1-Pentanethiol: A Thorough Look at Its History, Properties, and Applications
Historical Development
Organic sulfur compounds have generally carried a mixed reputation, from stinky oils playing roles in industrial chemistry to pivotal molecules intertwining with the early days of organic synthesis. 1-Pentanethiol stepped into the spotlight as chemists expanded their horizons beyond alcohols and simple hydrocarbons. In the early twentieth century, research into the reactivity of carbon-sulfur bonds grew as industries needed molecules with unique solubility, reactivity, and odor properties. Industrial applications for thiols, often called mercaptans, drove investment into refining production methods and understanding their place in the world of chemical reactions. The past several decades have shown a clear trend toward not only recognizing thiols for their potent, sometimes overwhelming, odors but also appreciating them as crucial intermediates in the manufacturing of pharmaceuticals, flavors, and even certain polymers.
Product Overview
1-Pentanethiol is a colorless to pale yellow liquid that instantly grabs attention with its strong, pungent odor – the kind you won’t easily forget, unfortunately. This compound fits snugly in the alkylthiol family, carrying a five-carbon straight-chain backbone and a single thiol group at the end. While the odor often takes center stage in descriptions, for those working in the lab or a plant, performance takes precedence. 1-Pentanethiol holds a particular niche as a reagent in organic synthesis, a modifier in lubricant additives, and a building block in scent creation. It’s not exotic, but it’s indispensable to anyone who relies on purposeful manipulation of sulfur chemistry.
Physical & Chemical Properties
1-Pentanethiol boils at around 119-120 °C, which comes in handy for distillation or separation from mixtures in industrial settings. It forms a slightly less dense layer on water, as its density sits at about 0.84 g/cm3. You won’t find it dissolving readily in water, but it blends well with most organic solvents thanks to its uncharged, hydrophobic carbon chain. The thiol group brings reactivity, especially with oxidants or alkylating agents. From a practical point of view, the volatility means workers must mind the vapors, and the flash point is low enough that storage and transfer protocols need tight control. The odor threshold starts at parts per billion – reminders that even a leak the size of a pinhole will not go unnoticed.
Technical Specifications & Labeling
Chemical suppliers list 1-pentanethiol with the CAS number 110-66-7 and often check for a purity of 98% or greater. Impurities matter, especially sulfur-containing byproducts, as they can affect downstream reactions or finished products. Containers come marked for flammability and toxicity, usually fitted with secure, sealed closures to contain leaks and minimize odor in warehouses or transit. Labeling on drums and bottles must include signal words like “Danger” and hazard symbols to warn of corrosive, toxic, and environmental hazards. On a chemical registry, alternative names feature prominently to avoid mix-ups.
Preparation Method
The classic route of making 1-pentanethiol involves reacting 1-chloropentane or 1-bromopentane with sodium hydrosulfide or sodium thiolate under anhydrous conditions. This method, called nucleophilic substitution, swaps out the halide for a thiolate group. Controlling moisture and temperature throughout the process keeps yields high and byproducts low. Some newer approaches utilize phase-transfer catalysis or continuous flow setups, which make scaling up safer and more efficient. Raw materials remain affordable, and process design often balances purity demands with economic feasibility – a real concern for those dealing with commercial-scale production.
Chemical Reactions & Modifications
Chemists appreciate 1-pentanethiol’s ability to form thioethers by reacting with alkyl halides, a staple in constructing more complex organic molecules. The thiol group oxidizes easily, producing disulfides, which pop up frequently in flavor chemistry and biology. In the right environment, under strong bases, deprotonation allows for nucleophilic substitutions or additions, broadening its value as an intermediate. Certain transition metals can activate the C–S bond, opening up possibilities for coupling reactions crucial in fine chemical synthesis. For folks interested in materials science, introducing the thiol onto polymers or surfaces imparts adhesion, chemical functionality, and reactivity, which feeds research into coatings and self-assembled monolayers.
Synonyms & Product Names
You’ll see 1-pentanethiol marketed and referenced under several names: n-pentanethiol, normal-pentanethiol, n-amyl mercaptan, amyl mercaptan, and pentyl mercaptan all trace back to the same compound. International chemical directories keep these synonyms side-by-side, essential for regulatory filings, shipping documents, and laboratory inventories. The term “mercaptan” appears in flavor, fragrance, and oil and gas industries, while “thiol” sticks with the more academic and pharmaceutical crowd.
Safety & Operational Standards
Anyone who’s spilled even a fraction of a milliliter can vouch: 1-pentanethiol’s odor overpowers a workspace and lingers in clothing, skin, and ventilation for days. Inhalation at low concentrations causes headaches, nausea, and respiratory discomfort. At higher levels, central nervous system effects can’t be ignored, so effective local exhaust ventilation and upgraded personal protective equipment matter more than with many simple solvents. Handling spills requires containment – neutralizing with bleach or hydrogen peroxide, absorbing onto inert material, and ventilating the area. For shipping, 1-pentanethiol counts as a hazardous material, demanding UN identification and compliance with regulations set by agencies like OSHA, REACH, and the Department of Transportation. Chemical safety data sheets spell out fire, toxicity, and environmental risks, along with practical measures to minimize harm.
Application Area
1-Pentanethiol’s uses cross boundaries: it pops up in the flavor and fragrance industries because natural and synthetic food aromas demand authentic sulfur notes, even at levels barely detectable. In petroleum refining, this compound modifies lubricant properties by attaching to metal surfaces, cutting down friction and wear. It also serves as a sulfur donor in making pesticides and fungicides, helping plants fend off disease. For the lab-trained, 1-pentanethiol offers a go-to reagent for introducing a thiol group into bioactive molecules or for building blocks in custom syntheses. Surface chemists leverage its affinity for metals, building self-assembled monolayers that tweak surface properties on anything from microchips to biosensors, an area growing rapidly with the miniaturization push in technology.
Research & Development
The toolbox for manipulating sulfur compounds keeps growing, and 1-pentanethiol lands in the middle of ongoing research into reaction mechanisms and selective transformations. Academic groups study its reactivity with transition metals, aiming to discover methods for site-selective C–S bond activation – a step toward building more complex molecules with efficiency. Material science pushes the boundaries by tethering thiols onto gold or silver nanoparticles, opening up fields in biosensing and nanomedicine. Changing the way thiols interact with catalysts could transform how drugs, agrochemicals, or specialty chemicals are built. Every couple of years, a new synthesis or application emerges, giving new life to an otherwise infamous molecule.
Toxicity Research
Controlled exposure studies show that while 1-pentanethiol won’t kill you at air concentrations typical in well-ventilated labs, chronic exposure above safety thresholds gives rise to headaches, dizziness, kidney, and liver stress. Animal tests point to central nervous system depression, weight loss, and, with enough sustained exposure, organ toxicity. Eye and skin irritation rank high as reasons for prompt decontamination. Environmental releases matter because persistence and bioaccumulation create risks for aquatic life, pushing national authorities to set strict emission limits. Risk assessments argue for tighter engineering controls and regular air monitoring. I’ve seen companies turn to third-party consultants for workplace testing, keeping everyone both safe and compliant with newer, more cautious guidelines.
Future Prospects
Cleaner and more efficient manufacturing routes will likely change how 1-pentanethiol gets to market, as green chemistry principles start to drive investment and research. Interest grows around renewable feedstocks and processes that decrease hazardous byproducts. In applications, look for expansion in electronics, as new sensors and microchips need surface modifications tied to thiol chemistry. Biotechnology stands ready for breakthroughs as functionalized surfaces and novel reagents let scientists fine-tune cell behavior or develop sharper diagnostics. With tightening environmental and occupational health regulations, innovation in detection, containment, and odor control won’t lag. Those keeping pace with both the regulatory horizon and technical breakthroughs are covering their bases for years to come.
What is 1-Pentanethiol used for?
The Smell Behind the Science
Step into any chemical laboratory where sulfur compounds get daily attention, and the distinct aroma of 1-Pentanethiol might be the first thing to hit your senses. It brings a sharp, unpleasant scent that no one in their right mind could ever forget. I remember my first week mixing organic molecules in grad school—the day I uncapped a bottle and learned that “smelly” sometimes means “effective.” The stink is a sign of a strong thiol group, a chemical handle that makes this molecule indispensable in several industries.
What 1-Pentanethiol Actually Does
Looking past the odor, 1-Pentanethiol works as a building block. Many specialty chemicals start their journey with simple thiols. Manufacturers put it to work making additives for lubricants, plastics, and rubber. Why? The sulfhydryl group on 1-Pentanethiol grabs hold of metals, especially when products need improved durability or more resistance to wear. Think about how rubbers in car tires or conveyor belts last a little longer; that’s sometimes thanks to this compound creating stronger bonds at the molecular level.
Chemists often rely on 1-Pentanethiol as an intermediate. It goes into synthesizing more complex molecules, including some pharmaceuticals. In the lab, it’s a go-to for protection reactions. This lets chemists temporarily hide reactive groups so they can tweak other parts of a molecule without unwanted side reactions. This single feature allows drug designers to make life-saving medicines with fewer steps and higher purity.
Fuel Additives and Odorants
Oil and gas professionals have a long history with 1-Pentanethiol because it acts as a stench marker, especially in natural gas, where leaks could be deadly. That sharp odor may set off alarm bells, but it’s what saves lives. The process isn’t complicated; adding a small amount lets detection reach even the faintest whiff before major trouble lands. It still strikes me as remarkable that the simplest warning system—our nose—remains one of the safest ways to protect entire communities.
For fuel, 1-Pentanethiol sometimes gets mixed in to prevent engine knocking. The sulfur atom can react with certain components in gasoline, helping engines run more smoothly. Keeping engines in top shape isn’t just about fewer breakdowns; it cuts down on wasted fuel and lowers emissions. As we shift to cleaner alternatives, getting the chemistry right in older fuel technologies still matters.
Risks and Looking for Safer Alternatives
Working with 1-Pentanethiol comes with real safety issues. The compound quickly irritates eyes and skin, and inhaling the vapor’s never recommended. When I worked in the lab, gloves and plenty of fresh air kept things bearable. Yet scaling up to factory-sized processes turns small risks into major hazards. Safety guidelines and regular training make a big difference, but so does searching for safer substitutes wherever possible.
Green chemistry researchers keep pushing for new thiol alternatives—compounds that can offer the same chemical benefits without the same health or environmental cost. The future may see more widespread use of bio-based thiols, or innovative ways to engineer protective equipment and safer handling processes, especially as regulations get tighter around hazardous chemicals.
Why It Still Matters
One whiff of 1-Pentanethiol stays with you, but so does its impact across different sectors. From making products last longer, to making workplaces safer, to leaving room for innovation in green chemistry, the story of this pungent compound stretches much farther than the nose would suggest. Anyone working in chemistry, manufacturing, or environmental health eventually learns to respect it—not just for what it is, but for what it makes possible.
What are the safety precautions when handling 1-Pentanethiol?
Why 1-Pentanethiol Demands Careful Handling
If you’ve worked with chemicals in a lab or industry, you know some substances make their presence known just by smell. 1-Pentanethiol lands right in that category with an odor that gets your attention fast. It isn’t just the scent that’s a concern. The compound’s reactivity, flammability, and potential for harm to skin, eyes, airways, and the environment give good reasons to step up protective habits.
Direct Exposure: Clothing and Skin
Handling 1-Pentanethiol without protection invites trouble. Liquid contact on skin leads to irritation and gives the compound a pathway into the bloodstream. Gloves built for chemical resistance, such as nitrile or neoprene, block most of that risk. Safety goggles that wrap around the eye, not just open safety glasses, prevent splashes from reaching the eye. A simple coat or apron, worn properly and consistently, stops accidental spills from soaking into your clothes.
Breathing Risks: Smell Isn’t the Only Warning
You can smell 1-Pentanethiol before it hurts you, but relying just on odor misses the mark on safety. Vapors get absorbed rapidly by the lungs and cause symptoms like headaches or nausea, and the process can happen quickly in small, poorly ventilated spaces. Reading the safety data sheet, you’ll find the need for proper fume hoods or local exhaust systems. Respirators make sense during spills or when the process means fumes linger in the workspace.
Fire and Spill Hazards: Not Just an Irritant
This compound flashes at low temperatures and burns with a risk for toxic gases. Open flames, hot surfaces, or static electricity near work areas turn routine handling into an emergency. Fire blankets, foam extinguishers, and a clear walk path bring down the danger. Practicing spill drills—not just reading about them—teaches muscle memory, so quick reactions come naturally when needed. Immediate cleanup using absorbent materials, followed by ventilating the space, gives the best shot at reducing damage.
Environmental Impact: Drain and Disposal Mistakes
Letting any volume of 1-Pentanethiol slip into the drain or soil can throw off local water and land ecosystems. The compound sticks around in waterways, affecting aquatic creatures before breaking down. Container labeling, strict inventory tracking, and sticking to hazardous waste disposal services keep these substances out of the environment. From personal experience, local regulations will hammer you with fines if you play loose with chemical waste.
Preparation and Training
People skip safety steps under pressure or out of habit. Routine training patches those risky gaps. Reviewing emergency response plans and recognizing the scent and appearance of the chemical can make a difference during a real incident. Keeping safety showers, eye-wash stations, and emergency numbers within sight beats hunting for help when time’s already ticking.
Leadership and Culture Matter
Culture around chemical safety starts from the top. In labs and small companies I’ve worked for, supervisors who model careful handling and never cut corners get buy-in from newer staff. Nobody wants to be the weak link. Clear expectations, real stories about slipups, and recognizing people who take extra steps encourage everyone to keep high standards.
What is the molecular formula and structure of 1-Pentanethiol?
Digging Into the Details
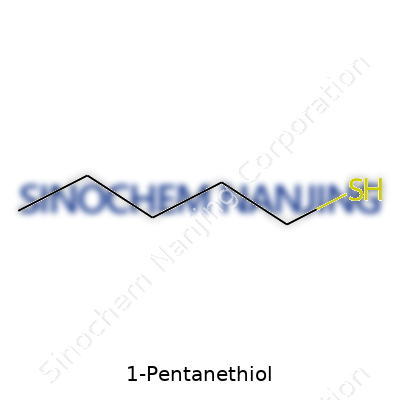
1-Pentanethiol stands out because it mixes the familiar lineup of carbon and hydrogen atoms with a sulfur atom, creating a chemical that’s more than just its name. You get the formula C5H12S. The structure resembles a simple chain of five carbon atoms hooked together, stretching from one end to the other, topped off with a thiol group (–SH) at the very tip.
Plenty of students and professionals, from biologists to chemical engineers, bump into chemicals like this day to day. If you ever notice a strong, often unpleasant odor coming from a lab bottle, chances are you’re close to a thiol. That sulfur atom doesn’t just tweak the smell, though—it plays a big role in how the molecule reacts and binds with other substances. Nature uses this kind of chemistry in everything from garlic’s flavor to key metabolites in your body. I remember my own early days with organic chemistry, battling with the sharp scent and sticky feel of thiols. Not fun, but definitely memorable, and proof that even simple molecules have a big presence.
How 1-Pentanethiol Is Built
The carbon backbone in 1-Pentanethiol links together in a straight line—no fancy loops or branches for this molecule. Each carbon, except for the last one, clings to two or three hydrogen atoms. The fifth carbon ties up to a sulfur atom, which then grabs a lone hydrogen itself. If you were to draw it out, the structure reads like this: CH3-CH2-CH2-CH2-CH2SH.
That “SH” group is what gives it both its function and reputation. Sulfur packs a punch, making 1-Pentanethiol reactive with metals and powerful enough to change the course of certain chemical processes. This explains why it often ends up in labs focused on organic synthesis, chemical manufacturing, or tests that need precision in measuring sulfur content. Anthracnose, a plant disease, can be checked with sulfur-containing compounds. I’ve seen researchers rely on these molecules to calibrate analytical machines or scramble the results of enzyme reactions, all thanks to this single atom swap from oxygen (in a typical alcohol) to sulfur.
Why the Structure Matters
Understanding structure lets chemists predict and control reactions. Replace the –SH group with an –OH, and you have 1-pentanol, an alcohol with tamer behavior and applications in flavoring and solvents. Switch it out for an –NH2, and you step into amine chemistry, taking you into pharmaceuticals and polymer science. The tiniest change in molecular structure shifts the role a molecule can play.
1-Pentanethiol isn’t a blockbuster chemical for everyday consumer goods. Instead, it steps up in select industries—especially where sulfur chemistry matters. Petrochemical outfits use thiols in trace analysis, geochemists rely on them as markers, and some advanced materials science teams harness their reactivity to bind metals or tweak surfaces. Remembering real-world handling, proper ventilation and careful storage stay critical. Even a few drops can fill a lab with a heavy scent, underscoring the importance of safety, not just curiosity.
Looking to the Future
As research pushes forward, 1-Pentanethiol and similar compounds may find fresh roles in biotechnology and nanomaterials. With the rise of custom molecule building, the basic carbon-sulfur backbone might turn up in sensors, environmental testers, or new types of adhesives. The lesson here: keep your eye on the details—structure, smell, and reactivity all matter. Small chemicals can drive big changes out in the real world.
How should 1-Pentanethiol be stored?
Why Proper Storage Really Matters
Anyone who’s spent time in a chemistry lab has likely come across materials with strong odors and a knack for causing trouble if left in the wrong place. 1-Pentanethiol falls right into that group. This compound, with its intense, skunky smell and volatile nature, has a reputation that lingers long after the bottle leaves the shelf. Handling it with casual carelessness turns an ordinary day into a headache, or worse, a safety incident.
The Troubles With 1-Pentanethiol
Every chemical user learns quickly: some molecules like fresh air, some don’t. 1-Pentanethiol hates heat and sunlight. Warmth means more vapors, and those vapors are not just a nuisance—they’re flammable. One spark in the wrong spot lays out the perfect recipe for a fire. That’s why temperature and ventilation rank high on the list of storage priorities. Keep this stuff above room temperature for long, and you’re rolling the dice on your safety and the safety of your colleagues.
Odor travels fast with thiols. One misplaced bottle at the back of a warm cupboard sends that stench straight across the hall, turning a busy work space into a place no one wants to be. It’s not just about comfort, though. Lingering vapors suggest exposure risks that demand real attention, not a rushed "air out the room" approach.
Key Storage Strategies Drawn From Hands-On Experience
- Temperature control: Cool, dry spaces work best. Standard practice places 1-Pentanethiol in a flammable storage cabinet, away from sunlight or heat sources. Lab refrigerators not used for food or drink handle the task well, provided spill containment sits inside the unit.
- Tight seals and robust containers: Loose lids or damaged bottles spell disaster. Always check closures before stashing the bottle. Glass or high-resistance HDPE containers keep leaks at bay while providing chemical compatibility.
- Dedicated space for thiols: Keep thiols separate from oxidizers, acids, and common bases. Mixing up storage shelves brings extra risk, with potential for violent reactions if spills occur.
- Labeling: Legible, resistant labels tell everyone what’s inside the bottle. Multi-language warnings and hazard symbols throw in an extra layer of protection for diverse teams.
- Ventilation: Even a closed bottle will leak vapor if the seal isn’t strong. Ventilated storage helps catch stray fumes before they cause headaches or worse. Labs with dedicated chemical hoods keep vapors moving away from working areas.
Addressing the Challenges Before They Hit Home
Beyond physical storage, good habits make all the difference. Fresh gloves every time, careful transfer of liquids, and checking for leaking bottles save time and avoid cleanups. In my own work, the extra minute spent double-checking a cap meant skipping a half-day’s disruption for odor control. Spills happen, even with trained staff; spill kits for organosulfur compounds hang near storage areas, so nobody scrambles to figure out next steps when an accident strikes.
Supervisors don’t have to micromanage, but they do have to set clear expectations. Regular safety walkthroughs and spot checks encourage everyone to take chemical risks seriously, not just in writing but in daily actions. After all, the payoff comes in fewer injuries, less lost material, and a workplace everyone actually wants to spend time in.
Building Better Chemical Storage Practices
Protecting people and investments starts with choosing solid storage setups and training everyone to care for hazardous chemicals. Investing up front in proper cabinets, periodic inspections, and straight talk about risks beats scrambling during an emergency. In every place I’ve worked, open communication and shared respect for safety rules turned headache-prone areas into quiet corners—no drama, no smell trailing down the hall. Keeping 1-Pentanethiol from turning into a problem takes real attention, not just a spot on the chemical shelf. If labs and classrooms keep these lessons in mind, everyone walks away healthier, safer, and a little less annoyed by that unforgettable thiol smell.
What are the typical applications of 1-Pentanethiol in industry?
What Makes 1-Pentanethiol Stand Out?
1-Pentanethiol doesn't always make the headlines, yet it's essential to a variety of work behind the scenes. Most people would rather avoid its sharp, unpleasant odor. Chemists and engineers know this compound for its sulfur atom and five-carbon backbone. It’s a strong building block if you value flexibility in synthesis or want a molecule that brings sulfur into your project.
Flavors, Fragrances, and a Little Bit of Chemistry
Take the world of flavors and fragrances. While you probably won't find 1-pentanethiol directly in your perfume, traces often show up during the development of particular aromas. Its strong scent helps chemists understand how sulfur changes a fragrance. Some rare food and drink processes also tap into these sulfury compounds to give products a signature flavor—think of how a slight hint of onion in a dish can make all the difference. Chemists only use 1-pentanethiol in minute quantities, well below safety thresholds. The key lies in navigation: balancing regulatory requirements, taste panels, and production risks.
Industrial Chemistry and Surface Science
I remember my days in a materials chemistry lab. We relied on thiols to form self-assembled monolayers (SAMs) on metal surfaces, mostly gold. 1-Pentanethiol’s unique chain length gave us just the right spacing for our sensor prototypes. These SAMs act as gatekeepers that control how sensors detect chemicals in the environment. Working with different alkanethiols, the five-carbon chain of pentanethiol offered properties we couldn’t get from shorter or longer molecules—like a balance between stability and flexibility.
This approach supports sensors used for pollution monitoring, medical diagnostics, and electronics. By tailoring surface properties, researchers enhance everything from corrosion resistance to electrical performance. The experience of actually watching these molecules line up “shoulder to shoulder” on a gold wafer brought the textbooks to life. It’s practical stuff that ends up in smarter devices.
Paints, Lubricants, and Polymer Additives
Manufacturers in coatings add 1-pentanethiol to their blends. Its sulfur atom improves adhesion, especially where metal meets paint. In the machining world, this molecule slips into lubricants as an additive, helping reduce wear by forming a film over hot metal surfaces. Some synthetic rubber productions lean on it as a cross-linking agent, locking chains together just enough to add toughness and bounce.
This isn’t theoretical. Breaking a part in the field because a lubricant failed costs real time and money. The extra layer of chemistry from 1-pentanethiol often prevents those breakdowns. Its molecular signature changes the performance on a real-world shop floor.
Safety Always Matters
Working with 1-pentanethiol means thinking about safety too. Its vapors shouldn’t linger in a closed room, and skin contact leads to irritation. Lab techs and plant workers gear up with gloves and proper ventilation. Anyone handling these chemicals needs training and respect for the risk—this isn’t something to take lightly.
Finding Solutions and Future Uses
Practical challenges keep evolving. Greener chemistry pushes the search for less hazardous alternatives, but 1-pentanethiol isn’t going away soon. Its strong reactivity and reliability underpin modern manufacturing, research, and product design. Better ways of handling waste, recycling residues, and developing less pungent options could take some pressure off safety teams and improve working conditions. For now, the many hats 1-pentanethiol wears in industry make it a compound worth respecting—and understanding beyond its smell.
| Names | |
| Preferred IUPAC name | pentane-1-thiol |
| Other names |
n-Pentyl mercaptan 1-Mercaptopentane Pentane-1-thiol |
| Pronunciation | /paɪˈtæn.ə.θaɪ.ɒl/ |
| Identifiers | |
| CAS Number | 110-66-7 |
| Beilstein Reference | 1216944 |
| ChEBI | CHEBI:35783 |
| ChEMBL | CHEMBL16363 |
| ChemSpider | 12269 |
| DrugBank | DB14004 |
| ECHA InfoCard | 100.004.894 |
| EC Number | 211-548-4 |
| Gmelin Reference | 72654 |
| KEGG | C02573 |
| MeSH | D010414 |
| PubChem CID | 8057 |
| RTECS number | SK8400000 |
| UNII | Y45U55H37M |
| UN number | UN2347 |
| Properties | |
| Chemical formula | C5H12S |
| Molar mass | 104.21 g/mol |
| Appearance | Colorless liquid |
| Odor | Disagreeable, garlic-like |
| Density | 0.832 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 2.80 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 10.6 |
| Basicity (pKb) | SB: 3.56 |
| Magnetic susceptibility (χ) | -74.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.439 |
| Viscosity | 1.14 mPa·s (20 °C) |
| Dipole moment | 1.516 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 205.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -117.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3547.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H314, H331, H400 |
| Precautionary statements | P210, P261, P273, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P330, P362, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-0-W |
| Flash point | 51 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | 1-1.8% |
| Lethal dose or concentration | LD50 oral rat 1320 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 132 mg/kg |
| NIOSH | SN 4550000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Pentanethiol: "1 ppm (skin), 8-hour TWA (OSHA) |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Methanethiol Ethanethiol Propanethiol Butanethiol Hexanethiol Octanethiol 1-Pentanol |

