An Editorial Look at 1-Naphthylthiourea: Yesterday, Today & Tomorrow
Historical Development
Stories about chemicals tend to twist through decades. 1-Naphthylthiourea came out of the early twentieth-century boom in synthetic organic chemistry, part of a wave that brought with it both new medicines and novel industrial challenges. Researchers keen on pest control discovered its rodenticidal properties in the 1940s. For a while, it played an outsized role on farms and in cities seeking to curb rat infestations. Over the years, as the downsides of acute toxicity showed up in laboratory animals and unlucky humans, regulators and chemists reevaluated its use. By then, stories from industrial plants and universities around the world had already cemented its place in chemical history, showing just how quickly understanding evolves about what seems like a promising chemical.
Product Overview
1-Naphthylthiourea stands out among thioureas because its molecular structure—anchored by a naphthalene ring—brings a distinctive set of behaviors. Folks who work in chemical analysis often point to its use as a reference compound in research and education, a nod to how frequently it pops up when teaching about aromatic substitutions and nucleophilic reactions. Through the years, versions of the compound have seen use not just in rodenticides, but in analytical chemistry, paint manufacturing, and sometimes as a laboratory reagent.
Physical & Chemical Properties
Its appearance usually reminds seasoned chemists of fine white powder or sometimes pale crystals. The melting point is relatively high compared to simpler thioureas, tipping over 140°C, which speaks to the stability of the aromatic backbone. In water, it refuses to dissolve much. Its real solubility comes out in organic solvents like ethanol and acetone, making it a useful option for organic synthesis. That naphthalene group brings heightened chemical stability but also sustains certain reaction vulnerabilities—especially to strong acids and oxidizers, which can split the thiourea group and break the molecule down to simpler fragments. One odd thing: its odor is usually faint, not as assertive as many sulfur-organic molecules, but after a few years in the lab, anyone can spot the whiff of naphthalene.
Technical Specifications & Labeling
Across decades, suppliers label 1-Naphthylthiourea by its CAS number (86-88-4) and its chemical formula (C11H9N2S). Purity grades run the gamut from technical to analytical, with specialized uses requiring precise quantification of impurities, especially heavy metals and isomeric naphthyl compounds. Labeling must reflect its toxicity and environmental risks. On my own shelves, the containers wear prominent hazard warnings: harmful if swallowed, suspected carcinogen, and dangerous to aquatic life. Being up front with these warnings is not just about regulatory compliance—it’s about keeping colleagues safe in the real world, not just in paperwork.
Preparation Method
The most common laboratory approach begins with 1-naphthylamine, a compound once manufactured in volumes for dyes. The synthesis runs through a reaction with ammonium thiocyanate in an acidic aqueous medium. That classic method set the standard for a generation of undergraduate chemists; the reaction delivers a decent yield, and the product can be purified by recrystallization. Industrial manufacturers tweak the scale, sometimes streamlining purification with solvent extraction or continuous crystallization. There’s satisfaction in watching crude crystals dissolve and reform, signaling the steady progress from ambiguous slurry to pure product. Other nitrogen sources and sulfur donors have been explored, but the old method, simple and reliable, seems to hold its ground in most labs.
Chemical Reactions & Modifications
1-Naphthylthiourea takes on a few notable transformations. Under gentle heating with acids, the thiourea motif can hydrolyze, giving off hydrogen sulfide—a hazardous byproduct that requires careful ventilation. Oxidizing agents sharpen the picture: the molecule cleaves, and the sulfur atom oxidizes to sulfate or other forms, while other fragments behave unpredictably, sometimes forming naphthoquinones. Chemists with an eye for molecular construction tweak the naphthalene ring or alter the thiourea, aiming for new derivatives to study biological or electronic properties. The sturdiness of its aromatic framework, paired with the reactivity of its thiourea group, offers a springboard for making related compounds with medicinal or technical promise.
Synonyms & Product Names
The name 1-Naphthylthiourea usually says it all, but other labels turn up: 1-naphthalenylthiourea, Anturat, ANT, and sometimes the acronym NTU. Chemical databases and textbooks sometimes get lost in translation, with minor spelling variants or prefix swaps, but any researcher familiar with aromatic thioureas knows what’s in the bottle. With such a storied chemical, the synonyms trace decades of academic and industrial language, adding to the archive of chemical knowledge.
Safety & Operational Standards
Lab life teaches that handling 1-Naphthylthiourea brings real hazard. Absorption isn’t limited to ingestion—contact with skin or inhalation poses serious risks. Most protocols call for gloves, goggles, and fume hoods, and those rules save lives: stories of laboratory accidents underline the consequences of complacency. Workplace exposure limits stem from animal studies revealing hematologic and renal effects. Waste shouldn’t hit regular drains or landfills, because aquatic toxicity can echo through ecosystems. Proper training matters here, not just a nod to procedure; folks who take shortcuts risk more than their own health. The legacy of this compound, as with many synthetic chemicals, stands as a reminder to treat even familiar powders with real respect.
Application Area
Its role as a rodenticide stands out most famously. Users once sprinkled it across farms, warehouses, and city infrastructure. With toxicity showing up in non-target animals (even pets and livestock), its use started shrinking. In modern labs, it plays a quieter part as a reference standard in analytical chemistry and in some specialized synthetic reactions. Some manufacturers employ it in pigment and dye research, owing to the naphthalene backbone’s chromatic potential. Occasionally, medical researchers probe its biological properties, looking past its flagged toxicity to see where its structure might inform new treatments or study biochemical pathways. These days, regulations keep the reins tight, driving many users to alternatives.
Research & Development
Decades ago, research interests focused on its promise as a specialized rodenticide that, in theory, reduced resistance in rat populations. Today’s scientific journals tell a different story. Environmental scientists map its persistence in soil and water, using modeling and analytical chemistry to trace degradation. Synthetic chemists revisit the molecule, searching for analogs with lower toxicity and more favorable safety profiles. Medical researchers, recognizing the molecular similarity to other bioactive thioureas, investigate derivatives for anticancer, antiviral, or antifungal activity—always mindful of risk. Progress feels slow but steady, shaped by pressure for safer, more targeted solutions that meet modern health and environmental standards.
Toxicity Research
If you leaf through toxicology textbooks from the past fifty years, 1-Naphthylthiourea earns many pages. Early rodent studies flagged kidney damage, methemoglobinemia, and other blowback from acute exposure. In humans, reported poisonings led to symptoms ranging from hematuria and jaundice to more severe systemic toxicity. Regulatory agencies shifted their recommendations, limiting or banning use outright in many contexts. Modern toxicologists turn to cellular assays, proteomics, and animal models to parse out exactly how the molecule disrupts cellular function. Environmental monitoring has shown persistence and mobility in certain soil types, raising concerns about groundwater contamination and harm to aquatic life. The debate over acceptable risk continues, with most experts leaning toward minimizing exposure altogether.
Future Prospects
Future stories about 1-Naphthylthiourea may sound different from its past. Its role as a rodenticide seems likely to fade further as safer, targeted biological options come on line. Where it shines now, and will probably remain relevant, is in research—helping illustrate reaction mechanisms, enabling synthetic modifications, and serving as a marker in analytical chemistry. Its structure serves as a starting point for chemists working at the boundaries of medicinal and materials discovery. To my mind, its value comes in context: as an object lesson about balancing innovation against risk, and as a concrete example showing how science revises itself over time. My bet is that future research will keep pulling it out as a control or a caution, letting it live on more as a tool than a direct commercial product.
What is 1-Naphthylthiourea used for?
Tracking Rats, Protecting Public Health
Few folks outside pest control circles ever hear about 1-Naphthylthiourea. On paper, it looks like just another tongue-twister in the line-up of chemicals, but its presence signals a serious push against rodents. City workers, researchers, and even some rural communities rely on it as a proven tool to measure rat populations. It’s not about making a place rodent-free overnight—it’s about learning how big the problem runs before jumping to quick fixes.
This chemical works as a rodenticide, and it does the job quietly compared to the noise of traps or the hassle of constant monitoring. Professionals use bait with the compound in specific sites. Rats that ingest enough of it die quickly, giving crews a surprisingly clear marker of how many are living underfoot. For big cities, knowing where rats hide tells department heads how to plan sanitation campaigns and where to put real money on prevention. It shapes budgets. It’s tough to argue with the cold facts 1-Naphthylthiourea gathers.
Why the Stakes Run High
Rodent-borne diseases never really leave the headlines for long. Leptospirosis, hantavirus, and salmonella—these turn up in neighborhoods where rats share surfaces with kids and kitchen pantries. In places with stretched resources, disease tracking can mean the difference between an outbreak and a quiet year. 1-Naphthylthiourea plays a part in that protection net.
People deserve to know what chemicals slip into their surroundings. The history of 1-Naphthylthiourea isn’t spotless. The compound can harm other animals, including pets and even some wild species that matter to the balance of local ecosystems. In humans, direct exposure spells trouble—scientific studies point to toxicity risks if someone eats or handles it without gloves or masks. The World Health Organization and the U.S. Environmental Protection Agency keep close tabs on how and where it’s used. Safety rules aren’t decoration—they’re the backbone of any public health push.
Balancing Safety and Results
Settling the rat fight with 1-Naphthylthiourea takes careful work. Companies train crews well. They stick to marked locations. Neighborhood councils ask the obvious questions: can dead rats get scooped up before stray cats or birds eat them? Do residents know about application dates?
Better solutions come when people talk honestly about risks and rewards. Communities in Europe experiment with integrated pest management, cutting baiting with broad environmental shifts—like fixing trash collection and sealing up cracks where rats invade. In American cities, agencies publish data on application times and locations so folks stay in the loop. Parents and pet owners worry less when information is public instead of buried in policy papers.
Research never stops. Newer, less hazardous rodenticides push forward as scientists dig for answers. Some folks swear by plain traps and better cleaning; others look to biotech for a future with rodent birth control. But for pockets of the world, 1-Naphthylthiourea holds its ground as a critical checkpoint. Admitting both its usefulness and its risk—without hype or hand-waving—sets a fair table for debate. That’s the only way real progress sticks.
Is 1-Naphthylthiourea toxic to humans or animals?
Looking at Toxicity: More Than a Lab Story
A lot of folks outside research labs might never run into 1-Naphthylthiourea—or as chemists shorthand it, ANTU—but for those in pest control, animal research, or toxicology, it’s a name with baggage. This compound found early fame as a rodenticide. It seemed like a neat fix for rat control, yet things look different once you know where and how this chemical lands in the real world.
People sometimes treat chemicals as magical tools—dose, and problem solved. ANTU’s story pushes back on that thinking. Its quick action against rats isn’t its only legacy; the biggest mark lies in what it does to other living creatures. The compound can cause lung damage in rats, even at surprisingly low doses. Most chilling, this doesn’t just stop with rats. Cats, dogs, and some wild animals can get caught in the fallout. Fatal lung swelling and fluid build-up show up soon after exposure. From personal experience handling pest control cases, I’ve met pet owners who lost beloved animals from indirect contact. Heartbreak follows when families deal with unintended poisonings.
Impacts on People: Not Just a Distant Danger
Some folks might shrug and say people don’t eat rat bait on purpose, so what’s the worry? Still, ANTU can harm anyone who handles it carelessly. The chemical can enter the body through inhalation, swallowed dust, or even skin contact. Even small lapses—dust on a glove, powder on a doorknob—can matter. Major health risks kick in after ingestion, with symptoms like headache, vomiting, body aches, and, at high doses, serious lung trouble. The science draws a clear line: this stuff isn’t just tough on rats. It poses a danger to humans, especially kids and workers who lack safety resources or training.
The Data: Science Points to Caution
Plenty of studies back up these real-life stories. The U.S. Environmental Protection Agency and international agencies recognize ANTU as hazardous. Rats exposed to even a tiny amount often die from severe pulmonary edema. Lab tests on other animals—mice, fish, birds—show toxic effects too. There’s evidence it can pass into soil and water, threatening local wildlife. Human toxicity reports aren’t as frequent, but cases pop up wherever safety slips.
Why We Need Better Choices and Safer Habits
The world doesn’t need to rely on dangerous old pesticides. Companies can produce rodent control options that don’t bring the same risks for pets, people, or wildlife. For anyone handling chemicals, safe storage and clear labels matter more than flashy claims about effectiveness. Wearing gloves, using proper containment, and never mixing chemicals with food or drink reduces harm. Community education can save lives; folks need real talk, not jargon.
Places that ban ANTU or limit its availability don’t just do it for regulations’ sake. They act on public health data and hard lessons learned over decades. As someone who has worked with both pet owners and public health educators, I see firsthand how much relief comes from knowing homes, parks, and playgrounds are free from these hidden dangers. The discussion around ANTU reminds us to bring both caution and compassion into choices about chemical use.
What are the storage recommendations for 1-Naphthylthiourea?
Protecting People and Preserving Power
Storing chemicals like 1-Naphthylthiourea, a potent rodenticide, calls for steady respect and attention to detail. Nearly every lab worker, researcher, or pest-control technician has witnessed what improper storage can do during those yearly cleanouts. Open bags, faded labels, and powder leaking onto shelves—none of that says “safe,” especially for a compound recognized as hazardous by OSHA and listed as an acute toxin by the CDC.
Keep It Cool, Dry, and Dark
Heat and moisture cause headaches, not just for people, but for chemicals, too. Stash 1-Naphthylthiourea in a spot that keeps both out. High temperature can speed up decomposition or sometimes spark slow chemical reactions. Dampness and spills, on the other hand, lead to clumping, possible hydrolytic breakdown, and maybe even release of fumes. Pick a solidly-built, non-metal locker with a steady room temperature. A locked chemical cabinet or climate-controlled storeroom prevents tampering and keeps conditions reliable, which lines up with the good laboratory and industrial hygiene practices called for in federal guidelines.
Original Packaging Is More Than Just Branding
Manufacturers provide containers designed to limit exposure to air, light, and physical contact. Transferring the powder to unmarked or makeshift containers leaves everyone guessing, especially if the new container reacts with the compound or allows vapors to escape. Stick with the supplied bottle, tube, or drum. Keep the label visible and easy to read, so everyone—seasoned staff or newcomers—knows what’s inside. If a label starts to fade, print a new one instead of risking misidentification, mixing, or accidental exposure.
Segregation: Not Everything Plays Well Together
1-Naphthylthiourea belongs far from acids, oxidizers, and reducing agents. Simple shelving mistakes—stacking incompatible chemicals together—have caused many an accident. Some substances, if leaking or combined by accident, can cause fires or noxious gases. An old habit from veteran chemists serves well: group chemicals by hazard class, then double-check with the SDS or chemical compatibility chart on the wall. Store this compound in a dry container, on a separate shelf dedicated to toxic substances, away from food, drink, and flammables.
Personal Protection Starts in the Storage Area
An ideal setup includes gloves and goggles within reach, and a sink or eyewash station nearby in case of an emergency. Spills can occur even with careful handling. Good ventilation removes any lingering dust or vapors, which is crucial for any chemical labeled toxic by inhalation or skin absorption. Regular training on these measures helps, especially since workplace accidents often follow complacency, not outright carelessness.
Inventory and Inspection: Small Habits, Big Impact
No one remembers every expiry date or inventory count, especially with a compound like 1-Naphthylthiourea that can quietly degrade or become unstable over long periods. Periodic checks identify compromised containers and discourage stockpiling beyond practical use. Records should note arrival dates and last-inspected dates, satisfying regulatory and safety requirements.
Safer Storage, Healthier Outcomes
All this effort reflects hard-won lessons in chemistry and health protection. Mistakes in chemical management don’t just put researchers or technicians at risk – they can spark regulatory trouble and community concerns. Treating 1-Naphthylthiourea with the respect outlined above keeps workplaces, neighborhoods, and the environment safer, turning standard safety advice into smarter daily practice.
How should 1-Naphthylthiourea be handled safely in the laboratory?
Understanding the Risks
Working with 1-Naphthylthiourea stirs up some real concern for safety among lab folks, and for good reason. This compound has a reputation for being toxic, especially to kidneys, and it doesn’t play nice if inhaled, ingested, or touched. I remember my first introduction to this chemical during graduate studies—before touching a gram, we huddled around a dusty Material Safety Data Sheet and listened to our mentor explain how carelessness lands researchers in the emergency room.
Personal Responsibility and PPE
A careless slip could send this chemical onto skin, clothes, or even a benchmate. I always reach for nitrile gloves, a full-length lab coat, and chemical splash goggles before popping open the bottle. Forgetting any of those makes handling it feel like driving without a seatbelt—unnecessary risk. Those of us with long hair tie it up tight; you never know what could dangle into a beaker.
Fume hoods have saved more than their share of lungs. Usually, I handle 1-Naphthylthiourea under proper ventilation, no exceptions. It makes a huge difference in keeping those fine particles or vapor from wandering into the air we breathe. Fume hoods also give a psychological line—you respect the space, keep distractions low, and always keep your hands steady.
Respecting the Label: Storage and Containment
Before adding even a pinch to a flask, I double-check the label—no faded writing, no mystery containers. This habit prevents mixups and protects everyone else who might grab the bottle later. Storing it away from incompatible substances, in tightly sealed containers, helps the entire department avoid disaster. I’ve cleaned up spills before, and a leak can lead to an expensive call to hazardous response teams.
Keeping Hands Clean and Spills Smaller
Contamination creeps up in surprising ways. Routinely, I wash hands with soap immediately after handling, not just at the end of a session. Once I learned traces of the compound can stick around even after glove removal, my faith in quick rinses vanished. Mop up spills right away—never delay, even if you’re juggling three tasks or running late for class.
I run through spill response steps in my head like a pilot with a checklist: evacuate if it’s airborne, grab the spill kit, protect skin with gloves, keep absorbent material ready, and dispose of everything in the proper waste container. Practicing these habits, as drilled into me by a tough but kind lab manager, makes reactions automatic.
Training Makes the Difference
Nothing beats training from someone who sees lab safety as more than a box to check. In every lab, someone sets the tone by modeling best practices, sharing stories of what went wrong, and explaining the why behind rules. I still remember seeing posters in the hallway showing the outcomes of chemical exposure—real scars, not theoretical risks.
Solutions Beyond Rules
Safety doesn’t improve by rulebook alone. Management can hire safety officers or encourage peer-to-peer check-ins before starting dangerous procedures. Regular safety drills and honest debriefs after close calls create a culture that values vigilance. Easy-to-read sheets and reminders posted in the lab nudge even veterans to double-check habits. Bringing safety gear closer to benches—extra gloves, goggles, chemical showers—cuts excuses. These steps show the system cares about every person, not just regulations.
What is the chemical structure or formula of 1-Naphthylthiourea?
Understanding 1-Naphthylthiourea at the Molecular Level
Looking at 1-Naphthylthiourea, you find a molecule that links a naphthalene ring system, which feels familiar to anyone who’s spent time with aromatic hydrocarbons, with a classic thiourea group. The naphthalene part looks like two fused benzene rings—think back to the thick, inky smell of mothballs or some old-fashioned soaps, which sometimes use related compounds. The thiourea side brings sulfur and nitrogen into play. The structure, in shorthand, is C10H7–NH–CS–NH2. Its full chemical formula reads C11H10N2S.
That molecule carries a naphthyl group at the 1-position attached to a thiourea moiety. In two-dimensional terms, there’s a naphthalene ring, where its first carbon hooks up with an NH group, which then connects to a central carbon double-bonded to sulfur, and again single-bonded to another NH2. Try sketching it—start with the double-ring, then work your way over. Suddenly, you see why this molecule grabs lab workers’ attention.
Why This Structure Matters
Thinking about real-world stakes, chemical structures guide everything—properties, hazards, uses. 1-Naphthylthiourea isn’t just some abstract string of atoms. Each part means something. The naphthyl brings stability, and an ability to dissolve in fats, which lets 1-Naphthylthiourea slip into biological membranes. The thiourea group, with its touch of sulfur, tweaks how it reacts with metals or enzymes. That’s why 1-Naphthylthiourea pops up in toxicology discussions, especially as a rodenticide or as a test for detecting certain metals.
The placement of the naphthyl group matters. Swap it to the 2-position and you land on another isomer, with different toxicity and reactivity. Chemists sweat these details because a small shift in structure brings huge real-world consequences. From firsthand experience in environmental labs, I’ve seen unexpected persistence of naphthalene derivatives in soil and water. Their ring structures help them dodge microbial attack. This sticks around—potentially poisoning water supplies or harming non-target species. Having the right formula isn’t trivia. It affects regulations, disposal rules, remediation tactics.
Safety and Health Implications
Working around 1-Naphthylthiourea, you quickly see health risks surface. The molecule’s structure makes it a skin and respiratory irritant. In some cases, exposure has led to acute poisoning—labs track this compound closely. Animal studies flag it for its toxicity to the kidneys and other organs. World Health Organization reports line up with what’s found in toxicology textbooks. Handling procedures—gloves, hoods, and strict inventory logs—come thanks to its particular chemical makeup.
Looking Toward Safer Solutions
As chemical safety conversations pick up, pushing for alternatives gains traction. Understanding the molecular structure guides safer design. Swapping out the naphthalene ring or the thiourea segment, chemists can look for similar function with less harm. Industry regulations lean heavier on green chemistry, urging companies to ditch persistent, hazardous molecules, including 1-Naphthylthiourea, when possible. Progress requires looking at structure and predicting outcomes—a lesson that echoes through every chemistry class and industrial accident report.
Figuring out the structure and formula of 1-Naphthylthiourea gives more than a line in a chemical catalog. It’s a roadmap for safer handling, better policy, and innovative replacement. The next time you see this name pop up, the chemical structure answers more than just a test question—it holds the story of how we interact with the compounds that shape our world.
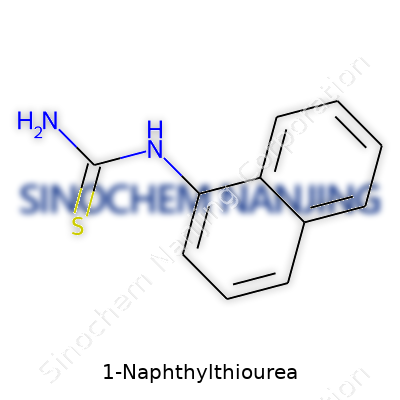
| Names | |
| Preferred IUPAC name | (naphthalen-1-yl)thiourea |
| Other names |
α-Naphthylthiourea ANTU 1-Naphthalenethiourea |
| Pronunciation | /ˈwʌnˌnæfθɪlˌθaɪ.oʊˈjʊəri.ə/ |
| Identifiers | |
| CAS Number | 86-88-4 |
| Beilstein Reference | 1209527 |
| ChEBI | CHEBI:9507 |
| ChEMBL | CHEMBL74687 |
| ChemSpider | 13260 |
| DrugBank | DB08548 |
| ECHA InfoCard | ECHA InfoCard: 100.004.561 |
| EC Number | EC 200-537-6 |
| Gmelin Reference | 82248 |
| KEGG | C14159 |
| MeSH | D009277 |
| PubChem CID | 15749 |
| RTECS number | YS8400000 |
| UNII | RU9J89S67W |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C11H10N2S |
| Molar mass | 199.28 g/mol |
| Appearance | White to pale yellow crystalline powder |
| Odor | Odorless |
| Density | 1.33 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 1.89 |
| Vapor pressure | 0.000008 hPa (25°C) |
| Acidity (pKa) | 13.08 |
| Basicity (pKb) | 7.68 |
| Magnetic susceptibility (χ) | -44.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.729 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 160.8 J/mol·K |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic if inhaled, causes damage to organs, harmful to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H332, H400 |
| Precautionary statements | P261, P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Autoignition temperature | 410°C |
| Lethal dose or concentration | LD50 (oral, rat): 53 mg/kg |
| LD50 (median dose) | LD50 (median dose) for 1-Naphthylthiourea: 40 mg/kg (oral, rat) |
| NIOSH | MW4800000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.3 mg/m3 |
| IDLH (Immediate danger) | IDLH: 5 mg/m3 |
| Related compounds | |
| Related compounds |
Thiourea Diphenylthiourea 1-Naphthylamine Naphthalene Phenylthiourea |

