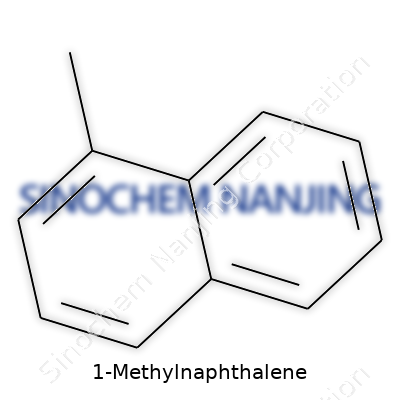
Understanding 1-Methylnaphthalene: Beyond the Basics
Historical Development
Delving into the history of 1-methylnaphthalene pulls us back to the industrial roots of aromatic hydrocarbons. Chemists started paying real attention to derivatives of naphthalene in the late 19th and early 20th centuries, thanks to the surging coal tar industry. Back then, coal tar distillation offered a treasure chest of chemicals, and 1-methylnaphthalene quickly became a point of interest for both researchers and industry. The rise of synthetic dyes and advanced organic chemistry built the groundwork for widespread use. Over decades, demand shifted with technology, but its story ties directly to growth in organic synthesis and energy sectors.
Product Overview
Most folks outside the lab might never hear of 1-methylnaphthalene, but this compound has quietly supported the progress of many modern products and experiments. As a structural cousin to naphthalene, the “methyl” group gives it distinct chemical traits. Its market today stretches from research reagents to niche industrial uses. The oil and fuel testing sectors put it to work assessing cetane numbers—basically, helping engines run smoother with the right kind of fuel blend. Scientists see it as more than just a solvent or additive; it's a key marker in performance measurements and molecular design.
Physical & Chemical Properties
On the bench, 1-methylnaphthalene presents a heavy, oily liquid form, colorless when pure but turning pale yellow as impurities sneak in or with age. Its notable point lies in its higher boiling temperature compared to naphthalene, hovering near 245°C, which means it evaporates far more slowly under ambient conditions. Smelling this compound often gives a reminder of mothballs, but more pungent and tar-like. Insoluble in water, it dissolves well in organic solvents. The stability of its aromatic rings lets it persist through many chemical processes, but under enough heat or with the right catalyst, the methyl group opens doors to more reactive sites.
Technical Specifications & Labeling
Looking at labeling and specs, regulatory guidance has shaped how chemists handle 1-methylnaphthalene. Packaging declares CAS number 90-12-0, a warning for fire risk, and instructions to store away from oxidizers and ignition sources. Industry standards demand low impurity levels, pushing for purity above 98% in most lab uses. This high bar protects both experiments and personnel from surprises during reactions. There’s special attention to accurate measurement because inaccurate dosage or unknown contaminants can spoil entire production batches or research runs.
Preparation Method
Traditionally, chemists extract 1-methylnaphthalene from coal tar, using fractional distillation and refined separation steps. In modern setups, they often employ catalytic methylation of naphthalene, choosing aluminum chloride and methyl chloride as key reagents. These steps require skill to control temperature and reaction rates, helping to limit the formation of 2-methylnaphthalene and other byproducts. After the reaction, separation and purification demand careful recrystallization or vacuum distillation, especially when pure compound fetches premium value for fuel tests and research.
Chemical Reactions & Modifications
Chemists treat 1-methylnaphthalene as a platform for creating advanced aryl compounds. That methyl group opens a window for side-chain halogenation, oxidation, and functional group swaps. Chlorination gives rise to products used in agricultural chemistry, while oxidation of that methyl can yield 1-naphthoic acid, a building block for dyes or pharmaceuticals. There’s active research into coupling reactions for new organic frameworks, targeting electronics and advanced materials. Its structure keeps things relatively selective—much more so than poly-substituted aromatic hydrocarbons.
Synonyms & Product Names
In technical language, you’ll hear 1-methylnaphthalene called alpha-methylnaphthalene, or sometimes by the ambiguous “methylnaphthalene” in less precise circles. Other names, like 1-methyl naphthalene or Naphthalene, 1-methyl-, pop up in literature and material safety data sheets, reflecting regional and company-specific naming customs. For anyone working with this compound, clarifying which isomer makes all the difference, since similar names hide real differences in behavior.
Safety & Operational Standards
Safety training drills the message that 1-methylnaphthalene brings both fire risk and health hazards. Its flash point sits above room temperature, but vapors can ignite with a strong enough spark. Skin and eye contact bring irritation; inhalation causes nausea or headaches, especially in poorly ventilated spaces. Occupational standards call for gloves, goggles, and proper fume hoods. Long-term exposure raises concerns about chronic health effects, echoing general rules for aromatic hydrocarbons. Lab managers stress proper waste disposal, as spills can linger in groundwater and soil. Emergency protocols don’t just belong on paper; chemical users need to drill them regularly.
Application Area
Most practical uses cluster in analytical chemistry, where 1-methylnaphthalene sets benchmarks for engine fuel performance. It sits as the zero point on the cetane scale, with heptamethylnonane on the other end. Testing blends of diesel means evaluating ignition characteristics, so maintaining pure reference standards becomes critical in this industry. In materials research and organic synthesis, the compound serves as both a precursor and reagent for advanced aromatic structures. Some specialty dyes, pesticides, and pharmaceutical intermediates rely on this molecule’s adaptable core, even as professionals work hard to manage associated risks and environmental concerns.
Research & Development
R&D labs keep pushing boundaries by exploring new transformations of 1-methylnaphthalene. Recent projects look at greener methods for methylation, lower-waste purification, and the development of highly selective catalysts. Innovations on the analytical side, like chromatographic detection, sharpen accuracy and speed, feeding into energy and automotive sector demands. Fundamental research follows the fate of its breakdown products in various environments, offering insight into environmental impact and potential remediation strategies. Scientists pay attention to both the raw chemistry and real-world effects, mindful of changing regulations and shifting market needs.
Toxicity Research
Research into toxicity reveals a need for caution. Animal studies point to moderate acute toxicity, with most danger arising from chronic respiratory or dermal exposure. Aromatic hydrocarbons like 1-methylnaphthalene present risks of central nervous system effects, organ damage, or carcinogenicity after repeated contact. Regulatory bodies in Europe and the US outline exposure limits and promote ongoing review as new data emerges. Scientists studying fate in the environment point to slow degradation, raising alarms for accumulation in ecosystems and possible long-term effects on wildlife and groundwater. Toxicity research keeps evolving as understanding deepens, but the message remains: control exposure, use protective methods, and monitor air and water where chemicals could build up.
Future Prospects
Looking ahead, 1-methylnaphthalene stands at a crossroads shaped by both technology and regulation. Cleaner fuel requirements and new engine designs make reference standards more vital in global automotive markets. At the same time, pressure increases for safer production, storage, and waste handling, pushing the chemical industry toward greener processes and less toxic alternatives where possible. Academics and industrial chemists look for ways to unlock more value from spent naphthalenes, aiming at sustainable cycles and higher efficiency. As environmental stewardship becomes more than just a buzzword, the responsible use and disposal of 1-methylnaphthalene will shape its relevance for years to come, challenging chemists to innovate while putting health and safety front and center.
What is 1-Methylnaphthalene used for?
What Stands Out About 1-Methylnaphthalene
Most people don’t think much about chemicals unless they work with them every day. For folks like me who’ve spent years consulting with engineers and researchers, 1-methylnaphthalene is more than just another name in a safety manual. This compound lives on the edge of several important sectors, especially in testing fuel quality and development. The story of 1-methylnaphthalene is, in many ways, the story of keeping engines honest and reliable.
Dive Into Diesel: Why 1-Methylnaphthalene Matters in Fuel Testing
Anyone working in fuel labs knows the cetane number tells you how quickly and easily a diesel fuel ignites. Curious minds developed two chemicals for cetane testing—one that burns easily and one that drags its feet. 1-methylnaphthalene plays the bad guy in this scenario, as it resists spontaneous ignition more than most substances. In my experience, every time a refinery wants to double-check diesel quality, they turn to the testing scale set up by this compound as a reference standard.
I once toured a testing lab inside a major refinery, and the lead technician joked that 1-methylnaphthalene works like the “bench press” for diesel—if your fuel fires up quicker than this stubborn chemical, you’ve got good stuff. Regulators and industry standards trace much of their measurement reliability to how this compound behaves in controlled burns.
Other Roles: From Research to Specialty Chemicals
The headlines focus on fuel, but I’ve seen industrial chemists use 1-methylnaphthalene in research as well. In laboratories, it serves as a solvent for certain organic reactions. It shows up in specialty manufacturing as a core ingredient to synthesize dyes, resins, and even some insecticides. I once spoke to a coatings manufacturer using it as a precursor for pigment development, pushing for brighter, longer-lasting industrial paints.
Its stability and aromatic nature make it attractive—not just for how it resists breaking down, but for its interactions in complex synthetic schemes. This chemical serves industries looking for specific molecular patterns that are hard to replicate with other materials.
Health, Safety, and Environmental Checks
My years working with engineers have taught me something important: every useful chemical brings challenges. 1-methylnaphthalene isn’t toxic in small doses, but like most aromatic hydrocarbons, it needs careful handling. People exposed through workplace accidents or poor ventilation report dizziness and skin irritation. Spills demand quick containment, as aromatic hydrocarbons can persist in soils and water. This reality drives ongoing efforts among environmental agencies and manufacturers to tighten controls, improve personal protective equipment, and invest in safer processes.
Researchers explore bio-based options and recovery techniques. Here is where public health and innovation meet. Some startups try to design safer analogues. Meanwhile, industry leaders push for recycling, closed systems, and better training. Reading industry reports and talking to plant workers, it is clear that driving down accidental exposure rates happens only through proactive management and constant vigilance.
Building Toward Safer and Smarter Use
Every time I hear a mechanic praise a quiet-running diesel engine, I recall the unsung role of 1-methylnaphthalene in keeping the standards clear and high. Its value shows up not in showy headlines but in dependable engines, trusted quality tests, and safer manufacturing. As more data comes in and greener solutions develop, this chemical’s story evolves—proof that the building blocks of technology deserve a closer look from all of us.
What are the safety precautions when handling 1-Methylnaphthalene?
Getting to Know 1-Methylnaphthalene
1-Methylnaphthalene flows through many labs and industrial plants. Anyone around this chemical quickly discovers its pungent, oily odor and learns its hazards aren't something to brush off. It takes just a short read through a safety data sheet to see inhalation or skin contact can mean headaches, nausea, or skin rashes. Long-term exposure rings alarm bells for liver and kidney health. Because of all this, everyone handling this chemical deserves honest, practical protection.
Personal Experience Handling Aromatic Chemicals
Spending time in research labs and industrial workplaces taught me real-world habits matter more than rules on posters. I’ve seen new employees walk through their first day thinking gloves are a hassle, until they watch a coworker’s skin break out from spilled naphthalene. Lessons like that stick. In my experience, the best protection comes from wearing chemical-resistant gloves, splash-proof goggles, and a long-sleeve lab coat or coveralls. Cotton offers some protection, but if you have access to nitrile or neoprene, don't think twice—grab those gloves.
Ventilation Isn’t Just a Box to Check
No one likes working in stuffy labs. 1-Methylnaphthalene evaporates at room temperature and releases vapors that linger. At my last job, the fume hood always ran, no matter how small the task looked. I learned to check the sash height and airflow settings myself, never trusting assumptions. Colleagues who skipped venting ended up nauseous—or with lingering headaches. If you ever get a whiff of chemical odor away from the hood, it’s time to walk back, rethink, and fix what’s wrong.
Fire Hazards: More Than Just Warnings
This compound lights up at fairly low temperatures. Stories stick with you—like the engineer who set down an open bottle near a hot plate. Room filled with smoke fast. There wasn't a fire, but it could’ve ended far worse. Keeping it away from heat sources, open flames, and sparks happens every day, not just during “special” work. Good fire extinguishers and a clear escape plan never gather dust.
Moving and Storing the Chemical
Heavy containers and cracked bottles spell spills. In my own stints at the bench, carrying bottles in secondary spill trays made cleanup manageable after a slip. Storage always means tight-sealed, labeled containers kept in cool, ventilated cabinets—never in the sun or next to anything reactive. More than once, I found mysterious fluid pooling at the bottom of a cabinet someone hadn’t checked. Never assume the last person left things tidy; always double-check the seals yourself.
Common Mistakes and Smart Solutions
People sometimes rush and pour this liquid without thinking. That’s where clear labels and regular safety reminders hold real value. I’ve seen firms use color-coded bottles and enforce double-checks on chemical IDs. This stops the mix-ups before trouble starts. Spills do happen, so I always reach for absorbent pads and a respirator before cleaning, dumping any idea of improvisation.
Cultivating a Safety-First Mindset
No practice replaces the habit of looking out for each other. Colleagues taking five minutes to help with PPE or walk through steps reduce accidents and increase trust. I learned more from open safety chats than any dry safety course. Building a relaxed, watchful attitude catches mistakes early. If something feels off—the funky smell, an unlabeled bottle, a strange-looking glove—speak up right away.
Handling 1-Methylnaphthalene always demands respect, not just rules. Practical steps rooted in real experience protect everyone, turning a risky chemical into just another tool on the shelf.
What is the chemical formula and molecular weight of 1-Methylnaphthalene?
Chemical Identity: More Than Just Numbers
For most folks, a compound like 1-Methylnaphthalene might sound like something far removed from everyday life. Truth is, a basic grasp of its chemical make-up helps us appreciate the complexity behind materials science, energy applications, and environmental science. The chemical formula for 1-Methylnaphthalene is C11H10. That means it has eleven carbon atoms and ten hydrogen atoms. Pretty straightforward, yet that arrangement defines its behavior in industries and laboratories worldwide.
Molecular Weight Isn’t Just a Stat
Molecular weight serves more purposes than just filling up a data chart. In this case, 1-Methylnaphthalene clocks in with a molecular weight of 142.20 grams per mole. Anyone working in research or manufacturing can tell you that weighing chemicals accurately keeps experiments reliable and products safe. For example, blending fuels or analyzing pollutants demands precision down to the decimal. One slip there, and the whole process can go sideways.
Why Knowing the Details Matters
Many industrial lab techs and environmental analysts depend on the numbers tied to compounds such as 1-Methylnaphthalene. The boiling point, flash point, and reactivity all depend on the structure and size of the molecule. For me, sitting through undergrad labs, nothing underscored the importance of molecular weight quite like calculating just how much material to use in a reaction or dose an instrument. Get that number off, and results turn into a guessing game.
Health and Safety: Awareness Is Key
Chemicals bring a certain risk if they’re handled carelessly. 1-Methylnaphthalene isn’t an everyday household name, but it shows up in heavy industry, especially when testing fuel quality or studying combustion byproducts. Agencies like OSHA and NIOSH have set guidelines for concentrations, precisely because these details help protect workers. I remember a friend in a fuel-testing lab who once underestimated the volatile nature of naphthalene derivatives—quick evacuation, lots of paperwork, and a lesson he won’t forget.
Environmental Perspective: Small Details, Big Impact
Even trace chemicals can make a dent in environmental health. Atmospheric chemists track compounds like 1-Methylnaphthalene to get a handle on air quality. Even in urban runoff and industrial discharge, knowing the exact formula and weight helps in tracking contamination and planning cleanups. These fine details bridge the gap between chemistry and community safety. It’s not just theory; I’ve seen local water testing efforts hinge on identifying and quantifying stubborn residuals like PAHs, of which this compound is one.
Fact-Based Solutions and Responsible Use
To reduce risks or improve efficiency, best practice comes down to precise information and careful management. Keeping up with proper labeling, storage, and disposal all depend on a rock-solid handle of chemical identity. This also means training, not just for doctoral-level scientists, but for every person who might move a drum or handle a sample. Reliable formulas and accurate weights provide the facts needed to make safe decisions. Manufacturers, regulators, and researchers can push for safer methods and greener alternatives, but none of it sticks without good data—and that starts with the basics. 1-Methylnaphthalene reminds us that chemistry’s details echo through health, industry, and the environment alike.
How should 1-Methylnaphthalene be stored?
Storing Chemicals with Care: My Take on 1-Methylnaphthalene
Most people don’t come across 1-Methylnaphthalene at home. In places where this chemical gets used, though — research labs, the petrochemical industry, or testing facilities — the way it’s kept makes a big difference for safety and quality. Over the years, I’ve seen what happens when attention slips. A leaky drum or a poorly labeled container often leads to ruined product or serious safety scares. It’s easy to talk safety, but handling real-world chemicals often exposes the details.
Understanding the Hazards
1-Methylnaphthalene isn’t your average lab solvent. It’s a flammable liquid with a strong odor, and breathing in the vapors can irritate your lungs and eyes. Spills stick around; skin contact causes redness or worse. Burning it creates black, stinging smoke. People regularly ignore how sneaky vapor release can be — this chemical can quietly fill the air in a closed space and reach dangerous levels. To avoid problems, recognize that it’ll find its way into trouble if you give it the chance.
Solid Storage Starts with the Right Containers
Using the wrong container remains one of the most common mistakes. Steel drums with tight-fitting, chemical-resistant lids hold up well, as long as they’re properly sealed and not rusty. Polyethylene lines many drums, adding a layer against leaks. Avoid using glass, which breaks easily or reacts with harsh cleaners. Decent storage also means labeling every drum with the proper chemical name and hazard symbols. Handwritten notes or taped labels fade or fall off. I once found a barrel marked only with a faded label — no one wanted to touch it, which shows how simple mistakes slow everything down.
Temperature and Ventilation: Saving Product and Health
1-Methylnaphthalene likes to vaporize above room temperature, so keeping it cool matters. Store it in a shaded, dry spot away from sunlight, heaters, or boiler rooms. Shelf life drops and vapor pressure jumps when it gets too warm, making leaks or bursts more likely. Simple thermometers help track temperatures and recognize when storage space runs too hot during summer.
Ventilation matters as much as temperature. Fume hoods or vented cabinets remove vapors before they build up, sparing staff long-term exposure and reducing fire risk. Every year, fire investigators report on accidents caused by fumes meeting an open flame or even a static spark.
Don’t Forget Compatibility and Spill Prep
No flammable chemical belongs near oxidizers, acids, or compressed gases. Mixing the wrong chemicals creates bigger problems than a single spill. A dedicated storage cabinet for flammables, grounded to avoid static, makes a reliable line of defense.
Good spill kits stand close by, packed with absorbent granules, gloves, and goggles. Most places skip regular spill drills. Practicing a quick cleanup — even just once a year — helps everyone deal with real leaks, instead of panicking when time slips away.
Continuous Checkups
Regular inspections spot rusty containers, slow leaks, or poor labels before trouble spreads. Workplace rules demand checks, but real improvement comes from building habits. Data from the Chemical Safety Board shows that consistent visual checks catch 80% of near-misses before they get ugly. Routine matters more than paperwork.
What Works Best
Simple storage habits often work better than complicated procedures. Keep chemicals in sturdy, labeled steel drums in a separate, ventilated, cool room. Organize regular checks, and never let up on practicing spill response. These small steps shield people, protect the environment, and keep the company running smoothly. The safest labs succeed not because they spend money, but because they sweat the details every single day.
What are the physical properties of 1-Methylnaphthalene?
Looking Closer at 1-Methylnaphthalene
1-Methylnaphthalene shows up in the lab as a faint yellow liquid with a strong, chemical odor. It’s a hydrocarbon with a simple structure: just a naphthalene ring and a single methyl group tucked onto it. Not many folks outside chemistry think about this material, but those who’ve handled diesel fuels or studied combustion know why it draws attention.
Boiling and Melting Points Shape Its Uses
The boiling point hovers near 245°C. That is considerably higher than water, and that extra heat tolerance matches up with its role in testing diesel cetane values. Pouring it out of a bottle, you notice it resists vaporizing unless pushed. Its melting point sits much lower, around -22°C. At most room temperatures, it flows freely, letting you handle it as a liquid rather than warming or cooling the container.
Solubility, Density, and What That Means in the Field
It flatly refuses to mix with water, floating there as a separate layer if you try. The density, about 1 gram per milliliter, means it feels a little heavier in your hand compared to many solvents—but it’s about the same as diesel. I’ve watched samples poured into test tubes, seeing them form distinct layers, with water at the bottom and a thick band of 1-methylnaphthalene right on top. In practice, workers spill a little and see it bead up on a benchtop, easy to wipe away, not dissolving or spreading like alcohol would. It likes dissolving in organic solvents, especially aromatic ones, so you find it mixing easily with benzene, toluene, and similar liquids. That’s part of why it survives through the refining process and why chemists can safely blend or separate it from close relatives.
Flash Point and Handling Realities
The flash point, usually around 114°C, demands respect. A hot metal tray or open flame makes it catch fire even if it doesn’t look angry. In a refinery or lab, this property sits front and center—anyone nearby needs to keep sources of ignition locked down. I remember training sessions where we compared 1-methylnaphthalene to lighter aromatics. Its higher flash point makes it a little safer than some compounds, but there’s no relaxing. Airflow, storage temperature, and clean procedures matter just as much with this liquid as with any flammable solvent.
Vapor Pressure and Air Quality
The vapor pressure doesn’t spike at room temperature. There’s much less of it moving into the air than, say, gasoline. Still, ventilation stays important. The strong, tar-like smell gives away its presence the instant you open a bottle, and inhaling those fumes long-term poses risks. I’ve seen folks underestimate this—windows open, hood running, but the odor lingers. Experience teaches respect for these fumes, especially since chronic exposure connects to headaches and even longer-term health trouble. Regulatory agencies have flagged risks and recommended strict handling procedures as a result.
Why Physical Properties Matter Day-to-Day
1-Methylnaphthalene’s physical quirks shape its story: strong odor, solid heat stability, reluctance to mix with water, and enough flammability to require real safety protocols. The density and solubility make it useful in testing fuels and painting an honest picture of how diesel will behave under the hood. Less attention gets paid to its everyday risks—the fire hazard, potential exposures, and lingering contamination. The solution often lies in rethinking storage and lab habits and in updating safety measures to match what the material can actually do, not what the paperwork says. Experience from the floor suggests that small adjustments in handling, ventilation, and cleanup turn the difficulties of 1-methylnaphthalene into something manageable and predictable, not mysterious. Proper labeling, employee education, and ramped-up personal protection keep accidents rare, and those are fixes anyone in industry or research can put in place without waiting for outside rules to change.
| Names | |
| Preferred IUPAC name | 1-methylnaphthalene |
| Other names |
1-Methylnaphthalin alpha-Methylnaphthalene 1-Methyl-naphthalene 1-Methyl naphthalene |
| Pronunciation | /ˈwʌnˌmɛθilˈnæfθəˌliːn/ |
| Identifiers | |
| CAS Number | 90-12-0 |
| Beilstein Reference | 528078 |
| ChEBI | CHEBI:36053 |
| ChEMBL | CHEMBL14323 |
| ChemSpider | 6923 |
| DrugBank | DB01878 |
| ECHA InfoCard | 100.011.287 |
| EC Number | 202-204-7 |
| Gmelin Reference | C 10 |
| KEGG | C02342 |
| MeSH | D008613 |
| PubChem CID | 7029 |
| RTECS number | RN3675000 |
| UNII | D0X6KVF51U |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID4020708 |
| Properties | |
| Chemical formula | C11H10 |
| Molar mass | 142.20 g/mol |
| Appearance | Colorless to yellowish oily liquid |
| Odor | Aromatic |
| Density | 1.02 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 3.87 |
| Vapor pressure | 0.067 mmHg (25 °C) |
| Acidity (pKa) | 12.85 |
| Basicity (pKb) | 14.66 |
| Magnetic susceptibility (χ) | '-77.8×10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.616 |
| Viscosity | 2.97 cP (25°C) |
| Dipole moment | 0.34 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 14.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6110 kJ/mol |
| Pharmacology | |
| ATC code | V19CA02 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS08 |
| Signal word | Warning |
| Hazard statements | H226, H304, H315, H319, H335, H336, H351, H411 |
| Precautionary statements | P210, P273, P280, P301+P310, P302+P352, P305+P351+P338, P308+P313, P403+P235 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 79 °C |
| Autoignition temperature | 525 °C |
| Explosive limits | Explosive limits: 0.8–5.0% |
| Lethal dose or concentration | LD50 oral rat 1840 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 1-Methylnaphthalene: 1,840 mg/kg (oral, rat) |
| NIOSH | RN1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1-Methylnaphthalene is "No OSHA PEL established |
| REL (Recommended) | 75 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
2-Methylnaphthalene Naphthalene Acenaphthene Anthracene Phenanthrene |

