1-Naphthylethylenediamine Hydrochloride: Deep Dive into an Influential Chemical
History and Development
1-Naphthylethylenediamine Hydrochloride didn’t enter the field overnight. The story starts in the early days of organic chemistry, a time when chemists stood hunched over workbenches, often guided by curiosity and a lack of sophisticated detection instruments. The naphthyl family has always intrigued researchers, its aromatic structure showing up in all sorts of reactions since the late 19th century. As research advanced through the 20th century, scientists sought intermediates to detect trace substances—think water quality testing or azo dye coupling—and the ethylene bridge linking the naphthyl ring to two amine groups caught attention. This structure, simple yet effective, made the compound valuable, especially after it proved useful in colorimetric analyses for trace nitrite detection. Widespread adoption followed, with laboratory suppliers standardizing inbound shipments, and the compound soon became a staple on reagent shelves across the globe. Today, the history behind this chemical helps frame its trusted place in labs and industry.
Product Overview
1-Naphthylethylenediamine Hydrochloride appears as a white to faintly off-white crystalline powder. It’s not a household name, but chemists recognize it as a sensitive indicator for nitrite ions. Its structure—naphthyl group tethered to an ethylenediamine backbone—enables it to react cleanly with diazonium salts, producing intense colored compounds. Suppliers package it in airtight bottles, careful to limit exposure to light and moisture. The compound appeals to lab professionals because it delivers consistent, reproducible results in colorimetric reactions. Its shelf life stays reliable so long as bottles remain tightly sealed and stored in cool, dry spaces. Researchers appreciate the subtle difference between quality grades—analytical standards top the list while technical grade products support less critical work.
Physical and Chemical Properties
Looking at this compound through the lens of practicality, you’ll find a crystalline solid with a melting point hovering around 250°C (decomposition). It dissolves readily in water, and the hydrochloride salt form improves stability compared to the free base. The chemical formula, C12H14N2·HCl, signals the extra weight from the hydrochloride addition. Room light starts to degrade the powder over long periods, and moisture can trigger slow hydrolysis, so storage conditions matter for long-term purity. The basic amine groups grant the compound moderate nucleophilicity, which accounts for its behavior in both synthetic and analytical settings. One whiff of the powder—if you risk it—gives a faint, amine-like odor, though safety protocols always recommend avoiding unnecessary exposure.
Technical Specifications and Labeling
Labeling speaks clearly nowadays. Bottles state not just the compound name but also batch number, date of manufacturing, recommended storage temperature, purity (usually over 98% for analytical products), and hazard statements. Safety pictograms warn users about toxicity and possible environmental hazards. In daily use, these bottles stay in the “reagents” section of chemistry stockrooms, each featuring a certificate of analysis documenting all measured impurities, moisture level, and method of assay. Regulations in Europe, North America, and Asia dictate labelling standards and import documentation. Careful compliance with these details matters in audits and ensures reproducibility in research and QA labs alike.
Preparation Method
Factories start synthesizing this compound by condensing 1-naphthylamine with ethylenediamine, often relying on controlled acidic conditions to prevent side reactions and maximize yield. Hydrochloric acid helps both with solubilizing starting materials and with forming the hydrochloride salt during isolation. After the main reaction finishes, technicians filter and wash the crude product before subjecting it to crystallization from water or alcohol. Final steps include drying under vacuum and sieving to produce a fine, uniform powder. Laboratories avoid exposure to open air during this phase, working quickly to prevent moisture uptake and degradation. On the bench, researchers sometimes prepare very small amounts fresh from precursors to guarantee performance for highly sensitive tests.
Chemical Reactions and Modifications
The standout reaction for 1-Naphthylethylenediamine Hydrochloride happens during diazotization. When introduced to a nitrite-containing sample in acidic medium, then treated with sulfanilic acid, a pink dye forms instantly—a visual indicator that lets even non-specialists spot tiny nitrite traces. This reaction stands at the core of the Griess assay, one of the most widely used colorimetric nitrite tests. Chemists have looked at modifying the basic structure to improve stability or shift the color response, adding functional groups or lengthening the carbon chain in quest for better sensitivity or selectivity. Despite many efforts to tweak the molecule, the original derivative keeps its place as the gold standard for routine analysis.
Synonyms and Product Names
Search through chemical catalogues and you’ll find a range of names: 2-(1-Naphthyl)ethane-1,2-diamine dihydrochloride, N-(1-Naphthyl)ethylenediamine hydrochloride, and the shorter NED. On safety documents you sometimes see C.I. 13015, especially in industrial settings where color index numbers make sense for tracking batch stocks. Labs may refer to it just as “naphthyl ethylenediamine hydrochloride,” which avoids confusion created by slightly different systematic names. Online ordering systems recognize all the variants, so finding what you need rarely causes trouble if the structural information matches.
Safety and Operational Standards
Even though seasoned chemists use this compound almost daily, they don gloves and goggles every time. It absorbs readily through skin and can trigger acute toxicity. Regulations flag it for immediate medical attention if ingested, inhaled, or if dust enters the eyes. It also lingers in water, so waste management teams must treat or incinerate contaminated liquids and solids. Material Safety Data Sheets (MSDS) require keeping antidote information and emergency procedures on hand. In practice, working with such compounds demands well-maintained fume hoods and accident-prevention training for staff. Disposal means sending all washings and paper wipes to a designated chemical waste stream; no shortcuts allowed.
Application Area
1-Naphthylethylenediamine Hydrochloride earns its keep in water quality testing. Municipal labs check nitrite contamination using colorimetric kits powered by the Griess reaction, ensuring public drinking water falls within strict safety guidelines. Food scientists lean on its sensitivity to check preservatives—sodium nitrite in meat products, for example—since regulatory bodies keep an eye on dietary nitrite exposure. Environmental monitoring programs depend on this dye response while tracking river, lake, and groundwater pollution. In the research world, labs turn to the compound for quantifying reactive nitrogen species during studies of oxidative stress, human disease, and plant metabolism. Skilled technicians value the compound because it produces quick, visible, and reliable results, sidestepping the need for high-end equipment in many standard protocols.
Research and Development
Scientists continue exploring the full capabilities of this compound. Biomedical researchers focus on adapting the assay to microfluidic chips or multiplexed sensors, aiming to cut sample sizes and automate results. Some groups combine the compound with alternative dyes, hoping for sharper color gradients or higher throughput. Ecologists want to see more robust test strips for field deployment, extending environmental surveillance to less developed regions. In the pharmaceutical realm, researchers aspire to use derivatives as intermediates for advanced drug synthesis, opening new paths in medicinal chemistry.
Toxicity Research
Safety studies keep expanding. Early toxicology flagged risks from ingestion and chronic exposure, prompting a shift to sealed containers and strict handling practices. Animal studies explore acute LD50 values and look for longer-term carcinogenic or mutagenic potential. Regulatory agencies in Europe and North America require routine review of toxicity data, incorporating published case reports and new findings from university and industry groups. Researchers look at both direct occupational risks and global environmental footprints, mapping breakdown products and monitoring water systems for trace residues. Growing public scrutiny pressures labs and suppliers to demonstrate not only safety in controlled workplace conditions but also a clear strategy for waste management.
Future Prospects
Looking ahead, industry watchers predict rising demand for sensitive, field-ready testing kits. Advances in portable spectrophotometry and smartphone integration could make the nitrite test powered by this compound accessible far from a traditional laboratory. Scientists see potential for developing greener synthesis paths to cut waste and hazard levels. Research funding increasingly supports next-generation analytical reagents—compounds with lower toxicity and equivalent sensitivity—to replace legacy chemicals where safety or supply issues arise. The durability and simplicity of 1-Naphthylethylenediamine Hydrochloride keep it relevant in a shifting market, with ongoing innovation focused both on convenience and safety.
What is 1-Naphthylethylenediamine Hydrochloride used for?
The Stuff Behind Water Testing
Anyone who has done high school chemistry or worked in a lab probably remembers color-changing reactions. One standout compound that brings out striking results in these tests is 1-Naphthylethylenediamine Hydrochloride. In the world of water and wastewater analysis, this chemical doesn’t get much attention outside scientific circles, but when it comes to detecting pollutants like nitrites, it steps into the spotlight. The moment this compound reacts with nitrite ions through a process called diazotization, it produces a deep pink azo dye. That color means something—a real, measurable change in a sample that can make the difference between clean water and something that could put public health at risk.
Keeping a Close Eye on Public Health
Clean water isn’t just about taste or clarity. Too much nitrite in water creates long-term health concerns, especially for infants and pregnant women. 1-Naphthylethylenediamine Hydrochloride helps scientists identify problems before they make it to the tap. Through simple color tests, environmental techs can gauge just how much nitrite water contains. The usefulness of this compound extends well beyond academic curiosity. Water quality teams in treatment plants, local labs, and environmental agencies depend on clear, immediate feedback when they run their tests. If results turn dangerous shades of pink, those teams know action can’t wait.
Simple Tools for Tough Jobs
Not every region has access to high-end gear. In my own experience helping out with local water testing campaigns, we relied on easy colorimetric methods. It didn’t cost much, and we didn’t need advanced training to gather meaningful results. 1-Naphthylethylenediamine Hydrochloride plays a central role in these tests because the reactions are reliable and the steps are clear. In rural clinics and developing regions, this sort of tool makes laboratory testing accessible, even with a shoestring budget. Sometimes the biggest contributions to public safety begin with compounds like this—quiet workhorses behind the scenes.
The Science in Urban Air Quality
Pollution tracking doesn’t stop at water. Urban research teams run similar tests on air samples to hunt down nitrogen dioxide. 1-Naphthylethylenediamine Hydrochloride reacts in these setups, providing quick visuals that technicians use to flag problem spots around cities, industrial zones, or traffic-heavy corridors. Quality-of-life bumps up when researchers catch spikes early and hold polluters accountable. This compound stands at the crossroads of science and environmental justice.
Building Trust through Reliable Testing
Misreporting or misreading environmental data erodes trust. I have seen small towns face public backlash after delays in pollutant reporting. The good news is that clear, consistent chemical reactions make it easier to back up findings. 1-Naphthylethylenediamine Hydrochloride allows labs to check water and air without relying on massive budgets or complex equipment. Precision matters in these cases, and the repeatable color changes help reduce mistakes.
Stronger Checks and Smart Policy
Governments and health organizations can do more by supporting easier access to these chemicals, pushing for up-to-date training, and encouraging transparency about results. Labs should double-check reagent quality, stick to best practices, and share up-to-date guides for using colorimetric tests. Collaboration with the public boosts awareness, especially when explaining what the results mean in real terms. The practical strengths of 1-Naphthylethylenediamine Hydrochloride help close gaps in safety and trust—one test at a time.
What is the chemical formula of 1-Naphthylethylenediamine Hydrochloride?
The Basic Facts: Understanding the Formula
1-Naphthylethylenediamine hydrochloride carries the chemical formula C12H14N2·HCl. Many recognize this compound in analytical chemistry labs, especially those who test for nitrite concentrations in water or food products. The hydrochloride part means the main molecule, 1-naphthylethylenediamine, bonds with hydrochloric acid, creating a salt form that dissolves well in water. This property makes it valuable in colorimetric assays, such as the Griess test, which turns a solution pink or red when nitrite is present.
Real-World Uses: Where Everyday Meets Chemistry
People working in environmental labs or food safety often depend on the accuracy of this chemical. Take, for instance, water testing during city-wide contamination scares. Public health officials rely on reagents that react dependably. 1-Naphthylethylenediamine hydrochloride fits this job, thanks to its stable formula and predictable reactions with nitrite.
In my own experience, working with this substance started with a simple purpose: ensure tap water met safety standards before reaching community taps. Even a subtle hint of contamination sets off alarms, demanding rapid, reliable tests. This compound, straightforward in makeup, lets technicians go from raw water samples to results in just a few steps.
Accuracy and Safety: The Stakes Behind the Numbers
Getting the formula right isn’t just about classroom memorization; lab safety rides on this. Mislabeling or misidentifying 1-naphthylethylenediamine hydrochloride can harm research, slow down emergency response, and expose workers to unexpected risks. In manufacturing, a single misplaced atom or mislabeled bottle leads to waste or worse—dangerous results for users downstream.
There’s a reason accuracy stays non-negotiable. The hydrochloride forms increase solubility, reducing risks tied to dust inhalation or accidental spills when compared to the free base. More consistent handling routines make safety training easier for new lab techs who might be nervous around chemical names that sound intimidating.
Sourcing and Quality Concerns
Quality control in suppliers influences the reliability of the final formula. Not every seller maintains the same purity standards. Labs that cut corners on supplier vetting sometimes end up with impurities, leading to false positives or negatives when results matter most. I’ve seen the aftermath—data thrown into question, reruns required, hard-earned trust lost.
Chemists recommend buying from suppliers with clear Certificates of Analysis and transparent sourcing. Remembering that C12H14N2·HCl isn’t just a string of letters and numbers; it represents countless hours of labor and a chain of trust from manufacturer to user.
Better Choices, Solving Bigger Problems
Instead of guessing, chemists seek robust documentation and clear labeling. Labs introduce internal quality checks, running positive and negative controls alongside each batch. Some institutions now push for greater transparency, urging manufacturers to disclose every step from synthesis to packaging.
Education also plays a big part. Instead of keeping protocols locked behind paywalls, sharing clear instructions and safety data keeps mishaps at bay. Chemistry, after all, serves real people. Whether researchers in crowded labs or inspectors walking up to a field well, getting the formula right for 1-naphthylethylenediamine hydrochloride keeps the science honest and the public safer.
How should 1-Naphthylethylenediamine Hydrochloride be stored?
Why Care About Storage?
A friend once had a small bottle of chemicals spill in the back of his closet. He had stored it there because space ran out on his shelves in the garage. Chemical smell filled the place for weeks, and nobody wanted to open that closet for months. That mishap drove home a basic idea. It’s easy to underestimate the risk of storing chemicals like 1-Naphthylethylenediamine Hydrochloride, yet small mistakes can snowball into dangerous events. Beyond the risk to health and property, proper storage just protects your investment. At five or six figures a drum, blowing an entire order through poor storage cuts deep into any budget.
Handling a Sensitive Compound
Anyone who’s worked in a lab knows some chemicals need careful handling from the minute they arrive. 1-Naphthylethylenediamine Hydrochloride, often used in diazotization labs or for nitrate analysis, reacts quickly with moisture, air, and sunlight. Exposure ruins not just its potency, but sometimes creates extra hazards.
Real-world evidence shows solutions and powders left out will clump, brown, or form dangerous byproducts within weeks. Research from chemical safety boards points directly to improper storage as a leading cause of lab accidents featuring this compound. Data from the National Institutes of Health links dozens of poisoning cases yearly to accidental inhalation or skin exposure from degraded product. These facts highlight the real risks behind “just stuffing it on the shelf.”
What Truly Works in Practice
Safe storage for 1-Naphthylethylenediamine Hydrochloride starts with a dry environment. Humidity in the air speeds up degradation fast. I’ve learned the hard way that putting silica gel packs in containers does more than just absorb moisture: it extends a chemical’s useable lifespan by months. For anyone who’s short on ideal lab space, investing in a small desiccator cabinet beats losing a batch to clumping or contamination.
Temperature matters as much as moisture. Most labs set this compound in cool cabinets, away from direct sunlight or heat sources. One summer, a shipment ended up on a windowsill for a day, and the whole lot changed color. That single mistake cost my team countless hours and hundreds of dollars. Manufacturers often specify 2-8°C in their literature for a reason — the compound breaks down beyond that range. For field kits, a portable cooler with ice packs becomes a small but essential expense.
Air exposure gets less attention than it deserves. Many folks just screw the lid back on after sampling, but air leaks in slowly. Double-bagging in airtight polyethylene bags cuts down on oxidation. Using bottles with good gasket seals also slows changes in the compound. I’ve seen teams paralyzed by a single batch going off. Preventing that is almost always cheaper than replacing spoiled stock or, worse, dealing with a spill.
Reducing Risk Day-to-Day
Chemical safety training materials at universities, pharmaceutical plants, and food labs all echo the same message. Keep containers tightly closed, clearly labeled, and out of reach of unauthorized staff. Segregate reactive chemicals. Never mix acids or oxidizers in with your storage area. Some labs go further by setting up colored bins or lockable cabinets to track inventory, adding another layer of oversight that stops slips before they become disasters. In my experience, visually simple checklists help catch small lapses before they spiral.
Many professionals overlook regular inventory rotation. Older stock tends to degrade quickly. Laboratories that cycle out dated containers every quarter catch problems before they grow. Unused leftovers risk turning into dust or residue hazards, which nobody wants to clean up after months of neglect.
Solutions That Stick
The most effective storage solution combines low humidity, stable temperature, airtight closures, and careful segregation. Long-term, investing in purpose-built cabinets pays off. For teams handling small quantities, using labeled, double-sealed containers with clear inspection records keeps everyone safe. Regular review of storage practices, clear responsibility assignments, and honest communication lower the odds of costly or dangerous mistakes. Experience, vigilance, and attention to detail give the best shot at trouble-free chemical handling every day.
What are the safety precautions when handling 1-Naphthylethylenediamine Hydrochloride?
The Risks in the Container
Anyone who’s spent time in a chemistry lab knows the line between curiosity and caution can get blurry, especially with chemicals like 1-Naphthylethylenediamine Hydrochloride. This compound often shows up in analytical chemistry. It packs a punch: reports point to irritation of eyes, skin, and the respiratory tract—not to mention environmental hazards if spills escape the lab. One whiff of the dust, and your sinus passages know it.
Personal Safety Comes First
At the bench, plain prescription glasses won't cut it. Goggles that actually fit and seal against splashes stay on until the last bottle is capped. I’ve seen too many folks reach for lab coats only after a problem lands in their lap. This chemical stains skin with purple marks that stick around for days—proof enough gloves remain a must, along with closed shoes and long sleeves.
Once, I had a friend who skipped the gloves to pour a few grams onto the balance. Less than two minutes later, he felt tingling and irritation. The warning on the bottle isn’t just there for legal reasons. Direct contact means irritation, possibly worse. Good ventilation—like a chemical fume hood—clears up vapors before you inhale them, a lesson I learned early after underestimating a compound’s volatility.
Set-Up and Storage Make a Difference
Open containers slowly and avoid working above head height. In one of my first years, I stored the powder in a crowded cabinet and later found residue stuck to the shelf and the outside of the jar. Now I double-check for tight lids, keep containers labeled, and never store reactive chemicals beside acids—these powders don’t always play nice.
Always protect the storage shelf with a plastic tray. These trays catch drips and make clean-up easier. Never leave the material exposed to humid air; moisture clumps the compound and can trigger decomposition. The desiccator spent under the bench, close at hand, actually proves its worth.
If Spills Happen
No matter how careful anyone tries to be, a spill eventually happens. I once fumbled a spatula, spilling powder across the benchtop. A quick dash for the paper towel just smeared everything. A portable vacuum designed for chemical dust avoids spreading particles. After clean-up, a 10% bleach solution breaks down the last traces.
For clothing and skin, waiting is never a solution. If this powder lands on you, get under the tap fast—ten solid minutes. Soap helps lift it off; don’t rub hard enough to break the skin, just enough to clear the stain.
Disposal Can’t Be an Afterthought
Too many labs cut corners by tipping used solutions into the sink. It’s dangerous and against regulations in most places. Excess and waste sit in a labeled, sealed container, handed over to chemical disposal experts at set intervals. On campus, I learned doing this protects not just the wastewater system but downstream communities and wildlife.
Culture of Respect in the Lab
Every small habit—pulling on gloves, working under the hood, double-checking the bottle—shapes the safety culture around you. Colleagues notice. Students mimic these moves. Even in places short on funding or fancy equipment, attention to basic precautions saves time, money, and worry. Respecting the risk keeps everyone safer, which builds real trust in the lab.
Is 1-Naphthylethylenediamine Hydrochloride hazardous to health?
Understanding the Chemical
1-Naphthylethylenediamine Hydrochloride shows up in research labs, mostly used in chemical analysis like the Griess reaction for detecting nitrites. Most people never see this white or light tan powder outside a controlled setting. Still, knowing the risks makes sense for anyone who spends time near chemicals, whether in a lab or teaching students.
Health Hazards: What Does Science Say?
Diving into the data, you won’t find many stories of this compound sending people to the hospital, but respected sources like Sigma-Aldrich or the National Center for Biotechnology Information don’t shrug it off either. Direct contact with skin or eyes can cause irritation, redness, and pain. Breathing in the dust may irritate the lungs and throat, leading to coughing, shortness of breath, or more serious respiratory discomfort for sensitive people. Swallowing the compound could upset the stomach, possibly leading to nausea or vomiting.
Some animal studies raise questions about mutagenicity—how a substance might change DNA. 1-Naphthylethylenediamine Hydrochloride hasn’t been studied as much as industrial chemicals like benzene or formaldehyde, but chemical relatives have shown troubling effects on cells. That prompts serious caution. The safety data sheet from major suppliers ranks it as harmful if swallowed or inhaled, and recommends gloves, goggles, and a tight workspace protocol.
Personal Experience in the Lab
I remember a summer in college spent tracking water pollution, where I used 1-Naphthylethylenediamine Hydrochloride alongside a spectrum of other reagents. In those days, lab safety got drilled into our heads: always work in a fume hood, put gloves on before opening bottles, label every flask twice. A classmate ignored the no-food-in-the-lab rule and ended up with a skin rash. The cause wasn’t proven, but it hammered home that unseen chemical dust doesn’t care about excuses.
Even now, those training habits matter. Not just for your own safety, but for everyone sharing a workspace. Lab accidents don’t only affect the person mixing chemicals. They hit the custodians, the students, the researchers who use the same tables next semester.
Preventing Harm: What Works in Practice
The basics go a long way. Chemical gloves protect your hands, lab coats keep spills off your clothes, and goggles guard your eyes. Let’s not forget a good fume hood. Proper ventilation makes a difference with powders like these, stopping those fine particles from ending up in your airways.
Proper storage adds another layer of protection. Keep this compound dry, away from acids and bases, and locked up. Post clear safety information where it matters. Don’t rely on memory—use a checklist each time, and teach others to do the same.
Disposal comes next. No pouring leftover solutions down the sink or tossing solid waste in the trash bin. Follow your local hazardous waste protocols, even if it takes extra time or paperwork. Polluted drains and landfill runoff don’t just harm wildlife—they boomerang back into drinking water, hurting communities.
A Path Forward
Many chemicals aren’t dangerous until they’re misused, and 1-Naphthylethylenediamine Hydrochloride fits that pattern. With training, vigilance, and a strong respect for what you’re handling, the risks can be managed. Keeping up-to-date with current information gives an edge—regulations and research findings don’t stand still. Teachers and lab managers should demand regular safety briefings, no matter how familiar staff might feel with a chemical. Nobody plans accidents, but with good habits, you can keep them rare.
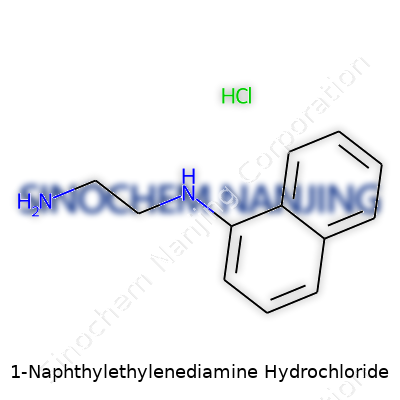
| Names | |
| Preferred IUPAC name | N¹-(naphthalen-1-yl)ethane-1,2-diamine hydrochloride |
| Other names |
N-(1-Naphthyl)ethylenediamine dihydrochloride 1-Naphthyl-ethylenediamine hydrochloride Naphthylethylenediamine hydrochloride 1-Naphthyl-1,2-ethylenediamine dihydrochloride 1-Naphthyl-ethylendiamine hydrochloride |
| Pronunciation | /wan-naf-thil-eth-uh-leen-dye-am-een hye-droh-klor-ide/ |
| Identifiers | |
| CAS Number | 50562-22-6 |
| Beilstein Reference | 120928 |
| ChEBI | CHEBI:38449 |
| ChEMBL | CHEMBL1496 |
| ChemSpider | 79507 |
| DrugBank | DB13373 |
| ECHA InfoCard | 05c3094a-6c27-4c5c-b2f1-1a20c3a1dcb1 |
| EC Number | 212-189-2 |
| Gmelin Reference | 75463 |
| KEGG | C08441 |
| MeSH | D008357 |
| PubChem CID | 85994 |
| RTECS number | SE8225000 |
| UNII | 9J9733875D |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID0044047 |
| Properties | |
| Chemical formula | C12H15ClN2 |
| Molar mass | 199.68 g/mol |
| Appearance | White to light purple crystal powder |
| Odor | Odorless |
| Density | 0.91 g/cm³ |
| Solubility in water | Soluble in water |
| log P | 0.11 |
| Acidity (pKa) | 10.25 |
| Basicity (pKb) | 6.3 |
| Magnetic susceptibility (χ) | χ = -57.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.704 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 197.5 J/mol·K |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 Health:2 Flammability:2 Instability:0 |
| Flash point | > 235 °C |
| Autoignition temperature | 210 °C (410 °F; 483 K) |
| Lethal dose or concentration | LD50 (oral, rat): 312 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50: 418 mg/kg |
| NIOSH | SN15750 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| Related compounds | |
| Related compounds |
Naphthylamine Ethylenediamine Naphthalene Sulfanilic acid Nitrite Diazonium salts |

