2,2,3-Trimethylbutane: Editorial Commentary on Its Place in Science and Industry
Historical Development
Folks who study the history of hydrocarbons often stumble on some overlooked oddballs, and 2,2,3-Trimethylbutane is one of them. It goes by the snappier nickname "Triptane." This molecule started turning heads back in the era of World War II, when fuel quality meant the difference between ground crews working overtime and bombers staying in the air. Chemists dug deep to boost the octane of aviation gasoline, and Triptane emerged from bubbling test tubes and high-pressure reactors as a radical solution. Suddenly, adding it to fuels helped engines run smoother, with less of the dreaded knock that chews up pistons. The story wasn’t just about finding new molecules, but about how basic organic synthesis found a place on the front lines, enabling planes to fly higher and further. Triptane’s tale reflects a pattern in industrial chemistry—necessity leads to new molecules, which reshape technologies and become silent workhorses in complex systems.
Product Overview
You rarely see 2,2,3-Trimethylbutane on store shelves because it's a specialty hydrocarbon with a niche audience. Industrial chemists, fuel engineers, and folks designing high-performance engines know Triptane as a powerful octane booster. Out in the world, it barely appears outside certain fuels, research labs, and a few chemical synthesis pathways. Given its structural makeup—a heavily branched paraffin—Triptane sets benchmarks for anti-knock properties and helps researchers model how fuel blends behave. Cars on the street don’t get direct doses, but in the era of precision fuel engineering, its story keeps repeating: small tweaks in molecular structure yield outsized impact.
Physical & Chemical Properties
Triptane stands apart with a density lighter than water and a boiling point around 80 to 81 degrees Celsius. While it comes across as another clear, colorless liquid to the naked eye, chemists appreciate its impressive volatility and relatively low flashpoint. This branching in its structure makes it less prone to knocking in internal combustion engines, which put it on a pedestal for octane rating standards. Its chemical behavior shows the same stability you see in other alkanes—resistant to acid and base attack, no willingness to react with strong features like double bonds, and happy to stick to the paraffin club of saturated hydrocarbons.
Technical Specifications & Labeling
The story of Triptane in the lab is one of careful handling. You can spot it by its distinct hydrocarbon odor, which still reminds some technicians of classic laboratories. Labels warn about flammability, but the rules follow the same templates in place for countless organic solvents and fuels—no open flames, proper grounding, good ventilation. Detailed labeling also reflects its hydrocarbon status, highlighting both C7H16 as a formula and 2,2,3-Trimethylbutane as a name. Modern inventory systems track each bottle because of both safety and tight regulatory controls around volatile organics.
Preparation Method
Making 2,2,3-Trimethylbutane isn’t a kitchen recipe, but the old-school approach involves alkylation. Chemists take isobutane and shoot it through with isobutylene, often coaxed by strong acid catalysts under pressure. Over the years, tweaks to catalyst formulations and purification steps let engineers raise yields and cut waste. This method found new relevance as demand for selective alkylation rose in refining, as refineries pushed to squeeze every drop of high-octane fuel from their cracking units. Today’s producers still rely on these solid foundations, even as researchers hunt for greener, less energy-intensive processes.
Chemical Reactions & Modifications
While Triptane doesn’t react the way alcohols and esters do, chemists still manage a few tricks. Classic combustion remains at the top of the list—feed it oxygen and it burns hot, with clean blue flames producing carbon dioxide and water. Under stronger chemistry, it resists oxidation but can break down under the punishing conditions inside a reformer or during catalytic cracking. Triptane’s stable framework means it’s more often a target in studies about thermal decomposition and reformulation, rather than a widely-used starting point for building more complex molecules.
Synonyms & Product Names
Ask for Triptane in industrial circles and someone brings out a bottle labeled 2,2,3-Trimethylbutane or even its systematic IUPAC alternative. Chemical wholesalers in the research sector also toss around identifiers like its CAS number 564-02-3 or terms like "high octane isoheptane." Outside the lab, few know it by any name, reflecting its quiet, behind-the-scenes role alongside better-known hydrocarbons like iso-octane.
Safety & Operational Standards
Working with Triptane calls for a steady hand, good ventilation, and sharp awareness about flammable liquids. Its volatility matches other light alkanes, meaning vapors can catch fire if given half a chance. Proper grounding and bonded containers prevent accidental sparks, and most protocols emphasize using chemical fume hoods or explosion-proof refrigeration. Inhalation remains a top concern—breathing in these vapors can leave people dizzy or worse, and PPE like gloves and goggles is just standard practice. Regulatory agencies mostly treat it within the same hazard bands as other flammable hydrocarbons, but special attention goes to its role in confined fuel systems where vapor pressure control matters most.
Application Area
Everyday life in the modern world runs on refined fuels, and Triptane has a special place in designing them. Its standout octane rating gave aviation and experimental engines a chance to push the limits of power and efficiency during key historical periods. Though no longer mass produced like in the peak aviation era, specialized fuel blends for competition engines or scientific calibration still draw on it. Fuel researchers use Triptane as a benchmark, comparing new molecules and predicting future blends’ abilities to resist knock. It pops up in the testing suites of refinery labs and advanced powertrain development programs, a reliable reference point for cutting-edge combustion studies.
Research & Development
Research around 2,2,3-Trimethylbutane keeps circling back to its role as a gold-standard octane reference. Its behavior in combustion chambers allows for detailed studies in pre-ignition, emissions control, and advanced fuel formulations. Teams working on cleaner-burning engines turn to Triptane to set boundaries and prepare the ground for alternative fuels. New catalysts and greener synthesis methods are slowly changing the way chemists approach its manufacture, especially as demand for sustainable processes drives the shift in industrial chemistry. Its use in small-scale syntheses as a model compound also supports publication after publication in fuel chemistry, with each study nudging the field toward higher-efficiency, lower-emission outcomes.
Toxicity Research
Public health and environmental teams don’t often single out Triptane in studies—the hydrocarbon shares many hazard patterns with its alkane cousins. Animal testing and vapor exposure research build up a picture of moderate acute toxicity at high doses, mostly worrisome for people inhaling concentrated fumes. Chronic exposure brings the usual worries that come with volatile hydrocarbons: headaches, dizziness, and longer-term impact on the central nervous system. Regulatory guidance focuses on minimizing workplace exposure and airtight storage. Soil and groundwater impact gets less attention, since fuel-grade hydrocarbons tend to evaporate before they seep too far—though the environmental story always leaves room for improvement as monitoring methods evolve.
Future Prospects
With electric cars racing around highways and battery factories outpacing gasoline refineries, some predict the old hydrocarbon chemistry might fade into history. That said, the story isn’t so black and white. Triptane’s lessons about branching, molecular structure, and fuel optimization still fuel new ideas in synthetic chemistry. As researchers hunt for the next generation of clean fuels and lubricants, the experience gained from molecules like Triptane builds a foundation. Alternative energy trends will keep shaping how hydrocarbons feature in both transportation and chemical manufacturing, but for now, small-volume specialty chemicals like this one still make contributions—both as a historical game-changer and as a yardstick for technical progress.
What is the chemical formula of 2,2,3-Trimethylbutane?
No Smoke and Mirrors—It’s All About the Atoms
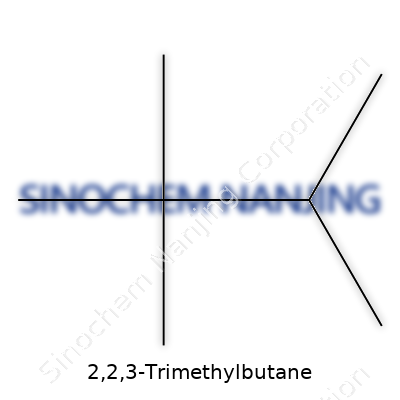
2,2,3-Trimethylbutane might sound like a mouthful, but its structure reflects a pattern that people in chemistry labs know by heart. This molecule falls into the family of alkanes, a basic group of hydrocarbons forming the backbone of fuels and everyday organic chemistry. The chemical formula, C7H16, comes from understanding the backbone and counting each carbon and hydrogen atom, one by one.
Breaking Down the Name and Structure
I remember the first day learning about systematic names like this. The numbers and dashes might seem cryptic until you sit down with a pencil and sketch the carbon skeleton. “Butane” means a four-carbon chain at the core. The prefix “trimethyl” flags three extra methyl groups—little clusters each containing one carbon and three hydrogens. Their numbers, 2,2,3, aren’t just labels, they map out precisely where those methyl groups anchor onto the butane base.
Draw out the chain. Start with carbon number one, then two. On the second carbon, you stack two methyl groups. On the third carbon, you place the last methyl group. That leaves seven carbons total (the original four, plus three from methyl groups) and fills in the hydrogens to reach sixteen. The molecular formula comes to life: C7H16.
Why These Details Matter Outside of Textbooks
It makes sense to ask why these formulas draw so much attention. Knowledge of chemical formulas helps guide safe storage, effective handling, and even laws around shipping. You probably won’t run into pure 2,2,3-Trimethylbutane pumping gas into your car. Yet, its skeleton connects to the way gasoline is refined and the reasons behind octane ratings. A fuel’s knock resistance links to the branching of molecules just like this. Branching often makes a molecule less likely to pre-ignite, keeping engines running smoother and more efficiently. For example, isooctane—a close chemical cousin—acts as the gold standard for the octane rating scale.
There’s another piece to this. Knowing the formula and structure allows researchers to predict properties like boiling points and volatility. A straight chain and a branched molecule with the same formula might behave differently in a reaction or even cook off faster under the sun. This kind of curiosity—looking beyond the label to the atoms—leads to better process safety and smarter regulation. The Environmental Protection Agency, for instance, needs this groundwork to assess health impacts of volatile compounds in air and water.
Supporting Safer Science with Reliable Information
I’ve come across misinformation, even in lab settings, that could stem from a mix-up of similar-sounding molecules. Getting the formula wrong, even by a single atom, opens the door to mistakes. Every training session or research project benefits from a culture of checking assumptions. Trustworthy reference databases like PubChem or the CRC Handbook make life easier by laying out exact formulas, structures, and properties.
Clarity in details drives confidence for industry workers, teachers, and students. Nobody wants confusion when handling chemicals with real-world risks. Double-checking formulas is as vital as wearing goggles or labeling bottles.
Room for Improvement
Helping more people access and understand chemical formulas goes beyond the chemistry department. It could look like simpler visuals in textbooks, more hands-on activities, or better training for anyone working around chemicals. Reliable fact-checking and open access to authoritative databases keep everyone on the same page, reducing errors and raising standards for safety and performance.
What are the physical properties of 2,2,3-Trimethylbutane?
Why 2,2,3-Trimethylbutane Catches Interest
Organic chemistry shows off its variety through molecules like 2,2,3-Trimethylbutane, known to some as triptane. Beyond the technical formula, this compound stands out for a few clear reasons. It belongs to the group of alkanes, meaning it only contains single bonds between its carbon atoms—no complicated rings or double bonds. Its name already tells a story: three methyl groups stuck onto a butane skeleton, creating a shape more crowded and compact than straight-chain alkanes.
What Sets Its Physical Characteristics Apart
2,2,3-Trimethylbutane behaves differently from longer or less-branched cousins. Its structural compactness causes the melting and boiling points to dip lower than many other C7 hydrocarbons. The boiling point hovers around 80 degrees Celsius, which lines up with the trend seen among branched alkanes. The tight packing knocks down the molecule’s ability to stick to its neighbors through intermolecular forces. Put this liquid in your hand, and you’ll notice it evaporates quickly, giving off a faint, gasoline-like scent. I remember working in an undergrad organic lab and being surprised by just how quickly small hydrocarbons slip away if lids aren’t tight.
On a hot day, trimethylbutane turns into vapor without much encouragement. Its melting point sits around -30 degrees Celsius, so under normal circumstances, nobody will see it as a solid. That fact sets it apart compared to longer or less-branched molecules, which might solidify on a chilly morning. Density for 2,2,3-Trimethylbutane sits a touch below water, near 0.69 grams per cubic centimeter at room temperature. That means any spill floats, a detail that matters for storage and cleanup—no need to fish it out from the bottom of a container.
Real-World Impact of Its Properties
Because of the tidy structure and low boiling point, trimethylbutane finds itself in fuel science conversations. Its high octane number drew a lot of attention during early aviation fuel research. Engineers need fuels that resist engine knocking; trimethylbutane fits much better than straight-chain alkanes when smooth running matters. People rarely encounter the pure substance outside a laboratory or the fuel industry, but knowing how it behaves gives insight into bigger industrial processes.
Potential Hazards and Handling
Small, vaporous hydrocarbon molecules carry certain risks. Quick evaporation leads to easy inhalation and, in a closed room, can drive up flammability dangers. From personal experience, working with volatile compounds brings out the value of good ventilation and closed storage. A splash on skin washes away with soap, but vapors linger—proper gloves and fume hoods save a lot of headaches. Its flash point sits quite low, so any spark nearby can result in fire. People in labs or refineries focus on handling procedures to dodge accidental ignition.
Room for Better Practices
Safe management starts with education. Students and workers need hands-on training on vapor containment and fire prevention. Equipment that tracks spills or vented vapors makes a real difference. Industries can lean toward better engineered containment and automatic detection to reduce mistakes. Sharing first-hand experiences about surprise spills or unexpected evaporations helps incoming generations understand the challenges. Following up on regular risk reviews ensures old routines don’t lead to overlooked hazards.
What are the common uses of 2,2,3-Trimethylbutane?
Everyday Chemistry in Action
Walk into any garage or peek under the hood during an oil change, and you’ll run into a blend of science and practicality. 2,2,3-Trimethylbutane, a mouthful of a name for sure, plays a behind-the-scenes role in the fuels and chemicals that keep industries and vehicles running. Most people don’t realize it, but this hydrocarbon shapes products that folks rely on, especially in the context of high-performance fuels.
Racing Fuels and Performance
Some years ago, I spent afternoons at local drag strips working with auto enthusiasts fine-tuning engines. Premium gasoline blends always come up, and 2,2,3-Trimethylbutane, a highly branched alkane, keeps popping up in the conversation. It’s all about those octane ratings. Fuels with higher octane are less likely to cause knocking, which damages engines. This chemical raises the octane number, so drivers and race teams can push engines harder without bouncing pistons or fried spark plugs. Refineries blend it into gasoline to meet strict performance requirements, especially for aviation or motorsports.
Fuel Research and Testing
Refineries and university labs don’t just guess at fuel properties—they test, and they test a lot. Pure hydrocarbons are like the standard yardsticks for measuring combustion, volatility, and environmental impacts. 2,2,3-Trimethylbutane sees use in research and certification labs as a reference fuel component. In classes and labs, I have seen students use samples like this to learn how to balance efficiency and emissions. Since it burns cleanly and resists premature ignition, scientists can collect data on how modern engines handle stress. This helps carmakers design better engines and fuels, drawing from real chemical evidence.
Petrochemical Synthesis
Beyond the racetrack and the fuel pump, 2,2,3-Trimethylbutane steps into the world of chemical synthesis. It’s valued because its structure resists breaking down under heat and pressure, giving chemical engineers a reliable backbone for building more complex substances. Manufacturers use it as an intermediate—essentially a stepping stone—in making adhesives, sealants, and specialty lubricants. These applications turn up in everything from airplane hydraulics to machines that shape metal. From my time shadowing at a plastics company, I saw how specialty hydrocarbons feed entire chains of production.
Environmental Considerations and Safer Handling
Efficiency brings responsibility. From experience in working in industrial environments, storing and handling flammable chemicals comes with serious risks. 2,2,3-Trimethylbutane evaporates easily, and even small spills create fire hazards. Modern storage tanks and transport systems use vapor recovery and containment to cut emissions and reduce accidents. Regulatory bodies keep a close watch on how much gets released into the air. Engineers are always on the lookout for replacement compounds and safer process controls, not only for workplace safety but also for long-term environmental protection. While alternatives are still in the works, investing in stronger ventilation and leak detection truly pays off, keeping workers and neighborhoods safer.
Supporting Real-World Solutions
Getting the balance right between performance, cost, and responsibility means giving attention to chemicals like 2,2,3-Trimethylbutane. Companies involved in fuels, manufacturing, and research know that careful selection, storage, and blending of hydrocarbons helps deliver better products. Setting up transparent sourcing rules and investing in green technologies makes a difference for the future. Chemical knowledge, backed by real data and thoughtful engineering, lies at the center of practical solutions that benefit both industry and society.
Is 2,2,3-Trimethylbutane hazardous or toxic?
Digging Into 2,2,3-Trimethylbutane
Once in a while, chemistry brings up names that look more like a secret code than anything familiar. 2,2,3-Trimethylbutane is no exception. Its catchy nickname, triptane, surfaces in stories about gasoline additives and advanced aviation fuel. Walk into any well-equipped chemistry lab and you’ll find it labeled as a hydrocarbon—a pretty standard fuel ingredient used to help engines resist knocking. Last season, a friend working in an industrial lab passed on this question: Just how risky is this oddly-named compound? That kind of curiosity sticks with you.
What the Science Says About Risks
Hydrocarbons cross paths daily in cars, fuels, paint thinners, even garden tools. Most folks rarely give a thought to the fumes they’re risking in a warm garage, but years of handling all sorts of chemicals has taught me to look past the label and dig into the data. Triptane isn’t considered a highly toxic hydrocarbon. Organizations like the European Chemicals Agency and the US National Library of Medicine report that it acts a lot like its petroleum cousins: light, volatile, and flammable. That last part deserves respect—flash points around -18°C mean it catches fire more easily than gasoline and can turn a careless spark into a dangerous fire before anyone has a chance to grab a fire extinguisher.
Breathe in too much vapor and you’ll likely begin to feel dizzy, maybe get a headache, or even feel some nausea. It wouldn’t surprise most seasoned techs, since hydrocarbons often push back against careless handling with mild symptoms that grow with repeated exposure. Triptane fumes shouldn’t linger unventilated, just as you don’t want to sniff around open cans of gasoline all afternoon. Direct skin contact rarely leads to more than mild irritation, but soaking your hands in any fuel-grade solvent always runs the risk of drying, redness, or rashes. Poisoning from accidental swallowing—rare but possible in home workshops with poor storage—creates far bigger headaches for medical staff: nausea, vomiting, even pneumonia from aspiration.
Where Problems Start
High octane sounds impressive on paper, but it leads chemical companies to handle triptane in bulk. Accidental spills or explosions do more than create momentary scares; fires involving volatile hydrocarbons release thick, toxic smoke and can spread at shocking speed. Chemical plants and refineries build extensive safety systems to lower these risks, but not everyone gets a factory tour or sees those fire suppression plans up close.
Another angle that rarely makes headlines—environmental hazards. As a hydrocarbon, triptane pooling on the ground means trouble for wildlife and water. Birds and fish have trouble bouncing back once fuels seep into their environment. Chemistry textbooks tend to skip these grim realities, but anyone working cleanup after fuel spills—myself included—remembers badly oiled feathers and slicked rivers. Even with triptane’s relatively low acute toxicity, the compound affects environments in ways that simple safety labels can miss.
Practical Steps for Safety
Most responsible workplaces use triptane in controlled settings with solid training and strict rules. Safety goggles, chemical-resistant gloves, and strong ventilation move from “nice to have” to “common sense” once you work with fuel additives. No one should pour triptane or similar hydrocarbons down the drain, and proper storage in flame-proof containers makes a difference every time. Governments and industry groups share practical advice, often for free—local fire departments and environmental agencies can point out best practices they want everyone to follow.
Sometimes it’s easy to overlook hidden risks in chemistry’s long list of compounds. 2,2,3-Trimethylbutane isn’t the world’s most toxic or mysterious chemical, but it calls for respect. Treating it with practical caution, good ventilation, and real training limits most danger, both in the lab and outside. Nobody wants to add a hydrocarbon fire or environmental mishap to their work experience, trust me on that.
How should 2,2,3-Trimethylbutane be stored and handled?
Real World Handling: No Room for Guesswork
I once joined a facility tour where basic chemical safety rules sometimes slipped through the cracks. That place stored 2,2,3-Trimethylbutane in a way that caused a real stir—a few open drums too close to a heat source. They didn't just skirt the rulebook. They put their workers in harm’s way.
2,2,3-Trimethylbutane has a low flash point. It catches fire much easier than folks expect. A single spark, a hot pipe, or even direct sunlight can push fumes into ignition. Real people, real workplaces need to treat this stuff like gasoline—even though it feels harmless in the moment.
Concrete Safety Steps
A drum of this hydrocarbon belongs far from open flames and hot equipment. Even heat from the afternoon sun presses the risk. Store tanks and drums in cool, shaded rooms with steady airflow. Good ventilation keeps vapor from settling and lowers the odds of a bad day. Use grounded containers—static charge builds up fast, and it won’t wait for someone to double-check the work later on.
Many accidents come from cutting corners. Friend of mine worked with a team that thought plastic bottles could serve as storage for convenience. Leaks showed up in days. Only containers rated for flammable liquids earn a spot in the storage room. Keep them closed, check the seals, and stack nothing above the allowed height. A toppled drum can turn a simple spill into a full-scale disaster.
Health Hazards Hit Quick
The smell of 2,2,3-Trimethylbutane won’t always give you much warning. At higher levels, breathing its vapors can knock your senses off balance—headaches, dizziness, and worse if you stay in the cloud. Goggles hold off the splash, and gloves keep skin free from irritation and rashes that can drag on for weeks.
Always use a fume hood or work outdoors where the air moves freely. Spraying, pouring, or mixing inside small rooms builds up vapor as fast as someone turns their back. Proper respirators stop folks from breathing in more than their bodies can handle—plain dust masks don’t cut it.
Tough Talk on Emergency Response
Years ago, I met a plant manager who insisted fire drills wasted time. A flash fire from a spilled drum in his warehouse changed that attitude fast. Every team should run through what-ifs at least twice a year. Know the fire extinguishers that crush chemical fires, teach folks where the exit routes go, and label every drum in bold print. If a spill hits the floor, sand and absorbent pads keep it from spreading, never water. Workers should trust their training and call the fire team for anything bigger than a puddle.
Why Vigilance Matters
I’ve seen supervisors rely too much on luck instead of planning. Nobody feels blessed getting a face full of fumes or dealing with a warehouse fire after work hours. 2,2,3-Trimethylbutane needs respect—regular walkarounds, honest talks with staff, and budgets for strong containers. Safety drains resources up front, but it keeps businesses alive and neighbors out of trouble. Smart handling protects families and futures, no shortcuts taken.
| Names | |
| Preferred IUPAC name | 2,2,3-Trimethylbutane |
| Other names |
Triptane 2,2,3-Trimethylbutane 2,2,3-Trimethylbutan |
| Pronunciation | /tuː tuː θriː traɪˈmɛθɪlˌbjuːteɪn/ |
| Identifiers | |
| CAS Number | 594-76-3 |
| Beilstein Reference | 1718737 |
| ChEBI | CHEBI:12087 |
| ChEMBL | CHEMBL15419 |
| ChemSpider | 13949 |
| DrugBank | DB01935 |
| ECHA InfoCard | 100.007.905 |
| EC Number | 204-493-5 |
| Gmelin Reference | 103286 |
| KEGG | C06588 |
| MeSH | D014260 |
| PubChem CID | 6566 |
| RTECS number | EJ5950000 |
| UNII | 6MV2V90TJW |
| UN number | UN1262 |
| CompTox Dashboard (EPA) | DTXSID6040155 |
| Properties | |
| Chemical formula | C7H16 |
| Molar mass | 114.23 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 0.690 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 2.82 |
| Vapor pressure | 3.5 kPa (at 20 °C) |
| Acidity (pKa) | 56.0 |
| Basicity (pKb) | pKb ≈ 17 |
| Magnetic susceptibility (χ) | -9.47e-6 |
| Refractive index (nD) | 1.382 |
| Viscosity | 0.573 mPa·s (at 20 °C) |
| Dipole moment | 0.0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -198.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5467.9 kJ/mol |
| Hazards | |
| Pictograms | GHS02 |
| Signal word | No signal word |
| Precautionary statements | P210; P233; P240; P241; P242; P243; P261; P271; P301+P310; P303+P361+P353; P304+P340; P305+P351+P338; P311; P370+P378; P403+P233; P403+P235; P405; P501 |
| NFPA 704 (fire diamond) | 1-4-0 |
| Flash point | −18 °C (closed cup) |
| Autoignition temperature | 415 °C |
| Explosive limits | 1.1% - 6.0% |
| Lethal dose or concentration | LD50 (oral, rat): 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 12,200 mg/kg (rat, oral) |
| NIOSH | RN3675000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 120 mg/m3 |
| IDLH (Immediate danger) | IDLH: 900 ppm |
| Related compounds | |
| Related compounds |
Isobutane Neopentane 2,2-Dimethylbutane 2,3-Dimethylbutane Pentane |

