Insightful Look at 1-Naphthylamine Hydrochloride
Historical Development
The story of 1-naphthylamine hydrochloride follows the arc of synthetic organic chemistry in the late nineteenth century. Chemists working with coal tar spotted naphthalene’s potential, and before the dawn of widespread petrochemicals, managed to transform it into a functional amine. The hydrochloride salt came next, largely out of need for better solubility in laboratory prep and industry. As companies raced to make better dyes, rubber antioxidants, and rubber accelerators, the hydrochloride form offered real practicality. Over time, its applications spilled into other sectors, riding the waves of industrialization and regulation.
Product Overview
This compound falls into the aromatic amines family but stands apart for its utility and handling. 1-naphthylamine itself is an oily substance in pure form, prone to oxidation and harder to work with. Hydrochloride addition offers a crystalline, much more manageable salt. Whether you work in textiles, pharmaceuticals, or lab research, this salt form delivers consistent quality and properties over the raw base. Its role in dye intermediates and pigment production relies on its stability and reactivity in both acid and base environments. Product packaging almost always gauges moisture tightly. Purity checks focus on minimizing o-toluidine and other unwanted aromatic aminated materials due to strict regulatory pressure.
Physical & Chemical Properties
1-naphthylamine hydrochloride shows up as stable, nearly white to pale pink crystals, depending on trace impurities or handling conditions. These crystals dissolve easily in water, creating clear solutions characteristic of ionic aromatic salts, and hold up well through light exposure if stored correctly. The melting point usually reads between 241–243 °C, which is higher than the free amine, a result of ionic bonding with hydrochloric acid. With molecular formula C10H9N·HCl and a molar mass just around 179.65 g/mol, it’s more straightforward for inventory and calculation work compared to more complex salts. Labs and factories track the product with infrared or NMR spectra, tracing the aromatic fingerprint and salt formation, referencing reputable sources such as the Merck Index or peer-reviewed material safety data sheets.
Technical Specifications & Labeling
Professional manufacturers supply quality control results with every batch. Minimum purity sticks near 99%, and even minor metal or aromatic impurities raise eyebrows during audits. Labels provide full chemical names, hazard pictograms, and safety phrases, consistent with global GHS regulations. Data sheets often also note melting range, solubility in various solvents, and outlines for safe disposal. Adjustments to these standards rely on customer application—labs need the cleanest material, while heavy industry prefers the affordable, compliant grades. Certificates of Analysis track batch-to-batch consistency, which matters when final products can wind up in soft toys, industrial rubber, inks, or research animal testing.
Preparation Method
Production relies on naphthalene as a starting point. Nitration produces 1-nitronaphthalene, which reduction with iron filings and hydrochloric acid in aqueous solution or catalytic hydrogenation yields the free amine. Bubbling dry hydrogen chloride through the freshly formed 1-naphthylamine precipitates the hydrochloride—a step as traditional as it is efficient. Crystallization from water followed by careful drying rounds out the industrial workflow. This basic setup persists despite some attempts at eco-friendlier hydrogenation, especially due to the high conversion rate and ease of separation relative to other aromatic amines. Safety management keeps a close watch on the reduction step, since temperature runaways or vented hydrogen create tangible risks if not handled by experienced teams.
Chemical Reactions & Modifications
Once in hydrochloride form, the amine group stands ready for diazotization—an entry point into countless aromatic dye syntheses. Reaction with nitrous acid produces the diazonium salt, opening pathways to coupling reactions and formation of azo compounds central to textile and ink coloration. Electrophilic substitution at the remaining aromatic positions allows for downstream modifications, such as sulfonation or acetylation. In the pharmaceutical lab, the free base liberated from the salt can undergo N-alkylations to serve as building blocks in research chemicals and experimental drugs. Its chemistry provides insight, too, into the metabolic fate in toxicology since the naphthyl structure is subject to oxidative ring transformations by cytochrome P450 enzymes.
Synonyms & Product Names
The chemical appears widely as 1-naphthylamine hydrochloride, α-naphthylamine hydrochloride, or C.I. 37235 in dye indexes. Older European and US catalogs often use “naphthylamine hydrochloride” without specifying isomers, so it pays to verify the position on the ring. The pharmaceutical industry sometimes uses “naphthalen-1-amine hydrochloride” in research papers. International suppliers rely on UN numbers from shipping regulations and a clutch of regional product codes, so buyers should pay attention to avoid mix-ups with the 2-naphthylamine variant, which carries different risk profiles under REACH or TSCA.
Safety & Operational Standards
Safety teams apply some of the tightest controls on compounds like this, given the robust evidence linking aromatic amines to health risks. Standard operating procedures call for gloves, goggles, and industrial ventilation in all handling and transfer operations. Current workplace exposure limits reflect research connecting naphthylamine to bladder carcinogenesis. Disposal rules treat the salt as hazardous waste in nearly every country, with incineration required for disposal to prevent groundwater contamination. Manufacturing protocols restrict ambient dust and spills, since skin or respiratory uptake can occur even at low levels. Health monitoring, with urine analysis and cancer screening, appears in workplaces where exposure seems plausible, often in markets with strong labor protections.
Application Area
The main use shows up in azo dye synthesis for textiles, leather tanning, and paper industries. Its reliability as a coupling agent stands out in colors like Congo red and naphthol yellow, valued for lightfastness and wash resistance. Other industries use it in rubber chemicals—helping control vulcanization rates in tire and gasket production. Analytical labs exploit its reactivity as a reagent to measure oxidative agents or metals in environmental samples. Basic research in molecular biology sometimes uses this salt due to its ring system's mimicry of polycyclic amines found in metabolic pathways. Regulations around food, toys, and cosmetics sharply limit applications, given international concern over residue or contamination.
Research & Development
Researchers still study this compound for new analytical methods and as a probe for understanding amine metabolism in living organisms. Development focuses on finding alternatives in dye chemistry, given rising restrictions in most developed economies. Collaborations between university labs and manufacturers aim to design less hazardous analogues but still rely on 1-naphthylamine derivatives for comparative toxicology and structure-activity research. Environmental chemists research ways to degrade the aromatic ring without persistent pollutants, using photocatalysis or engineered microbes in lab settings.
Toxicity Research
Decades of epidemiology link long-term 1-naphthylamine exposure, especially inhaled or absorbed over years, with bladder and urinary tract cancers. Early dye workers saw high incidence of malignancy, pushing regulatory authorities to classify these compounds as potent carcinogens. Current studies use test animals, cell cultures, and chemical modeling to map the breakdown products and DNA interactions. National Institutes of Health publications and IARC monographs include 1-naphthylamine hydrochloride in their lists of confirmed human carcinogens, which underpins nearly all modern handling regulation. Environmental toxicologists look closely at its persistence and accumulation, flagging it for strict discharge limits into wastewater.
Future Prospects
In the years ahead, the search continues for safer replacements. Regulations grow tighter, nudging both industry and academia toward green chemistry and process alternatives. Some nations have phased out aromatic amine dyes in favor of metal-complex or reactive dyes, though 1-naphthylamine derivatives persist in specialized roles. Ongoing toxicology refinements build evidence for revised workplace standards and environmental release thresholds. At the same time, chemists mine the rich reactivity of the naphthylamine structure, adapting it for sensors in analytical chemistry and retaining a narrow but persistent use in fundamental research. Experimentation with enzyme-driven modification and rapid detection techniques may curb risks and harness more sustainable pathways for the coloring and materials industries.
What are the main uses of 1-Naphthylamine Hydrochloride?
Navigating Industrial Chemistry with 1-Naphthylamine Hydrochloride
People outside chemical labs rarely hear about 1-Naphthylamine Hydrochloride, but it plays a solid part in more things than you’d think. Working in industrial settings, I’ve watched this compound move from barrel to batching tank, and even though it never crosses most people’s minds, plenty of products owe their bright colors or sharp reactions to this tricky little molecule.
Building Blocks in Dye Manufacturing
Let’s start with color. 1-Naphthylamine Hydrochloride turns up as an early stepping stone in the path to azo dyes. These dyes don’t just show up in fabric—shirts, dresses, upholstery, even some colored plastics take on their hue thanks to processes that often begin with compounds like this. Since the dye industry has tightened safety and quality standards since the 20th century, many factories still rely on trusted chemical reactions where 1-Naphthylamine Hydrochloride reacts with diazonium salts to create a rainbow of synthetic dyes. As someone who’s spent afternoons troubleshooting dye batch inconsistencies, I’ve found the reliability of these classic reactions makes real-world manufacturing much smoother.
Analytical Chemistry and Research
Back in my college days, I learned quickly that detecting traces of certain chemicals calls for pinpoint reactions. 1-Naphthylamine Hydrochloride comes into play in lab tests for nitrite ions. Researchers test water, food, or biological samples for these pollutants using colorimetric analysis, relying on the distinct color change this compound provides. It’s not dramatic, but it’s a bread-and-butter approach in environmental and health labs around the world. If a batch of municipal water has questionable nitrite levels, chances are 1-Naphthylamine has been part of the confirmation process along the way.
Pharmaceutical Pathways
Drug companies use 1-Naphthylamine Hydrochloride as a chemical starting point for crafting some medicines, especially those featuring complex rings or specific nitrogen groups. It takes a special knack to optimize those synthesis routes, and I’ve worked with folks who chart every step, making sure each transformation goes off properly. The roots of cancer research even trace back to studies of these aromatic amines, pushing modern scientists to improve both synthesis and safety practices over the decades.
Ongoing Concerns and Safer Solutions
With all this utility, there’s a flip side. 1-Naphthylamine and its salts carry established health risks, especially with long-term exposure or careless handling. I’ve seen plant workers suit up with gloves and respirators before even opening the container. Agencies like IARC call out this chemical as a possible human carcinogen, prompting safety professionals to train staff, set exposure limits, and push for greener alternatives. Research into plant-based dyes or less hazardous intermediates is gaining ground, and some firms try enzyme-catalyzed syntheses where possible, aiming for a safer workplace and cleaner footprint.
Wrapping a Role Around Industry and Labor
For now, 1-Naphthylamine Hydrochloride keeps its spot in the toolkit for synthetic dyes, analytical procedures, and drug chemistry. Workers upstream and downstream see its impact. As regulators and innovators keep driving toward safer and smarter processes, this compound sits at an interesting crossroads—part old-school workhorse, part modernization motivator. If factories and labs keep investing in protective gear, tight protocols, and new chemical engineering methods, the benefits of what this compound creates remain within reach, with much less risk riding alongside.
What safety precautions should be taken when handling 1-Naphthylamine Hydrochloride?
Respecting a Tricky Chemical
1-Naphthylamine hydrochloride isn’t one of those substances you toss around the lab without a second thought. This compound has a long history in dye manufacturing, and scientists have studied its health risks for decades. Studies tie its exposure to bladder cancer among workers from the dye industry, and the chemical itself can irritate the skin, eyes, and respiratory tract. People sometimes underestimate what can go wrong in a lab or factory—until it gets personal. I once saw a colleague splash a similar compound on their hand; even that brief moment left them with red, burning skin that took days to calm down. That memory hasn’t faded, and it sticks with me each time I see folks cut corners for the sake of speed.
Personal Protective Equipment (PPE) Isn’t Optional
If you ask anyone who’s spent time with industrial chemicals, those thick nitrile gloves and splash goggles become second nature. Even short exposure can cause trouble. Lab coats matter too—not just for looking professional but for protecting regular clothes and keeping skin covered. I always reach for a face shield when there’s a risk of splashing, not just goggles, because sometimes a quick reaction isn’t enough. Picking the right mask or respirator keeps microscopic particles or fumes out of your lungs, because even if 1-naphthylamine hydrochloride dust isn’t billowing out, inhaling small amounts can cause real problems over time.
Air Circulation Saves More Than Just Comfort
Ventilation isn’t just a technicality for inspectors to check off. An open window or standard lab fan doesn’t move enough air to keep this compound out of your breathing zone. Every time I’m in a lab with chemicals flagged as carcinogens, I head for a certified chemical fume hood. That way, even accidental spills or open containers won’t fill the air with invisible risks. Most labs and workspaces have them—there’s little excuse for skipping this step. In places where a dedicated fume hood isn’t possible, at least some mechanical local exhaust beats doing nothing.
The Value of Training and Good Habits
Anyone who handles hazardous substances needs more than a data sheet—they need training in emergency procedures and a routine of clear, careful habits. I remember a safety officer drilling us on location and use of eyewash stations and chemical showers until everyone in the room could get to them in their sleep. In those rare moments when something spills, muscle memory kicks in. Mistakes shouldn’t cost anyone their sight or health. Workers should know not only how to clean up safely, but also why certain steps matter. For example, never using bare hands, never working alone, and never putting chemical-soaked gloves back on the shelf.
Safe Storage and Waste Disposal
Proper storage involves more than finding space in a cabinet. Strong acids or bases don’t belong next to 1-naphthylamine hydrochloride. Sturdy, clearly labeled containers prevent accidental mixing or spills. I prefer storing such compounds below eye level, so lifting and retrieving isn’t awkward or dangerous. Used gloves, bottles, or residues should go straight into hazardous waste containers, not regular trash bins or down the drain. Companies and institutions have rules for a reason: local regulations and international standards exist to keep these risks away from public water supplies and landfill.
Caring for People Beyond the Rules
Textbooks and safety sheets help, but people in labs or factories often build their own unwritten code: always watch out for the quiet coworker, double-check bottles before grabbing, and speak up fast if you spot a shortcut. Staying safe with 1-naphthylamine hydrochloride, or any hazardous chemical, means listening to past mistakes and treating every precaution as personal. In this way, what starts as a checklist becomes a culture—one where everyone expects to go home safe and healthy, every shift.
How should 1-Naphthylamine Hydrochloride be stored?
Understanding What’s at Stake
Storing chemicals the right way keeps people safe and protects the environment. With 1-naphthylamine hydrochloride, this becomes even more critical. This compound, found in dyes, research labs, and some specialty manufacturing settings, brings along toxic properties. I’ve seen what can happen when folks get casual with handling; respiratory issues and skin burns start showing up pretty fast when chemicals don’t stay in the right place.
Keeping Out Moisture and Light
Humidity can mess with a lot of chemicals, and 1-naphthylamine hydrochloride reacts strongly to water. Anyone who’s spent time in a wet, muggy storage room knows how quickly dry powders clump together or degrade. This stuff absorbs water and breaks down, creating dangerous by-products. The solution starts with an airtight container—glass or robust plastic with tight seals. Forget cardboard boxes and bags. Those won’t hold up. This chemical should always sit away from sinks, drains, or any spot that might get splashed or sweaty.
Ventilation and Smell
In labs and storerooms, bad air makes tough jobs even riskier. Strong ventilation keeps chemical fumes from building up, and anyone storing 1-naphthylamine hydrochloride knows the value of a good exhaust fan. I’ve walked into spaces where the air felt heavy with irritants—it doesn’t just hurt your lungs, it makes accidents more likely. That means a storeroom needs real airflow, not just an open window. Doors should stay closed to limit how much air escapes into common areas, but fumes never stay trapped if fans do their job.
Chemical Compatibility
Mixing the wrong chemicals spells trouble fast. Once, I watched a rookie toss incompatible bottles on the same shelf, and we scrambled to clean up a spill when a cap loosened. 1-naphthylamine hydrochloride can attack metals, acids, and oxidizers. It belongs on shelves built for strong chemicals, never stacked alongside bleach, peroxides, or acids. Labels need to stay clear, facing out, checked often for damage. No one wants a faded label hiding something deadly.
Temperature Control
Temperature swings ruin chemicals. That’s the difference between a solid, stable powder and a dangerous mess. Hot storage rooms cause decomposition and evaporation, leading to toxin release. Cold, dry, dim places help chemicals last longer. I always pick rooms with thermometers and keep logs. Inspections might feel tedious, but the alternative—explosions, leaks, ruined chemicals—costs much more.
Personal Safety Gear
Forgot your gloves or goggles? One splash or spill can remind you fast. I learned that lesson early, after a close call with another irritant chemical. With 1-naphthylamine hydrochloride, gloves, goggles, and long sleeves cut down the risk. Stores should always stack safety gear close to entrance doors. Everyone who enters needs to suit up, even for a quick check or inventory count.
Reliable Record Keeping and Training
It’s easy to lose track of old bottles in a crowded storeroom. I’ve seen people get hurt from forgotten cans of outdated or leaking stock. Good practice means every bottle of 1-naphthylamine hydrochloride stays listed, checked for expiration, and inspected after handling. Regular drills and clear safety sheets near the shelves keep habits sharp.
Thinking Ahead
Some labs use digital tools and alarms to prevent mistakes. Cameras and sensors spot leaks right away. Waste containers for 1-naphthylamine hydrochloride need clear labels and regular pickups, not just a bin dumped in the corner. Safety depends on teamwork: one careless move puts everyone at risk. With the right habits, anyone storing this chemical can work smarter, safer, and help prevent health and environmental problems before they start.
What is the chemical formula and molecular weight of 1-Naphthylamine Hydrochloride?
Chemical Formula and Molecular Weight
1-Naphthylamine Hydrochloride, with the chemical formula C10H9N·HCl, appears frequently in chemistry discussions for a reason. Its molecular weight stands at 179.65 grams per mole. This detail isn’t just for lab notebooks – it’s essential for accurate measurements in any scientific setting. Any process that involves this compound, from reactions in a research lab to calculations for production batches, starts with this information.
A Closer Look at the Compound
This chemical, formed by the reaction of 1-naphthylamine and hydrochloric acid, becomes a stable crystalline powder. It dissolves well in water, making it a candidate for a range of chemical syntheses. Years ago, I worked in a chemistry lab where understanding the solubility of chemicals could make or break a project’s timeline. A slip in the math could delay progress by days or even weeks. Correctly calculating how much 1-Naphthylamine Hydrochloride to add meant everything ran smoother.
Why Molecular Data Matters
Manufacturers dealing with dyes, pharmaceuticals, or organic synthesis use the molecular weight to figure out exact proportions. Imagine preparing a batch of dye. If the amount of 1-Naphthylamine Hydrochloride is off, not only does the color shift, but safety might be at risk. Health and safety protocols get built on precise calculations, such as the right amount to use when handling potential toxins. The National Institute for Occupational Safety and Health (NIOSH) advises careful workplace monitoring of aromatic amines, including 1-naphthylamine derivatives, due to proven links with increased risk of cancer for workers exposed over time.
Production, Handling, and Safety
Factories process thousands of kilograms of 1-Naphthylamine Hydrochloride every year. Its stability as a hydrochloride salt helps reduce some risks, but this doesn’t mean users can skip out on personal protective equipment. I remember a site visit to a pigment factory where employees knew every safety measure by heart — gloves, masks, regular medical checks. The molecular weight plays into these safety routines too; it dictates how much can be released or stored safely, and what ventilation or filtration systems need to handle.
Environmental and Health Considerations
Concerns about aromatic amines like 1-Naphthylamine Hydrochloride extend well beyond the production plant. Waste disposal requires strict control, since improper handling has contaminated groundwater in some regions. The Environmental Protection Agency (EPA) and international watchdogs keep updating limits based on new research, which often draws on molecular data to inform risk. At the same time, safety training for workers helps identify symptoms early and avoid long-term hazards. I’ve gone through those sessions, learning how immediate reporting and correct procedures can make all the difference.
Next Steps for Safer Chemistry
Chemical manufacturers and research groups keep pushing for safer alternatives or better control methods. Some look at ways to modify the 1-naphthylamine structure to make less hazardous compounds, but solutions tend to require stepwise progress. Ongoing research, real-world training, and advances in safety technology all play a part in reducing risk. People on the shop floor, in the lab, and at the design desk each use molecular weight and formula data to get things right — not just for productivity, but for health and environmental care.
Is 1-Naphthylamine Hydrochloride hazardous or toxic?
Why Worry About 1-Naphthylamine Hydrochloride?
1-Naphthylamine hydrochloride makes a regular appearance in the chemical industry, especially in dye manufacturing and research labs. Many people don’t think much about chemicals they can’t buy at the store, but it pays to look closer before handling anything this powerful. Safety should not be optional, especially for anyone with skin in the game on factory floors or university benches.
Toxic Effects: Not Just a Lab Story
I used to dismiss half the cautionary tales about old dye chemicals. Then I started working near a plant that handled naphthylamines and saw colleagues suffer skin irritation that went beyond just “annoying.” Turns out, 1-naphthylamine hydrochloride can irritate eyes, nose, even the lungs if dust or fumes get in the air. Gloves and goggles help, but training matters even more: some workers washed hands all the time, others figured hot water would “just take care of it.” Unchecked, careless contact led to burns, rashes, and headaches.
Long-term exposure raises the stakes. Studies have linked the parent compound, 1-naphthylamine, to bladder cancer—one big reason it got swept out of most consumer dye markets decades ago. Unlike a rotten smell or flashy color, cancer risk sneaks up, and folks exposed over weeks or years may never suspect the real cause. In the early 20th century, dye factories saw high rates of urinary diseases among workers who weren’t shielding themselves or keeping things clean.
Down the Drain and Into the Ground
Chemicals rarely stay boxed in once spills or runoff happen. Even if someone is tidy at their own bench, accidents and sloppy disposal can seep into soil and water. Those old pipes running under chemical plants are a favorite route for leaks. 1-Naphthylamine breaks down slowly in the wild, so fish and aquatic insects pick it up fast—another reason regulations keep getting tighter.
The risk to the general public stays low if facilities run tight ships, but lapses have lasting aftershocks. You don’t want to explain to your town why the river can't support fishing anymore, or why water smells funny.
Taking Responsibility, Improving Practice
I’ve seen the safest workplaces keep meticulous records, update safety practices, and refresh hazard signs before anyone complains. This approach limits unexpected trouble, like a clogged vent turning into a fume hazard. Strong ventilation, chemical-resistant gloves, and showers close to work spaces save real lives. Simple steps—like banning eating and drinking in lab areas—head off more trouble than extra meetings ever could.
Some companies use alternatives now, switching away from chemicals with clear ties to cancer. Switching isn’t free, but watching someone’s health fail costs even more in the long run. In schools and labs, instructors hold more sway than rules or warnings; one good talk about what naphthylamines did to earlier generations changes how students view their beakers.
Keep Learning, Keep Vigilant
If anybody downplays the hazards tied to 1-naphthylamine hydrochloride, take that as your signal to dig deeper. Health agencies worldwide have classified this chemical as potentially carcinogenic. The weight of research supports limits and protective measures, not casual handling. Knowledge, updated protocols, and honest conversations keep everyone ahead of the curve.
Trust grows when managers put worker safety before shortcuts and back up safety policies with resources. That sends a signal to everyone: their lives matter more than cutting corners or chasing quick numbers.
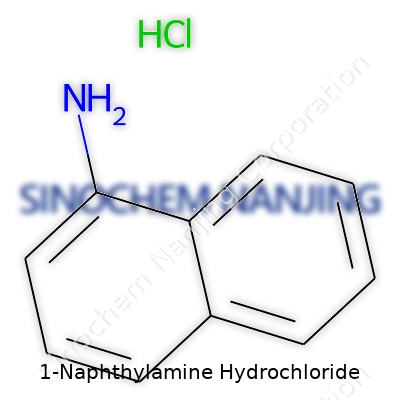
| Names | |
| Preferred IUPAC name | naphthalen-1-amine hydrochloride |
| Other names |
1-Naphthylamine hydrochloride α-Naphthylamine hydrochloride 1-Aminonaphthalene hydrochloride |
| Pronunciation | /waɪn-ˈnæfθɪl.əˌmiːn haɪˌklɔːraɪd/ |
| Identifiers | |
| CAS Number | 2243-62-1 |
| 3D model (JSmol) | `3D model (JSmol) string for 1-Naphthylamine Hydrochloride: CNc1cccc2c1cccc2.Cl` |
| Beilstein Reference | 83750 |
| ChEBI | CHEBI:60686 |
| ChEMBL | CHEMBL1521 |
| ChemSpider | 10053 |
| DrugBank | DB11433 |
| ECHA InfoCard | ECHA InfoCard: 100.007.818 |
| EC Number | 207-128-8 |
| Gmelin Reference | 82103 |
| KEGG | C19309 |
| MeSH | D009281 |
| PubChem CID | 61358 |
| RTECS number | GH1400000 |
| UNII | 2176FS3P3H |
| UN number | 1667 |
| CompTox Dashboard (EPA) | DTXSID5020743 |
| Properties | |
| Chemical formula | C10H10ClN |
| Molar mass | 163.63 g/mol |
| Appearance | White to light brown crystalline powder |
| Odor | Odorless |
| Density | 1.305 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.14 |
| Acidity (pKa) | 2.74 |
| Basicity (pKb) | 9.18 |
| Magnetic susceptibility (χ) | -49.0e-6 cm³/mol |
| Refractive index (nD) | 1.752 |
| Viscosity | Viscous liquid |
| Dipole moment | 6.12 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 129 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -10.6 kJ/mol |
| Hazards | |
| Main hazards | Carcinogenic; toxic if swallowed, inhaled, or absorbed through skin; causes skin and eye irritation. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H350: May cause cancer. H317: May cause an allergic skin reaction. |
| Precautionary statements | P260, P264, P270, P280, P301+P312, P302+P352, P304+P340, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0-Health Hazard |
| Flash point | 130°C (266°F) |
| Autoignition temperature | 385 °C |
| Lethal dose or concentration | LD50 oral rat 500 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1470 mg/kg (Oral, Rat) |
| NIOSH | NIOSH: RN3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Naphthylamine Hydrochloride: 0.1 mg/m³ |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | IDLH: 50 mg/m3 |
| Related compounds | |
| Related compounds |
1-Naphthylamine 2-Naphthylamine Naphthalene Aniline 2-Naphthol |

