2-(2,4-Dichlorophenoxy)Propionic Acid: A Look Beyond the Label
Historic Routes of Discovery and Use
2-(2,4-Dichlorophenoxy)propionic acid, often recognized in agronomy circles due to its functional role in weed management, has a backstory that reaches into the heyday of synthetic herbicide development. During the mid-20th century, as agriculture began looking for answers to labor-intensive weed control, researchers dove headlong into the world of substituted phenoxy acids. This class of chemicals offered a new route for improving crop yields in the face of persistent weed competition. The journey toward mass application found its champions in farmers looking for more efficient land stewardship, and the compound quickly earned a passport to broadacre fields, orchards, and research plots alike.
What’s Inside the Bottle?
This acid stands as a crystalline solid at room temperature, sometimes appearing as an off-white powder. Water solubility terms alone can’t capture the whole picture because, while it dissolves to a degree, it prefers organic solvents when forming concentrated solutions. Chemically, it carries two chlorine atoms on its aromatic ring and a characteristic ether linkage, contributing both to its selectivity against unwanted plants and its persistence in the soil environment. These molecular features don’t arrive by accident—each tweak and modification over the years brought about fresh understanding of how the compound interacts in real fields, under sunlight, with the biomes living in and around roots.
Specifications and Labeling—More Than Small Print
Reading a technical sheet or a label, you find specifics about purity, melting point, and concentration—all points that shape end use and application. Farmers and researchers rely on this information to judge application rates that balance weed control and crop safety, but every bottle tells just part of the story. Stable shelf life and clarity on breakdown products matter just as much, because every drop that misses its target or drifts in the wind has consequences beyond the intended acreage.
Preparation: From Theory to Factory Floor
The creation of 2-(2,4-dichlorophenoxy)propionic acid starts with the synthesis of 2,4-dichlorophenol, which reacts with propionic acid under carefully controlled conditions. Industrial chemists keep a close watch on reagent purity and reaction temperature, not just for yield but to guard against unwanted byproducts. Their experience helps avoid costly waste and ensures consistency across thousands of metric tons, especially given the ever-tightening regulations on trace contaminants in crop protection products.
Pathways for Chemical Modifications
Chemists don’t often leave a molecule alone—tweaking side chains, playing with ring substitutions, or combining with salts to make the substance more effective or safer for users and the environment. Modifications in the molecule’s structure let formulation scientists offer granular or liquid versions to match everything from solo application by backpack sprayer to mix-in-the-tank logistics of row crop farming. Every adjustment draws on years of field trial results and regulatory feedback, pushing for broader application windows, faster breakdown times, or safer handling in storage sheds and transport trucks.
Names and Alternatives on the Market
This compound appears in literature under several banners, from 2,4-DP to dichlorprop, and even under international trade names in different corners of the world. Switching from one synonym to the next, users trace a trail of studies and application guides, revealing how researchers across continents and decades tackled persistent weed problems with this molecule as a central piece in the puzzle.
Safety and the Daily Realities of Field Use
Farmers and workers face a balancing act, weighing the value of weed control against risks of handling synthetic chemicals. Direct contact can cause irritation, and improper storage or spills may bring about risks for groundwater or aquatic systems. My own family farm always handled chemical storage with a seriousness born of experience—ventilated lockers, careful training, eyewash stations nearby—because safety isn’t a checklist item, it’s a way of thinking. Regulatory agencies set boundaries and exposure limits after years of toxicological review, but it’s field-level education and repeated reminders that reinforce careful, respectful handling on each farm.
Fields of Impact—Where It Shows Up
From wheat fields in North America to rice paddies overseas, 2-(2,4-dichlorophenoxy)propionic acid finds its role as a proven tool against broadleaf weeds that sap nutrients and water. The story doesn’t stop at row crops. Turfgrass managers, orchard growers, and even forestry professionals call on this product when seeking precision in selective weed management, aiming to boost productivity and safety in tough-to-navigate conditions.
Research—Seeking Smarter Solutions
Labs around the world continue to probe how 2-(2,4-dichlorophenoxy)propionic acid interacts with crop and weed alike, running genetic screens, field trials, and metabolic breakdown studies with an eye for surprises. New research explores how formulation tweaks may lower environmental load or reduce carryover that risks hitting the next season’s sensitive crops. Collaborations between public researchers and input suppliers push for a clearer map of breakdown pathways, seeking the sweet spot of efficacy and environmental stewardship. These studies drive not only adoption on the ground but also shape what gets approved by regulators and how stewardship guidelines frame real-world practice.
Toxicity—Walking the Science Tightrope
Toxicological profiles demand respect. Ingesting, inhaling, or excessive skin contact with this compound brings health concerns ranging from mild irritation to more severe systemic effects, depending on dose and frequency. Decades of animal and environmental studies guide current exposure thresholds, ensuring they stay within boundaries proven to keep workers and non-target life safe. My own view, shaped by talking with seasoned applicators and reviewing the latest health advisories, is that informed users rarely treat storage or handling as routine. They recognize cumulative exposure risks and follow label guidance, not out of compliance, but out of shared responsibility for community and local ecosystems. Regulators review new findings regularly, sometimes tightening allowable residue levels or restricting certain uses based on emerging results.
The Road Ahead—Meeting Challenges and Expectations
As populations grow and farms look for ways to do more with less input, the future for 2-(2,4-dichlorophenoxy)propionic acid sits at a crossroads. New pest pressures and increasing scrutiny on active ingredient residues make continued research and stewardship essential. Emerging alternative herbicide chemistries, biological controls, and precision farming tools may complement or, in some scenarios, replace older synthetic acids. Looking ahead, I see a space for this compound—one where new data and smarter application technology keep its usage within science-backed safety margins, supporting robust yields while protecting people and the environment for the long haul.
What is 2-(2,4-Dichlorophenoxy)Propionic Acid used for?
What Is It, Really?
2-(2,4-Dichlorophenoxy)propionic acid, usually called 2,4-DP, stands out as a synthetic chemical mainly targeted at weed control. If you’ve ever had to battle wild grass or tough weeds in a crop field, you probably know these invaders don’t leave with polite requests. Farmers and groundskeepers turn to this herbicide to cut down specific broadleaf plants that crowd out useful crops. The backbone of this compound is a structure tailored for selectivity: it means 2,4-DP aims for unwanted growth while leaving most desired plants alone.
How It Shows Up in the Real World
Picture a golf course. Lush, rolling greens easily lose their shine when weeds begin creeping along the edges. Maintenance crews spray 2,4-DP in measured amounts, and that helps keep those weeds from spoiling the scene. Out in wheat or barley fields, it’s much the same story. Farmers walk a fine line—using enough 2,4-DP to push back the weeds but holding back enough to keep soil and crops safe. That balance doesn’t get written into instruction manuals; it comes from years of watching how each season’s weather and soil change the playing field.
Decades ago, harsher herbicides left behind problems—crops wouldn’t grow right, neighbors would complain, and water runoff became a worry. Newer compounds like 2,4-DP fixed some of those headaches. The selective targeting saves money, cuts risk for the folks applying it, and lightens the environmental punch. That said, no chemical comes without downsides. Runoff from fields sprayed with weed killer can sneak into streams and lakes, sometimes causing trouble for fish and water plants.
The Safety Questions
People have grown more careful about what lands on their food and in their drinking water. Nobody loves the idea of too much chemical sticking to their salad. Studies from groups like the EPA and European Food Safety Authority dig into these questions year after year. Research shows 2,4-DP breaks down in soil faster than some older weed-killing chemicals, which takes some pressure off the ecosystem.
That’s only part of the story, though. Long-term exposure, like what might happen to farm workers, sparks tough debates about health. Some studies link repeated exposure to pesticides in general with a higher risk of certain illnesses. The numbers on 2,4-DP specifically point to low risk if it’s used the right way, but mistakes and accidents have happened. This gets everybody—scientists, regulators, farmers—arguing about best practices, tighter rules, and safer substitutes.
Is There a Better Way?
No one tool solves every weed problem. Biological controls, crop rotation, and planting cover crops can help fight weeds without chemicals. But those options take more labor and sometimes don’t scale well to big farms. The debate keeps moving, with scientists looking for herbicides that break down more quickly, hit only what they should, and don’t show up where they shouldn’t.
Experience teaches that chemicals like 2,4-DP aren’t a magic solution. They fit as one part of a bigger approach to managing weeds and protecting food supply. New research keeps pushing for reduced use, better timing, and sharper safety training. For anyone with feet in the dirt, reading labels and understanding the land goes just as far as any jug of 2,4-DP.
Is 2-(2,4-Dichlorophenoxy)Propionic Acid safe for humans and the environment?
Understanding 2-(2,4-Dichlorophenoxy)Propionic Acid
2-(2,4-Dichlorophenoxy)propionic acid, better known as 2,4-DP, turns up in many weed killers. For years, landscaping teams, golf course managers, and everyday homeowners have relied on this herbicide to control unwanted plants. Most folks using these products rarely pause to ask if this compound is truly safe for people or the planet.
Health Concerns that Deserve Attention
Whenever I spread fertilizer on the lawn or clear the driveway with a weed killer, I always check the label. Herbicides with names like 2,4-DP spark questions for good reason. Safety sheets warn about keeping the spray away from kids and pets. The reason? Some studies show exposure—especially if handled without gloves or a mask—can irritate the skin, eyes, and respiratory system. Lab research on animals connects long-term exposure to higher risks of liver and kidney damage. Some links to hormone disruption also start to show up in repeated testing, which hits home for families that want to avoid unseen health risks.
People who work with 2,4-DP regularly, like groundskeepers or agricultural workers, face more risk than the average person. Protective gear and following manufacturer directions shrink the dangers, but accidents happen. That's not just theory. Researchers from the National Institutes of Health tracked pesticide applicators and found that even well-trained adults sometimes report headaches, dizziness, or nausea after handling weed killers that contain 2,4-DP.
The Environmental Impact Isn’t Small
Walking along a creek after heavy rain, the chemical smell sometimes lingers, proof that what lands on the grass doesn’t always stay there. Runoff from lawns and fields carries 2,4-DP into streams and lakes. There, it can do more than knock down weeds. Fish and aquatic insects can be sensitive. Research from the United States Geological Survey documents the breakdown products of 2,4-DP in water and soil, showing they linger for weeks or months. Some residue even shows up in remote places where it was never sprayed, circling back through water and wind.
That's not the kind of cycle anyone wants for the environment. Birds and pollinators—bees, butterflies, and beetles—lose out when habitat disappears thanks to unchecked spraying. Reports also tell of resistant weeds cropping up, making farmers use even more herbicide in a frustrating spiral.
Better Choices and Safer Habits
People often ask what to do with a stubborn weed problem. Reaching straight for chemical weed killers can feel like an easy fix, but options exist. My own experience swapping out the herbicide for hand-weeding and mulching keeps the yard in check while skipping the chemical risks. Organic landscaping services keep popping up for the same reason—less reliance on harsh weed killers.
Some regions now require buffer strips near waterways, fewer spraying days during rain, and public notification when herbicides get used. These efforts come from the growing understanding that controlling one problem—the weeds—should not create new dangers for people or the ecosystem.
More folks question what’s in their backyard spray bottles, and those questions push companies to design safer products. Staying informed, asking questions, and choosing alternatives can help protect both our families and the green spaces around us.
How should 2-(2,4-Dichlorophenoxy)Propionic Acid be stored and handled?
Treating Farm Chemicals as Serious Business
I remember walking into the chemical shed on my uncle’s farm for the first time. Rows of bottles—names I couldn’t pronounce, warnings in bold red writing, and a pungent scent always hanging in the air. Those labels tell you these aren’t products to sling around carelessly. 2-(2,4-Dichlorophenoxy)propionic acid, better known as a selective herbicide, comes with heavy requirements for proper storage and handling. Ignoring them brings real risks, not just to the crops but to ourselves, neighbors, pets, and even drinking water.
Safe Storage Starts with Common Sense
Any container of 2-(2,4-Dichlorophenoxy)propionic acid finds its best home on a cool, dry, and well-ventilated shelf—never basking in sunlight or exposed to moisture. Heat breaks it down faster, and excess humidity increases the chance of corrosion or leaks. Ordinary home shelves or sheds sometimes fall short. Dedicated, locked cabinets crafted from sturdy, chemical-resistant material keep the acid away from children, curious pets, and accidental spills.
Mixing this chemical with food, feed, seed, or medicine in storage only invites unnecessary trouble. Cross-contamination picks up speed that way. Best to stand each product separately—no sharing shelves. A waterproof label should always remain clear and intact, making identification quick even if the bottle grows dusty.
Good Habits Keep Trouble Away
People get tempted to cut corners. It might seem smart to reuse empty milk jugs or soda bottles. That shortcut sets up a disaster down the road, especially because herbicides can look like harmless liquid if left unlabeled. Always reach for the manufacturer-provided containers. No transferring, no improvising.
If a bottle cracks or leaks, sponge up spills with absorbent—and safe—materials, such as sand or clay granules, not rags you’ll later toss in regular trash. Scrubbing with bare hands shouldn’t cross anyone’s mind. Gloves and goggles make real sense. Research from the National Institute for Occupational Safety and Health links long-term herbicide exposure to skin rashes and eye irritation, so personal protective equipment proves more than overkill.
Ventilation and Preparation
Stuffy, cramped sheds trap vapors. The right location opens to a breeze or has exhaust fans installed, so breathing gets easier, and fumes don’t linger. If working outdoors, stay upwind. Once, during my college days, a careless mix in a cramped barn left me coughing for days—a lesson not soon forgotten.
Before handling, experts point to the value of training. The US Environmental Protection Agency recommends reviewing safety data sheets before every season—not just reading them, but talking through what to do in the event of a spill, splash, or fire. Every coworker or family member should know these basics, not only the farm manager.
Disposal Means Responsibility
Modern farming owes a debt to environmental stewardship. Never pour leftovers down drains, into fields, or garbage cans. Community hazardous waste programs exist for a reason. Following their process costs a little more time, but it keeps soil and rivers cleaner for everyone.
Sticking Together for Safety
Every time we handle 2-(2,4-Dichlorophenoxy)propionic acid with care, we protect both our crops and our community. Real safety comes from consistent, honest efforts—not shortcuts. Clear labeling, protective gear, smart storage, and safe disposal aren’t suggestions. They’re the line between regret and responsibility in the long run.
What are the potential side effects or hazards of using 2-(2,4-Dichlorophenoxy)Propionic Acid?
Understanding the Risks
2-(2,4-Dichlorophenoxy)Propionic Acid, sometimes known in agricultural circles and chemistry classes, plays a key role in fields as a herbicide. It looks like a cousin to 2,4-D, which has stirred its own debates for years. If you’ve worked around farms or even well-tended suburban lawns, you’ve probably come across chemicals like this. With repeated use, growers notice crops grow cleaner while weeds get knocked out, but the side effects can pile up for people, animals, and the ecosystem.
Potential Health Side Effects
Spending time around this chemical—handling it, spraying it, or even breathing the air after application—can irritate skin and eyes. Folks report red, itchy skin or a burning feeling in the throat after exposure. It gets worse if the wind picks up and sends droplets or vapors into places they don’t belong. In some rough cases, people suffer from headaches, dizziness, or nausea. If someone doesn’t wash it off right away or inhales too much, symptoms stick around longer than anyone would like.
America’s National Institutes of Health flags chemicals like this as tricky. Over time and with high doses, they start chipping away at liver function and kidney health in animal studies. No one wants to gamble with those risks. The International Agency for Research on Cancer considers some of these kinds of herbicides as possible carcinogens. Science hasn’t given a unanimous answer for every detail, but the suspicion stays.
Environmental Hazards
This chemical doesn’t stay put after spraying. It seeps into soil, drains with rainwater, and rides streams that lead straight into lakes or rivers. Aquatic life takes a hit—fish exposed to chemicals like this don’t thrive. Insects and worms living in the soil struggle or vanish, which breaks up the food web. No-till farming caught on partly to reduce runoff, but even then, traces end up where they shouldn’t. Drinking water supplies sometimes reveal tiny amounts of 2-(2,4-Dichlorophenoxy)Propionic Acid, which worries folks living upstream and downstream alike.
Real-World Solutions
Farmers and land managers keep looking for ways to protect crops while protecting themselves and the broader ecosystem. It starts with proper training and gear. Wearing gloves, goggles, and breathing masks during mixing and application keeps the chemical off skin and out of lungs. Safe storage matters—a locked shed away from water supplies and food keeps accidents down. Labels and manuals sound boring, but following them keeps people safer and cuts down on careless spills.
Switching up weed management can chip away at the issue. Crops get rotated, weed barriers go down, or mechanical weeders replace some of the chemicals. Plant scientists keep working on crops that resist pests without as many sprays. In some towns, local governments put limits on when and how these chemicals go into public spaces. This way, playgrounds and picnic spots stay safer for families.
People benefit from staying informed. Talking to local extension offices helps landowners know what’s in their soil and water, and health clinics can walk folks through symptoms and provide checkups for those who are at risk. Honest conversations lead to better practices—and help protect not just crops, but everyone who lives near them.
What is the recommended dosage or application rate for 2-(2,4-Dichlorophenoxy)Propionic Acid?
The Role of 2,4-DP in Weed Management
For years, 2-(2,4-Dichlorophenoxy)Propionic Acid—often called 2,4-DP or dichlorprop—has played a solid role in agricultural weed control, especially in cereal grains and turf. Farmers use it to fight off pesky broadleaf weeds that rob crops of nutrients and sunlight. The catch is, success hinges on getting the application rate just right.
Why Dosage Makes Such a Difference
Applying too much 2,4-DP risks crop damage and can cause herbicide carryover in the soil, harming next season’s growth. Too little, and weeds bounce back, forcing repeated sprays and higher costs. My experience walking wheat fields after a heavy-handed spray left patches yellowed around the edges drove home how easily over-application sets a farmer back. The soil pays the price, and so does the yield at harvest.
Recommended Application Rates: What the Science Shows
Research has consistently landed on a rate between 0.56 and 1.12 kilograms of active ingredient per hectare (about 0.5 to 1 pound per acre) for cereal grains and grasslands. The actual spot within this range depends on the specific weed species present, their growth stage, and crop type. Young, actively growing weeds usually require less. Larger, harder-to-control species demand the higher end of the scale.
For creeping Charlie, dandelion, and other stubborn lawn invaders, residential turf usually gets between 0.84 and 1.12 kilograms per hectare. The pulse of academic literature supports splitting applications where pressure is high, using two lighter treatments spaced a few weeks apart rather than dumping it all at once. This reduces risk to the grass and gives better knockdown of weed seedlings.
How Environmental Factors Shift the Equation
Weather isn’t just a background detail here. Wind, humidity, and rainfall both before and after spraying can swing results a few notches in either direction. A summer squall an hour after application can wash much of the herbicide off target plants, wasting product and letting weeds recover. On hot, dry days, spray solution evaporates faster and the risk of drift grows, which means less herbicide lands where intended. I’ve seen neighbors save money and protect waterways by checking hourly forecasts the morning of a treatment rather than just spraying because the calendar says so.
Best Practices for Safety and Sustainability
Label instructions from manufacturers and local extension offices pack in valuable, real-world advice. The EPA and Health Canada both require buffer zones near water and recommend no-spray times ahead of rain or irrigation. Simple tools like flat-fan nozzles and hand-held drift shields do more than high-tech solutions, especially on small acreages where overspray can become a bigger headache.
Rotating herbicides and never exceeding recommended amounts helps slow the stubborn march of resistant weeds. It’s tempting to double down when results lag, but repeated overuse just shortens the tool’s working life. Relying on integrated management—like using crop rotation, cultivating in early spring, or keeping competitive stand densities—keeps the tool fresh for years instead of burning it out after one or two seasons.
Solutions that Stick
Sticking to the established rate, watching the weather, and incorporating non-chemical tactics adds up to both better weed control and a farm that works with nature, not against it. The right dose respects the power of the chemical without letting it run the show. Years in the field have shown that careful planning, field scouting, and keeping up with university guidelines help everyone get more from each spray and avoid both crop injury and environmental fallout.
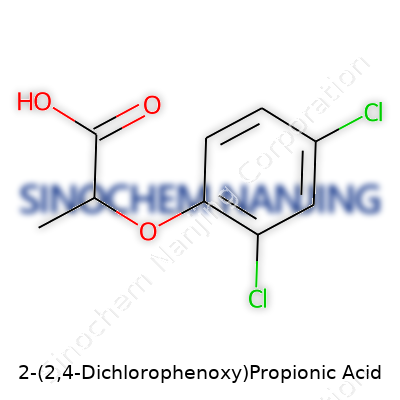
| Names | |
| Preferred IUPAC name | 2-(2,4-dichlorophenoxy)propanoic acid |
| Other names |
2,4-DP Dichloroprop Dichlorprop 2-(2,4-Dichlorophenoxy)propanoic acid |
| Pronunciation | /tuː baɪ ˌtuː fɔːr daɪˌklɔːrəˈfiːnɒksi prəˈpɒnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 120-36-5 |
| Beilstein Reference | 1281197 |
| ChEBI | CHEBI:1707 |
| ChEMBL | CHEMBL1511 |
| ChemSpider | 7277 |
| DrugBank | DB02926 |
| ECHA InfoCard | 03e789b0-e84a-44f2-a3f7-2b8973c7942e |
| EC Number | 201-956-0 |
| Gmelin Reference | 136607 |
| KEGG | C06742 |
| MeSH | D004051 |
| PubChem CID | 9483 |
| RTECS number | UF8225000 |
| UNII | C8F1G60XGJ |
| UN number | UN3334 |
| CompTox Dashboard (EPA) | DTXSID6023275 |
| Properties | |
| Chemical formula | C9H8Cl2O3 |
| Molar mass | 233.06 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.42 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 1.1 x 10^-5 mmHg (25°C) |
| Acidity (pKa) | 3.1 |
| Basicity (pKb) | pKb = 13.15 |
| Magnetic susceptibility (χ) | -38.9e-6 cm³/mol |
| Refractive index (nD) | 1.565 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -538.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3349.2 kJ/mol |
| Pharmacology | |
| ATC code | C01CA03 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05, GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H410 |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P337+P313, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 132°C |
| Lethal dose or concentration | LD50 oral rat 825 mg/kg |
| LD50 (median dose) | 685 mg/kg (rat, oral) |
| NIOSH | XP9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
2,4-Dichlorophenoxyacetic acid 2-(2,4-Dichlorophenoxy)butyric acid MCPA (2-methyl-4-chlorophenoxyacetic acid) 2,4-Dichlorophenol 2,4,5-Trichlorophenoxyacetic acid |

