5-Mercapto-1H-Tetrazole-1-Acetic Acid: A Closer Look at an Unsung Chemical Cornerstone
Historical Development
Chemistry has plenty of unsung but foundational players, and 5-Mercapto-1H-tetrazole-1-acetic acid fits right into that picture. The tetrazole scaffold first got attention in the early to mid-20th century when energetic materials and pharmaceuticals started to lean on nitrogen-rich heterocycles for both stability and reactivity. For those working in academic labs in the 1960s and 70s, tetrazoles offered a gateway into the world of medicinal chemistry, particularly as substitutes for carboxylic acids in drug design. With its thiol group and acetic acid tail, this compound opened fresh territory, giving chemists a handle for further functionalization, metal coordination, and bioisosteric replacements. Lab notebooks tell a story of researchers boosting yields, sharpening purification methods, and expanding the use of tetrazole derivatives from small bench-scale reactions to commercial applications.
Product Overview and Synonyms
5-Mercapto-1H-tetrazole-1-acetic acid, sometimes called MTA or tetrazoleacetic acid, doesn't often make popular science headlines, but it keeps industries humming quietly underneath it all. Chemists and formulators searching chemical catalogs will spot synonyms like 2-(5-mercapto-1H-tetrazol-1-yl)acetic acid. Whether referenced by abbreviation or full name, this compound draws attention for how effectively it bridges nitrogen-based heterocycle chemistry and sulfur-rich functional groups. The substance usually appears as a pale crystalline powder, though it takes on new faces through salts or metal complexes. Most people outside of a research setting never notice it, yet it plays a distinct role in synthetic campaigns, advanced coordination chemistry, and specific specialty chemicals.
Physical and Chemical Properties
Chemical intuition and practical lab experience teach you the value of understanding physical form and reactivity without always needing an exhaustive database entry. In the jar or flask, 5-Mercapto-1H-tetrazole-1-acetic acid presents as a solid that dissolves in polar solvents like water and methanol, often displaying slightly acidic properties due to the carboxyl group. When you work with it on the bench, a slightly sulfurous odor lingers. Despite its relative stability on the shelf, you have to watch out for light, moisture, and elevated temperatures since these factors can trigger slow decomposition or changes in crystal hydration. The molecule contains a dense cluster of nitrogen within the tetrazole ring, and this arrangement governs many of its bonding and reaction dynamics. On the micro-scale, the thiol group can act as both a reducing agent and a ligand site, muddying the waters for anyone hoping for straightforward oxidation or protection strategies.
Technical Specifications and Labelling
The reality of using this material involves deciphering small-print details on labels and certificates. Purity and moisture content can cause headaches during sensitive syntheses. In the supply chain, you’ll spot technical details—percent purity, residual solvents, and batch-specific physical descriptions—often as a checklist on packaging. Labelling regulations remind researchers to handle material under established chemical hygiene rules, with attention to storage in cool, dark, and dry locations. If a student tries to sneak some out of the jar in a humid lab, you learn quickly how hygroscopic the powder can be. Reliable suppliers put these details front and center, making it easier for researchers or process chemists to trust what they’re pulling off the shelf.
Preparation Method
Setting up the synthesis of this tetrazole derivative, you quickly realize that the pathway must navigate several competing reactions. Classic routes begin with cyanoacetate and hydrazine or azide reagents, cycling through cycloaddition and functionalization. Introducing the thiol group means balancing aggressive nucleophiles, robust acid handling, and sometimes high temperatures. Preparation can demand purification steps involving recrystallization or chromatography, where the choice of solvents and conditions makes or breaks the project. It’s the sort of synthetic challenge that hones a chemist’s attention to procedural specifics and calls for solid troubleshooting when a reaction stalls or yields slump. Over the years, procedural tweaks—alternative solvents, milder reducing agents, optimized isolation steps—shaved hours off syntheses and improved reproducibility, letting research groups scale up or diversify derivatives more reliably.
Chemical Reactions and Modifications
In the wild world of chemical reactions, the ability to exploit both the tetrazole ring and the thiol side chain makes this compound a Swiss Army knife for organic synthesis. The acidic hydrogen on the carboxylic acid can be swapped out with base-promoted esterification or amidation, providing a way to bolt on new appendages. The mercapto group reacts with alkyl halides or forms disulfides, allowing surface functionalization on metals or polymers. Coordination chemistry benefits too, since the sulfur and nitrogen atoms offer complexing sites for transition metals—a fact that found its way into countless research proposals for new catalysts or sensors. Tinkering with reaction conditions can coax this molecule into serving as a nucleophile, electrophile, or ligand, depending on the needs of the moment. This adaptability is a far cry from old, single-purpose reagents that dominated lab shelves decades ago.
Safety and Operational Standards
Any experienced chemist recognizes the responsibility to manage risk while handling multifaceted reagents like 5-Mercapto-1H-tetrazole-1-acetic acid. Training drills it in early that even seemingly mild thiols can cause skin and respiratory irritation, and though the molecule won’t win awards for acute toxicity compared with classics like cyanides or heavy metal salts, gloves and fume hoods set the standard. I’ve seen labs bounce back from incidents where complacency led to accidental spills or minor exposures—nothing focuses the mind better than the sharp odor of volatile sulfur wafting across the bench. Employers and institutions require compliance with regional chemical safety directives (like OSHA or REACH), and these rules demand detailed secondary containment and emergency response plans. Bottom line: respect for the potential hazards keeps everyone healthier, lets research continue uninterrupted, and sharpens a culture of safety that never goes out of style.
Application Area
Talk to industry and university researchers using this tetrazole, and you’ll uncover a spectrum of targets and end uses. Metallurgists favor it for forming robust metal complexes, often in corrosion inhibition or electroless plating baths. In pharmaceutical design, the tetrazole ring borrows space from carboxylic acids as a bioisostere, which can improve metabolic stability or tweak solubility in promising new drug candidates. A few advanced polymer chemists also find ways to anchor this molecule onto backbones to impart adhesive or cross-linking functionality. In my own experience on collaborative projects, the molecule functioned as a linker in developing diagnostic probes, connecting a fluorophore on one end and a targeted ligand on the other, all while maintaining robust aqueous solubility and resistance to degradation.
Research and Development
Chemistry never stands still, and research keeps opening new avenues for derivatives of 5-Mercapto-1H-tetrazole-1-acetic acid. I’ve seen labs around the world dig into new methods for synthesizing this class with greener solvents and reduced waste, a shift driven by mounting pressure for sustainable chemistry. Published literature reveals a steady push to discover rare metal complexes with enhanced catalytic properties, using the multifaceted ligation potential of the tetrazole-thiol system. Materials scientists are finding ways to immobilize sensors on surfaces, using the unique binding capability of the mercapto side chain. Pharma-focused teams report new prodrugs and delivery vehicles resembling this scaffold, aiming to sidestep issues with classic acid moieties. Each time a fresh patent or article lands on my desk, the incremental improvements make it clear that innovation builds steadily—one synthetic tweak or analytical method at a time.
Toxicity Research
Assessing toxicity goes well beyond reading a few safety data sheets, especially when compounds enter potential commercial streams. The tetrazole core doesn’t trigger red flags at the same level as older classes like nitroaromatics or alkylating agents, but the thiol group gets closer attention because of its impact on skin and lung tissue. Formal research tracks both acute and chronic effects on cell lines and animal models, and there’s a growing expectation for rigorous long-term studies before any pharmaceutical or agrochemical derivative sees the light of day. Responsible companies increasingly turn to computational predictive models—QSARs, docking simulations—to anticipate and screen new analogues before wet-lab work begins. Stories circulate in industry circles where ignoring early toxicity warnings led to costly late-stage failures, and these cautionary tales prompt a culture where careful toxicological evaluation forms a non-negotiable part of early R&D.
Future Prospects
Looking ahead, I see several promising directions for 5-Mercapto-1H-tetrazole-1-acetic acid. The growing demand for sustainable chemical platforms encourages greener synthesis routes and streamlined purification protocols, trimming environmental footprint. Computational chemistry will likely play a deeper role in predicting both reactivity and biological interactions, letting researchers build smarter derivatives before hitting the wet bench. In electronics and materials, the molecule’s affinity for metal ions lays the groundwork for innovative sensing platforms and nanostructured catalysts. Pharmaceutical research stays hungry for fresh scaffolds to take on drug resistance, metabolic instability, or solubility headaches, and the unique combo of tetrazole and thiol groups opens up territory yet to be fully explored. Support for open science and data-sharing among global labs will help cross-fertilize breakthroughs, making sure this compound has a longer and wider scientific life than many of its ancestors. The compound has already shown resilience and adaptability; as new problems show up, history suggests resourceful researchers will find new ways to put this multifaceted building block to work.
What is 5-Mercapto-1H-Tetrazole-1-Acetic Acid used for?
What Is 5-Mercapto-1H-Tetrazole-1-Acetic Acid?
5-Mercapto-1H-Tetrazole-1-Acetic acid, often seen in labs as a white or off-white powder, has earned its keep among chemists for good reason. It stands out with its strong sulfur group, which brings unique reactivity—think of it as a little chemical hook that grabs what others can’t easily reach.
Applications in Pharmaceuticals and Research
Pharmaceutical researchers value this compound for building new drugs. Its tetrazole ring mimics the carboxylate group found in many amino acids, which are the building blocks for proteins and often central to modern medicine development. This opens doors to new ways of creating molecules that last longer in the body or bind better to their targets. Chemists rely on it for these modifications because it plays nicely in tough reaction conditions, holding together when heat rises or solvents change.
Building Better Molecules
Medicinal chemistry isn’t just about putting atoms together—it’s about finding shortcuts and clever ways to get more effective drugs. 5-Mercapto-1H-Tetrazole-1-Acetic acid closes gaps there. It acts as a connecting piece in reactions called “bioisosteric replacements,” where the tetrazole swaps in for a carboxylic acid. This tiny change can extend how long a drug lasts, boost how well it dissolves, and sometimes sidestep patents, giving companies a new path for competitive drugs.
Corrosion Inhibition and Industry Use
Metal parts in factories rust and decay faster than most people expect, especially around water. Engineers found that chemicals like 5-Mercapto-1H-Tetrazole-1-Acetic acid, thanks to their sulfur content, latch onto metal surfaces and build a shield that fights corrosion. In oil refineries or water treatment, adding even a small amount can help protect hundreds of thousands of dollars' worth of equipment. Real-world impact like this saves jobs, resources, and energy spent on replacing machinery.
The Environmental Question
There’s a flip side to every powerful tool. With compounds like this, safe handling takes priority. Exposure to tetrazoles can irritate eyes and skin, even in small doses. Sulfur in the wrong place can feed bacteria that cause foul smells or toxic byproducts. Labs and factories using this compound need tight controls, monitoring waste streams, and solid training for workers. Green chemistry calls for exploring safer solvents and designing better disposal methods. As someone who’s spent long hours around active chemicals, these steps aren’t just policy—they’re the difference between health and lasting damage.
Solutions to Move Forward
The way forward means blending science with responsibility. Drug makers can research safer analogs and invest in rigorous toxicology testing. Industrial users need investment in better filtration and real-time leak detection. Leaders in research and production can share best practices more freely, pushing for higher industry standards. People trust products that come from transparent, careful handling, not shortcuts. As demand grows, these measures protect both workers and the environments where they live.
What is the molecular formula and molecular weight of 5-Mercapto-1H-Tetrazole-1-Acetic Acid?
Breaking Down the Structure
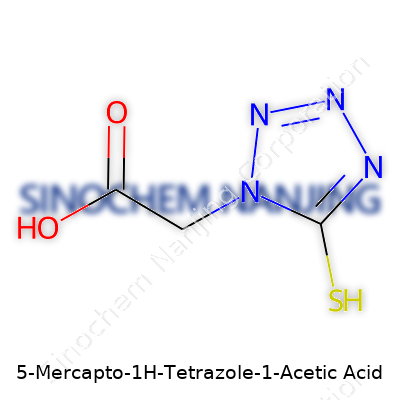
Organic chemistry brings plenty of mouthfuls, and 5-Mercapto-1H-Tetrazole-1-Acetic Acid is one of them. Underneath that long name, there's a structure worth unpacking. The molecular formula—C3H4N4O2S—tells a story of three carbons, four hydrogens, four nitrogens, two oxygens, and a sulfur atom all linked in a tight arrangement. This formula does more than tell us what’s in it; it points directly to its properties, uses, and, often, safety issues chemists think about.
Calculating the Molecular Weight
To get the molecular weight, pull data directly from the periodic table. Carbon weighs about 12.01, hydrogen 1.01, nitrogen 14.01, oxygen 16.00, and sulfur 32.07. When added, C3 (3x12.01 = 36.03), H4 (4x1.01 = 4.04), N4 (4x14.01 = 56.04), O2 (2x16.00 = 32.00), S1 (1x32.07 = 32.07). All together, 36.03 + 4.04 + 56.04 + 32.00 + 32.07 lands at 160.18 g/mol. This isn’t just trivia. Chemists and biochemists use this number to measure out precise quantities, check purity, and prepare solutions in the lab.
Scientific Relevance and Applications
Why care about the details of a molecule’s weight and formula? Experience in the lab quickly teaches that small errors add up. One missed decimal or swapped atom leads to failed reactions, wasted materials, or worse, dangerous outcomes. Getting the composition right ensures researchers develop pharmaceuticals, materials, or teaching samples that actually do what they’re supposed to.
Looking at this acid, colleges and companies often investigate its activity as a ligand in metal coordination chemistry. In past lab sessions, unexpected reactivity linked back to the mercapto (–SH) group, which can bind to metals or act as a nucleophile. This means researchers using 5-Mercapto-1H-Tetrazole-1-Acetic Acid need to appreciate not only its written structure but also how those unique groups act once dissolved or heated.
Importance to Everyday Research
Lots of folks don’t realize how much precision matters until they’re mixing beakers and waiting on a reaction that never happens. The formula spells out exactly what’s present, whether someone aims to analyze by NMR, set up crystallization, or just confirm a shipment from a supplier. A reliable label means fewer surprises and safer handling, which feeds back into higher trust in published results.
On more than one occasion, double-checking the molecular weight before use has helped prevent overdoses of a compound or mislabeling in a database. In the rush of experimentation, clarity about what’s actually in the vial or bottle keeps projects on track. Risks—like inhaling unknown powders or mixing incompatible chemicals—drop off when the facts sit clear at the top of a datasheet.
Moving Forward: Best Practices
Bringing things into the real world, keeping a habit of cross-verifying molecular formulae and weights against trusted databases (like PubChem or ChemSpider) helps maintain accuracy. Reliable sourcing, clear labeling, and updated safety sheets based on precise molecular details all support safe and productive research environments. Teaching newcomers to pause and check their numbers—instead of racing to the next step—builds better habits and fewer mistakes over a career.
Knowledge about 5-Mercapto-1H-Tetrazole-1-Acetic Acid starts at the formula and weight. With that in hand, researchers stand ready to explore complex behavior, create new materials, or support medicine safely—one molecule at a time.
How should 5-Mercapto-1H-Tetrazole-1-Acetic Acid be stored?
Why Storage Matters for Sensitive Chemicals
Storing chemicals never feels like just a box-ticking exercise. I’ve seen new lab techs rush through the process, but with a compound like 5-Mercapto-1H-Tetrazole-1-Acetic Acid, shortcuts can backfire. This compound packs reactive sulfur and nitrogen groups. In an academic lab, I once saw cleanup become chaos because a similar sulfur-containing tetrazole absorbed moisture and started clumping. The right storage decisions help avoid these headaches, reduce exposure risk, and make sure experiments go as planned.
Temperature, Moisture, and Light: The Real Enemies
Moisture keeps sneaking in, especially in humid climates. This chemical pulls water from the air, making it less stable and harder to handle. To keep the powder dry, I use airtight bottles with desiccants tucked inside cabinets away from lab sinks and dishwashers. Cold temperatures slow down unwanted reactions and limit decomposition. Most reference guides—including those from trusted sources like Sigma-Aldrich—advise refrigeration at 2–8°C for extended shelf life. I’ve worked in spaces where chemicals sat at room temperature for weeks, and signs of breakdown popped up fast.
Light doesn’t mix well with many tetrazole derivatives. I’ve watched labels fade on bottles stored in the open, and the compounds inside slowly shift color. Using amber bottles or wrapping clear containers in foil protects against UV and keeps chemistry reliable. This step might sound simple, but many accidents start small before building up.
Avoiding Cross-Contamination and Mixing Hazards
This acid can react unpredictably if it meets incompatible chemicals, especially oxidizers and strong bases. During my time in a teaching lab, I caught a rookie mistake—someone shelved it beside nitric acid. Even a slow leak spells real trouble. Dedicated shelves for sulfur- and nitrogen-based materials keep everyone safe, and labeling with both chemical names and hazard pictograms cuts confusion for those less familiar with complex structures.
Accidental contact with skin or inhalation risks spike if crystals scatter during handling. Wearing gloves, lab coats, and protective goggles is straightforward, but still gets skipped in busy labs. Spills become less likely when bottles with wide necks or scoops sized properly for the material are used. I’ve had to dust fine powders off benchtops too many times—thoughtful handling and storage setups pay off daily.
Documentation and Safety Go Hand-in-Hand
I once helped inventory a university’s storeroom and found chemicals with labels long washed off or faded. Accurate, clear labeling saves so much time and helps everyone—especially after a stressful day or during inspections. Material safety data sheets on hand, whether printed or digital, keep critical facts close for both emergencies and routine checks.
For smaller labs or home setups, regular audits make a huge difference. I set monthly reminders to check for containers with condensation, signs of decomposition, or faded labels. An ounce of prevention stops costly replacement and helps everyone sleep better at night, knowing the lab isn’t sitting on a ticking time bomb.
Thinking Forward: Safer Labs Start with Proper Storage
Storing 5-Mercapto-1H-Tetrazole-1-Acetic Acid reminds us that chemistry isn’t just about reactions in flasks. Care in storage shields the work, the researchers, and the discoveries yet to come. Reliable practices create trust, safeguard the environment, and keep everyone focused on the science—not on preventable emergencies.
What are the safety precautions for handling 5-Mercapto-1H-Tetrazole-1-Acetic Acid?
Why Caution Matters
5-Mercapto-1H-Tetrazole-1-Acetic Acid has earned a spot in many research labs for its role in organic synthesis, metal chelation, and electronics work. Anyone who’s spent much time around this compound knows it brings some headaches along for the ride. Safety isn’t just about rules on a wall. It’s about going home in one piece every night, and letting the research take center stage instead of the accident reports.
PPE Isn’t Optional
Once you crack open a bottle of this stuff, there’s no excuse for skipping personal protective equipment. Proper lab coats, chemical splash goggles, and high-quality nitrile gloves are standard for a reason. 5-Mercapto derivatives often release sulfur-containing odors — you want to keep that smell off your skin and out of your lungs. Anyone who’s ever had to scrub the stink off their hands after a spill knows how much it lingers.
Never handle this acid in street clothes. Fabric pulls in vapors and dust, and you don’t want to take that souvenir home. Closed-toed shoes and long sleeves keep stray drops away from arms and feet. I’ve seen nasty chemical burns on ankles because someone thought “five minutes” without proper gear was no big deal.
Making Ventilation a Priority
Solid or liquid, this compound doesn’t belong in the open air. Set up shop under a certified fume hood every single time. Fume hoods eat up harmful vapors before they drift into breathing space. Most sulfur-rich compounds sting the nose almost immediately, so that first whiff often means the hood fan isn’t on, or you’re working too far out of the sash.
Clean work surfaces before and after use. Dust and residue give this acid a way to sit around for days, and nobody wants to wipe their arm through a hidden patch. Good housekeeping takes five extra minutes, but it saves hours at the doctor’s office.
Storage Knows No Shortcuts
Moisture and heat will break down 5-Mercapto-1H-Tetrazole-1-Acetic Acid, releasing dangerous fumes and risking spontaneous degradation. Store these chemicals away from oxidizers, strong acids, or bases. I’ve seen labs clear out half a fridge after one unlabeled bottle started corroding its cap and leaking.
Lock samples in tightly sealed containers. Use secondary containment trays to catch accidental spills. Keep materials away from sunlight and sources of ignition; one power surge can fork over a whole summer’s work in flames.
Spill Preparedness and Waste Handling
Spills are a “when,” not an “if.” Anyone handling this acid should know where the nearest spill kit sits and how to use it. Have neutralizing agents, absorbents, and disposable towels within reach. Small spills demand fast action: absorb, contain, and dispose in labeled hazardous waste bags. Never pour it down the drain or toss it in the regular bin — environmental damage happens fast and doesn’t stay in the lab.
Label all waste containers with the exact chemical name and date. Routine checks for leaks or overfilled bins save time and trouble. I’ve seen too many researchers pay the price for trusting an unlabeled bottle in the back of the storage cabinet.
Training Isn’t Just Paperwork
It’s tempting to treat chemical handling training as a box-ticking exercise. Real skill comes through hands-on walkthroughs, in-the-moment questions, and honest stories about what goes wrong when people get lazy. No one ever plans to make a mistake, but routine drills and clear protocols lower the odds of disaster. A good lab culture rewards questions and shared responsibility, not shortcuts.
Working with tough chemicals carries risk. Awareness and preparation keep accidents on the rare side, letting the science — and not the incident reports — do the talking.
Is 5-Mercapto-1H-Tetrazole-1-Acetic Acid available in different purity grades or packaging sizes?
The Substance and Its Role in Research
5-Mercapto-1H-Tetrazole-1-Acetic Acid lands in the crosshairs of scientists for a reason. Its unique structure gives it a strong pull in synthetic chemistry and analytical labs. I remember a time in grad school when just getting a small sample of an uncommon compound felt like a victory, especially if it met the purity level our group demanded. In pursuit of results, the grade of this chemical counts for more than one might expect. Errors in synthesis or data often point back to tiny impurities in a source reagent.
Purity: The Non-Negotiable Variable
Whether targeting pharmaceutical intermediates or conducting binding studies, the difference between a 95% pure batch and a 99% pure batch can show up in final outcomes. Even a 1% unknown contaminant throws off chromatograms. I’ve watched a colleague lose days, painstakingly tracking the signal of an unwanted byproduct—later, we traced it right back to the reagent grade. Buying 5-Mercapto-1H-Tetrazole-1-Acetic Acid in multiple grades now becomes essential, not luxury.
Companies now recognize this. Established suppliers provide the acid in research, technical, and analytical grades. Laboratories working towards publication or regulatory approval reach for the highest chromatographic purity they can afford. Sometimes, fresh synthetic projects only call for the basic grade. Specialized pharma and biotech companies will insist on detailed Certificate of Analysis documentation, verifying trace impurity levels or identifying residual solvents.
Packing Choices: Scaling From Bench to Bulk
It’s a scenario I’ve lived through—budget meets ambition. Sometimes, your protocol demands only a few milligrams. Other times, a scale-up experiment chews through grams in weeks. Access to 100 mg, 1 g, or 25 g bottles side-by-side can be the difference between being stuck and surging ahead. Distributors do respond accordingly. Large chemical companies and catalog suppliers such as Sigma-Aldrich and Alfa Aesar won’t squeeze you into a one-size-fits-all model. Smaller glass vials work just as well for those running pilot reactions or analytic validation.
For academic teams, cost still weighs heavily on decisions. Buying the smallest offered quantity has saved my own group more than once, particularly when trying out a compound for the first time. Industrial buyers, in contrast, work out long-term purchase contracts measuring in kilograms. There, packaging upgrade options—HDPE drums or bulk resin bags—reduce waste and shipping fees.
Why Standards Matter
Traceability forms the foundation for safe, clean chemistry. Trusted chemical suppliers won’t just sell you any bottle. Batch numbers and material safety details back each shipment. Regulatory compliance—whether for GLP labs or Good Manufacturing Processes—depends on having these records. It’s comforting as a bench chemist to know what’s on your shelf came with honest documentation, and that any deviation reflects your experiment, not the starting material.
Meeting New Demands
The chemical landscape keeps shifting. Environmental pressures and increasing global demand push suppliers to provide more options. Custom purity grades or a wider menu of packaging sizes have become normal in the catalogue. I’ve noticed small biotech startups ordering “just enough,” while eco-minded research teams favor recyclable packaging over traditional plastic. Sometimes, requests even lead to community improvements—a supplier might add a low-metal-content variant in response to rising demand from electronics researchers.
Conclusion
Choices in purity and packaging sizes for 5-Mercapto-1H-Tetrazole-1-Acetic Acid matter for reasons rooted in real laboratory experience. Sourcing the right grade safeguards experiment integrity, stretches budgets, and supports safety. Open communication between buyers and producers keeps moving innovation forward, one well-labeled bottle at a time.
| Names | |
| Preferred IUPAC name | 2-[(1H-tetrazol-5-yl)sulfanyl]acetic acid |
| Other names |
1H-Tetrazole-5-thiol-1-acetic acid 5-Sulfanyl-1H-tetrazole-1-acetic acid 1-(Carboxymethyl)-5-mercaptotetrazole 1H-Tetrazole-5-thiol acetic acid |
| Pronunciation | /ˈfaɪ mərˈkæpˌtoʊ wʌn eɪtɛˈtræzoʊl wʌn əˈsiːtɪk ˈæsɪd/ |
| Identifiers | |
| CAS Number | 940-46-5 |
| 3D model (JSmol) | `35715/mol/LnMzSbp/XDPkzhu5awsFQCIvLQFqNVkTCrRjJ7C9qfY=` |
| Beilstein Reference | 1858739 |
| ChEBI | CHEBI:85171 |
| ChEMBL | CHEMBL151872 |
| ChemSpider | 25483680 |
| DrugBank | DB08233 |
| ECHA InfoCard | ECHA InfoCard: 100.122.163 |
| EC Number | EC 260-396-1 |
| Gmelin Reference | 121307 |
| KEGG | C14025 |
| MeSH | D017311 |
| PubChem CID | 124478 |
| RTECS number | XZ3150000 |
| UNII | 76TMK3D64F |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C3H4N4O2S |
| Molar mass | 162.17 g/mol |
| Appearance | White to Off-White Powder |
| Odor | Odorless |
| Density | 1.87 g/cm³ |
| Solubility in water | slightly soluble in water |
| log P | -1.3 |
| Acidity (pKa) | pKa = 2.2 |
| Basicity (pKb) | pKb: 11.34 |
| Magnetic susceptibility (χ) | -33.2 × 10^-6 cm³/mol |
| Refractive index (nD) | 1.700 |
| Dipole moment | 2.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -307.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | Std enthalpy of combustion (ΔcH⦵298) of 5-Mercapto-1H-Tetrazole-1-Acetic Acid: **-1167 kJ/mol** |
| Pharmacology | |
| ATC code | D03AX12 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302+H312+H332, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P333+P313, P362+P364, P337+P313, P501 |
| Flash point | > 242.3 °C |
| Lethal dose or concentration | LD50 (Oral, Rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): >2000 mg/kg (rat, oral) |
| NIOSH | SN1784000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 500 mg |
| Related compounds | |
| Related compounds |
1H-Tetrazole 5-Amino-1H-tetrazole 5-Methyl-1H-tetrazole 5-Phenyl-1H-tetrazole Mercaptoacetic acid |

