1-Naphthonitrile: Insights, Utility, and the Road Ahead
Historical Development
The story of 1-naphthonitrile runs alongside the rise of organic chemistry as a major driver of both industry and scientific discovery. Over a century ago, chemists noticed the fascinating reactivity patterns of naphthalene, a coal tar derivative, and quickly learned that adding a nitrile group to its ring unlocked a flood of new possibilities. Early days saw painstaking extraction and isolation from crude mixtures, relying on techniques that would earn a modern chemist’s skepticism for their lack of precision. Demand for dyes, pigments, and pharmaceuticals eventually pushed researchers toward more reliable and consistent synthetic methods. The compound’s journey has always tracked the needs of chemical manufacturing, proof that the drive for better materials rarely stands still.
Product Overview
Known also as α-naphthonitrile, this compound aligns closely with the world of synthetic intermediates. Its core, a fusion of aromatic naphthalene and a nitrile group, sets it apart as both robust under mild conditions and reactive in carefully controlled situations. Companies and research laboratories keep supplies on hand as they design new molecules, especially in pharmaceuticals and agrochemicals. For those designing complicated molecular architectures, 1-naphthonitrile becomes less a product, more a reliable stepping stone.
Physical & Chemical Properties
With its solid state and slightly yellow crystals, the appearance of 1-naphthonitrile does not call much attention. It floats along with a melting point in the range of 60 to 62 degrees Celsius, a boiling point above 300 degrees, and a distinctively aromatic, sharp odor that lingers in the lab. Chemists respect its chemical stability; for most common storage practices, it holds up well, yet its nitrile group keeps it alert to transformations with nucleophiles, acids, and bases. The compound dissolves best in organic solvents like ethanol, ether, and benzene but refuses to mix into water easily. That separation of worlds comes in handy during purification steps, making workup a bit less of a headache.
Technical Specifications & Labeling
On packaging, clarity about purity levels and trace contaminants cannot be overstated. The stakes rise for applications in pharmaceuticals or electronic materials, where even small impurities introduce unexpected failures or safety questions. Standard labels stick to common identifiers, such as its CAS number and recognized synonyms, because the audience needs to know exactly what’s in the bottle. Safety and handling instructions must remain prominent, since exposure risks go hand-in-hand with the molecular structure of any nitrile.
Preparation Method
Running through standard protocols, most routes to 1-naphthonitrile tap into dehydration of 1-naphthylamine or direct cyanation reactions. Over decades, synthesis has shifted away from high-waste methods that taxed both the environment and plant operators. In recent years, transition metal catalysts, such as copper and palladium, have taken the lead, allowing for cleaner, more scalable methods. High-pressure and microwave-assisted reactions have trimmed reaction times and minimized the drudgery of long heat cycles. That shift nods to modern green chemistry principles, reflecting a serious commitment to reducing chemical footprints where possible.
Chemical Reactions & Modifications
The carbon-nitrogen triple bond of the nitrile stands as both a functional handle and a challenge, allowing for hydrolysis to naphthoic acids, reduction to amines, and substitution reactions with a host of nucleophilic partners. In lab notebooks, chemists write about transformations that seem simple in principle but demand finesse. Every change to the core structure introduces new options for further derivatization, such as forming dyes, pigments, and biologically active molecules. What keeps 1-naphthonitrile attractive is that balance of reactivity and survivability—a trait appreciated wherever repetitive chemical steps could spell disaster for weaker cores.
Synonyms & Product Names
Over the years, the name game has grown long. Alongside "1-naphthonitrile," one runs into "α-naphthonitrile," "1-cyanonaphthalene," and sometimes even a less formal "naphthyl nitrile." In academic references, name choice leans toward clarity and IUPAC standardization. For commercial products, common names, purity grades, and sometimes proprietary labels appear on packaging.
Safety & Operational Standards
1-Naphthonitrile commands respect for its moderate toxicity profile and volatile aroma. Direct inhalation or swallowing triggers irritation and, if mishandled, more severe toxicological impacts—especially in enclosed or poorly ventilated areas. Labs that handle this substance seriously prioritize gloves, goggles, and fume extraction. Regulators flag nitriles as a family for close observation, and rightfully so, with the risks extending well past the benchtop into waste streams and groundwater concerns. Waste handling routines must address not just employee safety, but also ecological consequences, signaling a wider responsibility than the lab door can contain.
Application Area
Industry taps into 1-naphthonitrile primarily for synthesis of colorants and intermediates for medicines. Some specialty polymers start from its simple ring, growing complexity through controlled transformations. Research circles use the compound to experiment with new reaction mechanisms or to build blocks for functional materials. The agroscience world leans on it for steps toward active ingredients that help farmers control weeds and insects. At the same time, pharmaceutical development often borrows intermediates built from this core to reach antiviral, antifungal, and anticancer candidates.
Research & Development
Chemists rarely stop searching for new tricks with old molecules. For 1-naphthonitrile, that means work never ends to streamlining methods, pushing selectivity, or finding routes that fit modern sustainability pressures. Teams look into cleaner catalytic cycles, solvent replacement, and integrated continuous-flow systems. Academic publications still exit presses every year, highlighting tweaks that shave off costs, improve yields, or reduce waste. The push also extends to digital chemistry, where machine learning analyzes reaction patterns to spot shortcuts that manual methods might miss.
Toxicity Research
Work with nitrile-bearing compounds always prompts health questions. Researchers have mapped metabolism, distribution, and excretion routes in mammals for 1-naphthonitrile. Studies link exposure to respiratory irritation, central nervous system effects, and, in high doses or prolonged contact, liver and kidney impacts. Regulators balance real-world exposure levels against protective thresholds, publishing recommended exposure limits and setting protocols for workplace monitoring. For now, serious accidental poisonings remain rare, but routine vigilance acts as the first and best line of defense.
Future Prospects
Looking forward, interest in 1-naphthonitrile will only grow as industries push for molecules that deliver specialized performance at lower environmental cost. Synthetic chemistry is evolving—shifting toward atom economy and solvent-less processes, which align well with how this compound can be prepared and modified. The compound’s scaffold remains ideal for fine-tuning electronic materials, new drug candidates, or advanced chemosensors. Renewed focus on green transformations and sustainable manufacturing could open fresh methods for producing—or even recycling—aromatic nitriles. As scientific priorities shift and laboratories embrace automation, the role of intermediates like 1-naphthonitrile will evolve, serving as a linchpin for innovation across multiple sectors.
What is the chemical formula of 1-Naphthonitrile?
A Closer Look at 1-Naphthonitrile
1-Naphthonitrile goes by the formula C10H7CN. This structure shapes more than textbook answers—it plays a role in real labs, industry, and everyday innovation. You can spot that CN at the end, which means a cyanide group sits on a naphthalene ring. Years spent reading up on aromatic compounds and smelling the oddly sharp aroma of nitriles in labs have shown me how a tiny shift, like the addition of a CN group, changes everything. This isn’t the gridwork of a generic chemical. It’s how chemists build pharmaceuticals, dyes, and specialty materials that show up in all kinds of essential products.
The Reason Formula Details Matter
The right formula does a lot more than win a trivia game. C10H7CN isn’t just a shorthand—it tells you how carbon, hydrogen, and nitrogen connect to create a specific mix of chemical behaviors. Knowing exact formulas helps manufacturers scale up production without toxic errors. In pharmaceuticals, tiny mistakes trigger dangerous side effects. Decades of science history, especially after some early industry blunders, stress why following the letter of the formula isn’t optional.
Folks outside the lab might glance past this, but the formula helps make standards across continents. Tracing each atom matters to regulators, safety officers, and R&D teams. I’ve worked with teams who rewrite protocols on the spot if one atom in a formula proves wrong. Suddenly, a batch worth thousands might go to waste or, worse, turn hazardous.
Digging Deeper into 1-Naphthonitrile’s Role
Plenty of industries take this formula and run with it. C10H7CN crops up in the dye industry and helps synthesize specific intermediates for pigments that color clothing, plastics, and more. In advanced pharmaceutical labs, researchers use 1-Naphthonitrile as a starting block for more complex chemical designs. Its skeleton lets scientists attach new groups in the right locations, building up drugs or agrochemicals fit for high-stakes jobs, like anti-fungals and smart materials. Amid all this, the purity that comes from knowing the formula straight up affects research outcomes. One mishap with the molecular formula means millions in wasted supplies or failed patents.
Solving Problems with Better Information
Problems crop up fast in the chemical world when folks guess about formulas. Accurate details save time, money, and keep people from harm. Testing labs often double and triple-check chemical identities, comparing base formulas like C10H7CN with sample results. If a shipment doesn’t match, it gets quarantined. There’s a huge push now for all suppliers and researchers to adopt digital verification tools. Blockchain in chemical supply chains helps track every ingredient and label, cutting down on misidentified materials.
Transparent traceability, open-access chemical databases, and updated digital labeling solve plenty of issues right at the start. Teams can catch errors early, avoiding bad batches, chemical accidents, and even lawsuits over faulty labels. This approach doesn’t just fix a single incident—it raises the whole field’s reliability, helping everyone from students to senior chemists trust what’s really in the bottle.
What are the common uses of 1-Naphthonitrile?
Industrial Dye Building
Factories often look to 1-Naphthonitrile when blending the ingredients for industrial dyes. The industry relies on compounds that deliver rich and stable color. I remember reading a trade magazine that covered how much work goes into producing dyes for everyday goods, from textiles to plastics. Quality matters for workers who want their batches to turn out the same shade every time, and 1-Naphthonitrile steps in here. Chemists use it as a starting point, connecting it with other chemicals to build up more complex dye molecules. The strong naphthalene base makes it dependable when compared with lighter aromatic compounds, reducing the risk of fading or chemical breakdown.
Pharmaceutical Manufacturing
Pharmaceutical researchers know the value of the right building block. 1-Naphthonitrile shows up in labs aiming to develop new therapeutic compounds. The chemistry can feel tricky, but the compound’s single cyano group can be swapped out or transformed, giving it a lot of flexibility. Researchers often use 1-Naphthonitrile to design molecules that target cell receptors or serve as scaffolds for anti-inflammatory or antifungal candidates. I’ve watched pharmacy students struggle with the complexity of multi-step syntheses, but this nitrile keeps things simpler when they want a naphthalene ring without extra baggage. Real-world approvals and commercialization still take years, but this chemical forms the backbone for many early-stage discoveries.
Agrochemical Synthesis
Farmers don’t always realize how complex the process is behind the weed-control products they trust. One important ingredient is a naphthalene derivative like 1-Naphthonitrile. Chemical companies use it to create several types of pesticides and herbicides because the naphthalene ring holds up well outdoors. My neighbor, whose family runs a large vegetable farm, once showed me how they tested new formula batches. They needed chemicals tough enough for sun, rain, and soil microbes, but safe for crops and end users. The stability from the naphthalene nucleus means that derivatives of 1-Naphthonitrile can hang on through these conditions, helping maintain farm productivity and manage invasive weeds.
Specialty Polymers and Materials Science
Polymer labs sometimes reach for naphthalene-based compounds like 1-Naphthonitrile when aiming to add durability and unique properties to new materials. The chemical’s rigid ring structure makes the resulting polymers less prone to breaking down under heat or stress. During my last visit to an R&D center, a materials scientist explained how small tweaks at the molecular level can change the performance of an entire product line—from stronger coatings to specialty plastics for electronics. Ingredients that bring both structural strength and chemical resistance open doors for engineers in electronics, automotive, and consumer goods.
Environmental and Safety Notes
Many industrial chemicals require careful handling. 1-Naphthonitrile is no exception. Safety data lists it as an irritant and toxic at significant exposures. Factories invest in proper ventilation, training, and emergency protocols. Communities benefit most when companies put environmental controls at the center of operations. The chemistry world needs voices combining knowledge and real front-line experience, sharing best practices on safe use and disposal. Smarter regulation and reliable data on routes of exposure can keep workers and neighborhoods safe, while allowing industries to tap into the benefits of compounds like 1-Naphthonitrile.
Is 1-Naphthonitrile hazardous or toxic?
Understanding the Basics
1-Naphthonitrile pops up in laboratories and chemical catalogs far more often than most people realize. Its chemical roots link back to naphthalene, that same stuff many folks recognize from mothballs. But this molecule swaps out a hydrogen atom for a nitrile group, changing how it behaves and how it might affect health.
Hazards in the Lab and Beyond
People handling 1-Naphthonitrile in the lab can’t ignore its risk profile. The dust and vapors that rise during weighing or heating can trigger eye and skin irritation. I’ve opened containers packed with finely ground nitriles and seen how easily static pops powders straight into the air. Without gloves, expect a nasty rash or itch after any careless touch. The compound’s smell carries a sharp edge, and that’s no surprise—nitriles rarely signal anything mild to breathe.
Inhalation may not land you in the hospital instantly, but headaches, nausea, and breathing difficulty do happen. Chronic exposure piles on the risk too. Animal studies show that long-term contact damages organs, especially the liver and kidneys. I recall colleagues scrambling to improve ventilation after one summer incident, fans whirring louder for days because one tech didn’t spot a tiny spill soon enough. Even if acute toxicity sits lower than industrial solvents like acetonitrile, the risk isn’t zero.
Toxic Effects and the Science Behind Them
Once 1-Naphthonitrile soaks through skin or slips into lungs, the body breaks it down using the same metabolic tools it uses for other cyanide-releasing chemicals. Most folks know cyanide has a rough reputation, even in trace amounts. While 1-Naphthonitrile doesn’t dump huge amounts of cyanide, metabolic byproducts strain the body’s systems, especially when exposure isn’t caught early.
The EPA and other health agencies don’t ignore these effects. Animal tests track LD50 values (how much kills half a lab animal sample) and point toward moderate toxicity. That aligns with personal care: Most chemists treat any nitrile like it’s going to hurt them if they don’t pay close attention. Carcinogenic risk remains uncertain, but nobody wants to find out the hard way years down the line.
Why Safe Handling Matters
Long sleeves, goggles, gloves, and a decent respirator or fume hood—none of these count as optional when dealing with 1-Naphthonitrile. If you think small-scale work doesn’t matter, try clearing a headache after breathing nitrile dust for an afternoon, or listening to colleagues complain about sore throats the next morning. Even minor spills can build up on surfaces. Rags and solvent wipes help, but good habits limit bigger problems.
Paths Toward Safer Use
Switching out 1-Naphthonitrile for friendlier chemicals doesn’t always fit research or manufacturing needs, but smarter engineering does. Local exhaust systems cut exposure fast. Proper waste management ranks high, since dumping these compounds down the drain slides risk onto the community and environment. Routine training refreshers and honest hazard labels keep newcomers out of trouble. Having once cleaned up after a careless tech left a bottle uncapped, my respect for solid protocols only grew.
For any group handling chemicals, transparency about risk and a focus on healthy work culture beat shortcuts every time. Detailed records make future tracking easier. Keeping first aid kits stocked, with clear instructions on what to do after a spill or splash, stays smart because nobody aims to be the case study turned cautionary tale.
How should 1-Naphthonitrile be stored?
Everyday Responsibilities in the Lab
1-Naphthonitrile carries enough risk to demand serious respect. Back in my graduate research days, one wrong move near the organics storage taught me to never underestimate how quickly a volatile compound can change the atmosphere. 1-Naphthonitrile won’t shout danger like an acid bottle, but it packs plenty of risk for anyone with skin or lungs nearby.
Trust in Data, But Always Double-Check
The safety data sheet for 1-Naphthonitrile lists harmful vapors, risk of fire, and toxicity on direct contact. This chemical falls into the category of substances you want nowhere near a heat source. I remember the sharp smell left behind even hours after a sealed bottle broke—those fumes stick around and latch onto your senses, and they signal more than just inconvenience.
Not all research spaces come with the perfect, custom-fit storage. In shared labs, folks cut corners. Some tuck these bottles into a crowded cabinet, beside bases and acids. That’s a recipe for chaos. One day of careless stacking can crack a seal, and one heat lamp out of place will do the rest.
Maximum Precaution, Minimum Drama
Direct sunlight and warmth help 1-Naphthonitrile vaporize. Storing it below 25°C slows those reactions. A well-marked, ventilated, and lockable cabinet works best. Shelves with a solid lip keep the bottle in place. Improvised storage on open shelving offers a real risk; if another person bumps into the bottle, it can drop or break. Accidents rarely make appointments—they arrive when you rush a job or cut a corner.
You also spot the difference in labs that store chemicals by hazard class. In those spaces, you never find flammable organic compounds near oxidizers. It’s a subtle point, but organizing by chemical compatibility limits the blast radius after an accident. Once, someone stacked acetone under a perchloric acid shelf, and a slow drip ended in a mess nobody wanted to clean.
The Role of Information
Clear labeling and date checks should sit at the core of storage routines. Every time I opened a bottle with an unknown date, I worried. If the seal’s broken, waste it safely. Fresh containers matter: decomposition and volatilization can create higher risks over time. Chemical cabinets require regular checks. Anyone who’s ever found a sticky, leaking bottle understands the headache of chemical residue cleanup.
Keeping a neat log of inventory goes beyond compliance. It protects the entire team and keeps the facility from sinking into disarray during inspections. When staff change over, transparent records stop the next user from stumbling into a dangerous mess.
Protecting Yourself and Others
Nobody can predict every freak accident, but clear protocols go a long way. Gloves, goggles, and a fitted lab coat won’t fix everything, but they buffer against the worst. I’ve seen how quick a glove can tear on an unnoticed chip on a bottle—double layers matter with some chemicals.
Train each user. If someone new joins, walk them through storage practices for hazardous organics. Oversight stops careless mistakes from spreading. Invest in up-to-date safety equipment—proper vent systems, spill containment trays, even a fire extinguisher built for chemical use. You can skimp on fancy software, but never on core safety tools.
Room for Improvement
Too often, storage errors come from rushing or a lack of training, not malice. Most operators just need reminders and the right tools. Investing in professional development saves far more time and money in the long haul than any shortcut. Survival in a laboratory means treating routine tasks, like chemical storage, as life-or-death practices. Nobody regrets safe habits until a problem arrives—by then it's too late.
What are the physical properties of 1-Naphthonitrile?
Getting to Know 1-Naphthonitrile
There’s something interesting about many organic compounds, but 1-Naphthonitrile has a style all its own. You see, its molecular formula, C11H7N, gives it a sturdy skeleton — a naphthalene ring with a cyano group, tucked away at the first position. It’s not some theory-hoarded chemical; it’s found use in real-world settings, from dye production to laboratory work. Ask anyone who’s spent time around benchtop glassware and you’ll get an earful about its sharp aroma and unique behavior.
The Physical Traits That Stand Out
Let’s be straightforward. 1-Naphthonitrile shows up as a white to pale yellow solid, depending on its level of purity and how much it’s been exposed to light or air. If you tried picking it up, you’d notice it feels powdery. It melts between 60 to 64°C, which you could easily hit with a water bath or a heat gun in the lab. It boils at about 306°C, far above what most folks would reach outside laboratory equipment, so it hangs on tight before turning to vapor. That high boiling point hints at strong molecular forces holding things in place — typical of aromatics with cyano groups attached.
Solubility sets the stage for practical use. 1-Naphthonitrile hardly budges in water. Splash it in and you’ll see crystals separate instead of dissolving. Its real home is in organic solvents like acetone, chloroform, or ethanol. Someone with calloused hands who’s tried to purify it will remember the challenge — filtration followed by recrystallization, often in hot ethanol, gets the job done.
Why These Details Matter
These physical properties are more than just trivia. In manufacturing, handling chemicals with a low melting point can mean easier processing. Low water solubility also means no headaches about run-off into water systems during use and disposal, though this compounds the responsibility for proper waste handling. Technicians need to know about its boiling point and volatility — 1-Naphthonitrile gives off a distinctive odor, and chronic exposure can be irritating. In tight quarters with poor ventilation, folks feel it in their throats. Health and safety protocols must be applied wisely, and that’s no place for shortcuts.
Those with hands-on chemistry experience keep an eye out for stability. 1-Naphthonitrile is fairly stable at room temperature, but heat or strong UV can break it down, triggering side reactions that complicate clean-up or product quality. Not surprisingly, those same cyano groups that make splitting from water tough also push up its persistence in the environment. Chemical hygiene and waste management practices make a difference.
What Can Be Done About Risks?
Anyone serious about safety in the workplace keeps their material safety data sheet (MSDS) close at hand. Good ventilation, protective gloves, and reliable goggles aren’t just recommendations — they’re practices learned the hard way. Facilities using aromatic nitriles benefit from well-designed fume hoods. Clean-up becomes easier when people understand the compound’s lack of water solubility and plan spills with that in mind.
Chemists and manufacturers can also keep an eye on greener alternatives, especially if large-scale use gets planned. This includes searching for chemicals with similar usefulness but lower persistence in the environment. Beyond that, proper collection and incineration of waste can cut down long-term impact, and continued study helps everyone understand just what these molecules mean for workplace wellness and the planet in the long run.
Bearing Out the Science in Everyday Work
Anyone working with 1-Naphthonitrile knows the details add up. From its powdery appearance and stubborn insolubility to its volatility and handling challenges, these traits make or break smooth lab and factory workflows. Focusing on the nuts and bolts — not just the data points — brings out a better understanding and a safer experience with this useful, if sometimes prickly, chemical.
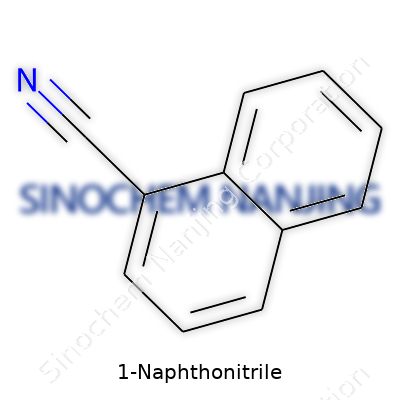
| Names | |
| Preferred IUPAC name | naphthalene-1-carbonitrile |
| Other names |
alpha-Naphthyl cyanide 1-Cyanonaphthalene 1-Naphthyl cyanide 1-Cyano-naphthalene |
| Pronunciation | /ˈnæf.θə.nɪˌtraɪl/ |
| Identifiers | |
| CAS Number | 86-53-3 |
| Beilstein Reference | 1208737 |
| ChEBI | CHEBI:35725 |
| ChEMBL | CHEMBL16110 |
| ChemSpider | 10240 |
| DrugBank | DB08352 |
| ECHA InfoCard | ECHA InfoCard: 100.018.829 |
| EC Number | 202-125-1 |
| Gmelin Reference | 52978 |
| KEGG | C06535 |
| MeSH | D014607 |
| PubChem CID | 7006 |
| RTECS number | QJ0525000 |
| UNII | 4F010789SV |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C11H7N |
| Molar mass | 129.16 g/mol |
| Appearance | White to light brown solid |
| Odor | Odorless |
| Density | 1.16 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.022 mmHg (25 °C) |
| Acidity (pKa) | 25.3 |
| Basicity (pKb) | 4.02 |
| Magnetic susceptibility (χ) | -64.0·10^-6 cm³/mol |
| Refractive index (nD) | 1.613 |
| Viscosity | 1.613 mPa·s (25°C) |
| Dipole moment | 4.02 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 206.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 272.7 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4687 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | sg%03G |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P305+P351+P338, P321, P333+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-1-0 Health:1 Flammability:1 Instability:0 |
| Flash point | Flash point: 146°C |
| Autoignition temperature | 526°C |
| Lethal dose or concentration | LD50 (oral, rat): 490 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 1040 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 25 mg/m³ |
| IDLH (Immediate danger) | IDLH: 250 mg/m³ |
| Related compounds | |
| Related compounds |
1-Naphthylamine 1-Naphthol Naphthalene 2-Naphthonitrile Naphthylacetic acid |

