10-Azaanthracene: Science, Story, and the Road Forward
Historical Development
The story of 10-Azaanthracene spins out from classic anthracene chemistry, tracing its roots back to the bustling years when chemists craved new aromatic frameworks. Early synthesis work took cues from coal tar discoveries and the ambitious plans of the Wöhler era. Real momentum kicked in through the twentieth century, when the need for nitrogen-containing polycyclic aromatics ramped up with interest in dyes, pharmaceuticals, and organic electronics. German and British labs, hungry for new scaffolds, started tweaking the anthracene backbone, leading to this intriguing compound: a three-ring aromatic with one CH swapped for a nitrogen at the 10-position. What stood out wasn’t novelty for its own sake, but the realization that introducing nitrogen unlocked a new range of electronic and reactive behaviors. Each decade added new wrinkles—improved syntheses, expanded uses, growing awareness of both promise and challenge in scaling up production. Research never really slowed, as every advance folded into another cycle of applications and questions.
Product Overview
10-Azaanthracene shows up as a yellow to light-brown solid, easy to spot on a chemist’s workbench thanks to its color and faint, somewhat sharp odor typical for similar aromatic compounds. Not a household name, it still finds its way into many laboratories, especially in fields aimed at pushing the limits of aromaticity and electron-rich systems. Its aromatic core holds together surprisingly well, resisting breakdown under normal conditions. For anyone who’s worked with similar materials, handling it feels familiar, making it approachable for both academic and industrial researchers willing to experiment beyond simple anthracene.
Physical & Chemical Properties
The compound’s melting point sits in the range typical for high-stability aromatics, generally holding well above 100°C. Solubility tells the main story—it goes easily into common organic solvents but resists water. This opens up quick purification routes but limits how it mixes with more polar procedures. Chemically, swapping a nitrogen into the structure tweaks electronic properties: you get a different pattern of electron flow, slightly lower electron density in some positions, and a change in how radical cations and anions form. As a solid, it mostly keeps to itself, but in solution, it starts to show off more: strong fluorescence under UV, sensitivity to acid or base, and an ability to bind protons or metals via its lone nitrogen pair. These properties really matter when you design new ligands, sensors, or organic electronics.
Technical Specifications & Labeling
Purity jumps out as the main concern—nobody wants unknown contaminants when they’re optimizing a photophysical measurement or catalysis pathway. Most labs trust suppliers who offer purities above 98 percent, detailed spectral data, and batch analysis. Labels often highlight the compound’s aromatic nitrogen content and storage advice—keep out of moisture, limit light exposure, and avoid extremes of temperature. No one wants a sample of 10-Azaanthracene going yellow then black in a dark drawer just because a cap came loose. Even so, real-world preparations don’t always live up to ideal specs, so folks regularly run their own NMR, MS, or elemental checks, tying in trust but verify as an everyday lab mantra.
Preparation Method
Synthesis reflects a mix of old and new thinking. Early routes borrowed from classic Friedländer or Skraup-type reactions, drawing on readily available starting materials like anthranilic acids, aminophenyl ketones, or their derivatives. The modern chemist leans on cross-coupling and condensation, sometimes aided by catalytic metals, solvents that encourage cyclization, or controlled heating. The long-standing challenge involves managing both yield and purity; you want maximum throughput but hate seeing byproducts that clog up separation. Green chemistry advances have nudged things forward, with some groups swapping out noxious reagents for greener alternatives and focusing on atom economy. Each generation of improvement matters. Cleaner reactions mean less waste, happier downstream users, and more defensible safety and environmental records.
Chemical Reactions & Modifications
10-Azaanthracene responds well to substitution and coupling chemistry, especially at the positions next to nitrogen or at the ring periphery. Halogenation, nitration, or reductive alkylation turns out to be manageable. These tweaks produce families of derivatives that appear in analytical probes, ligands for metal complexes, or even building blocks for organic semiconductors. Its lone nitrogen changes the way electrons flow during reaction, supporting both electrophilic and nucleophilic attacks under controlled conditions. For the organic chemist, this opens up a bench full of options—tuning light absorption, shifting redox properties, or adding hairier functional groups to chase after specific applications. Over time, these modifications have turned 10-Azaanthracene from a curious molecule to a springboard for more complex architectures.
Synonyms & Product Names
The molecule answers to a few names: 10-azaanthracene stands out in IUPAC circles, but chemists also call it 10-azanaphthacene, sometimes even slipping into older naming like 9-azaanthracene if count starts at a different ring. These names pop up in older literature or supply catalogs, reminding everyone to double check structures rather than just labels. That calls for vigilance in lab orders and literature searches, which anyone who’s chased a reference for hours can appreciate.
Safety & Operational Standards
Lab safety for 10-Azaanthracene grows from both its aromatic core and nitrogen substitution. Most seasoned chemists treat it with the same respect they give anthracene or phenazine derivatives: gloves, eye protection, and careful avoidance of inhalation or dust creation. Papers and supplier docs weigh in on respiratory effects and the messy possibility that aromatic amines can trigger long-term toxicity. Waste management dovetails with best practices for persistent organic pollutants—proper containment, labeling, and avoidance of drain disposal. Many university and industry EHS protocols list aza-aromatics as materials to handle in fume hoods, not just open benches. Each of these steps supports both personal safety and environmental stewardship—lessons hammered home at every lab safety refresher or after-action review.
Application Area
Applications run wide, shaped by a mix of chemical robustness and flexibility. In the lab, 10-Azaanthracene stands in as a template for the design of new materials—its conjugated system engages photochemists, phenomenologists, and those engineering organic conductors or semiconductors. The pharmaceutical angle draws on its resemblance to bioactive polycyclics, paving the way for new drug analogs or investigation of enzyme inhibitors. Metal-organic complexes based on this skeleton find use as probes or catalysts, leveraging the nitrogen’s ability to coordinate and stabilize transition metals. Academic studies branch out into sensing technology: the molecule’s strong fluorescence underpins sensors for protons, metals, and radicals. These applications all benefit from the balance of stability and reactivity baked into the aza-anthracene system.
Research & Development
Ongoing research keeps stretching boundaries. Green chemistry inspires new synthesis methods and functionalization tricks. Teams working on organic electronics continue to test 10-Azaanthracene as a scaffolding for improved light-to-electricity conversion or as an efficient emitter in OLED devices. Environmental analysis groups deploy it as a probe, mapping trace metals or environmental pollutants thanks to sensitive fluorescence response. The pharmaceutical world continues to test derivatives in screens for antimicrobial, anticancer, or enzyme-modulating activity, even if most efforts remain at the preclinical or lead-hopping stage. Spillover from these efforts often lands back in method development—better separations, smarter sensors, or more sustainable chemical pathways bringing round after round of incremental but real-world improvement.
Toxicity Research
Toxicity represents the flip side of 10-Azaanthracene’s promise. Aromaticity combined with nitrogenation often prompts long-term health questions, echoing concerns that have surrounded related aromatic amines for decades. Studies point to the need for careful handling, especially for chronic exposure: respiratory risk from dust, possibility of DNA interaction, and unclear pathways for breakdown in environmental systems. While direct epidemiology is sparse, structure-activity parallels suggest it belongs in the family you want to treat with serious care. Regulations covering waste disposal, workplace exposure, and environmental monitoring contribute to a framework that supports responsible use. Ongoing research aims to clarify breakdown products, metabolic fate, and routes for remediation in case of accidental releases. The stakes feel real in any environment where safety isn’t just a checkbox but an expectation built on lived experience.
Future Prospects
Looking forward, 10-Azaanthracene’s future stands up to scrutiny from both scientific and societal needs. Technology continues to reward molecules that put versatility and sustainability within reach. Anyone working at the interface of organics and electronics sees ongoing value in customizing conjugated scaffolds. The combination of ease of synthesis, stability, and accessible modification suggests more growth in advanced materials—from thin-film devices to molecular sensors. Pharmaceuticals may lag, but the search for new frameworks never really stops. Attention to green chemistry and occupational health drives the search for cleaner, safer syntheses and whole-life stewardship. Looking at the broad investment in research, collaboration across synthetic, analytic, and toxicological teams signals that the challenges and promise of aza-aromatics, embodied in 10-Azaanthracene, remain far from exhausted.
What is 10-Azaanthracene used for?
Looking Beyond the Chemical Formula
Pick up a bottle labeled 10-Azaanthracene in a lab and it probably looks like just another yellowish powder destined for a back shelf. So why do organic chemists keep this compound on their radar? Let’s get right into it. 10-Azaanthracene stands out because it contains a nitrogen atom embedded in its three-ring structure, swapping out a carbon. This simple swap has big effects on both reactivity and application.
Making New Materials Happen
Modern research doesn’t just chase knowledge for the sake of it; scientists hunt for molecules that help create better materials. 10-Azaanthracene helps researchers make building blocks for organic electronics. These aren’t esoteric gadgets—think about flexible screens or new ways to convert sunlight into energy. In my grad school years, colleagues tested aza-derivatives for light-emitting diodes and organic field-effect transistors. The nitrogen atom shifts the molecule’s electronic properties enough to give new possibilities for energy transfer and charge mobility.
Useful in Pharmaceutical Research
Let’s say you want to develop a new drug. Molecules like 10-Azaanthracene give you a launching pad. Acquiring new chemical “scaffolds” means that drug companies can try out new ways to block or enhance signals inside the body. Heterocycles, or ringed molecules containing nitrogen, pop up again and again in top-selling medications. Chemists use azaanthracene as a starting point for more complex molecules, letting them tweak solubility, binding strength, or the avoidance of unwanted side effects. Researchers have published several papers showing how azaanthracenes can hook up with other atoms to create inhibitors for enzymes tied to cancer or inflammation.
Understanding Chemical Interactions
Chemists don’t work in a vacuum. Every experiment leans on what’s worked—or failed—before. 10-Azaanthracene offers a way to examine how electrons move through ring systems. In practice, that means it turns up in fundamental chemistry labs that focus on photochemistry or testing new reactions under UV light. Its fluorescence and altered reactivity teach students and researchers about reaction mechanisms in real life, not just on paper.
Environmental Sensors and Detection
One unexpected angle: scientists have found that 10-Azaanthracene-based compounds help in detecting pollutants or dangerous chemicals. Certain derivatives light up differently in the presence of heavy metals or other contaminants. I remember a project that tested modified azaanthracenes as quick detectors in river samples—the stuff works, and a change in color or fluorescence gives a clear, cheap early warning about water quality.
Challenges and Solutions
Handling 10-Azaanthracene comes with challenges. The nitrogen atom makes it less stable under strong acid or heat than regular anthracene. Some researchers run into waste issues, since old-school purification gets finicky. Instead of tossing more solvents at it, green chemistry suggests milder conditions, solvent recycling, and smaller batch runs to cut down on waste. Plus, with new purification tools, it’s becoming easier to recycle and reuse starting materials.
No Silver Bullet, But Real Value
10-Azaanthracene doesn’t make headlines, but it earns its shelf space. Researchers in my field respect molecules like this for their versatility and the doors they open—sometimes in electronics, sometimes in health, sometimes in environmental applications. What keeps them valuable is not just what they do alone, but how they help push other discoveries ahead.
What are the physical and chemical properties of 10-Azaanthracene?
Getting to Know 10-Azaanthracene
10-Azaanthracene tells a unique story among aromatic compounds. Picture a structure similar to anthracene, but one of the central carbons is swapped out for a nitrogen atom. That tiny substitution opens up an entirely different world for performance. The presence of nitrogen brings a subtle twist to both stability and reactivity—qualities anyone handling chemicals learns to respect quickly.
Physical Characteristics That Stick Out
Pull a sample of pure 10-Azaanthracene out of storage and you’re likely to see pale yellow crystals. They don’t throw any bold colors like some of their cousins, but their understated appearance hints at purity—usually a sign of careful synthesis and isolation. This compound melts at about 157-160°C, a mark familiar to any chemist who collects melting points to check sample cleanliness or to compare against published values. That point above 150°C means no casual melting in the lab, and you’re working with a solid under normal room conditions.
Run a nose test next to nearly any dry aromatic and you’ll pick up a faint smell, sometimes almost sweet. 10-Azaanthracene strikes a mild aroma, though not as strong as pure anthracene. It doesn’t dissolve easily in water. Instead, try organic solvents if you want it in solution—think chloroform, dichloromethane, or even a bit of toluene. For practical chemical work or material applications, this matters, since those aren’t solvents you’d use in everything. Having a molecule this stubborn against water outlasts a spill or a rinse in the sink, so safety protocols always stay in play.
Chemical Character and Reactivity
The nitrogen atom doesn’t just sit there passively. This tiny change tunes the electron density throughout the aromatic rings. It makes the molecule more reactive than plain anthracene, mainly when you’re talking about reactions with acids, bases, or oxidizers. In our lab work, any nitrogen-embedded aromatic often shows a tendency for more selective chemistry. The basic site at the nitrogen can coordinate to metals or accept protons, so in coordination chemistry this opens doors. Cancer researchers, for instance, have pushed aza-aromatics in DNA intercalators for years, and 10-Azaanthracene finds use in that experimental sphere.
The stability of 10-Azaanthracene can’t go unmentioned. Leave it on the bench and it’ll hold up to room air for a while, but throw it in with strong acids or powerful oxidizers and the story shifts. I’ve seen it handled bare-handed in undergraduate labs—still, gloves and eye protection win out every time, since you never truly know the byproducts when someone improvises a reaction.
Where the Science Takes Us
Electronic properties play a starring role. Swapping carbon for nitrogen tweaks the electron flow across the molecule. In my experience, aza-aromatics like this one wind up in the design of organic electronics or light-emitting materials. These tweaks to the aromatic “backbone” actually helped kickstart work on improved organic LEDs and solar cell materials. The research world shifted to these molecules in the search for better conductivity, longer lifetimes, and stability under different lighting conditions. People who don’t spend their days mixing chemicals might not know it, but molecules like 10-Azaanthracene are in the backdrop of that progress.
Looking Toward Practical Use
Not every synthetic chemist keeps 10-Azaanthracene on the shelf, but for those who do, it’s because of its ability to bridge fields—everything from medicinal chemistry to material science. Accessibility does leave some room for improvement in supply chains and greener synthesis, especially since some standard routes use heavy metals or strong acids. Chemists interested in safety and sustainability look to move away from high-risk reagents and toward more benign conditions, maybe utilizing catalytic methods or solvent swaps.
How should 10-Azaanthracene be stored and handled safely?
Respecting Chemistry Means Respecting Risks
10-Azaanthracene catches attention among researchers for its role in organic synthesis and material science. I’ve spent hours in labs where chemical safety marks the line between productive work and a trip to occupational health. With compounds like 10-Azaanthracene, a cavalier approach courts disaster. Thinking it’s “just another organic” ignores the reality that mismanagement could mean exposure to hazardous dust, fire, or unwanted reactions that mess with your health and the lab.
Understanding Its Hazards
This compound, sharing family traits with anthracenes, comes with its share of safety warnings. Those who’ve handled its crystalline powder know it can irritate skin and eyes. Studies point to harmful effects from inhalation over long periods. Safety data sheets flag it as hazardous, and the mere fact that it’s used in research—not consumer products—should urge caution. My own experience has taught me never to underestimate aromatic amines for toxicity.
Storage: Preventing Trouble
Safe storage doesn’t start with a locked cupboard—it starts with respect for chemical reactivity. Store 10-Azaanthracene in a tightly sealed glass or compatible plastic container. Some researchers use PTFE liners in the cap for extra protection. Moisture and direct sunlight break down this compound over time, so a cool, dry spot away from bright light works best. Flammable storage cabinets help reduce fire risks. I have seen fires start from neglecting ignition sources or letting chemicals sit too close to heat-generating equipment.
Clear labeling is more than bureaucracy. I remember the confusion during an audit when one bottle lacked proper labeling—time wasted, hazard increased. Keeping storage containers marked with the chemical’s name, hazard symbols, and the date received prevents mix-ups and keeps inspections short and smooth.
Handling: Safety Starts Before the Experiment
No shortcut beats the safety basics. Proper PPE makes the biggest difference: nitrile gloves, safety goggles, and a lab coat are minimal requirements. A dust mask or respirator means cleaner lungs, especially when weighing powders. I always make sure the balance stays inside a ventilated enclosure. Splash risks mean using trays and working away from the edge of the bench; nobody wants crystalline dust drifting onto skin or into equipment.
Some solvents and bases can trigger unwanted reactions with 10-Azaanthracene, so checking for incompatibilities before combining chemicals pays off. Spills happen, so absorbent pads and appropriate containers for chemical waste sit close at hand. After all, no researcher aims for chemical residue on shared benches.
Disposal: Doing the Right Thing for the Workspace—and the Environment
Proper disposal protects both lab crews and groundwater. After each run, I place wipes and leftover material in a chemical waste container labeled for organic solids. The temptation to toss things in regular trash never pays off. Local waste disposal rules shape the process, but waiting for a hazardous waste pickup costs far less than dealing with soil contamination claims.
Common Sense and Culture
Caring for chemical safety isn’t about fear; it’s about thoughtful routines. Weekly safety checks, easy access to spill kits and a culture where team members flag hazards go farther than any printed guideline. Stories circulate about incidents where a shortcut led to big problems. My years working in busy research settings taught me that shared responsibility—eyes on each other, raising concerns—builds a safer lab for everyone.
Is 10-Azaanthracene hazardous or toxic?
Understanding 10-Azaanthracene
10-Azaanthracene catches the eye in research labs where chemists build new molecules for electronics, sensors, or possibly medicines. Its structure, a tweak of the anthracene skeleton with a nitrogen atom, opens the door to lots of creative work in organic chemistry. The main question people have isn’t about how to make it, but whether it’s safe to handle in the first place.
Gaps in Toxicity Data
Search for hard toxicology numbers on 10-Azaanthracene, and things turn frustrating quickly. It rarely appears in the kinds of regulatory documents that spell out exactly how toxic a compound looks in animal studies or environmental tests. Compare this to anthracene itself, tracked by the EPA and widely cited for low acute toxicity but with some flags for long-term health or ecological risk. For 10-Azaanthracene, scientists mostly rely on broader patterns: nitrogen-containing polycyclic aromatic compounds tend to carry greater reactivity. Some display carcinogenic properties, cause mutations in cell cultures, or disrupt aquatic life.
Why Uncertainty Matters
Ignoring uncertainty isn’t smart, especially in chemical labs. I spent years in research where someone always wanted to push safety to the side because one compound “didn’t look dangerous.” Then later, stories rolled in about skin rashes, mysterious coughs, or headaches. Too much skepticism toward risk led to rushed handling and accidents. Even with substances showing mild acute effects, repetitive exposure in poorly ventilated spaces tends to build health burdens over time. Where regulatory data falls short, the best bet is a healthy dose of respect for the unknowns.
Practical Steps in Handling
Lab safety isn’t complicated. Put 10-Azaanthracene under a fume hood. Gloves, long sleeves, goggles, and a lab coat may not feel heroic, but they make a big difference when working with chemicals that could absorb through the skin. Good housekeeping—wiping surfaces, prompt cleanup of spills, waste kept in clearly marked containers—does more than any paperwork to lower risk. Keep material safety data sheets handy, even if they offer limited details, and stay ready to update them as more information surfaces.
Long-Term Impact Outside the Lab
Volumes of discussion swirl around the environmental effects of similar aromatic molecules. They can stick to soil and sediment, persist in water, and resist breakdown. Some pass through water treatment plants and come back to haunt wildlife through slow accumulation. If 10-Azaanthracene follows this pattern, lab disposal practices should matter as much as lab safety itself. A drain or a trash can isn’t an exit strategy. Work with hazardous waste services that know their business and never treat research leftovers as harmless just because there’s little published data.
Building a Safer Culture in Chemistry
Policy moves slow, so much of the responsibility falls on people in the trenches—students, postdocs, lab techs. A culture that asks questions about safety, pushes for transparency from suppliers, and shares red flags when something feels off ends up safer. Real trust forms in labs not when everyone shrugs at the unknown, but when people show they care about protecting their own health and the world outside.
Looking Ahead
Until regulators and researchers agree on full toxicity profiles, the approach with 10-Azaanthracene demands caution and humility. Every new compound deserves a period of careful respect. That’s not just policy, it’s practical wisdom learned from years of close calls and stories nobody wants to repeat.
Where can I buy 10-Azaanthracene and what is its price?
A Closer Look at 10-Azaanthracene Sourcing
Anyone searching for 10-Azaanthracene has probably come across a few hurdles. Unlike everyday chemicals you can order with a few clicks, 10-Azaanthracene requires more legwork. Only a handful of specialized suppliers carry it, and most require you to fill out some form of researcher verification or disclose your intended use. Sigma-Aldrich, Alfa Aesar, and TCI America list the compound in their catalogs, but they don’t stock it in their general chemical warehouses like basic solvents. You end up corresponding with their sales representatives or requesting a quote through online portals.
10-Azaanthracene typically gets used in academic labs or R&D teams, mostly in fields touching on organic electronics or material chemistry. Because of this narrow customer base, suppliers tend to produce it in small batches. Quantity options mostly fall in the milligram to gram range. If you ask about a kilogram, you’ll get a very different quote—and likely a lot of questions about your lab and project. That scrutiny follows the usual safety and compliance protocols tied to specialty chemicals.
The Real-World Price Tag
The price of 10-Azaanthracene depends heavily on purity and quantity. Prices swing from around $250 for 100 milligrams at over 97% purity, up past $1,000 for a gram. In my experience running a startup chemistry project, this price barrier forces smaller labs to double check whether alternatives exist, or to explore whether a synthesis route makes sense internally. Most university groups turn to small-scale purchases, often splitting quantities among several projects to stretch grant budgets.
For bigger buyers or those looking to scale a process, bulk discounts only show up once you ask about larger amounts, but it rarely becomes “cheap” in any sense. That reflects both tight supply and the complexity of making the compound. Not every specialty chemical producer is willing to set up for custom synthesis, especially if the chemical involves tricky steps or low yields.
Access Isn't Only About Price
Availability plays a big part in the story. Laboratories in North America or Western Europe generally place an online order and wait a week or two. Colleagues in South America, Southeast Asia, or Africa face longer shipping times, higher courier fees, and occasional customs headaches. I’ve seen researchers in less-connected regions partner with collaborators abroad just to get rare chemicals shipped into their country. Logistical barriers sometimes cost more—both in money and lost research time—than the sticker price itself.
Chemical suppliers could improve transparency not just in pricing, but in regulatory restrictions and delivery lead times. Adding more supply-chain clarity gives labs a better shot at planning research calendars without holding projects back for months due to shipping miscommunications or documentation snags.
Building a Better Path for Sourcing
For students and smaller labs hoping to stretch funds, open communication helps. Suppliers will sometimes suggest alternate compounds or advise when a substitution makes sense. Some academic consortia pool their orders for lower prices, forming informal buying groups to share rare chemicals. More broadly, researchers and chemical suppliers could work together to establish centralized resource banks for stockpiling specialty reagents and sharing internationally.
The price of 10-Azaanthracene paints a picture bigger than dollars and cents. Barriers in special chemical sourcing speak to the broader challenge of access in science, where geography and budget still shape what kinds of experiments are possible.
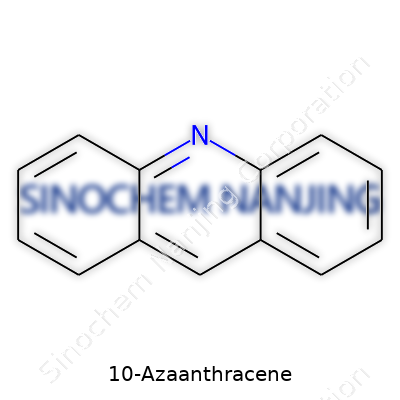
| Names | |
| Preferred IUPAC name | azaphenanthrene |
| Other names |
10-Azabenzo[a]anthracene 10-Azanthracene 10-Aza-anthracene 10-Azaanthracene |
| Pronunciation | /ˈtɛn ˌæzəˈænθrəˌsiːn/ |
| Identifiers | |
| CAS Number | 335-19-3 |
| Beilstein Reference | 29019 |
| ChEBI | CHEBI:75805 |
| ChEMBL | CHEMBL3348920 |
| ChemSpider | 65071 |
| DrugBank | DB08357 |
| ECHA InfoCard | ECHA InfoCard: 100.045.292 |
| EC Number | 620-36-4 |
| Gmelin Reference | 635197 |
| KEGG | C11368 |
| MeSH | D000936 |
| PubChem CID | 70970 |
| RTECS number | AT3676000 |
| UNII | IQ77B87I0C |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | 10-azaanthracene |
| Properties | |
| Chemical formula | C13H9N |
| Molar mass | 219.26 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.18 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.38 |
| Vapor pressure | 1.01E-6 mmHg at 25°C |
| Acidity (pKa) | 4.99 |
| Basicity (pKb) | 2.8 |
| Magnetic susceptibility (χ) | -77.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.715 |
| Dipole moment | 2.08 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 224.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4073 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D03AX12 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P280: Wear protective gloves/protective clothing/eye protection/face protection. |
| NFPA 704 (fire diamond) | 1-2-0-† |
| Flash point | 110°C |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (oral, rat) |
| NIOSH | NA |
| PEL (Permissible) | Not established |
| REL (Recommended) | 500 mg |
| Related compounds | |
| Related compounds |
Acridine Phenazine Anthracene |

