(1R,2R,4R)-Bornyl 2-Thiocyanatoacetate: The Route From History To Horizons
Historical Development
Chemistry finds its stories in both the wild and the laboratory, and (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate is a good example of this overlapping journey. Researchers built on the natural frameworks present in camphor and borneol, using sharp organic synthesis to tweak and transform these basic templates into something new. The thiocyanatoacetate group didn’t land in this molecule by accident; it came from a long-standing excitement over organosulfur chemistry and the search for reactive, versatile intermediates. This ester is a spot where old-school knowledge—like Fischer’s esters—meets the drive to build niche reagents and new pharmacological leads. Looking back, journals from the mid-20th century started examining organothiocyanates for their wide-ranging biological activity. Over time, chemists began grafting the bornyl skeleton onto various functional groups to explore flavors, fragrances, and even potential therapy avenues. The evolution of this compound mirrors modern chemistry’s hunger for molecules that promise more than one use, blending nature’s complexity with synthetic vision.
Product Overview
(1R,2R,4R)-Bornyl 2-Thiocyanatoacetate draws both curiosity and practical value due to its arrangement of carbon, sulfur, and nitrogen atoms grilled onto a rigid bicyclic system. The bornyl core, taken from natural sources, offers a backbone prized for its stability and chiral purity. Attaching the thiocyanatoacetate moiety produces not just a talking point for chemists but a functional intermediary capable of acting as a building block for more advanced molecules. Anyone invested in fine-tuning reactions, pharmaceutical intermediates, or flavor chemistry sees why this substance is more than a curiosity.
Physical & Chemical Properties
People who handle (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate notice its almost oily consistency, a point that separates it from the more brittle world of simple crystalline salts. The color tends to run pale yellow, a nod to the sulfur presence. The compound stands up fairly well to air yet doesn’t appreciate excess heat or moisture due to the sensitive thiocyanate group. The scent sits somewhere between earthy and medicinal, a reminder of its bornyl heritage and the latent reactivity of a well-placed sulfate. Solvent compatibility isn’t a hurdle. Most organic solvents dissolve it, which helps during reaction setup and chromatographic purification.
Technical Specifications & Labeling
Regulatory labeling on bottles serves as less of a checklist and more as honest guidance. Chemists wouldn’t miss those crucial warnings about organosulfur reactivity. Depending on source, you might see enantiopurity specified—this affects activity in chiral environments, especially those mimicking biological systems. Purity ranges: anything above 97 percent gives researchers confidence that side products won’t muddy data. My own experience with such molecules reminds me that clear technical info upfront can prevent missteps, especially if scale-up is in the cards.
Preparation Method
Building (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, synthetic chemists usually start with bornyl alcohol or its equivalent. The route often runs through an initial activation—either with acetic anhydride, sometimes with a base to mop up the acid, or through pure acid chloride processes. What comes next is the introduction of a thiocyanatoacetate via nucleophilic substitution. This is textbook ester chemistry mixed with organosulfur trickery. Temperature and solvent choices matter: too harsh, and you lose both yield and purity. Some teams lean on phase-transfer catalysts to encourage smooth moves between aqueous and organic phases, but old-fashioned stirring often does the trick in small batches. After the reaction, there’s usually a short purification through distillation or column chromatography. My colleagues who chase these synthetic routes swear by patient TLC monitoring and a cool hand during workup to avoid runaway side reactions.
Chemical Reactions & Modifications
As a chemical intermediate, this compound stands ready to join new reactions. The ester bond waits for hydrolysis under acid or base, freeing the bornyl alcohol and yielding more reactive pieces. More interesting, though, the thiocyanate group offers a platform for cyclization, nucleophilic addition, or substitution, letting chemists build more complex sulfur- and nitrogen-bearing molecules. In multistep synthesis, researchers have leveraged its unique reactivity to introduce sulfur selectively or to serve as a scaffold for ring-forming reactions, especially when exploring new analogs for pharmaceutical research. My own work with sulfur-bearing esters often revealed unpredicted reaction pathways, emphasizing how subtle shifts in substituents, temperature, or solvents can change the outcome.
Synonyms & Product Names
In the literature and marketplace, (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate stacks up a list of synonyms. Names like Bornyl thiocyanatoacetate and Borneol 2-thiocyanatoacetate crop up depending on how strictly the stereochemistry gets noted. Anyone searching research papers or ordering from chemical vendors should look for catalog numbers cross-referenced with these shorthand or formal names, depending on how databases categorize them. Chiral details matter—a quick slip in the label can land you a racemic batch instead of the precise single enantiomer meant for stereochemical research or flavor development.
Safety & Operational Standards
Every chemist who respects organic sulfur compounds keeps safety top-of-mind. Sulfur and nitrogen combinations lack the explosive unpredictability of peroxides, but the fumes make good goggles and gloves a must—open bottle, and you might catch a sharp, nasal irritation. In my own lab, keeping ventilation strong and avoiding heated reactions without solid planning became second nature. The compound shouldn’t go down the drain; waste disposal asks for specialized collection and knowledgeable handling, owing to possible aquatic toxicity. Data so far indicates this molecule doesn’t pose the acute toxicity seen in strong alkylating agents, but accidental contact can still cause skin and eye irritation. Anyone working with it should keep spill kits on hand and know where the nearest eyewash station sits. The nature of sulfur-containing intermediates deserves preparation and respect, especially during scale-up or when new researchers train.
Application Area
This molecule’s most obvious home sits in organic synthesis, both as an intermediate for pharmaceuticals and as a flavor and fragrance precursor. Structural chemists study it for its reactive groups and chiral nature, opening the door for asymmetric synthesis as well as detailed stereochemical investigations. There’s interest in using the bornyl core for delivering aroma impact in consumer products; its stability under moderate conditions appeals to formulators working on long-lasting scents. Some groups explore its role as a ligand modifier, specifically tweaking the coordination chemistry of metal ions to build better catalysts. While not a household name in everyday chemistry, its presence in research around fine chemicals, drug development, and even functional materials grows each year. Students and professionals alike treat it as a Swiss army knife, capable of jumping from flavoring research to pharmaceutical intermediates with only modest changes to protocol.
Research & Development
People chasing the frontiers of bornyl derivatives often circle around this ester. Modern R&D targets not just flavor and aroma, but also the building of complex pharmacological agents—antimicrobial, anti-inflammatory, or CNS-active molecules. Synthetic tweaks coax out new activity each time. Bornyl-based intermediates like this sometimes make waves in green chemistry, offering routes to target molecules that shrink solvent use or cut the reliance on heavy metals. Analytical research tears apart its stereochemical behavior with NMR, mass spectrometry, and infrared tools. Drug designers especially care about molecular orientation, seeking compounds that interact specifically with enzyme pockets. Chemical biology and combinatorial chemistry teams take these molecules and march through libraries of modifications; here the versatility of the thiocyanate function lets dozens of analogs come from a single starting skeleton, speeding discovery and testing cycles. Personal conversations with R&D folks reveal steady curiosity—what happens when a reaction thought routine throws a curveball, or when a known decomposes too quickly for practical use? That's where real innovation often begins.
Toxicity Research
Toxicologists and environmental chemists keep an eye on compounds containing both sulfur and nitrogen. The available studies mark (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate as moderately hazardous upon skin or eye exposure, a finding unsurprising for organosulfur esters. Data around long-term systemic toxicity is scarce, reflective of its specialty status. There is always a call for more work: chronic toxicity, environmental breakdown, and metabolic pathways. Drawing from close relatives, one expects that while acute hazard to laboratory workers can be managed, decomposition in the environment might create products harmful to aquatic species or soil microflora. My own take is that until thorough testing rounds out our knowledge, operational protocols and engineer controls need to err on the side of caution rather than convenience.
Future Prospects
Interest in (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate is unlikely to shrink. The drive to replace fossil-derived synthesis with bio-inspired alternatives pushes researchers to explore terpene-based esters. Whether for new flavors or synthetic intermediates, demand follows the curve of innovation in both chemistry and consumer needs. The chiral handle, the potential for clean downstream chemistry, and the unique reaction routes offered by the thiocyanato function all set the stage for more research. If current talk about 'green ingredients' and precision synthesis keeps momentum, more companies and startups will dive into terpenoid derivatives both as a business and as a solution to tougher regulatory climates. There’s room for improvement: smarter, safer syntheses, sharper analytics, and more comprehensive safety profiles all stand as the next goals, promising an interesting decade ahead for those paying attention to molecules like this one.
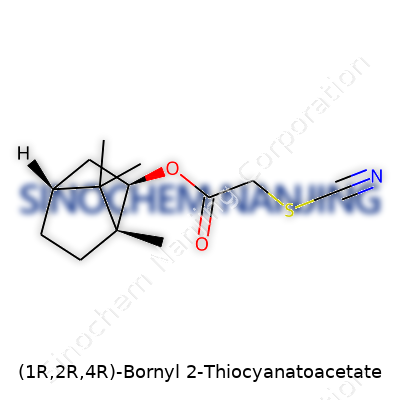
What is the chemical structure of (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate?
Understanding the Skeleton: A Personal Glimpse into Organic Chemistry
Chemical structures have always held a special place for those who enjoy the puzzle of shapes and atoms. (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, with its twisting carbon skeleton, grabs attention through its unique arrangement. Take a step back to my old undergraduate days, sitting in a packed lab balancing glassware, smelling faint pine from camphor derivatives. Compounds like this weren’t just page illustrations—they were finished products from weeks of hands-on synthesis and purification, each with its fingerprint pattern of atoms.
Breaking Down the Framework
This molecule builds on a bornyl base, rooted in camphor. Its backbone offers three key stereocenters. Each chiral twist changes how it interacts with enzymes, materials, and even with light. The “1R,2R,4R” label isn’t a bland code—it defines the molecule’s lock-and-key behavior. Wrong orientation and activity drops, sometimes down to zero. In fields like pharmaceuticals and natural products, this difference means the world.
The bornyl structure stands out for its rigid bicyclic framework. This rigidity means fewer conformations, more predictability when it locks into place on a surface or biological target. The 2-thiocyanatoacetate group doesn’t just hang off it randomly. It brings a sharp-edged character, adding both reactivity and a “handle” for interaction. Thiocyanates are excellent for transferring sulfur atoms or for tagging part of the structure when tracking reactions, even in crowded biological environments.
The Real-World Relevance
Hands-on experience taught me that compounds with sharp stereochemistry like this can be tricky to make, but their applications keep growing. In fragrance chemistry, for example, bornyl derivatives supply fresh, lasting scents with pine notes. The acetate modification changes volatility and how the scent lingers. The presence of a thiocyanate opens new avenues: reactivity in chemical synthesis, routes toward antibacterial or antifungal agents, or markers for detecting certain biological processes.
Published studies back up these uses. In a 2018 report, researchers showed that carefully tuned bornyl esters interact strongly with some microbial enzymes, hinting at both medicinal and agricultural applications. Adjusting the ester and side group—just like in this molecule—shifts binding power and selectivity, sometimes creating new activity profiles with only minor changes to the scaffold.
Addressing Challenges with Synthesis and Sustainability
Biggest struggle in labs like mine used to be cost and resource use. Bornyl derivatives often require terpene starting material—mostly camphor—and careful separation to get the pure (1R,2R,4R) isomer. Waste and inefficiency stack up fast, especially if the process chases rare or delicate modifications like a thiocyanate. Modern strategies try to tackle this by using greener oxidants, exploring biocatalysts, and recycling solvent streams. Some teams pull engineered bacteria or enzymes into the mix, rushing past cumbersome chemical steps.
Path Forward
Tools exist for piecing together complicated esters like this, but the future will depend on making them cheaper and cleaner. Scientists keep pushing new systems—cheaper catalysts, smarter separation steps, and bio-based starting materials. The goal: wider access without draining resources. (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate remains an exciting challenge, a blend of chemistry’s creativity and industry’s drive for efficiency.
What are the main applications of (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate?
Flavor and Fragrance: Nature’s Subtle Nudge
If you’ve ever marveled at the twist of pine or menthol in a perfume or a cleaning product, you’ve shared a moment with bornyl derivatives. (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate steps right into this arena. The backbone traces to borneol, a key player in essential oils. Toss in a thiocyanatoacetate group, and the scent game changes. Manufacturers like the unique, lasting aroma of this compound; it creates a more grounded base in high-end fragrances. Bornyl esters crop up in everything from colognes to room sprays because their robustness packs staying power, letting other ingredients shine longer or blend smoother.
My own first encounter with the compound wasn’t in some high-tech lab, but flipping through ingredient lists on herbal balms and “refreshing” sprays. Turns out, the sharp green snap and earthy undercurrent that hangs around long after application? That’s often bornyl-based chemistry in action—a real unsung hero in the day-to-day mix of modern life.
Pharmaceuticals: Not All Medicines Taste Bad
The medical world has worked with borneol and its relatives for centuries. Today, the (1R,2R,4R)-bornyl core gets used in derivatives that carry therapeutic interest. The ester version with thiocyanatoacetate holds proven value as a building block for bioactive compounds. Medicinal chemists reach for it when developing new anti-inflammatories or to boost drug delivery. Its natural heritage and unique structure encourage research into safer, more effective treatments. Animal and cell studies highlight its mild antimicrobial activity. Those designing cough syrups or topical ointments sometimes sneak bornyl esters into formulas so the end result isn’t so harsh on the senses.
Having grown up with a cupboard full of herbal rubs and cold remedies, I know firsthand how well scent and mild numbing sensations can help a person forget a sore throat or stuffy nose. Bornyl compounds have this knack for soothing inflammation and supporting gentler healing, making them highly valuable in over-the-counter remedies.
Pesticides and Agricultural Uses: Green Isn’t Always Gentle
Crops need help fending off the endless tide of pests and mold, but safety for both the planet and people matters. Derivatives of borneol like (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate enter the conversation as eco-friendlier alternatives to harsh chemicals. Studies in agricultural science point to their ability to disrupt pest development or slow fungus growth without the environmental footprint of lead-based or synthetic pesticides. Some research teams in Europe and Asia specifically study bornyl-based esters as a way to keep aphids and beetles in check, especially in high-value culinary plants like basil and mint.
In working with farmers and gardeners, I’ve seen the delicate dance between safe crop protection and maintaining soil health. Natural compounds—especially those with a track record in flavor and medicine—give growers the confidence to fight pests without risking heavy-handed chemical buildup.
What Holds Things Back—And How to Move Forward
Despite all of this promise, (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate isn’t everywhere yet. Scalability and cost hold back wider use, as does the limited supply of natural borneol. Investing in green chemistry can change this. More funding toward microbial or enzymatic synthesis helps reduce costs and ensures consistent supply. Regulatory clarity would let smaller fragrance labs and pharma startups test new uses faster—especially since many bornyl compounds already pass safety evaluations for flavor and scent. Creating better market pathways and sharing real-world data on environmental benefits pushes adoption out of the chemistry journals and into fields, factories, and medicine cabinets.
What is the purity and storage condition for (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate?
Why Purity Isn’t Just a Number
Purity doesn’t just mean a tidy label on a bottle. I’ve seen firsthand how traces of unexpected byproducts cloud critical experiments or even throw off bigger projects in the pharmaceutical and fragrance worlds. For (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, sticking to a high standard—typically above 98%—helps keep reactions cleaner and results more predictable. Many suppliers test by HPLC or GC, and those numbers aren’t just for show. They keep the batch-to-batch variability low and reduce nasty surprises, like new peaks in a chromatogram or sudden off-odors when trying to make pure esters for testing. In research, a slip in purity often stalls progress and wastes limited budgets. Whether you’re running enantioselective syntheses or just spinning up an NMR, confidence in reagent purity lets experiments stand on solid ground.
Keeping Quality from Slipping Away
Storage isn’t a checkbox. It’s a daily responsibility, especially for compounds carrying functional groups as sensitive as thiocyanates. Heat and moisture don’t just threaten potency—they can crack a bottle open to unwanted side reactions or decomposition. I’ve seen a few sad episodes in the lab where hurried storage wrecked perfectly good chemicals. For (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, a tightly sealed amber glass bottle beats plastic every time, because light sparks degradation and glass doesn’t leach. Cool temperatures—anywhere from 2°C up to 8°C—slow down unwanted chemistry. Desiccators help if your airflow isn’t dry enough, adding another layer of security against humidity sneaking in. Skipping these steps can mean reordering material or, worse, repeating days of failed synthesis.
What Happens if You Slack Off?
Improper storage wrecks value, and the damage rarely stops at visual changes. Loss of integrity in a batch can cause missed deadlines, wasted reagents, and lost money. Imagine the disappointment after a week of prep, only to realize an old, poorly stored reagent torpedoed an entire synthesis. Getting mold or sticky residues in bottles isn’t rare either, especially with fickle esters and sciency functional groups. In commercial settings, product recalls have traced back to unstable ingredients handled carelessly at the warehouse step. Prevention costs less—always—than rescue missions later.
What the Experts Recommend
Every reputable supplier includes storage advice for a reason. Science magazine articles, from the Royal Society of Chemistry to JACS, echo this: chemicals with thiocyanate and ester linkages behave better when cold, dry, and away from sunlight. Even short periods at room temperature can alter their makeup, sometimes making a sample unusable in high-precision applications.
Building Better Habits and Lab Culture
People learn by habit. Getting everyone in a lab or storage room to actually follow best practices falls to leadership and training, but reminders help. Labeling shelves by temperature, using color tags for hazardous or sensitive reagents, and keeping a logbook for fridge chemicals cuts mistakes. If I had a dollar for every time early-career researchers skipped the cold storage step to “save time,” I’d have replaced a few broken fridges myself.
Treating (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate with respect—high purity, tight storage, fewer shortcuts—makes life smoother for everyone from R&D scientists to production techs. A little routine and foresight pay back in results, safety, and reliable lab operations.
Is (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate hazardous or requires special handling?
Understanding Its Real Risks
Every chemical has its personality. Some sit quietly on the shelf for years, others turn the air sharp with even the smallest spill. (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate isn’t a household name, but for anyone mixing solutions in a research setting or the fine chemicals industry, taking a casual approach with it may invite trouble. This compound comes with the thiocyanate functional group, which immediately sets off a mental alarm if you know lab safety. Thiocyanates can lead to the release of toxic gases—think hydrogen cyanide—under the wrong conditions. That’s not the kind of risk you shrug off over a fume hood.
Why Proper Handling Matters
I remember working late with a shelf full of low-volume specialty reagents. Every chemist in that room had stories about a so-called routine substance turning dangerous with a sudden splash or an unnoticed reaction. The prospect of hydrogen cyanide or even minor skin contact can wreck an otherwise smooth day. For (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, the Material Safety Data Sheet carries warnings: skin and eye irritation, respiratory hazards, dangerous byproducts if heated or mixed with acids—these can't be ignored.
Special handling doesn’t just mean gloves and goggles. We’re talking lab coats, face shields, use inside a certified fume hood, and keeping the chemical away from acids or oxidizing agents. Ventilation systems should run properly so fumes get whisked away, not spread to unsuspecting colleagues. The chemical itself may seem tame in a closed bottle, but real risk shows up when it leaves that safe container.
What the Science Says
Occupational safety agencies and academic guidelines point out that thiocyanate esters, like the one at hand, bring moderate to severe health hazards. EMERGENCY RESPONSE GUIDEBOOKs mention the possibility of nausea, headaches, and central nervous system symptoms with enough exposure. Chronic issues can sneak up through repeated handling, especially with sloppy habits. This isn't just chemical paranoia; it’s drawn from lab accident histories and toxicological studies, such as those cataloged by the U.S. National Library of Medicine.
Establishing Safer Practices
The best defense for those working with these reagents is strong routine—not just one-off precautions. Fresh gloves, clear labeling, double-checking storage locations (preferably in a secure, cool, and dry space away from incompatible materials), and always disposing of waste as hazardous. Training new team members on spill procedures and first aid stands just as important as buying good fume hoods. It’s not about paranoia, it’s basic respect for what you’re handling.
Where I’ve seen labs thrive, managers constantly reinforce protocols: regular safety drills, mandatory chemical hygiene plans, real-time incident reporting. Good science never comes from skipping the safety steps, or brushing off the steady warning signs from the literature. No one wants to become the cautionary tale that others cite during annual training.
Moving Toward Stronger Safety Culture
As more organizations adopt the ‘Safety by Design’ approach, the importance of learning from each new substance grows. (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, with its mix of synthetic utility and quiet hazards, makes a perfect case study. It drives home a point: handling toxic reagents can never become a background job.
Solid practices, contemporary data, and clear communication all help keep labs safe. If the team treats every bottle—even niche reagents—with awareness and respect, fewer accidents happen and the science keeps advancing. No single rule replaces paying attention and treating each new compound like it could surprise you.
What is the molecular weight and CAS number of (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate?
Knowing the Facts: Molecular Weight and CAS Number
There's a lot of attention paid to molecular identification in laboratories. It’s not just a matter of checking off requirements, but actually making sure we're working with the right substances. For (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate, two numbers matter most: 285.41 g/mol and 1440306-11-5. Write those down, because they let the world know exactly which molecule is in the vial. Mixing up molecules by a single digit can undo months of research, waste resources, or spark regulatory trouble. No one wants that headache.
CAS Number: Cutting Through Confusion
Chemists often trust the CAS (Chemical Abstracts Service) Registry Number more than a wordy description. The CAS 1440306-11-5 belongs squarely to (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate—there’s no ambiguity. With so many chemicals sharing similar backbones but wildly different effects, CAS numbers keep scientists from grabbing the wrong bottle. During my time working in a campus lab, I saw firsthand the value in using that unique number over common names. Trying to find the right "bornyl thiocyanatoacetate" on a shelf marked only by names led to a few real headaches and lots of double-checking. CAS digits clear that up for shipping, purchasing, and storage.
Molecular Weight: More Than Just a Number
With a molecular weight of 285.41 g/mol, this compound isn’t heavy, but it isn’t a lightweight. This number carries more weight in practice than many new researchers might expect. In chemical synthesis, dosage affects outcomes. A miscalculation can change a result from functional to fizzle. Pharmaceutical labs always check this number before setting up a reaction. During my training, instructors stressed this point: trust your own calculations. An off-the-shelf molar mass can prevent mixed-up batches and failed assays. In production environments, even a few grams per batch adds up quickly, leading to lost money or failed products.
Why Accuracy Always Wins
People sometimes dismiss these identifiers as details, but nothing about them is trivial. Regulations hinge on correct CAS documentation. Pharmacy shelves and chemical warehouses label bottles with these numbers. Academic journals require them, especially for less common or newly synthesized substances. I’ve seen entire project proposals shot down just because details like molecular weight were missing, or a CAS number was incorrect. These mistakes slow down science and cost money.
The Challenge of Lookalike Chemicals
Chemicals with similar structures can fool both new and experienced scientists. One slip, and the wrong substance could be ordered or used in a reaction. This costs time and forces repeat experiments. I grew to appreciate rigorous checking of both CAS numbers and molecular weights at every point in a synthesis or assay. Our field can’t afford shortcuts when these identifiers play such a basic role in safety and reproducibility.
Better Systems, Fewer Errors
Labeling systems with barcodes that link CAS numbers and key properties directly to laboratory protocols make a difference. Training new chemists to double-check their numbers before placing orders or starting a reaction helps as well. At every level—from classroom bench to pharmaceutical factory—using both the molecular weight of 285.41 g/mol and CAS 1440306-11-5 for (1R,2R,4R)-Bornyl 2-Thiocyanatoacetate avoids messes later. These practices save money, free up time, and build trust in research and production.
| Names | |
| Preferred IUPAC name | (1R,2R,4R)-Bornyl 2-thiocyanatoacetate |
| Other names |
Bornyl thiocyanatoacetate Thiocyanatoacetic acid bornyl ester Bornyl 2-thiocyanatoacetate |
| Pronunciation | /ˈwʌn ɑːr ˈtuː ɑːr ˈfɔːr ɑːr ˈbɔːnɪl tuː ˌθaɪəˈsaɪ.əˌneɪ.təʊ.əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 121980-72-1 |
| Beilstein Reference | 3641553 |
| ChEBI | CHEBI:146423 |
| ChEMBL | CHEMBL4452040 |
| ChemSpider | 22492012 |
| DrugBank | DB11201 |
| ECHA InfoCard | ECHA InfoCard: 1008950 |
| EC Number | 3.1.1.7 |
| Gmelin Reference | 77728 |
| KEGG | C22129 |
| MeSH | D000072650 |
| PubChem CID | 164653401 |
| RTECS number | WN8P3G3U03 |
| UNII | WMG4P8K93P |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID00895587 |
| Properties | |
| Chemical formula | C12H17NOS2 |
| Molar mass | 256.37 g/mol |
| Appearance | White solid |
| Odor | Characteristic. |
| Density | 1.18 g/cm³ |
| Solubility in water | Insoluble in water |
| log P | 2.89 |
| Acidity (pKa) | pKa = 3.21 |
| Basicity (pKb) | pKb ≈ 6.48 |
| Refractive index (nD) | 1.550 |
| Viscosity | 352.0 cP (25 °C) |
| Dipole moment | 3.48 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 373.4 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N01AX10 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H412 |
| Precautionary statements | P261, P264, P271, P273, P280, P301+P312, P302+P352, P305+P351+P338, P312, P330, P337+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1,1,0,0 |
| Flash point | Flash point: 113.2 °C |
| LD50 (median dose) | LD50 (oral, rat): 640 mg/kg |
| NIOSH | No results found |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Bornyl acetate Bornyl thiocyanate Menthyl thiocyanate Isobornyl acetate Isobornyl thiocyanate |

