2-(2,4,5-Trichlorophenoxy)Propionic Acid: Unpacking the Legacy, Impact, and the Road Ahead
Tracing the Rise: A Look Back at Discovery and Uses
In the history of agriculture, 2-(2,4,5-Trichlorophenoxy)propionic acid, often known as 2,4,5-TP or Silvex, played a role that mirrored both innovation and controversy. Its story started during a time when chemists pursued reliable methods to control invasive plants. By the early 1950s, researchers saw real promise in this family of synthetic auxins, which mimicked plant hormones and triggered weeds to grow themselves to death. Instead of waiting for seasonal cycles or relying on labor-heavy clearing of fields, farmers and public land stewards could mix up a solution and tackle stubborn brush. The chemical does its job quickly and targets broadleaf plants, leaving grasses mostly untouched. This seemed ideal for right-of-ways beneath power lines and railroads, as well as rangeland management where brush threatened grazing area.
Getting to Know the Molecule
The backbone of 2-(2,4,5-Trichlorophenoxy)propionic acid comes from trichlorophenoxyacetic acid and adds a propionic acid group, tweaking its activity compared to similar molecules. On a shelf, you’d spot it as a colorless to pale yellow solid, fairly stable under normal storage but not keen on open sunlight or strong oxidizers. Its formula speaks to all that chlorine – each atom boosting the herbicide’s persistence and giving it considerable muscle against deep-rooted weeds. This chemical is only sparingly soluble in water, leaning instead toward organic solvents for dissolving and spray application. For users, these properties shaped how it could be made into concentrates, granules, or ready-to-use solutions.
Inside the Lab: How It Gets Made
Manufacturers start by reacting 2,4,5-trichlorophenol with 2-chloropropionic acid in a controlled environment, often under basic conditions to push the reaction along. During synthesis, it’s crucial to keep eye on temperature, purity of raw materials, and prevent uncontrolled byproducts. Technically, it’s not a complex reaction, but vigilance holds importance because unwanted dioxins – highly toxic contaminants – can slip in if temperature control slips. Over time, procedures and technology kept improving to check impurity levels, but the risk lingers in older production processes or poorly regulated operations.
Technical Specs and Honest Labeling
Any pack of 2,4,5-TP you’d have found once came with clear chemical information: the main active ingredient percentage, lists of inert fillers, solubility stats, and handling protocols. Labels flagged hazards tied to skin or eye contact and spelled out safe storage rules. As global scrutiny on chemical safety sharpened, regulatory agencies revised label rules to factor in environmental impact and human health data, not just field performance. Labels also started marking proof for dioxin testing, reflecting public concern sparked by high-profile contamination events. Honest labeling lets workers, farmers, and the public make educated choices – a standard that only gets more vital as information flows more freely.
How It Behaves and What It Can Become
Once this compound hits the environment, it can take paths beyond its maker’s intent. Sunlight and soil organisms eventually break it down, but that process drags out, especially where oxygen runs low. Lab studies have recorded various breakdown products, some less harmful, but persistence in soils or sediments poses worry for water tables and food safety. Chemists over the years have attempted modifications, adjusting the structure to dial back environmental persistence or shrink toxicity without giving up weed fighting power. Achieving that balance has proven tricky – drive down persistence, and the herbicide may need frequent re-applications, adding cost and work.
Other Names and How It's Recognized Worldwide
Across patents, regulation, and academic literature, 2-(2,4,5-Trichlorophenoxy)propionic acid goes by several aliases: Silvex, 2,4,5-TP, and less commonly, Fenoprop. These synonyms reflect its use and registration in different regions, and researchers have flagged the need for tighter harmonization of chemical names to avoid confusion. This runs deeper than paperwork. If a contaminated product travels or research draws from the wrong safety data sheet, mistakes can happen—reminding us that clear naming isn’t just a formality in the chemical world.
The Safety Question: Lessons Learned and Operations Improved
Few chemicals have led to such heated scrutiny over safety as the family containing 2,4,5-TP. Early on, few in the field wore anything more than boots and gloves. Toxicity cases and environmental fallout led to stricter controls in mixing, spraying, and disposal, and industrial producers moved to build dioxin measurement into every batch's release. Regulators in North America and parts of Europe banned or restricted its use after growing evidence that production impurities posed serious risks. Some international markets reacted later, but the lessons from mishandled chemicals shaped not just the rulebook but the culture around herbicide safety across many compounds. I’ve seen this shift reflected in today’s training sessions, where even the most seasoned applicators respect these products as tools that demand caution.
Real-World Impact: Where It Was Applied
Looking at county extension records and government contracts, 2,4,5-TP dominated brush control operations for decades, standing alongside its cousin 2,4-D in weed management. Utility companies leaned on it to keep rights-of-way clear. Ranchers and farmers eyed it for restoring grass on overgrown pastures. But overuse sparked pushback – drift damaged desirable crops, and residues appeared in unexpected watersheds. Field experience and lawsuits both called attention to how even well-intended applications could spill over into risks for neighbors or wildlife. As alternatives and new technologies showed up, reliance dropped, but a look at historic herbicide maps still shows its fingerprints spanning huge swathes of land.
The Engine of Progress: Ongoing Research and Shifts in Focus
Research on 2-(2,4,5-Trichlorophenoxy)propionic acid split into two paths. Some researchers hunted for breakdown strategies, bioremediation tricks, or waste handling routes to clean up legacy contamination. These teams have piloted fungi, bacteria, and advanced oxidation methods that target residues down to trace levels. Others still ask how selective herbicidal activity can be harnessed with less risk – Could tweaking the molecule or designing new blends reduce drift or collapse toxicity without banning chemical controls outright? Much of the world shifted toward integrated management, combining targeted chemicals with mechanical or biological tactics for sustainable results. Makers know future regulatory hurdles will only grow, pushing innovation and accountability.
Weighing the Toxicity: Science and Human Cost
At the heart of 2,4,5-TP’s reputation sits its association with dioxins, especially TCDD, which turned up as a contaminant during manufacture. Decades of toxicology research layered animal studies, epidemiology, and environmental health reports. Evidence has linked exposure to reproductive harm, immune disruptions, and potential cancer risks, with the dioxin impurity often commanding the spotlight. A generation faced fallout, especially those handling or living near large-scale applications. Agencies responded with bans, hazard communication, and long-term health monitoring in affected communities. Even now, monitoring and remediation for dioxins tied to past use shape public policy debates and set a higher bar for modern chemical approval worldwide.
Looking Ahead: Better Chemistry, Smarter Choices
Thinking about 2-(2,4,5-Trichlorophenoxy)propionic acid’s future, the conversation has shifted from revisiting past mistakes to driving progress in chemical stewardship. Modern weed control leans less on persistent synthetic herbicides, exploring both improved molecules and whole-system strategies for vegetation management. Regulatory frameworks now bring in stakeholder voices and set tighter standards for environmental monitoring before products reach the market. For today’s scientists and decision-makers, this chemical’s story offers a clear lesson: the drive for productivity must always be checked by hard science, transparent risk assessment, and the willingness to change course when risks outpace rewards. Where the field goes next hinges on learning well from chemicals like 2,4,5-TP, balancing innovation with a hard-won understanding of consequences, and putting health—human and environmental—at the core of every decision.
What is 2-(2,4,5-Trichlorophenoxy)Propionic Acid used for?
Breaking Down Its Purpose
2-(2,4,5-Trichlorophenoxy)Propionic Acid rarely pops up in everyday conversation, but in farming circles, it’s known by its short name: 2,4,5-TP. This chemical found its reputation mainly as a selective herbicide. Many farmers leaned on it to tame tough, unwanted brush and woody plants, giving crops a fighting chance against nature’s constant takeover. Having grown up in a farming community, I saw firsthand how chemicals like this ended backbreaking hours of weed pulling and allowed fields to stay productive.
Why It Was Popular
Broadleaf weeds put a real dent in crop yields. 2,4,5-TP made it easier for large-scale operations to manage their land, especially in pasture, rangeland, and along utility rights-of-way. People started using it heavily in the 1950s and 60s across the United States for just this reason. I remember old-timers at the co-op store talking about how regrowth of stubborn brush slowed to a crawl after a few applications. Across mid-century America, progress often meant using whatever tools fixed a problem quickly.
Complex History and Lingering Questions
Any conversation about 2,4,5-TP brings up more than weed control. Evidence started piling up about byproducts called dioxins. These aren’t just minor concerns—these pollutants link to health problems like cancer, reproductive issues, and disruptions in hormones. Things turned serious after reports tied the chemical’s production and use to dioxin contamination, not just in workers but in communities living close to treated areas. I’ve talked to families still worried about what might linger in the soil or the water table after heavy spraying decades ago.
The U.S. Environmental Protection Agency stepped in during the 1970s and cut back permitted use. By 1985, all registered uses of 2,4,5-TP in the United States disappeared. Sometimes regulations sound out-of-touch, but people on the ground saw changes for the better in health and less environmental risk. Across the globe, most countries followed suit, phasing out or placing heavy restrictions on similar herbicides.
Lessons From Its Legacy
The story of 2,4,5-TP opened a bigger discussion about chemical safety and transparent research. Blind faith in “miracle solutions” gave way to stricter rules about testing products before they hit the market. Not every patch of overgrown land needs a chemical shortcut. Today, integrated pest management, crop rotation, and even reintroducing grazing animals play a huge part in keeping land healthy. These methods never promise instant results, but they often protect both profits and health in the long run.
Some argue new herbicides face over-regulation, slowing innovation for farms facing tough weeds. I’ve seen the struggle from both sides. Yet seeing what happened with 2,4,5-TP, families push for long-term health over a quick fix.
Looking Forward
Every time another synthetic chemical enters the market, people remember what happened with 2,4,5-TP. Researchers spend more time checking what ends up in the air, water, and eventually the food chain. Public demands for safer alternatives have already pushed companies to develop products with fewer side effects. Many farmers now ask the tough questions themselves instead of waiting for disaster to strike. Earning trust takes work—clear science, honest conversations, and lessons from the past.
Is 2-(2,4,5-Trichlorophenoxy)Propionic Acid safe for humans and the environment?
Understanding the Risks
Many folks have crossed paths with herbicides in gardens, farms, or public parks—though most never stop to check what’s in the spray bottle. 2-(2,4,5-Trichlorophenoxy)propionic acid, usually called 2,4,5-TP or Silvex, has roots in weed control going all the way back to the 1940s. Its mission: kill unwanted plants fast. Its track record, though, raises plenty of questions, especially after the discoveries made about its risks.
How Exposure Happens
In practice, exposure often comes through skin contact, breathing dust, or eating produce that picked up a dose during spraying. In many places, the compound itself isn't used much anymore, thanks to tight restrictions rolled out in the 1970s and 80s. Regulators didn't just wake up one day and decide to ban it—reports of birth defects among farmers and towns downstream from spraying sites got the ball rolling. The worries intensified after studies uncovered that Silvex can often carry dioxins as chemical byproducts, known for a long shelf life in soil and a long list of health dangers.
Science Backs Up the Caution
The science shows alarm bells have some merit. Dioxins, hiding inside some batches of Silvex, were fingered as culprits for immune disruption and cancer, along with a range of hormone problems in humans and animals. The U.S. Environmental Protection Agency puts dioxins among the most toxic substances. Several peer-reviewed studies have noticed increased cancers and reproductive issues linked to areas where this herbicide was heavily used.
Take the story of Times Beach, Missouri—a town so badly contaminated after Silvex-laced waste was sprayed on dusty roads, its residents had to leave their homes behind for good. That episode wasn’t the only one, but it captured what can go wrong when regulators, chemical makers, and consumers don’t have the full picture.
Environmental Impact Goes Beyond Humans
Experience in agriculture tells me weeds are rarely the only things affected. Silvex sticks around in soil, moving slowly through fields, creeks, and rivers. According to studies by the World Health Organization, residue in soil can sit tight for months or even years, depending on sunlight, rainfall, and soil type. Wildlife pays the price, too. Toxins work their way up the food chain, hurting fish and birds even in places miles away from the spray site.
What Might Better Answers Look Like?
Folks often want to jump straight into the question: What’s the alternative? Organic and mechanical weed control methods fit into modern farming. Rotating crops, mulching, or even something as simple as pulling weeds by hand on smaller plots can make a big difference. Where chemicals still seem necessary, strict testing and labeling, along with third-party audits, help spot the warning signs before health gets traded for convenience again.
Why Experience Matters Here
My own years working with home gardeners and small farmers have shown that good decisions grow out of knowledge and caution, not just efficiency. Trust gets built up when consumers know what chemical histories stand behind the food on their plate or the playground under their feet. In my view, the story of Silvex isn't just about chemistry—it’s about asking the tough questions and looking for safer farming and pest control every step of the way.
How should 2-(2,4,5-Trichlorophenoxy)Propionic Acid be stored and handled?
Understanding the Risks
Long, complicated names like 2-(2,4,5-Trichlorophenoxy)Propionic Acid sound locked up in the world of chemists. The reality is, this is the backbone for certain herbicides, and it comes with more risk than people usually imagine. In my years working in an agricultural supply warehouse, I’ve seen first-hand what happens when someone ignores the warnings on these chemical drums. Rashes, headaches, and even stories of people winding up at the doctor showed up just from sloppy habits.
Why Storage Location Changes Everything
Too many old barns still keep chemicals on wooden shelves, next to seeds and even snacks. This chemical doesn’t just smell strange — it can contaminate the air if leaks start. Locking this stuff away keeps kids and pets out, but also cuts down on the chances of careless mixing. The smartest setup uses dedicated storage rooms with solid, metal shelving and a spill tray underneath every shelf. That kind of attention pays off the first time a container starts leaking, since it keeps drips out of the soil and stops fumes from building up where people breathe.
Container Choices and Label Smarts
Dealers almost always deliver these acids in thick, tight-sealing barrels or jugs. In my time helping farmers, I always stressed using the same containers for refills, and not switching them up. The risk isn’t just a lost label — it’s that other containers can react with the chemical, releasing gases no one wants inside their workroom. Even permanent markers fade, so strong, printed labels matter more than a quick scribble. If the label fades, confusion grows — and that leads to waste or accidents.
Temperature and Ventilation Count
Heat pushes chemicals like this one to break down and possibly build up pressure. That’s a recipe for disaster — I’ve seen older cans swell or leak when summers turn brutal. Unventilated spaces trap fumes inside, which gives workers headaches or burns. Storage rooms set around 20°C, with simple fans pulling fumes outdoors, make a big difference. If you ever step inside and catch a sharp whiff, the room’s missing good ventilation.
Handling: Staying Clean and Prepared
Every season, new staff wonder if gloves and goggles really matter. Short answer: yes, they do. I once saw a coworker wipe his eye after touching a jerry can and end up losing the rest of his shift to the nurse’s office. Nitrile gloves hold up better than those thin, cheap types, and eye protection stops drips from causing permanent damage. Good habits, like changing clothes after a shift and never eating in the chemical room, save trouble later on.
What to Do When the Worst Happens
Chemical spills feel like panic at first, but a couple of simple tools make cleanup calm. Absorbent sand or clay, stored in easy reach, soaks up most minor spills fast. Keeping clean-up kits in the storage area beats running around with a mop. I always made sure staff got basic spill training; fast response stops the chemical from reaching drains or mixing with other products.
Running a Safe Operation
Living around chemicals takes more than hanging warning signs. Good storage cuts risk, and simple handling rules keep people safe. My years in the industry proved that strong, clear habits save money, preserve health, and keep the peace with regulators. Every safe season starts with wearing the right gear, storing chemical drums right, and keeping eyes peeled for leaks. Common sense, not fear, drives results every year.
What are the potential side effects or hazards of 2-(2,4,5-Trichlorophenoxy)Propionic Acid?
Hazards at a Glance
Plenty of farm chemicals claim to make life easier for farmers, and 2-(2,4,5-Trichlorophenoxy)propionic acid—sometimes called 2,4,5-TP—once showed up in weed control efforts on pastures and crops. For all the talk about better yields, more manageable fields, and quicker weed kills, something keeps coming up: what lingered after the spray drifted or the residues soaked into the soil? Scientists flagged safety questions decades ago, and for good reason.
Soil, Water, and Residue Concerns
Compounds like this don’t often vanish quickly in the environment. Experiences on family farms show that the earth takes a beating from “persistent” chemicals—the story gets worse if those compounds seep into groundwater or end up riding dust into someone’s lungs. U.S. Environmental Protection Agency (EPA) research showed remnants sticking around in soil, risking runoff into streams. Some folks who rely on well water downwind of treated land face a real possibility of trace exposure.
Health Side Effects
Doctors and toxicologists worry about more than just chemical names—they look at how people and animals react to exposure. Evidence on 2,4,5-TP points to short-term health impacts: skin irritation, redness, eye inflammation, headaches, and nausea often top the list. Longer exposure or larger accidental intake raise the risk of liver, kidney, and nervous system effects. Animal studies, used because people can’t be research volunteers in dangerous trials, found birth defects and organ problems from chronic doses. The U.S. banned the substance in the late ’80s, spurred on by findings of dioxin contamination in the manufacturing process.
Dioxin: The Hidden Danger
Dioxin doesn’t show up in the ingredient label, but it can form as an unwanted byproduct. This chemical class triggers longtime fears among scientists, mainly because dioxins build up in fatty tissues and linger in the food chain—fish, beef, even dairy products reflect the risk when runoff makes its way through the ecosystem. Chronic low-level exposure links to immune suppression, reproductive issues, cancer, and developmental problems in infants and children. Guidance from the World Health Organization classifies some dioxins as carcinogenic. Real cases from the Vietnam War era, when related herbicides entered the environment, reinforced the health urgency.
Dealing With What’s Left
Many countries yanked this chemical off the shelf after cumulative research. Still, old stockpiles and contaminated soils stick around in barns and abandoned lots. Farmers and landowners must steer clear of using any leftover product, check labels in older chemical sheds, and report suspicious containers to the local environmental agency. State and federal clean-up programs work to keep buried risks sealed or removed, but the cost and scale put pressure on communities.
Lifestyle and Policy Approaches
Switching to safer herbicides remains the practical path for most. Some growers move toward integrated weed management: crop rotation, physical removals, and targeted spraying cut down the reliance on harsh chemistry. Policymakers should keep partnering with independent scientists and public health advocates, making sure banned chemicals don’t return by backdoor sales or illegal import. Public health alerts and environmental monitoring matter for everyone—not just farmers—since residues settle in shared water, foods, and dust.
Supporting the Shift
Programs offering training and compensation make it easier for family farms to change practices. Universities and cooperative extension offices need money for research, so they can share what works best and warn when old problems resurface. Together, these steps help communities look out for health, protect water, and move away from risky legacy chemicals.
What is the recommended dosage or application rate for 2-(2,4,5-Trichlorophenoxy)Propionic Acid?
Understanding Why Dosage Matters
It’s easy to look at a chemical name like 2-(2,4,5-Trichlorophenoxy)propionic acid and get lost in the jargon. This compound, sometimes called silvex, has a reputation stretching back decades in weed management. Anyone who’s farmed, maintained turf, or managed invasive brush faces the classic problem — the tug-of-war between plant health and weed control. Hitting the sweet spot with this chemical takes more than reading a label. Value comes from pairing know-how with accountability, because applying too little wastes effort and cash, but using too much risks moving toxins into the water or hurting nearby crops.
What Experts Recommend — and Why Reliable Guidance Beats Guesswork
Looking back at published recommendations, guidance from the U.S. Department of Agriculture and university extension services points toward 0.5 to 1.5 pounds of active ingredient per acre. That baseline comes from years of study, not a “one-size-fits-all” rule. Formulation strength, target species, and application method all change the math. Diluting too heavily or gunning for a faster “kill rate” can both backfire. In practice, anyone out in the field benefits from testing a small patch first, especially when weather, soil, and target weed age throw wrenches into the works.
Personal Experience and Practical Hurdles
I’ve watched more than a few landowners attempt to eyeball solutions in a rush to save time. Some read the label, break out battered kitchen scales, and figure out ounces per gallon. Others skip right to two or three times the suggested rate, expecting better results. More product on the ground rarely translates to better weed knockdown. Instead, the vegetation wilts, surrounding pasture yellows, and stormwater threatens to ferry residues away. Smart applicators trust calibrated sprayers, double-check nozzle patterns, and keep mixing tanks clean — because an ounce of prevention weighs less than regulatory headaches or a sick milk cow.
Safety Always Joins the Discussion
The science behind the numbers improved after silvex caught scrutiny for possible dioxin contamination and groundwater risk. Regulators placed strict limits, and advisory groups like the EPA pushed for careful record-keeping. Rinsing equipment, watching for wind drift, and staying well below the maximum allowed rate became more than rules on a page. Neighbors take notice. Ranchers and vegetable growers want clean water and safe feed. Those attitudes leave a stronger legacy than any harvest count for the year.
Better Choices and Smarter Management
Anyone reaching for a jug of herbicide owes it to their land and community to pause before pulling the trigger. Reading real-world case studies and talking with experienced extension agents always delivers more value than copying last year’s plan. A few hours of training can help avoid soil damage or wasted money. Soil testing, weather tracking, and following up on treated fields help everyone learn from surprises. Rotating control strategies, exploring non-chemical options, and keeping a detailed application log turn paperwork into peace of mind.The best solution means hitting the label-recommended rate, staying alert for changes, and never letting routine shortcut responsibility. With chemicals like 2-(2,4,5-Trichlorophenoxy)propionic acid, respect and steady hands carry forward longer than fast results.
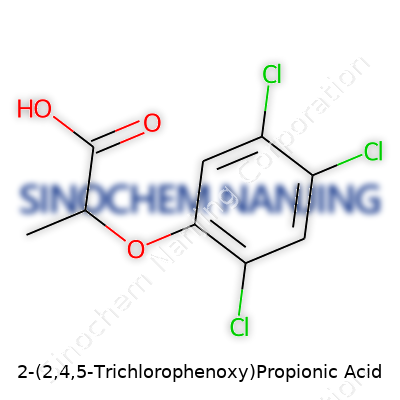
| Names | |
| Preferred IUPAC name | 2-(2,4,5-trichlorophenoxy)propanoic acid |
| Other names |
Silvex 2,4,5-TP Fenoprop |
| Pronunciation | /tuː tuː fɔːr faɪv traɪˌklɔːrəˈfiːnɒksi proʊˈpiːoʊnɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 93-72-1 |
| Beilstein Reference | 1366016 |
| ChEBI | CHEBI:2807 |
| ChEMBL | CHEMBL1226 |
| ChemSpider | 13717 |
| DrugBank | DB03531 |
| ECHA InfoCard | 13af02d7-7726-4d07-aaf8-7de91e9d5c15 |
| EC Number | 3.1.1.15 |
| Gmelin Reference | 81559 |
| KEGG | C13721 |
| MeSH | D015850 |
| PubChem CID | 9451 |
| RTECS number | UF9625000 |
| UNII | 25384U89QV |
| UN number | 3077 |
| Properties | |
| Chemical formula | C9H7Cl3O3 |
| Molar mass | 275.54 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.64 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.78 |
| Vapor pressure | 6.6 x 10^-7 mmHg (25°C) |
| Acidity (pKa) | 3.46 |
| Basicity (pKb) | pKb ≈ 11.6 |
| Magnetic susceptibility (χ) | -2.66e-6 |
| Refractive index (nD) | 1.597 |
| Dipole moment | 3.25 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 385.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -502.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3684 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | C01EC01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P273, P280, P301+P312, P305+P351+P338, P501 |
| NFPA 704 (fire diamond) | 2-2-0- |
| Flash point | 158°C |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | LD50 oral rat 700 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 1160 mg/kg |
| NIOSH | TY1575000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Fenoprop 2,4,5-Trichlorophenoxyacetic acid (2,4,5-T) 2,4-Dichlorophenoxyacetic acid (2,4-D) MCPA |

