2,2,2-Trinitroethyl Methacrylate: More Than Just a Name in Chemical Innovation
Tracing the Path: Historical Insights
Before lab benches gleamed with clean glassware and today’s sophisticated analytical tech, chemists relied on patience and gut-feeling alongside formula sheets. The story of 2,2,2-Trinitroethyl Methacrylate stretches back to an era obsessed with harnessing nitro derivatives for new explosives and energetic polymers. During the latter half of the twentieth century, chemists grew fixated on bridging organic monomers with functional nitro groups, hoping for stronger, more responsive materials. Trinitroethyl methacrylate is born from that intense drive, an offshoot of projects once steered by military funding now spilling out into civilian laboratories. The march of history shaped its story not through chance, but through persistent inquiry—someone always asking, “What else can this do?”
Getting Down to Brass Tacks: Product Overview
At a glance, 2,2,2-Trinitroethyl Methacrylate might look like just another chemical handle, but its performance shows purpose. By grafting methacrylate’s double bond onto the hefty trinitroethyl group, the molecule brings twin gifts: the reactivity of acrylate chemistry and the energetic punch of high-density nitro content. Veterans in polymer synthesis have eyed this compound for three main reasons: it brings new flavor to custom polymers, offers dense energy for specialty applications, and often challenges the comfort zone of safe handling. Its potential does not end in the catalog; this is a tool for chemists who want to go beyond “good enough.”
Tough Stuff: Physical and Chemical Traits
You get to know a chemical the way you get to know a tool. Every specific property—density, boiling point, solubility—says something about how it behaves when the gloves are on. Trinitroethyl methacrylate packs a moderate density and exists as a pale-yellow oily liquid at room temperature. It tends to avoid water, prefers organic solvents, and needs a gentle hand due to its sensitivity to shock and heat. Where regular methacrylate esters seem friendly, this version reminds you with every whiff of its nitro roots. That pungent tang, the faintly ominous shimmer, the knowledge that a spark could spell disaster—physical properties create both potential and responsibility.
Technical Fine Points and Labeling Reality
No shelf label can capture the full story. Regulations around toxic and energetic organic compounds dictate strict labeling language—flammable, explosive hazard, toxic if inhaled. Labels do little to underline personal risk, though. The flavor of these rules comes from hard lessons learned in the past, from incidents where compounds crossed from curiosity to catastrophe. Chemists who handle this stuff lean on updated Material Safety Data Sheets, which spell out storage, transportation, and spill management, sometimes more for the sake of insurance than daily practice. Even among experienced workers, the sight of the nitro designation on a label brings an extra moment of respect.
Clever Chemistry: How it Comes Together
Making trinitroethyl methacrylate demands a certain kind of patience. This preparation typically kicks off by nitrate-bombing ethanol derivatives under careful acid control, yielding trinitroethanol—an important starting material for this and other energetic compounds. In a second stage, skilled hands combine trinitroethanol with methacryloyl chloride, coaxing out the prized ester. The yields never quite feel generous, and the settings always require measured temperatures, dry glass, and a clear head. Anyone hoping for a casual, high-throughput route will find this synthesis slow-going, and probably just as well. This is chemistry at its most careful—where discipline stops disaster.
Chasing Transformations: Chemical Reactions and Tweaks
Chemists with restless hands rarely leave a new molecule untouched. Trinitroethyl methacrylate plays nicely with radical initiators, fitting snugly into traditional polymerization techniques. By nudging initiators and adjusting ratios, researchers adjust the flexibility, toughness, or energetic performance of copolymers. The methacrylate moiety welcomes grafting and copolymerization, a favorite trick for tinkerers pushing the limits of resin technology. For those obsessed with tweaking thermomechanical properties—or dialing back energetic risks—derivatization opens even more doors. Every new modification becomes a test: how much explosiveness can we trade for safety, or vice versa?
Synonyms and True Names
By any other name, the compound would still smell as sharp. 2,2,2-Trinitroethyl methacrylate sometimes travels under the more formal “trinitroethanol methacrylate” label in textbooks. Despite variations, the industry settles on a few core synonyms, making searching through research literature a little less like running a gauntlet. Remembering these synonyms comes in handy when chasing down hidden data or talking shop at conferences.
Realities of Handling: Safety and Conduct
The chemical safety bible never glosses over the word “trinitro.” Precautions hang thick in the air: grounded equipment, temperature monitoring, small-scale quantities, and walls covered in warning placards. Training for handling nitro-functional methacrylates stresses personal protective gear and strict ventilation, not just for peace of mind but for hard-earned self-preservation. One mistake—one overlooked static charge or brewing peroxide contaminant—cycles back every story you’ve seen in the accident books. In my own time around energetic compounds, nothing ever felt routine; every session had that extra tension that only comes from respect for risk.
Fields That Call for Trinitroethyl Methacrylate
This isn’t a chemical for everyone or every purpose. Most of its story so far belongs to specialized fields: energetic binders in explosives, research into high-density propellants, and the rare polymer formulations that flirt with risk for special rewards. Military labs and a handful of industrial chemists test the boundaries, pushing for better sensitivity tuning, improved binding strength, or new routes to smart energetic composites. A few daring academics dabble with it in the hunt for functional materials—think sensors, timers, or tiny, self-destructive elements for tamper-evident devices. Across the spectrum, almost every new application weights the scale: how much energy is too much for practical use?
Science on the Front Edge: Research and Development
Labs chasing innovation keep trinitroethyl methacrylate on a short list whenever new energetic polymers come up. Over the past decade, studies have focused on copolymerization with other acrylates, grappling with the balance between mechanical ruggedness and controlled energy release. The push for safer handling leads to research on polymer matrices that cage or stabilize this group, cutting back on the risk of accidental release. Teams publish papers touting advances in storage stability, toxicity reduction, and lower sensitivity to mechanical insults. For some in the industry, the research arms race revolves around finding just the right blend—not the most explosive, but the one that answers both safety and utility.
Walking the Line: Toxicity and Health
Nobody who spends time with nitro compounds can ignore their health downsides. Significant exposure history often reads like a warning pamphlet: headaches, dizziness, nausea, and—at high enough doses—much worse. Lab animals and cell assays tell a story of clear risk, flagging the compound’s impact on liver and blood chemistry. Over the years, occupational safety groups piled up evidence on the need for minimal exposure, especially through inhalation or skin contact, labeling the chemical in many places as a notable workplace hazard. The move toward encapsulation and closed-system handling owes its momentum to these findings.
Looking Ahead: The Future for Trinitroethyl Methacrylate
The future for 2,2,2-Trinitroethyl Methacrylate pivots on a single tension—chasing energy without courting disaster. As technologies hunger for more responsive smart materials and tailored energetic systems, this compound gets a closer look from both innovators and regulators. Every year brings tweaks in synthesis aimed at less hazardous byproducts, smarter polymer hosts, and routes that turn dangerous raw stuff into safe intermediates. The next breakthroughs likely won’t rely on sheer brute power but on careful, intelligent engineering: lower-impact derivatives, precision dispersal systems, and maybe even roles far removed from where it got its start. The hope is clear—tap the best this chemical offers without repeating the mistakes that gave energetic chemicals a bad name in the first place.
What is 2,2,2-Trinitroethyl Methacrylate used for?
What is 2,2,2-Trinitroethyl Methacrylate?
2,2,2-Trinitroethyl methacrylate doesn’t roll off the tongue easily, but in the world of chemistry, it grabs plenty of attention. This compound, rich in energetic nitro groups attached to a methacrylate backbone, finds a niche within both military and industrial science. I remember the first time I saw it in a research report—the mechanistic detail almost felt intimidating. As much as the chemical name sounds like something from a spy thriller, its real-world uses rely on both its explosive energy and its ability to bond tightly within polymers.
Use in Explosives and Propellants
The main reputation of 2,2,2-Trinitroethyl methacrylate springs from its role as an energetic binder for explosives and propellant compositions. Most commercial and research-grade explosives need a material that can both hold the mix together and boost energy output. The nitro groups deliver just that—contributing oxygen and energy. Military labs often turn to specialty binders like this for improving performance in rocket propellants or warheads, especially in cases where traditional plasticizers or binders lag behind in power or reliability.
Civilians rarely interact with this substance directly. It’s a tool reserved for trained chemists designing next-generation explosives. Published studies and patent filings point to its use in plastic-bonded explosives, where safety and handling matter almost as much as detonation power. The balance between stability and energy release keeps scientists up at night, as both underperformance and instability can spell trouble.
Material Science and Polymers
Beyond explosives, polymer chemists have experimented with 2,2,2-Trinitroethyl methacrylate to create specialty plastics with energetic properties. The methacrylate piece enables the molecule to hook onto other monomers in polymerization reactions. This yields plastics that carry built-in energy—bringing utility in controlled demolition charges or as initiators for reactive materials.
Industry has always chased after compounds that merge performance with reliability. In this case, the chemical’s ability to integrate into a plastic matrix makes it more useful than pure explosives powder. Once bound in a polymer, there’s less risk of accidental detonation during manufacturing or transport, compared to loose, powdery explosives. That added safety margin brings real-world value—not just in labs, but for workers on the ground.
Risks and Safety: No Room for Complacency
Speaking honestly, a nitro-based compound like 2,2,2-Trinitroethyl methacrylate always wears a warning label. Misuse or mishandling can deliver disaster, making careful storage and rigorous training mandatory. Regulators track sales and distribution closely, with good reason. Just hearing about accidents with energetic polymers at research institutions makes me realize how crucial solid protocols are. You don’t see this material at your local hardware store, and you shouldn’t.
Alternatives and Solutions
Chemists constantly look for substitutes that deliver high power with less risk. Green energetic materials—those with reduced toxicity and cleaner decomposition—have started to gain ground. Some labs test new binders based on glycidyl or azide chemistry, but trinitroethyl methacrylate persists where high energy and stability remain top priorities. Manufacturers now invest more in remote manufacturing processes, digital monitoring, and lower-scale pilot production runs—tools that lower risk without dropping performance.
The bigger lesson revolves around respect. Every energetic material deserves handling with care, clear rules, and honest talk about what might go wrong. Training goes a long way. Open discussion—between researchers, safety officers, regulators, and even the public—keeps everyone safer and the science moving forward, even when the chemical names keep getting longer.
What are the safety precautions when handling 2,2,2-Trinitroethyl Methacrylate?
Understanding the Real Risks
Working with 2,2,2-Trinitroethyl Methacrylate carries much more weight than a standard afternoon in a chemistry lab. This compound lives up to its name. As someone with experience handling energetic chemicals, I know that one loose moment can change a routine day into a hospital visit. This stuff finds its way into energetic polymers, explosives research, and specialty adhesives. It packs energy, and treating it like any ordinary chemical is a shortcut to regret.
Respecting Its Instability
Explosive sensitivity isn’t just textbook theory. Static discharge, friction, impact — all can trigger a violent reaction. Even storing this compound means vigilance. In labs I’ve worked in, we always kept explosive materials in small amounts, away from possible ignition sources. Glass containers, proper grounding straps, spark-proof tools: these become non-negotiable rules, not options. Most accidents I’ve seen happen when people rush or overlook the basics.
Personal Protective Equipment Is Not Optional
No one walks into the lab wearing only street clothes. Thick laboratory coats, antistatic gloves, and face shields guard against splashes and accidental ignition. Ventilated goggles give extra protection because even a tiny splash can do real harm. I always double-check my gear before handling a nitro compound. Colleagues forget their gloves, thinking they’ll just “do it really quick.” Shortcuts cause scars.
Keeping a Controlled Environment
Temperature control matters. Rooms get equipped with blast shields, pressure relief venting, and sensors. Labs with experience in energetic materials also use remote handling tools — long tongs, manipulators, or full glove boxes. I remember a mentor explaining that the lab bench should look empty except for what’s immediately needed. Less stuff nearby means less risk of accidental contact or static buildup. Good ventilation pulls fumes away, protecting your lungs from nasty vapors.
Emergency Planning Means Getting Out Alive
Chemical burns, shrapnel injuries, or toxic inhalation top the list of worries. Every lab I’ve seen with good safety records posts clear evacuation routes, keeps appropriate extinguishers on hand, and reviews emergency procedures monthly. It’s easy to get lulled into routine, but emergencies come when least expected. I never open a container without knowing the nearest exit and safety shower.
Waste Disposal: You Can’t Just Toss It
Unspent Trinitroethyl Methacrylate never goes in the regular trash. Labs lock up any leftovers in approved explosive-waste containers before certified disposal. Ignoring this step turns workplaces into ticking time bombs. Back in school, we had a scare with improper storage — only luck saved us that day. Regulatory guidelines come from real disasters, not red tape.
Final Thoughts on Responsibility
Handling energetic materials isn’t just about protecting yourself. Mistakes can endanger everyone nearby. Good training, honest respect for the risks, and a little humility have kept me safe so far. Caution isn’t overkill — it’s common sense. If you’re ever in doubt, stop and ask. Rushing or guessing is where things go wrong.
What is the chemical structure of 2,2,2-Trinitroethyl Methacrylate?
Understanding the Building Blocks
Walk into any laboratory where researchers set out to create new-age polymers and explosives, you’ll probably spot chemists keeping a careful eye on compounds like 2,2,2-Trinitroethyl methacrylate. This molecule grabs attention thanks to its energetic qualities and the meticulous balance it holds between stability and volatility. The structure behind these properties helps drive unique reactions and opens up possibilities in synthesis work that doesn’t come easy with standard monomers.
Breaking Down the Chemical Structure
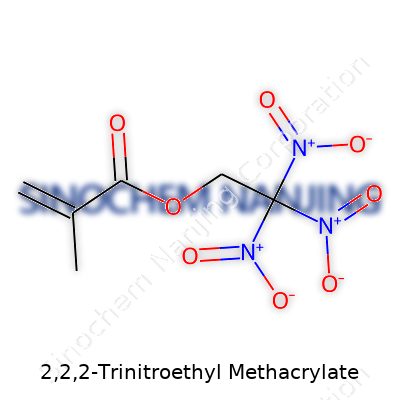
At the heart of its makeup, 2,2,2-Trinitroethyl methacrylate combines two essential segments. On one end sits the methacrylate group — a backbone in polymer chemistry, made up of a vinyl group (C=C) bonded to a methoxycarbonyl (COOCH3)— linked through an ester bond. The other half features the trinitroethyl group. That’s a two-carbon chain, capped off with three nitro (-NO2) groups attached to the same carbon atom, essentially forming —C(NO2)3.
If you’re picturing the structure, it looks like this:
- Methacrylate chunk: CH2=C(CH3)−COO−
- Trinitroethyl tail: CH2−C(NO2)3
Why This Matters in Applied Science
It’s easy to glance at formulas and not think much about what’s behind them. Spend any time around a team dealing with energetic materials, you start to see how nitro groups shape outcomes. Take the triple nitro decorations suspended from the ethyl group. These functional groups pump up sensitivity and boost the release of energy when the compound decomposes. There’s a fine line between something that stores energy and something that becomes hazardous, and chemistry here decides which side a material lands on.
Polymer chemists sometimes look for new monomers that bring both structure and energy to bear, especially for applications in propellants or special adhesives. Standard acrylics offer good weather-resistance or bond strength, but drop a trinitroethyl group in the mix, and you get a material that responds rapidly, making it attractive for specific tasks in defense or pyrotechnics. Still, the intense reactivity brings a set of risks. It calls for strong expertise, since improper storage or handling can end up catastrophic. I’ve seen people double-checking protocols, verifying the storage temperature, and insisting on shielding and grounding before opening even a gram.
Safety Always Comes First
Nitro-rich compounds have taught the scientific field more than a few hard lessons. Students and researchers learn by heart that nitro esters demand respect—dynamite and PETN use similar chemistry for good reason. In professional labs, following protocols matters as much as knowledge of reaction pathways. Routine safety checks and the right training make the difference. There’s no room for shortcuts.
What Can the Field Do Better?
Every discussion about energetic chemicals brings up safety, but the field moves forward when experts share best practices. Routine risk assessments, constant updates to handling protocols, and investment in safer analogues can lower the danger. Encouraging collaborative research lets people compare notes, improve synthesis procedures, and sometimes find molecular tweaks that tame the volatility without losing performance. At the end of the day, everyone—from the bench scientist to the end-user—benefits from candor, shared experience, and a willingness to treat powerful molecules with healthy respect.
How should 2,2,2-Trinitroethyl Methacrylate be stored?
A Chemical with an Edge
2,2,2-Trinitroethyl Methacrylate doesn’t exactly blend into the background among chemicals you find in a standard lab. Anyone who’s handled materials with nitro groups knows the kind of respect and caution they demand. I once watched a colleague, gloves trembling ever so slightly, tiptoe through setting up a storage cabinet after learning a new delivery included bottles of this very compound.
Heat: The Silent Saboteur
Warmth seems harmless, but this molecule thinks otherwise. 2,2,2-Trinitroethyl Methacrylate carries a reputation for thermal instability. Temperatures climbing even a few degrees past room temperature start raising the chance of nasty surprises. We keep it cool—literally. Ventilated storage areas with air conditioning represent more than comfort; they can help save lives. Refrigerators rated for hazardous chemicals put a reliable line of defense between the material and ambient heat. As an extra measure, I’ve always made sure our lab had a thermometer stuck to the inside of the chemical fridge door. Someone glancing at numbers twice a day beats discovering a problem too late.
Container Choice Isn’t a Footnote
The temptation to toss every liquid into the closest free bottle lands people in trouble. Glass stands out here. Polymers and some plastics don’t react well under stress from nitro compounds, sometimes breaking down and creating an even bigger mess. Sealed amber glass bottles block out light, a trigger for decomposition. While plastic bottles tempt with their durability against breaking, their permeability and reactivity cost more in risk. We label, date, and track these bottles so they don’t become mysteries buried in the back corner.
Avoiding Friends and Foes
Combining reactive chemicals never brought anyone peace of mind. In storage, proximity makes a difference. I learned that the hard way once, when a carelessly shelved oxidizer leaked and left scorch marks on a stainless steel shelf. With 2,2,2-Trinitroethyl Methacrylate, we always use a dedicated cabinet, far removed from acid, base, and especially anything flammable or reducing. Segregating energetic materials reduces the fallout from accidents.
Poor Ventilation Converts Worry into Crisis
Ventilation separates a safe lab from a hazardous one. I keep storage areas for explosive chemicals away from any places with stagnant air. Vents or even dedicated chemical hoods cut down on vapor buildup. People often believe sturdy doors and thick walls solve every storage problem, but it’s stale air and vapors that create risks you can’t see or smell until it’s too late. Sensors and alarms aimed at vapor detection can add another critical layer of oversight.
Attention to Records and Inspection
Letting stacks of paperwork ride gets tempting. Careful records on every bottle and inspection logs outpace any fancy gadget in keeping storage safe. I’ve opened too many safety audit sheets with half-filled lines and questionable dates. Inspections that check for discoloration, swelling, or pressure build-up take minutes and prevent irreversible accidents. Anything suspect comes out of circulation, period.
Training: The Foundation of Safety
All the best equipment in the world still leaves gaps if people shortcut protocols. No one should handle or store 2,2,2-Trinitroethyl Methacrylate without up-to-date, hands-on training. Safety drills, not just lectures, burn these best practices into everyday habits. Giving everyone a chance to walk through emergency plans means fewer people freeze if the worst happens.
Building Safer Labs, One Step at a Time
Storage of chemicals like 2,2,2-Trinitroethyl Methacrylate stays rooted in respect for the risks these compounds bring. Cool, segregated spaces, the right bottle, and rigorous attention to detail in record-keeping and inspections—for many labs, these aren’t extras. They shape the culture. Practical safety measures make a difference each day, keeping the unpredictable in check.
What are the potential hazards of 2,2,2-Trinitroethyl Methacrylate?
Understanding Where the Hazards Come From
2,2,2-Trinitroethyl methacrylate has a long, technical name that barely rolls off the tongue, but the risks packed into this molecule deserve straight talk. I’ve worked around energetic chemicals, and what stands out about this compound is its explosive nature, not just in a chemistry sense but in real daily workplace risks too.
The Explosion Factor
A product built from nitro groups and a methacrylate backbone looks like trouble. Nitro compounds often come with a volatility problem, and this one does too. Small shocks, friction, or static can trigger explosions. In the real world, even equipment that looks safe might spark just enough energy to set things off. Factories get extra jumpy about storage conditions, employee safety protocols, and handling methods.
People sometimes overlook how easy it is to get complacent. A rushed morning, a loose wire, or a missed spill on a work surface—these little lapses have led to real disasters. I’ve seen hazmat teams move with slow, steady motions just to avoid accidental ignition. That’s because the consequences reach beyond the lab; explosions can level buildings, destroy livelihoods, and leave workers with permanent injuries.
Chemical Exposure and Health Concerns
The toxicity brings its own set of worries. Inhalation, skin contact, or even eye exposure poses health risks. Nitro compounds often act as systemic toxins, impacting blood and organs. Strong evidence links high exposures to headaches, dizziness, respiratory distress, or even methemoglobinemia. Fast responses and proper protective gear save lives, but accidents rarely hand out second chances.
I have watched new workers assume gloves and goggles will shield them from every danger. The reality stays messier. Splashing, leaks, or unnoticed spills around seals and fittings lead to exposure without warning. Emergency rinses and first-aid kits become essential, not just compliance paperwork.
Contamination and Environmental Spill Risks
Control rarely stops at personal health. Disposal and accidental release raise larger alarms. Trinitro compounds break down slowly and can poison water sources. Contaminated soil or spilled material entering drains puts whole communities at risk. Even if the chances seem low, the long half-life and environmental stubbornness of these chemicals mean cleanup work can drag on for months or years.
Disposal regulations exist for a reason, yet enforcement can slip, especially in small firms under pressure. The aftermath of a chemical spill includes lawsuits, devastated habitats, and sick wildlife. Keeping contamination out of the local ecosystem matches the challenge of keeping it out of the human body.
Solutions That Make a Difference
Real prevention starts with engineering and training. Storing trinitroethyl methacrylate far from heat, light, or friction keeps everyone safer. Strict access controls and specialized containers matter. I’ve seen companies benefit from regular, honest safety drills. Staff who truly practice emergencies react better than those who treat drills as just another box to tick.
Choosing less hazardous alternatives changes the story, too. Modern chemistry often finds substitutes, but some processes still depend on the explosive punch of nitro methacrylates. Companies moving away from these stubborn chemicals do so with good reason.
At every level—worker, manager, community—people need honest facts, not just technical labels. Hazards from trinitroethyl methacrylate demand respect, tight procedures, and a willingness to push for safer choices whenever possible.
| Names | |
| Preferred IUPAC name | 2-methyl-2-[(2,2,2-trinitroethoxy)carbonyl]prop-2-enoic acid |
| Other names |
Methacrylic acid, trinitroethyl ester 2,2,2-Trinitroethyl methacrylate Trinitroethanol methacrylate |
| Pronunciation | /ˌtraɪˌnaɪtroʊˈɛθɪl mɛθˈækriˌleɪt/ |
| Identifiers | |
| CAS Number | 85492-82-6 |
| Beilstein Reference | 1208737 |
| ChEBI | CHEBI:132080 |
| ChEMBL | CHEMBL4198722 |
| ChemSpider | 23085694 |
| DrugBank | DB03271 |
| ECHA InfoCard | 03c18ccd-31bb-485b-a757-b3d5d094c4f1 |
| EC Number | 208-809-5 |
| Gmelin Reference | 146522 |
| KEGG | C19797 |
| MeSH | D015679 |
| PubChem CID | 10868298 |
| RTECS number | UF9100000 |
| UNII | FM823LWK2C |
| UN number | UN1344 |
| CompTox Dashboard (EPA) | DJ35QN5YHI |
| Properties | |
| Chemical formula | C6H7N3O8 |
| Molar mass | 363.15 g/mol |
| Appearance | Pale yellow liquid |
| Odor | Odorless |
| Density | 1.620 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 0.9 |
| Vapor pressure | 0.00015 mmHg (25 °C) |
| Acidity (pKa) | 8.13 |
| Basicity (pKb) | 13.43 |
| Magnetic susceptibility (χ) | -62.8 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.502 |
| Viscosity | 150 mPa·s (20°C) |
| Dipole moment | 4.64 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 367.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -30.8 kcal/mol |
| Std enthalpy of combustion (ΔcH⦵298) | −1385 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS09 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H315, H317, H319, H335, H341, H370, H373 |
| Precautionary statements | P210, P261, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P370+P378, P501 |
| NFPA 704 (fire diamond) | 4-3-2-W |
| Flash point | 134 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1320 mg/kg |
| LD50 (median dose) | LD₅₀ (oral, rat): 220 mg/kg |
| NIOSH | SN 4475000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Trinitroethyl methacrylate (TREM) Trinitroethyl acrylate Trinitroethyl ether Trinitroethanol Nitrocellulose Methacrylic acid Methyl methacrylate Glycidyl methacrylate |

