Looking at 1-Methylisoquinoline: More Than Just a Chemical Curiosity
Historical Development: Echoes from the Laboratory
The history of 1-Methylisoquinoline runs closely with the exploration of heterocyclic compounds, which saw a surge in attention through the nineteenth and twentieth centuries. Chemists ventured into the world of alkaloids, stumbling on structures like isoquinoline that showed up in nature, especially in medicinal plants. The methylated version, 1-Methylisoquinoline, came into sharper view in the context of just how tinkering with molecular structures grew into a playground for drug discovery. Decades ago, researchers built on classic nitration, methylation, and cyclization techniques, inching closer to reliable ways to make molecules like this. These days, any chemist serious about heterocycles recognizes the value of landmarks like isoquinoline derivatives for both medicinal and materials research.
Product Overview: More Than Meets the Eye
Stacked on the shelves of organic chemistry labs, 1-Methylisoquinoline might not draw the crowds that big pharmaceuticals do, but it plays a quiet role in exploratory chemistry. Its core lies in an isoquinoline scaffold with a methyl group tucked at the first position, a tweak that subtly shifts its chemical temperament. Industry and academic circles keep the compound on their radar because structural relatives have yielded several drugs and natural products. Experience shows that simple methylation often changes how a molecule interacts with biological systems, bringing out quirks that researchers love to puzzle over.
Physical & Chemical Properties: Character You Can Measure
1-Methylisoquinoline doesn’t linger much in the spotlight, but its physical traits deserve a close look. In the flask, it appears as a pale to colorless liquid at room temperature, sporting a melting point just below typical lab conditions and boiling at around 243–247°C. The faint, slightly sweet odor makes it stand out compared to harsher amines or pyridines. The methyl group added to the heterocycle pushes the molecule’s lipophilicity, which sometimes makes it easier to handle in organic solvents. The compound resists oxidizing agents more than some other isoquinolines, thanks in part to the stability the methyl group brings. Solubility in ethanol, ether, and similar organic solvents helps in synthetic workups, but water solubility remains low, nudging chemists toward organic extractions for purifications.
Technical Specifications & Labeling: What Sets It Apart in Stockrooms
Working in synthetic chemistry, one picks up a habit of checking bottles carefully. Pure 1-Methylisoquinoline sports a CAS number of 1739-84-0, a unique identifier for inventory and safety purposes. Material labels in reputable labs always highlight purity percentages, given some applications call for near-spectroscopic grade. Any reputable supplier provides NMR and GC-MS traces for researchers who want peace of mind before firing up a reaction. With light sensitivity recorded in some references, proper storage in amber glass keeps the compound stable. Clear hazard and handling text always tags the bottle, with pictograms that signal flammability and acute toxicity, in keeping with established laboratory protocols.
Preparation Method: Tinkering in the Lab
Chemists, myself included, can appreciate the mental gymnastics behind making 1-Methylisoquinoline. Traditional routes lean heavily on the Bischler–Napieralski cyclization method, where starting with an appropriately substituted phenylethylamine gives up the core ring. Methylation at the one-position carried out with methyl iodide and a strong base in a dry, inert atmosphere usually gets the job done. Careful control of reaction conditions—like dropping the temperature or picking a solvent that minimizes unwanted byproducts—leads to better yields. For researchers aiming for greener chemistry, one-pot reactions and microwave-assisted syntheses pop up in recent literature, each step sparking a little bit more efficiency or safety. Troubles with over-alkylation or demethylation show up from time to time, reminding everyone that even small changes in the procedure call for close monitoring and plenty of TLC spotting.
Chemical Reactions & Modifications: A Flexible Building Block
The beauty of a scaffold like 1-Methylisoquinoline comes through in its reactivity. That methyl group offers a useful handle for further functionalization, through halogenation or even oxidative processes that switch out the group for other moieties. Nitration tends to target the benzene part of the molecule, while sulfonation steers clear of the methylated nitrogen area. A favorite trick in our lab has been cross-coupling the compound with aryl halides, opening a door to custom-designed analogs. Hydrogenation targets the quinoline ring system and can produce tetrahydroisoquinolines, which have taken off in drug discovery circles. Companies playing in the pharma space eye such modifications as a pathway to turning simple scaffolds into new leads.
Synonyms & Product Names: More Than One Label
Ask different chemists and you might hear names like 1-Methyl-isoquinoline, alpha-Methylisoquinoline, or even N-Methylisoquinoline, depending on naming conventions, literature references, or the quirks of suppliers. These synonyms reflect the shifting trends in chemical nomenclature and speak to just how connected global research has become. Outside journal articles, some older product catalogs will revert to historic names based on the time period or regional tradition, which can trip up trainees who didn’t cut their teeth in the stacks of dusty chemical handbooks.
Safety & Operational Standards: Getting the Job Done Without Surprises
Years in the lab underline an important lesson: underestimating chemical hazards leads to accidents, not breakthroughs. 1-Methylisoquinoline calls for chemical goggles and gloves, with ventilation a must during distillation or prep work. The compound triggers mild to moderate irritation if it comes into contact with skin or mucous membranes, enough to make proper PPE non-negotiable. Inhalation of vapors during solvent removal or under inert atmosphere setups poses particular risks, so labs lean heavily on fume hoods. For larger scale work, spill kits and fire suppression gear keep hazards in check, as the compound can feed a solvent fire. Following chemical hygiene standards, waste handling sticks to strict protocols, with spent reagents destined for special disposal, not the sink. Training sessions on new chemicals, including 1-Methylisoquinoline, go beyond reading an MSDS. They build up lab-wide awareness that even “routine” chemistry means coordination and preparedness.
Application Area: What Makes It Useful
Chemists across fields tend to use 1-Methylisoquinoline as a starting material in complex syntheses. The compound’s profile aids in building up functionalized molecules for everything from pharmaceuticals to specialty dyes. In medicinal chemistry, the methyl group provides a way to tweak molecular interactions, sometimes leading to improved bioavailability or altered pharmacokinetics. Outside drug discovery, research teams have dabbled in using it to make ligands for metal complexes, setting the stage for advances in catalysis. Some segments of analytical chemistry value it as an internal standard, exploiting its unique mass and structure in method development. Each area draws something different from the compound’s versatility, but it rarely stands alone as an end product.
Research & Development: Where the Hopes Lie
In my experience with heterocyclic synthesis, few things excite a research group more than new analogs pieced together from simple scaffolds. The path from base compound to innovation runs through repeated testing and fine-tuning. Projects using 1-Methylisoquinoline frequently focus on building libraries of derivatives, screening them for biological activity against tough targets like kinases, GPCRs, or antimicrobial enzymes. Advances in high-throughput screening and machine learning speed up the identification of promising hits. Some projects pair this scaffold with modern click chemistry or photoredox methods, searching for leapfrog advances rather than incremental change. Funding agencies look for transformative potential, and researchers who showcase robust synthetic routes starting with 1-Methylisoquinoline often find a receptive audience.
Toxicity Research: Not Just a Footnote
Work with 1-Methylisoquinoline reminds chemists not to overlook toxicity, even when dealing with largely “routine” organic molecules. Acute toxicity studies show that the compound produces central nervous system effects in animal models at higher exposures, which matches expectations for aromatic amines and heterocycles. Prolonged exposure raises additional concerns, leading labs to err on the side of caution with chronic exposure limits. No widespread use in therapeutics means tight toxicity data aren’t always readily available, but related methylated isoquinolines have shown both mutagenic and carcinogenic potential in some contexts. Researchers looking at environmental impact track degradation products in wastewater streams, stressing the value of green chemistry and containment protocols. Institutional review boards expect a high bar for safety, and project leaders who ignore this side of the compound tend to get pushback from oversight committees and colleagues alike.
Future Prospects: What Comes Next?
Looking at how research priorities shift, the future for 1-Methylisoquinoline stands tied up with advances in medicinal and materials chemistry. The push for sustainable, green syntheses only grows stronger, and the challenge of streamlining heterocyclic production without using hazardous reagents brings out creativity in even the most seasoned chemists. The compound’s straightforward reactivity, paired with rapid exploration of analogs in pharmaceutical research, sets the stage for potential breakthroughs. AI-based modeling tools now make it easier to predict which tweaks to the scaffold might unlock new properties. Environmental considerations, such as better ways to treat and recycle waste from its preparation and use, nudge the field toward greater responsibility. Teams that treat 1-Methylisoquinoline as a launchpad—rather than an end in itself—are likely to uncover new applications, crop up in patent filings, and give shape to the next wave of molecular innovation.
What are the main uses of 1-Methylisoquinoline?
Applications in Drug Development
1-Methylisoquinoline shows up in labs more often than you might think. Chemists appreciate its structure because it lends itself to building complex molecules, especially when looking to make active pharmaceutical ingredients. Small changes to the isoquinoline core sometimes lead to medicine for pain, high blood pressure, and certain neurological disorders. In my own research circles, colleagues often hunt for new ways to tweak basic isoquinoline rings, hoping to hit on fresh drug leads. As companies chase after new therapies, the versatility of 1-Methylisoquinoline keeps it in demand for medicinal chemistry projects.
Role in Agrochemical Discovery
Farmers face a tough job, fending off pests and boosting crop yields. Chemical companies keep screening different molecules for promising activity. 1-Methylisoquinoline acts as a building block here as well, helping scientists design new herbicides or insecticides. Decades of agricultural progress owe a nod to heterocycles like isoquinolines, because changing one chemical group on a core like this sometimes shifts a compound from useless to essential. 1-Methylisoquinoline serves as a toolkit staple any time innovation in crop protection comes up.
Building Specialty Chemicals and Materials
Chemical manufacturers use 1-Methylisoquinoline as a way to make dyes, pigments, and other functional additives. The structure makes it possible to modify light absorption or give stability to some specialized polymers. Over the years, teams in specialty labs have found 1-Methylisoquinoline handy as a base for colorants or as a stepping-stone to more complex chemical products. Its role in creating these specialty materials doesn’t get as much press as the pharmaceutical angle, but it helps round out the value of this compound.
Use in Academic and Industrial Research
Many chemists first encounter 1-Methylisoquinoline as a model compound when learning advanced synthesis or testing new catalytic reactions. During my graduate work, the compound helped teach me about C-H activation and functional group transformations. Research teams in universities and companies often turn to it for testing reaction mechanisms or seeing how catalysts perform. This makes it a favorite in teaching labs and industrial screening projects as well.
Safety Concerns and Handling
Anyone using 1-Methylisoquinoline ought to wear gloves and eye protection and keep workspaces ventilated. The compound can irritate the skin or eyes on contact, and breathing in vapors causes headaches and dizziness. I’ve seen some people skip safety routines to save time, but that only brings risk. Standard lab safety practices and thorough training should always come before any use of volatile organics like this one.
Room for Improvement in Sustainability
Making and disposing of chemicals like 1-Methylisoquinoline raises questions about environmental impact. Many traditional methods require toxic solvents or produce waste that’s tough to manage. Switching to greener routes, such as using renewable raw materials or developing safer synthesis, brings real benefits. There’s room for researchers to design cleaner pathways so that chemical building blocks like this one fit better with global sustainability goals. Investing in these improvements not only helps the planet but also supports safer workplaces and communities.
What is the chemical formula and structure of 1-Methylisoquinoline?
Understanding 1-Methylisoquinoline: More Than a Formula
Chemistry books have a habit of giving us molecule names and formulas, but behind those letters and numbers, there’s a world to discover. Take 1-Methylisoquinoline, for example. This compound, with the chemical formula C10H9N, shows up as a small tweak to a centuries-old aromatic system. By attaching a single methyl group to the first position of the isoquinoline backbone, it brings a small but significant shift in chemical personality. I’ve looked at a lot of heterocyclic molecules in the lab, and that little methyl group—just one carbon and three hydrogens—really can make all the difference.
The Structure: A Closer Look
Aromatic rings seem simple at a glance, but a look at them under a molecular model reveals how small changes influence reactivity and behavior. Isoquinoline itself resembles naphthalene at first, fusing a benzene ring to a pyridine ring. Swap out one hydrogen on the nitrogen-containing ring—the one at the “1” position—and add a methyl group there. Draw it on paper and you get two rings fused together, with a -CH3 sticking off the first carbon next to the nitrogen. That methyl group at the first position blocks certain types of chemical attacks and nudges the molecule into new kinds of reactions. Such changes turn a standard compound into a tool for building more complex pharmaceuticals, dyes, or catalysts.
Why Structure Matters in Practice
Research and manufacturing both care deeply about the way a molecule fits together. Tinkering with the isoquinoline framework by adding a methyl at the first carbon changes everything from how it smells to what it can become. For instance, medicinal chemists often lean on derivatives like this when searching for anti-inflammatory or anticancer agents. One reason this strategy works comes down to stability and electronic properties. That methyl group adds extra electron density, making certain reactions easier and others tougher. You start to see this in the way 1-Methylisoquinoline behaves in the lab. It resists strong acids better than the parent compound and can form interesting complexes with metals.
Responsible Discovery and E-E-A-T Principles
Real expertise in chemistry means understanding not just a formula but what it means in the real world. Accuracy matters, and so does experience working with real samples in a real lab. Trust comes from consistently seeing how textbook concepts play out, whether preparing small batches for research or looking at safety data sheets when handling aromatic amines. Transparency includes discussing both the benefits and risks. 1-Methylisoquinoline gets mentioned in the context of dyes, drug synthesis, and material science, but it also appears in discussions about safe handling and environmental impact. Chemists rely on reputable sources, peer-reviewed data, and firsthand experience when working with such molecules.
Possible Applications and Responsible Use
1-Methylisoquinoline fits into a world of possibilities ranging from pharmaceuticals to industrial chemicals. The value lies in understanding both the promise and any pitfalls. Thorough research and careful process design lead to discoveries that help, not harm. Chemists with solid backgrounds contribute to safer workplaces and greener manufacturing. Sharing that practical insight builds public trust and advances responsible science.
Is 1-Methylisoquinoline hazardous, and what are the safety precautions?
The Nature of 1-Methylisoquinoline
1-Methylisoquinoline belongs to a group of chemicals called heterocyclic aromatic compounds. You’ll find it in labs that specialize in organic synthesis and sometimes in the pharmaceutical industry as a building block. This compound, like many in its category, has raised safety questions, especially for people who work with chemicals every day.
Hazards in Handling and Exposure
Anyone handling 1-Methylisoquinoline should understand that this isn’t just another bottle on the shelf. It gives off vapors that can cause respiratory discomfort and irritation to the eyes and skin. A strong chemical smell makes its presence tough to ignore. During graduate research, I remember colleagues working with similar isoquinolines—one neglected gloves during a hurried transfer and paid for it with a rash and persistent itch that stuck around. Even mild exposure can create big problems, especially after hours in a closed space.
Toxicology studies place this molecule on the “handle with caution” list. Animal studies haven’t declared it the most dangerous in the lab, but repeated exposure builds up. Some evidence suggests these nitrogen-based compounds may disrupt cellular function or cause longer-term organ stress. If spilled, it won’t just evaporate harmlessly. Liquid can stain benches, and residue lingers long after you think clean-up is done. The point is, even low-level exposure stacks up, and nobody wants to gamble with their health for the sake of convenience.
Sensible Safety Steps
I can’t count the number of times I’ve seen workers skip goggles or ignore the fume hood. Too many folks trust their habits over the science. For 1-Methylisoquinoline, the basics matter: gloves, goggles, a well-fitted lab coat—and keeping that reaction vessel under a hood. Standard nitrile gloves work for brief encounters, but change them if any splash or spill happens. Even a tiny leak out of a bottle creates a strong smell that seeps into clothing, especially after accidental drips.
Fire hazards often sneak up—this compound burns readily, releasing toxic fumes. Proper storage matters as much as protective gear; tuck it away from heat, out of direct sunlight, and never in large quantity beside oxidizers or acids. During one undergraduate project, improper cap sealing led to vapor buildup that almost knocked out a whole section of the storeroom. A reminder: don’t trust any chemical to stay put just because the bottle looks clean.
Information and Training—More than a Checklist
Relying on printed labels or minimum guidelines isn’t enough. To meet standards and build a real safety culture, employers must invest in hands-on training and refreshers—especially before new staff enter the lab. Digital safety sheets help, but walking through spill responses and real-alarm drills leaves a bigger impression. Exposure monitoring shouldn’t get brushed off as busywork. Folks should monitor work surfaces and, once in a while, air quality. If anything goes wrong—chemical in the eyes or on the skin—emergency stations must stay unlocked, clear, stocked, and regularly tested.
Seeking Safer Alternatives
Chemists hunt for alternatives to harsh chemicals, but sometimes a reaction won’t work with other building blocks. If 1-Methylisoquinoline must stay on the bench, the goal becomes strict control, strong oversight, and open conversation about any near-miss or incident. Passing off this step as “routine” welcomes mistakes, and in a lab, even small mistakes have real cost. Small changes—sharper focus on habits, updated checklists, and regular gear checks—lay the groundwork for fewer injuries and a healthier workplace.
How should 1-Methylisoquinoline be stored and handled?
Treating Chemicals With Respect
Working with chemicals in the lab is part of my daily routine, and experience teaches you quickly: small oversights cause big problems. 1-Methylisoquinoline looks unassuming, but nothing is worth risking the health of you or your team. I think back to my first year in a shared lab, where a careless cap left the air thick with solvent fumes. Nobody appreciates a migraine and an emergency evacuation. That’s why handling any organic solvent, methylisoquinoline included, always starts with awareness and planning.
Storage: Keep It Cool, Dry, and Secure
1-Methylisoquinoline is flammable, so letting bottles sit near a window or on an open bench invites trouble. Set aside a space inside a metal, flame-proof safety cabinet for all volatile organics. Keep the temperature stable and away from any strong sources of heat, sparks, or open flames, since vapors might catch fire before you know it’s happening.
Humidity tends to ruin reagents and may trigger strange chemical changes. Every time I’ve stored organics in a dry, well-ventilated place, bottles last longer and I haven’t lost valuable stock to contamination. Seal bottles tightly after each use, since even brief contact with air speeds up degradation.
Label Every Container Clearly
You’d think every scientist would label bottles, but in the rush of a long day, skipping the marker happens. Think about what matters if a spill or accident occurs. Emergency teams, coworkers, or students need to know what they’re dealing with. Write out the full chemical name, concentration, hazards, and the date received. I’ve seen colleagues avoid accidents thanks to simple, clear labeling and would never handle any compound without that habit.
Use Strong Personal Protection
There’s a temptation to rush a transfer and skip the gloves. Stay cautious—liquid organics, including methylisoquinoline, often absorb fast through skin and may cause irritation or worse. Nitrile lab gloves form a reliable barrier, much better than latex for most solvents. Wear safety goggles to protect your eyes from splashes; a minor slip can turn into an urgent medical situation.
Use a fume hood for handling and pouring. I’ve underestimated the harsh odor and potent vapors before, and quickly learned my lesson—ventilation stops exposure before it starts. In shared spaces, communicating with others about which hoods you’re using helps prevent cross-contamination or surprise encounters.
Handling and Cleanup
During my early days, a cracked bottle on a cluttered bench left me scrambling for absorbent pads. Always check the integrity of storage containers before opening or moving them. If you spot leaks or damage, transfer the liquid into a new, clean container quickly and record the incident. Keep spill kits within reach, and rehearse simple cleanup procedures so you act fast if anything goes wrong.
Dispose of waste following institution protocols and never pour leftovers down the sink. Collect used solvent in well-marked, sealed containers for professional disposal. This step protects both people and the environment, stopping hazardous runoff before it becomes a problem.
Building Safer Habits Together
Chemicals pose risks, but clear habits, training, and respect for what you handle keep work safe and productive. With 1-Methylisoquinoline—like any organic compound—routine matters more than bravado. Sharing knowledge with new team members and reviewing storage practices regularly turns safety from a box-ticking exercise into a shared responsibility.
Where can I purchase 1-Methylisoquinoline and is it available in different purities?
The Hunt for 1-Methylisoquinoline
Anyone searching for 1-Methylisoquinoline usually brings a solid reason to the table. This compound often finds its way into research labs and chemical manufacturing plants. An organic chemist once told me about spending hours poring over supply catalogs looking for just the right purity for a new process—the sort of headache that anyone in synthetic chemistry knows too well.
Where Does One Buy 1-Methylisoquinoline?
For most people outside the industry, buying specialized chemicals sounds like something only big companies do. Yet, plenty of chemical suppliers cater not only to research institutions but also to independent researchers and small biotech startups. Sigma-Aldrich, Thermo Fisher Scientific, and TCI America stock 1-Methylisoquinoline. You will see it listed in their online catalogs. They show purity right alongside the product, which helps buyers make quick decisions.
Dealing with a reputable supplier matters. Counterfeit or contaminated chemicals can derail experiments catastrophically. I learned the hard way some years back. A colleague received a “good deal” batch of a reagent from an unknown distributor, only to face failed reactions and a mess of troubleshooting. Established suppliers run stringent quality checks, and their certificates of analysis back up every batch.
Purity: What’s the Big Deal?
Purity might seem trivial to outsiders, but a fraction of a percent can swing a result. Research-grade 1-Methylisoquinoline usually ships at 95% or greater purity. Some suppliers push into the 98%+ range, especially for analytical labs where trace impurities interfere with sensitive equipment. For industrial settings, cost sometimes trumps purity, so you might see batches at 90–95%, especially if downstream processing will clean it up later.
Sometimes, only a pharmaceutical-grade material cuts it. That’s a different game altogether. Pharmaceutical manufacturers often work directly with custom chemical producers to validate every step, document traceability, and run frequent audits. Even trace impurities must be identified to ensure patient safety.
What Gets in the Way?
Regulation and shipping rules cloud the process. 1-Methylisoquinoline isn’t on the lists of controlled substances that gum up so many chemical purchases, but it’s still subject to hazmat regulations. Shipping hazards, import duties, and documentation sometimes slow things down, especially if buyers need rapid turnaround or delivery outside the main supply chains.
Sometimes, buyers hit minimum order size restrictions. Not everyone needs a multi-kilogram drum—graduate students and independent chemists groan at this sort of barrier. Some suppliers do offer “research quantities” in grams, at a premium compared to industrial volumes.
Safer, Smarter Buying
Anyone buying 1-Methylisoquinoline for the first time should check for a certificate of analysis, look up the supplier’s reputation, and clarify shipping and handling costs upfront. It pays to call and speak with technical support. The people on the other end often steer chemists around pitfalls: out-of-stock lots, problems with shelf life during long shipping times, or misunderstandings about purity needs.
While online ordering has made these chemicals accessible to a wider group, it’s smart to approach every purchase with care. Getting the right compound at the right purity sets the stage for safe, credible research and production.
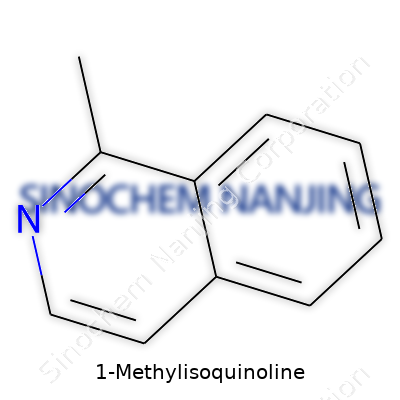
| Names | |
| Preferred IUPAC name | 1-Methylisoquinoline |
| Other names |
Isoquinoline, 1-methyl- 1-Methyl-isoquinoline NCI-C56498 Isoquinoline, methyl- 1-methylisoquinoline |
| Pronunciation | /ˈwʌnˈmɛθɪlˌaɪsəʊˈkwɪnəliːn/ |
| Identifiers | |
| CAS Number | 1721-52-2 |
| Beilstein Reference | 1340734 |
| ChEBI | CHEBI:15338 |
| ChEMBL | CHEMBL134003 |
| ChemSpider | 16712 |
| DrugBank | DB04208 |
| ECHA InfoCard | EC#: 207-965-4 |
| EC Number | 1.2.3.118 |
| Gmelin Reference | Gmelin 2052 |
| KEGG | C14670 |
| MeSH | D015859 |
| PubChem CID | 7069 |
| RTECS number | NL8925000 |
| UNII | 30655G6P77 |
| UN number | UN3439 |
| Properties | |
| Chemical formula | C10H9N |
| Molar mass | 143.19 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | aromatic |
| Density | 1.06 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 2.28 |
| Vapor pressure | 0.156 mmHg (25 °C) |
| Acidity (pKa) | pKa = 5.42 |
| Basicity (pKb) | 5.20 |
| Magnetic susceptibility (χ) | -68.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.612 |
| Viscosity | 0.982 cP (20°C) |
| Dipole moment | 2.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 185.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 49.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5464 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H411 |
| Precautionary statements | Precautionary statements for 1-Methylisoquinoline: "P261, P264, P271, P301+P312, P304+P340, P312, P330, P405, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 0, Special: - |
| Flash point | 93°C |
| Autoignition temperature | 850°F (454°C) |
| Explosive limits | Explosive limits: 1.3–7.5% |
| Lethal dose or concentration | LD50 (oral, rat): 670 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 1-Methylisoquinoline: 533 mg/kg (mouse, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200 mg/L |
| Related compounds | |
| Related compounds |
Quinoline Isoquinoline 2-Methylisoquinoline 3-Methylisoquinoline 1,2,3,4-Tetrahydroisoquinoline |

