, and even drivers hold a slice of the solution for the future of fuel.
Is 2,2,4-Trimethylhexane hazardous or toxic?
What We Know About 2,2,4-Trimethylhexane
You might come across 2,2,4-Trimethylhexane in a chemical safety data sheet, or if you’re working around fuels or solvents in an industrial space. It belongs to the broader group of hydrocarbons often found in fuels, lubricants, and some manufacturing environments. It looks and smells a lot like other colorless liquids that can pose health risks, though its exact effects on people aren't always top of mind unless you’re working with it often.
Concerns About Exposure
Most of what we know comes from studies of similar hydrocarbons. Direct, prolonged exposure to the skin or lungs can cause irritation. Breathing high levels for a long period may eventually affect the central nervous system. Short-term exposure—like a splash on skin or a whiff in the air—often brings dizziness, headache, or nausea. At the job site, ignoring ventilation or skipping gloves adds risk. Hands can get dry or cracked, eyes start to sting, and the body absorbs these molecules more than many folks realize.
Fire Risks and Workplace Safety
Flammability is a big topic. 2,2,4-Trimethylhexane will ignite under the right conditions. Open flames and static electricity near storage drums can spark trouble, so safety managers post warning signs and keep fire extinguishers handy. In workshops where fuel is transferred between containers, the smell hangs in the air. One slip or missed leak can have consequences well beyond the initial spill.
What Science Says About Toxicity
Looking at animal studies and the symptoms reported by workers, long-term effects seem possible. Hydrocarbons with structures close to 2,2,4-Trimethylhexane have shown liver, kidney, and even nervous system toxicity when long exposure is part of the routine. No one is volunteering to be a test subject, and the full list of threats isn’t complete yet. Some studies point to possible links with chronic headaches or breathing difficulties. The U.S. National Library of Medicine lists the substance as harmful and suggests avoiding unnecessary exposure whenever possible.
Regulation, Education, and Safer Practices
There isn't a federal exposure limit specifically set for 2,2,4-Trimethylhexane that matches more widely known solvents, but product labels and safety data sheets include hazard warnings. Occupational health specialists encourage frequent breaks, strong airflow, and personal protective gear like gloves and goggles. Spill kits and proper storage make handling this stuff less stressful. Companies that care about keeping people safe often invest in regular air quality monitoring and offer training drills, so new hires know what to do if a drum leaks or someone feels sick.
Better Choices for the Future
Everyone deserves a safe workspace. I’ve watched coworkers feel invincible, only to end up suffering from headaches or skin problems after months of exposure. Nobody should learn safety by accident. With green chemistry on the rise, some manufacturers look for safer alternatives that mimic the useful properties of 2,2,4-Trimethylhexane but with a lower risk profile. Open conversations about hazards, regular reviews of old storage tanks, and updating handling protocols turn good intentions into real protection.
How should 2,2,4-Trimethylhexane be stored?
The Risks Behind the Name
2,2,4-Trimethylhexane doesn’t show up in daily conversation for most people, but those who work in chemical labs or industrial settings know it well. This compound comes with some well-known hazards. It’s flammable, and like most hydrocarbons, it can release vapors that threaten both health and workplace safety. Stories of workplace accidents often start with small mistakes—like leaving a container uncapped or storing chemicals next to a heat source. People sometimes forget how quickly bad things happen and how long they stick around.
Practical Steps Matter Most
Putting this solvent in a tightly sealed metal drum offers a solid baseline. Glass and plastic may crack, react, or leak if a careless knock happens. Thick-walled metal containers have saved more than one technician from vapor leaks and fire. Storage away from direct sunlight and heat makes a big difference, too. It only takes a little warmth for volatile liquids to send fumes looking for a spark. Placing chemicals in a designated, well-ventilated storeroom shrinks that risk a lot. I learned this lesson early on helping out in a university research lab. Renovated cabinets and new warning labels only took us so far—the game-changer came from the regular walk-throughs and clear separation of flammable solvents.
Staying Vigilant, Not Paranoid
A safety data sheet feels like a lot to get through, but the simple instructions—store in a cool, dry place with plenty of airflow, away from ignition sources—never fail. That’s because these rules come from real incidents, not just theory. I’ve seen coworkers get complacent and leave solvent bottles too close to old electric equipment in a cluttered storeroom. All it took was one faulty switch to trigger a small fire. Regular safety audits and employee training keep these situations from escalating. Quick reminders about using the right container and checking labels help even experienced workers refocus.
Supporting Facts and Standards
The National Fire Protection Association assigns flammable liquids like 2,2,4-Trimethylhexane strict guidelines: store below the flash point, never stack near oxidizers, and make sure to ground all containers during transfer. OSHA and local regulations back this up with clear mandates because incidents have proven their value. According to the Chemical Safety Board, workplace accidents involving improperly stored flammable chemicals affect thousands each year. Insurance claims and hospital visits follow preventable oversights. These aren’t abstract risks; they’re written in lab accident logs and burned-out storage cabinets. Good habits—capped containers, labeled shelving, duplicate logs—keep these numbers down.
Solutions Don’t Require Fancy Equipment
Reliable storage starts with simple moves. Lock up solvents in a fire-rated storage cabinet, always double-check those labels, separate out incompatible compounds, and use containers designed for flammable liquids. If possible, set up spill-containment trays or use absorbent pads so that leaks catch early. Routine checks beat expensive sensor systems, especially in smaller shops. Assign responsibility to specific workers—if everyone cares a little, nobody skips the important steps.
Bringing Safety Home
Safe storage isn’t just a regulatory box to check. It keeps workspaces secure and allows folks to focus on their tasks without the fear of an accident around the corner. In my own experience, the places with clear organization, direct communication, and simple safeguards always felt safer, more efficient, and less stressful for everyone involved. People respect the chemical, stay alert, and trust that the system looks out for their well-being.
What are the physical properties of 2,2,4-Trimethylhexane?
What Sets 2,2,4-Trimethylhexane Apart?
2,2,4-Trimethylhexane shows up as a clear, colorless liquid, a member of the broader alkane family. With several methyl groups branching out from its backbone, its molecular structure gives it a lower density and higher volatility compared to some straight-chain cousins. For folks working with fuels or laboratory standards, these twists in the molecule make a real difference.
Boiling and Melting Points: Where It Shifts State
If you heat it up, 2,2,4-Trimethylhexane boils at about 135°C. That's lower than straight-chain nonane, which shows a higher boiling point. The branches interrupt tight molecular packing, so less heat is needed to push it into the vapor phase. If frozen, this compound turns solid around -91°C. Its melting point speaks to how loosely the molecules arrange themselves compared to less-branched alkanes.
Density and Solubility: Lightweight and Hydrophobic
In my lab days, pouring 2,2,4-Trimethylhexane left a familiar sensation. At room temperature, its density sits close to 0.72 g/cm³. That means it floats on water, and spills are easier to track than some denser chemicals. Like most alkanes, water wants nothing to do with it; the hydrocarbon backbone shrugs off polar interactions. On the other hand, it blends smoothly with other organic solvents and liquid fuels.
Viscosity and Volatility: Mobility in Action
Run this compound through tubing, and it flows with ease due to low viscosity. No surprise here—branching causes less internal friction than long, straight hydrocarbon chains. Because of its volatility, you can catch its faint paraffin odor not long after opening a container. In real-world terms, high volatility means evaporative losses if storage isn't airtight—a constant headache for chemists in warm labs.
Flash Point and Flammability: Safety Considerations
Safety teams always keep a close eye on flash points. For 2,2,4-Trimethylhexane, this number lands between 24–28°C. This low figure translates to serious caution during summer or in hot process areas. Any vapors above that temperature can catch fire if there's a spark or open flame. Precise venting, spill control, and proper labeling can't be skipped; this compound demands them.
Real-World Roles and Health Matters
Because of its volatility, 2,2,4-Trimethylhexane helps calibrate engines and fuel systems. It's also a reference point in environmental studies tied to petroleum releases. Breathing in too much vapor might yield headaches, dizziness, or mild narcosis—symptoms I've seen handled on shop floors by simply upping ventilation and using closed systems for transfers.
Making Work Safer and Smarter
Familiarity with physical properties clears up a lot of practical decisions. If a lab uses 2,2,4-Trimethylhexane, they store it in cool, flame-proof cabinets and favor glass bottles with proper seals. Glove and goggle use cuts down on accidental exposure. For spill remediation, foam and dry chemical extinguishers stand ready, never water—which just spreads a floating fire.
Looking Ahead: A Matter of Awareness
The more people know about compounds like 2,2,4-Trimethylhexane, the fewer stories circulate about accidents or waste. Facts on boiling point and flash point aren’t just figures on a chart; they shape every safe and successful interaction, from fuel testing to chemical handling in real life.
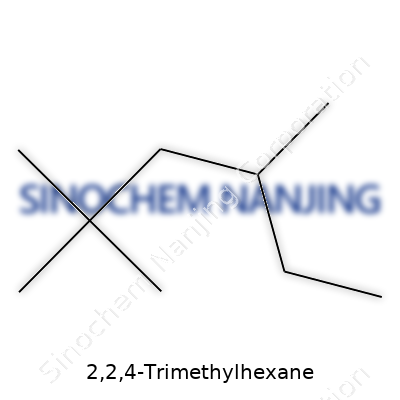
| Names | |
| Preferred IUPAC name | 2,2,4-Trimethyloctane |
| Other names |
Diisobutylmethane 2,4,4-Trimethylhexane |
| Pronunciation | /tuː tuː fɔːr traɪˈmɛθɪl ˈhɛkseɪn/ |
| Identifiers | |
| CAS Number | 1678-36-8 |
| Beilstein Reference | 2841599 |
| ChEBI | CHEBI:89231 |
| ChEMBL | CHEMBL15461 |
| ChemSpider | 11027 |
| DrugBank | DB14096 |
| ECHA InfoCard | 0739357f-466d-4df0-b87f-bbbb9ea6d3bb |
| EC Number | 202-534-2 |
| Gmelin Reference | 105639 |
| KEGG | C08399 |
| MeSH | D014255 |
| PubChem CID | 12088 |
| RTECS number | XN8225000 |
| UNII | Z1532M90G5 |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID6020145 |
| Properties | |
| Chemical formula | C9H20 |
| Molar mass | 114.23 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.690 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.8 |
| Vapor pressure | 2.93 mmHg (25°C) |
| Acidity (pKa) | 18.41 |
| Magnetic susceptibility (χ) | -8.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.405 |
| Viscosity | 0.69 cP (20 °C) |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 249.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -295.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –5463.4 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H304, H315, H336, H411 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P273, P301+P310, P303+P361+P353, P304+P340, P312, P331, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 59 °F (NTP, 2022) |
| Autoignition temperature | 210 °C (410 °F; 483 K) |
| Explosive limits | 1–6 %(V) |
| Lethal dose or concentration | LD50 oral rat 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5000 mg/kg (rat, oral) |
| NIOSH | RN0177 |
| REL (Recommended) | 35 mg/m3 |
| IDLH (Immediate danger) | 900 ppm |
| Related compounds | |
| Related compounds |
Isooctane 2,2,3-Trimethylbutane 2,3,3-Trimethylpentane 3-Ethyl-2,2-dimethylpentane 2,4-Dimethylhexane |

