1-Naphthyl Isothiocyanate: A Deep Dive into Its Role, Risks, and Realities
Historical Development
Long before chemical engineering carried the badge of modernity, researchers turned to aromatic compounds for versatility and branching pathways. 1-Naphthyl isothiocyanate (NITC) grew out of the robust organic dye industry of the nineteenth and early twentieth centuries, a less-publicized sibling to the naphthalene-derived colors found in everything from textiles to art materials. Organic chemists in that era were fascinated by the sharp reactivity of isothiocyanates, learning quickly how the naphthyl backbone could channel these reactions into functional materials and powerful laboratory tools. Historical patents and journals from the 1930s record isothiocyanates surfacing in research on chemical synthesis and protein modification, where their ability to form stable bonds with amines made them essential tools in mapping the boundaries of biochemistry.
Product Overview
Chemists and technicians often reach for 1-naphthyl isothiocyanate in situations calling for a selective reagent. The chemical bridges the gap between academic curiosity and practical necessity. Markets offer it as a fine crystalline powder, typically used as a laboratory reagent, investigative probe for biological systems, and even as a model compound in toxicology. The substance carries the gravitas of both legacy and innovation in chemical supply catalogs, reflecting its consistent demand and broad applicability over decades.
Physical & Chemical Properties
1-Naphthyl isothiocyanate manufacturers describe it as a faintly yellow to off-white solid. Its molecular formula is C11H7NS, with a molar mass of about 185.25 g/mol. With a melting point hovering between 56°C and 58°C, it shifts from crystal to liquid readily upon heating. The compound’s slight solubility in water, coupled with robust solubility in most organic solvents such as chloroform and acetone, enables straightforward manipulation during experimental procedures. Sharp, pungent odors accompany the powder, a reminder to keep the workspace ventilated and protective gear close at hand.
Technical Specifications & Labeling
Buyers and lab managers pay attention to purity, which usually exceeds 97% for research-grade samples. Labels must display the chemical’s name, formula, batch number, and hazard pictograms indicating its irritant and harmful nature. Signal words and storage recommendations appear on every bottle to satisfy international chemical safety regulations. Because misuse poses a risk, chemical suppliers attach detailed safety data sheets (SDS) and list the UN and CAS registration numbers. Commercial NITC often features stability under dark, cool storage and compatibility with sealed glass containers.
Preparation Method
The standard approach starts with 1-naphthylamine, a well-established aromatic amine. By treating it with carbon disulfide in an alkaline setting, the mixture yields a dithiocarbamate intermediate. Once this intermediate meets a dehydrating agent such as phosphorus oxychloride, isothiocyanation proceeds, giving 1-naphthyl isothiocyanate after extraction and purification. Chemists with sufficient training can run this method efficiently, but the steps demand skill in handling volatile and sometimes hazardous reagents, especially when it comes time to separate the product and manage the resulting waste.
Chemical Reactions & Modifications
1-Naphthyl isothiocyanate reacts heavily with primary and secondary amines, opening doors for attaching fluorescent tags or functional groups to proteins. This reactivity supports research in proteomics, where mapping active sites helps understand enzymes and disease processes. Under mild heating, it couples with alcohols or thiols, generating useful intermediates for further chemical innovation. In the hands of experienced chemists, the compound undergoes nucleophilic substitution, ring-opening, and condensation—flexibility that secures its spot on laboratory shelves worldwide.
Synonyms & Product Names
Scanning reagent catalogs or chemical inventories, you might see this compound labeled as Naphthalen-1-yl isothiocyanate, 1-Isothiocyanatonaphthalene, or 1-Naphthylthiocyanate. These subtle shifts in names sometimes confuse newcomers, but seasoned operators look for the formula, C11H7NS, or the CAS Number 86-57-7, to ensure they handle the correct material. This variety flows from differences in regional naming systems and supplier preferences shaped by years of trade and standardization.
Safety & Operational Standards
Experience teaches that 1-naphthyl isothiocyanate demands respect in daily handling. Strong irritant properties make gloves, goggles, and fume hoods non-negotiable in any workspace. Short-term exposure often brings stinging eyes and nasal passages, with skin contact causing rashes or burns. The compound’s dust chokes airflow if it escapes into the lab air, so containment strategies stand front and center. Disposal follows hazardous organic protocols—sealed, labeled waste in accordance with the Environmental Protection Agency (EPA) and local waste bylaws. Chemical hygiene plans in accredited labs emphasize drills and quick response in spill scenarios.
Application Area
Biochemists leverage 1-naphthyl isothiocyanate for site-directed labeling of proteins, where it forms covalent bonds, helping map enzyme active sites. Analytical chemistry labs exploit its isothiocyanate group in chromatography and spectroscopy, where its reactions generate markers for quantifying trace compounds. Toxicology teams run NITC through animal models and cell cultures, seeking clues about liver injury, cholestasis, and environmental toxicity. The breadth extends into materials science, where controlled modification enables the assembly of new polymers and smart materials for sensors or selective membranes.
Research & Development
Research targeting 1-naphthyl isothiocyanate flourishes at the crossroads of chemistry and medicine. Universities have published work on its precision as a liver toxin in rodent models, which enables exploration of protective drugs and restoration pathways in hepatology. Biotech startups and academic labs tinker with NITC in state-of-the-art protein fingerprinting, tying old-school chemical reactivity to the challenge of decoding massive proteomic databases. My own time in research circles taught me that a responsive chemical like this can run circles around more cumbersome reagents—less fuss, more data.
Toxicity Research
Toxicologists draw heavily on 1-naphthyl isothiocyanate’s capacity to induce cholestatic liver injury in mice and rats. The compound blocks bile flow and triggers cellular injury, mimicking real-world disease mechanisms. These models underpin a chunk of preclinical drug safety studies, where researchers sift through biomarkers, DNA damage, and emerging protective compounds. Repeated exposure data guides regulatory decisions, with chronic dosing flagged for possible carcinogenic and mutagenic outcomes. As new generations of scientists push for animal-free toxicity testing, systematic screening of NITC across in vitro platforms speeds up safer product design.
Future Prospects
1-Naphthyl isothiocyanate stands on the frontline of several research efforts, including the hunt for more precise labeling agents, safer alternatives for toxicology testing, and transformative chemical modification methods. Ongoing advances in green chemistry call for less hazardous synthesis, spurring demand for methods that cut environmental and health risks. Many anticipate breakthroughs in automated proteomic workflows, real-time biosensing, and predictive toxicology—all feeding off the adaptable chemistry that NITC delivers. Investments in safety, greener process engineering, and high-throughput screening look set to redefine how researchers harness this staple compound.
What is 1-Naphthyl Isothiocyanate used for?
What Sets 1-Naphthyl Isothiocyanate Apart
Let's talk about 1-Naphthyl Isothiocyanate, a compound that shows up mainly in research labs and testing facilities. People in pharmaceutical research and toxicology use this chemical not because it has a flashy public image, but because of its unique effects on the liver. In my own years working as a lab technician, I've watched researchers rely on substances like these to push our understanding forward. Sometimes it takes a compound that’s a little bit obscure to unlock something major in medicine or toxicology.
Why Researchers Reach for This Compound
1-Naphthyl Isothiocyanate acts as a tool to model liver injury in animals, especially in rats. Scientists introduce it to study what happens when bile stops flowing properly—a condition called cholestasis. By simulating liver damage, researchers can observe symptoms, try out potential treatments, and see which drugs actually slow damage down or even fix it. This kind of testing produces information that helps doctors treat real liver problems in people. Liver injury isn’t just a laboratory issue. Chronic liver disease affects over 1.5 billion people worldwide, leading to pain, long-term disability, and even early death.
Beyond Toxicology: Digging into Metabolic Pathways
This compound does more than mimic liver trouble. Researchers use it to expose previously hidden parts of the body’s metabolic machinery. By triggering oxidative stress and inflammation, they watch different proteins, messenger molecules, and genetic regulators spark into action or shut down. That insight helps pharmaceutical scientists design smarter, safer drugs for cholesterol, hepatitis, or autoimmune diseases. In my own experience with drug development teams, you rarely see breakthroughs unless someone is mapping out exactly how molecules interact inside the body. Compounds like 1-Naphthyl Isothiocyanate make that possible.
Environmental and Safety Concerns
Handling it means dealing with risks. 1-Naphthyl Isothiocyanate is classified as harmful and has potential to cause serious irritation or acute reactions if people breathe it in, spill it on skin, or get it in their eyes. The only reason workplaces allow its use is tight safety protocols—personal protective gear, ventilated hoods, containment, and rapid clean-up plans. During my years of lab work, I saw more trouble from labs that didn’t take chemical handling protocols seriously than from the chemicals themselves. Proper education, training and discipline remain essential every time a scientist uses a compound like this.
Building Safer and Smarter Testing Models
It’s always tempting to look for “natural” models in drug screening, but compounds like 1-Naphthyl Isothiocyanate help researchers build reproducible, reliable study systems. Today, scientists work hard to move toward alternatives using cells grown in dishes, computer models, or even organ-on-a-chip systems. Still, the level of precision and consistent results needed for regulatory approval often require animal testing with compounds like this to fill in critical knowledge gaps. It becomes a push and pull—developing smarter, less harmful tools while still learning as much as possible about liver health and disease. This chemical, for all its hazards, continues to play an outsized role in that learning process.
What are the safety precautions when handling 1-Naphthyl Isothiocyanate?
Understanding What’s at Stake
Handling 1-Naphthyl Isothiocyanate takes focus. This compound isn’t some benign powder or colorant. In my time around chemical labs, I’ve come to respect substances with strong odors and warning tags. This one can irritate skin, eyes, and airways in a hurry, and inhaling dust means trouble in the long run—think respiratory issues and sensitization, not just a brief cough. There’s a lot of research linking isothiocyanates to cellular effects, so the level of protection should go deeper than just “using gloves.”
Personal Protective Gear is Non-Negotiable
Start with the basics: nitrile gloves, chemical splash goggles, and a solid lab coat. I’ve seen what happens when someone thinks latex gloves or cheap goggles will do—they don’t. Round it out with a face shield if splashing could happen. Anyone with open wounds or sensitive skin shouldn’t handle this material directly. A year ago, a colleague ended up with a burning rash after a missed spot on their wrist. Don’t cut corners here. Respiratory protection, like a fitted N95 or P100 respirator, matters if powders could become airborne or work occurs outside a fume hood.
Ventilation Makes a Real Difference
It doesn’t take much airborne 1-Naphthyl Isothiocyanate to start smelling it, and that’s reason enough to stick with a working chemical fume hood. I remember times when a quick “just-open-a-window” attitude led to lingering odors, headaches, and more cleaning than anyone wanted. Fume hoods pull vapors and dust away from your face, stopping accidental inhalation and long-term health hits. It’s basic but often skipped when people work after hours or outside main labs.
Spill Response: Prepared, Not Panicked
Most spills are avoidable with small batches and careful pouring. Still, keep spill kits handy. These aren’t just for show—I once watched an intern try to scoop up a powder spill bare-handed because the kit “looked sealed.” Absorbent pads, neutralizers, and the right waste containers belong close to any workspace. Sweep or vacuum dust gently, using HEPA-equipped vacuums rather than dry brooms that send particles into the air.
Storage—Out of Sight, Not Out of Mind
1-Naphthyl Isothiocyanate calls for dry, cool storage. Use tightly sealed containers made of glass or compatible plastics. Label them clearly, noting the date received and any hazard symbols. Don’t store it near acids, bases, or anything that could trigger a reaction. In my experience, one unlabeled jar brings a whole audit down on the lab—and that means running around finding out where the ventilation system missed a leak.
Disposal: Care for Staff and Environment
Disposal isn’t just a quick toss in the bin. Specific rules exist for isothiocyanates, usually as hazardous waste under university or city guidelines. Contact a safety officer before disposing of even small amounts. I’ve learned over time that compliance means fewer late-night phone calls and less risk of fines.
Training and Regular Reminders
Anyone expecting to handle this chemical should know the risks and common sense steps before lifting a lid. Refresher training—actual walk-throughs, not just online quizzes—should happen yearly. I still have mental images of the “worst case” videos from my grad school years, and they shaped how I organize every procedure in the lab.
Safer Handling Comes from Shared Vigilance
Risk can drop fast with attentive teamwork. Speak up if a procedure or setup seems sketchy. Print out handling procedures and leave them near the bench instead of buried in a binder. Protecting fellow lab workers matters far more than shaving a few minutes off the day. That’s experience talking, not just a rulebook.
What is the chemical structure of 1-Naphthyl Isothiocyanate?
What Makes Up 1-Naphthyl Isothiocyanate?
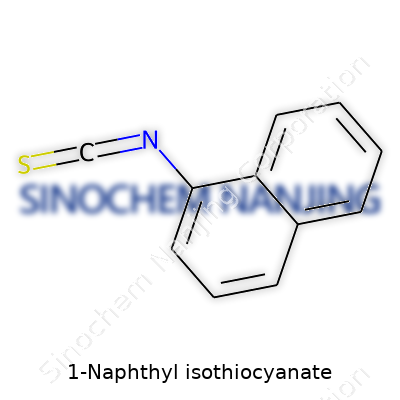
Chemistry never really gets old to those who spend time with it. 1-Naphthyl isothiocyanate (often shortened to 1-NITC or 1-NIC) carries a formula of C11H7NS. Breaking it down, this molecule builds on the rigid backbone of naphthalene. Picture two fused benzene rings, side by side, together making up that stable ring system. Imagine one hydrogen swapping places with an isothiocyanate group (–N=C=S) on the first carbon.
That isothiocyanate piece sticks off the ring, bringing along nitrogen and sulfur. Instead of being another simple side chain, this group comes loaded with chemical reactivity. The carbon found right in the middle of –N=C=S looks electrophilic. It means any nucleophile passing by will want to latch on. The sulfur at the far end, with its lone pairs, brings a whole new set of unpredictable reactions.
Why Shape Matters in the Lab
I’ve seen this molecule sway the fate of entire research projects. Most laboratories use 1-Naphthyl isothiocyanate for probing drug metabolism in the liver. Its naphthalene core lets the compound wedge into biological membranes or enzymes—so it’s unusually sticky for something so small.
Look closely: the naphthalene part makes the molecule nonpolar except at that isothiocyanate end. This contrast matters when you want to blend it with biomolecules or solvents. Those two connected rings add bulk, which helps it slip into spots that flat, simple molecules just can’t reach. And chemists have found that the spatial arrangement of atoms in 1-NITC shapes its toxicity and chemical fate inside the body. It’s not just about which atoms get attached—it’s about how they’re arranged.
Real-World Risks and Scientific Opportunities
The same structure that makes 1-Naphthyl isothiocyanate useful for research also makes it hazardous. Chronic exposure has linked this molecule to bile duct injuries in animals, and researchers watch its effects on the liver with a sense of caution. Handling the substance with care isn’t just good practice; it’s non-negotiable.
Despite risks, the unique chemical structure attracts curiosity among scientists. For instance, it targets enzymes in the liver—often leading to creative experiments about detoxification and metabolism. Because of its shape and buildup, researchers can fine-tune versions of the molecule to either reduce toxicity or amplify its ability to probe how drugs break down in cells.
Toward Safer Chemistry
Minimizing hazards becomes possible if you tweak the molecular structure. Reducing lipophilicity, changing the position of the isothiocyanate, or altering the naphthalene core could lead to friendlier molecules that still offer valuable insight into drug metabolism. Some chemists push for better personal protective equipment (PPE) and improved storage methods, keeping this compound—and its cousins—under tight control.
Clear understanding of the real shape and makeup of 1-naphthyl isothiocyanate doesn’t just add to textbook knowledge. It keeps chemical research honest, alert, and safer for those who work at the frontier between drug discovery and toxicology. Curiosity about this molecule pushes the boundaries of how we investigate the body’s response to drugs and environmental chemicals.
How should 1-Naphthyl Isothiocyanate be stored?
Not Just Another Lab Chemical
1-Naphthyl Isothiocyanate (NITC) brings up more than a tricky pronunciation; it asks for some solid respect in any lab or storage space. This isn’t just about following some checklist—there are real risks if this compound isn’t handled carefully. I remember visiting a university chemistry department years ago where they hadn’t locked down their storage practices. A mild leak sent a sharp, pungent odor down the halls, and someone ended up with a nasty rash. This stuck with me—and it drives home the point. Simple mistakes bring consequences, especially with substances like this.
Ventilation Matters
NITC fumes can go right through tightly packed shelves. Keeping containers in a well-ventilated spot goes beyond comfort. Constant airflow means anyone nearby stands less chance of inhaling irritants or encountering a buildup that could spark health problems. The Occupational Safety and Health Administration keeps repeating the same advice for a reason: healthy lungs and clear air give people better odds.
Container Choice Isn't Trivial
Students in labs sometimes figure any old bottle will do. That mistake comes fast when you deal with something that reacts with moisture or certain plastics. NITC holds up best in tightly sealed glass containers. Glass stays inert, so there’s less risk of a slow leak or ugly reaction. A tough screw cap, not some flimsy stopper, seals the deal. In my own lab, we only use containers with a PTFE (Teflon-like) liner. Those hold up, and that extra barrier helps trap fumes.
Temperature and Light Shape Longevity
Shortcuts aren’t worth it. Cool, dry, and dark storage spaces always earn choice status with strong chemicals. Heat speeds up decomposition and gets volatile compounds going. Even aging light bulbs sent NITC yellowing in one storeroom I saw, hinting at the breakdown happening inside the bottle. Choosing a locked chemical cabinet, away from radiators or windows, offers an easy win.
Label Like Your Life Depends On It
Handwritten labels fade. Stickers fall off. Yet, too many people skip relabeling or cover up essential details. Clear, durable labels—with hazard symbols, storage precautions, and a date—keep everyone alert. I’ve had coworkers grab the wrong bottle and almost cause a dangerous mix-up. Good labeling stopped things before they went further. At my last job, we switched to laser-printed, chemical-resistant labels—no more mystery containers.
Preventing a Chain Reaction
Storing NITC near incompatible chemicals turns accident into threat. Acids, strong bases, and oxidizing agents have no place within arm’s reach. If one container cracks, vapors or spillage make a bad situation escalate. Several safety data sheets flag this, and trusting those warnings saves money and misery. Many modern storage cabinets come with color-coded bins and shelving so even the busiest lab staff rarely mix things up.
Plan for the Worst, Hope for the Best
Even after following all the steps, things go sideways sometimes. Spill response kits, gloves, and eye protection sit close to any NITC stash in my lab. Routine checks catch corrosion or leaks before small problems get big. Training new staff on what to watch for—smells, stains, bulging containers—keeps everyone in the loop. Problems rarely start big; they start small, and careful eyes spot trouble early.
Chemical Safety Is Culture, Not a List
NITC isn’t going away, not with its regular use in research. Building real habits around safe storage—ventilation, the right containers, disciplined labeling, and training—keeps labs and workshops running safely. I’ve worked in places that cut corners, and others that drilled best practices into every routine. The safer shops always accomplish more. All those little steps, taken together, protect people and property, keep experiments on track, and make chemistry work for us, not against us.
What are the potential health hazards of 1-Naphthyl Isothiocyanate?
What 1-Naphthyl Isothiocyanate Is and Where People Might Encounter It
1-Naphthyl isothiocyanate pops up in chemical labs and sometimes in production settings. It isn’t something the average person bumps into at the grocery store. This compound often belongs in research spaces, especially those studying liver function. I’ve seen it handled behind layers of gloves and goggles, with fume hoods roaring to keep people safe. In academic work, it serves as a model chemical for examining liver injury.
How Exposure Happens in Research and Beyond
Direct exposure generally affects people working in scientific settings. Accidental spills, improper glove use, or leaky vials sometimes let this compound slip onto skin or into the air. A tiny amount can mean a big problem if it finds its way into someone’s mouth or lungs. Even a brief moment of carelessness around this chemical could start a chain of problems. I recall a colleague with a tiny splash on their glove, quickly washing their hands, but still reporting irritation that lingered for hours.
Impact on the Liver: Lessons from Studies and Labs
Liver toxicity stands out as the main worry. Scientists learned some of their hardest lessons from studies in rodents. Within days of exposure, animals develop cholestasis—where their livers stop moving bile like they should. It’s not just a matter of feeling queasy; the liver cells swell, bile ducts plug up, and whole systems misfire. In the human lab, symptoms might show as yellow skin or itching, but those signs stack up after deeper internal damage has taken place. I’ve seen researchers triple-check labels and containment for this reason alone.
Other Health Concerns: Skin, Eyes, and Breathing
This isn’t just a liver story. 1-Naphthyl isothiocyanate hits skin like a harsh irritant and can cause redness and discomfort. Eyes can smart and burn with even a slight touch. Dust or fumes from this chemical irritate the nose and throat, sometimes causing coughing fits or trouble breathing. Chemical safety sheets warn of its ability to trigger strong reactions in sensitive folks—but the reality is that anyone, not just the allergy-prone, can feel its sting.
The Cancer Link and Gaps in Human Data
Animal trials point to cancer risks after repeated exposures. Tumor formation often happened in livers after months of dosing. While direct human studies don’t fill this picture, what’s known about similar chemicals gives reason for caution. Regulatory agencies put it on watch lists based on these rodent results and the structure of the molecule itself, similar to other industrial toxins known for long-term harm.
What Protects People Working with This Chemical
Safety measures really define the difference between routine use and dangerous exposure. Good lab habits save skin and lungs. Full protective clothing—goggles, gloves, lab coats—aren’t optional. Proper waste disposal practices shut down risks of spreading even trace amounts. Facilities with strong ventilation systems catch stray fumes before workers breathe them in. Some labs keep a strict paper trail for every gram used, making sure clean-up leaves nothing behind.
Better Awareness and Safer Substitutes
Simple awareness goes a long way. Training sessions and clear labels help everyone spot where risk lives. Some research teams look for less toxic replacements, especially for training or routine monitoring. Early recognition of symptoms—knowing when to get medical help—cuts the worst outcomes short. Strong safety cultures protect not just those inside the lab, but the wider environment relying on what drains out of research pipes and trash bins.
| Names | |
| Preferred IUPAC name | Naphthalen-1-yl isothiocyanate |
| Other names |
1-Naphthylisothiocyanate Naphthyl mustard Naphthalene-1-isothiocyanate 1-Isothiocyanatonaphthalene Naphthalen-1-ylisothiocyanate |
| Pronunciation | /ˈwʌn-ˈnæf.θɪl aɪ.soʊˌθaɪ.əˈsaɪ.ə.neɪt/ |
| Identifiers | |
| CAS Number | 86-57-7 |
| Beilstein Reference | 1208222 |
| ChEBI | CHEBI:34712 |
| ChEMBL | CHEMBL1316 |
| ChemSpider | 11784 |
| DrugBank | DB07715 |
| ECHA InfoCard | 55408d6a-b46e-4fbb-ae0f-113cf4c091a3 |
| EC Number | 206-207-7 |
| Gmelin Reference | 808385 |
| KEGG | C06581 |
| MeSH | D009305 |
| PubChem CID | 7431 |
| RTECS number | QJ1425000 |
| UNII | 2942F2G00Z |
| UN number | UN3332 |
| CompTox Dashboard (EPA) | DTXSID2020904 |
| Properties | |
| Chemical formula | C11H7NS |
| Molar mass | 193.24 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | aromatic |
| Density | 1.161 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.90 |
| Vapor pressure | 0.0000673 mmHg at 25°C |
| Acidity (pKa) | 9.17 |
| Basicity (pKb) | 3.96 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.687 |
| Viscosity | 4.47 cP (20°C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.6 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | 205.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5177 kJ/mol |
| Pharmacology | |
| ATC code | V04CX33 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, may cause respiratory irritation, harmful to aquatic life. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P261, P264, P270, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P330, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-0 |
| Flash point | Flash point: 177 °C |
| Autoignition temperature | 500°C |
| Lethal dose or concentration | LD₅₀ Oral - rat - 3,400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 280 mg/kg |
| NIOSH | WN3675000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 0.1 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Naphthalene 1-Naphthylamine 1-Naphthol Isothiocyanic acid Phenyl isothiocyanate |

