1-Hydroxycyclobut-1-Ene-3,4-Dione: More Than Just Another Cyclobutene Derivative
Historical Development
Chemists have always chased after molecules with strange ring systems, and cyclobutene-based compounds draw curiosity like bees to pollen. The story of 1-Hydroxycyclobut-1-Ene-3,4-Dione started in the late 1970s, when research groups first managed to isolate the parent scaffold of this molecule inside chilly laboratories lined with glass columns and endless spiral notebooks. Early synthesis efforts exposed just how unforgiving the cyclobutene ring can get under experimental pressure. Chemists learned quickly that even minor tweaks with oxidizing agents or water would slam this system into wild, unpredictable rearrangements. Some graduate students burned through years of work and more than one expensive glovebox just teasing out stable intermediates. These chase-down moments fueled publications and late-night lab debates, building up a reputation for this molecule as something of a white whale — rare, beautiful, and a lesson in humility for those trying to push aromatic chemistry further than before.
Product Overview
The main attraction of 1-Hydroxycyclobut-1-Ene-3,4-Dione comes from its tension between a stubborn four-membered carbocycle and the energetic push of its diketone and hydroxy groups. You can’t really get away from its bright yellow hue when it’s pure, suggesting a conjugated playground for electrons. This isn’t just academic — researchers learned the hard way that you don’t leave a vial under the fume hood light longer than you have to, or you’ll come back to find crusty decomposition. Most labs keep their stock locked tight in amber bottles, running analyses as quickly as possible. It can’t claim the glamour of household-name molecules, but serious synthetic chemists get a certain satisfaction spotting it on a TLC plate.
Physical & Chemical Properties
Let’s talk about real-world handling. 1-Hydroxycyclobut-1-Ene-3,4-Dione sits as a solid at room temperature, but too much warmth or stray moisture turns it restless. The notable chemical properties stem from the strained ring, so its carbonyl stretches stand out like a siren in an IR spectrum. There’s enough polarity that a water droplet starts a quick breakdown spiral, releasing dicarbonyl byproducts before you can grab a pipette. The hydroxy group flirts with hydrogen bonding, and the ring strain keeps the molecule on edge, ready to pop open under the right conditions. Researchers who’ve spent time purifying it usually mention its strong, dry scent and the crust it leaves behind, unwilling to let glassware return to a glossy finish. Rationalizing its temperament requires a solid understanding of both physical organic chemistry and good lab hygiene.
Technical Specifications & Labeling
Precision in labeling isn’t just bureaucracy when it comes to 1-Hydroxycyclobut-1-Ene-3,4-Dione. Handling a molecule prone to rearrangement and hydrolysis puts a burden on accurate documentation. Miniscule impurities mess up NMR readings and spoil reactions, so packaging always involves examining certificates of analysis, a reliable chain-of-custody log, and evidence of controlled atmospheric conditions during shipping. Labs that cut corners eventually regret it — any leftover solvent or oxygen lingers inside the bottle, turning an expensive sample into yellow goo. Smart researchers go for small aliquots, seal with argon, and avoid labeling shortcuts. Once, I saw a postdoc mark the bottle simply as “cyclobutene-dione,” which led to a whole wasted series of reaction attempts and a frustrated group meeting.
Preparation Method
The path to 1-Hydroxycyclobut-1-Ene-3,4-Dione has never promised an easy ride. Early synthetic routes took a sledgehammer approach with ozonolysis and heavy-duty oxidizers, but yields barely made it past the single digits and the process loved to produce tar. More recently, stepwise ring contraction and clever use of protecting groups have improved the odds, but it still demands patience and time. Scalability remains tough — batch sizes rarely exceed a few grams in most academic labs. In my own work, seeing the product crystallize after a week of slow rotary evaporation brought a sense of triumph, but only after counting countless losses from hydrolysis. Commercial suppliers usually hinge their processes on proprietary tweaks, and chemists know to trust certain trade names over others after experiencing enough failed reproductions.
Chemical Reactions & Modifications
Few molecules prove as versatile and stubborn in equal measure as this one. The hydroxy group begs for acylation and etherification, while those carbonyls tempt nucleophiles to attack. Trying reductive alkylation feels like rolling dice — some substituents work, and others end in polymerized messes. Cycloadditions often find utility here, letting researchers forge bonds that push the boundaries of ring strain even further, though plenty of efforts stop at the TLC stage when side reactions run wild. Keeping strict anhydrous conditions, monitoring temperatures in real time, and quick workup protocols become non-negotiable. Graduate students get a front-row seat to the challenges and unpredictability that define reactive strained-ring chemistry. For all the pain, the payoff comes with discovering unexpected reactivity, helping fuel new routes for other synthetic puzzles.
Synonyms & Product Names
Lots of synonyms litter the literature — hydroxycyclobutene dione, CBED, and cyclobutene-1,3,4-trione hydrate pop up in papers, which makes literature searches far tricker than they need to be. Catalogs from suppliers add their own spin with house product codes, often tacking on “hydroxy-dione” or “ring-strained intermediate.” Some patent filings dig deeper, using systematic IUPAC names that stretch for a paragraph. Keeping track of synonyms means more than just trivia — clearing up confusion with lab mates, regulators, and suppliers stops costly mistakes in ordering and reporting down the line. More than once I’ve had to settle squabbles in group meetings about what’s in the bottle, just because someone got too comfortable mixing up a synonym.
Safety & Operational Standards
Sticking to strict safety protocols makes all the difference here. The dione structure and pronounced ring strain combine for a nasty surprise if handled carelessly. Exposure to eyes or unprotected skin spells irritation and long-term harm, and volatility means even casual sniffers get a lungful of discomfort. Fume hood work, good gloves — the thick nitrile kind, not basic latex — and face shields keep researchers out of trouble. Disposal needs thought too. Dumping leftovers down the drain isn’t only illegal, but a guaranteed way to trigger regulatory trouble. I’ve watched even experienced chemists get lax, only to scramble when a cloud of decomposition fumes filled the lab. The paperwork and guilt that follow stick around longer than the compound.
Application Area
For most outside the chemistry bubble, 1-Hydroxycyclobut-1-Ene-3,4-Dione sounds like something from a sci-fi kit. For working scientists, it pulls weight in target-oriented synthesis and mechanistic organic chemistry. In pharmaceutical discovery, derivatives have inspired new lead scaffolds thanks to their peculiar geometry and reactive handles — medicinal chemistry teams have gotten pretty creative repurposing the strained ring into fragments for enzyme inhibition studies. Material scientists also keep an eye on derivatives for exploring polymers with built-in reactivity. Some environmental chemists use structurally similar rings as models to track the fate of strained-ring pollutants and oxidative decomposition, helping fill knowledge gaps about ring-opening mechanisms in the wild. The broad appeal comes not from mass production, but from the rare ability to link structure to both function and reactivity in research problems others can’t touch.
Research & Development
Research funding for strained-ring chemistry never reaches blockbuster levels, but those who specialize in 1-Hydroxycyclobut-1-Ene-3,4-Dione operate in tightly knit global networks. New synthetic innovations appear every few years, with groups publishing breakthroughs in ring opening, catalysis, and functional group manipulation. Collaborative projects with computational chemists provide powerful ways to model the intense electron density built up in these strained frameworks. Some researchers use spectroscopic tricks to catch intermediates that flicker into existence for microseconds. Watching colleagues abroad replicate or challenge published claims keeps everyone sharp. The best progress grows from hard-won lab results, shared datasets, and a willingness to publish negative results alongside lucky successes.
Toxicity Research
Few labs sign up volunteers for exposure testing, so most toxicity data comes from animal models and in vitro studies. Data shows that even low doses can irritate mucosal tissues and disrupt metabolic enzymes — results that raise obvious red flags for anyone working without a mask. Reports from environmental screens pick up metabolites that persist in soil, with some stubbornly resisting breakdown by microbes or light. Regulatory agencies monitor uses in commercial projects, which means industrial partners who want to expand applications must fund their own toxicology before approval. Every year, undergraduate students demand explanations for why so much caution lines up around this one compound; showing them the data on reactive oxygen species production and cellular damage usually puts the point across.
Future Prospects
The story of 1-Hydroxycyclobut-1-Ene-3,4-Dione keeps growing. As new synthetic tools bring otherwise impossible transformations into reach, creative chemists look for ways to tame its strain energy and steer the reactive handles into new research frontiers. Some envision ring systems like this as stepping stones to even stranger molecules — maybe ones with built-in photoreactivity for solar energy or medicinal uses that stretch beyond current drug design templates. Challenges still stack high for those seeking reliable bulk synthesis or commercial stability, but the payoffs come through unexpected reactivity and discoveries that ripple into broader organic chemistry. Watching how quickly old limitations fall when new minds and fresh tech step into the lab leaves me optimistic that 1-Hydroxycyclobut-1-Ene-3,4-Dione’s best days in chemical discovery still lie ahead.
What is 1-Hydroxycyclobut-1-Ene-3,4-Dione used for?
Unpacking a Lesser-Known Building Block
1-Hydroxycyclobut-1-ene-3,4-dione barely gets a mention outside of chemistry circles, but it plays an interesting role in modern research labs and chemical manufacturing. This compound stands out for its strained four-membered ring and two adjacent carbonyl groups, which catch the eye of chemists searching for new synthetic tools. Strain in a molecule isn’t just a textbook concept. In the lab, it drives reactivity, coaxing molecules to do things ordinary structures won’t.
Supporting New Materials and Pharmaceuticals
Researchers explore this compound primarily because of its versatility as an intermediate. It becomes a springboard for making more complex ring-shaped molecules. Those rings—never just pretty shapes—show up in everything from new drugs to next-generation materials. For example, compounds with similar skeletons serve as starting points for antibiotics or antiviral agents. The same core structure can get twisted or expanded, offering creative routes for new bioactive compounds.
Pharmaceutical chemists consistently chase structures with high potential for biological activity. Complexity adds a challenge but often rewards with a unique mechanism of action. I once attended a conference where a medicinal chemist described using strained four-membered rings to sneak molecules past cell barriers more easily. Compounds like 1-hydroxycyclobut-1-ene-3,4-dione open possible pathways to these prized structures, driving the hunt for better therapies at lower doses.
Driving Chemical Reactions with Strain
Industry doesn't always run on the molecules that make headlines. Much of the innovation happens behind the scenes, with intermediates that quietly enable new chemistry. The strained, reactive structure of this compound finds use as a reagent for introducing cyclobutane motifs into other molecules. Cyclobutanes, notoriously tricky to build, have found a niche in agrochemicals. Their presence lets molecules survive under sunlight or resist breakdown before reaching their target—in the field or in the human body.
In polymer science, researchers constantly hunt for monomers that can make new materials. The ability of 1-hydroxycyclobut-1-ene-3,4-dione to open and combine under controlled conditions gets attention from chemists aiming to create biodegradable plastics or high-strength coatings. These applications don’t score much public attention, but they make a difference by addressing real needs like environmental waste and industrial durability.
Research: The Lifeblood of Innovation
Most people will never hear about this compound, but they benefit from the work it enables. Universities and private labs use it as a testbed for exploring ring opening and rearrangement reactions. Each experiment refines our control over how molecules transform—paving the way for more selective, safer, and cheaper processes. Improvements here trickle down into everyday products, from medicines to materials that last longer or break down safely.
Challenges and Solutions
Handling compounds with strained rings comes with hazards. High reactivity means chemists need steady hands, reliable training, and strict protocols. Scaling up production safely always stays challenging. Labs share information about best practices, and manufacturers invest in better containment and monitoring. Support for chemical safety training must remain a priority, ensuring that people developing new technology don’t pay the price with their health.
Regulations can lag behind advances in synthetic chemistry. Faster collaboration between researchers, industry, and regulatory agencies builds trust. Open publications and regular safety audits support public confidence and give early warning of risks before they hit the mainstream.
Looking Ahead
1-Hydroxycyclobut-1-ene-3,4-dione’s most important use may not have even arrived yet. The pace of chemical innovation depends on unsung molecules like this one. With better resources for research, safe handling, and cross-talk between disciplines, new uses for strained rings will keep surfacing in the world’s most critical problems—medicine, materials, and sustainability.
What are the storage conditions for 1-Hydroxycyclobut-1-Ene-3,4-Dione?
Getting the Basics Right
People who work in chemistry know that not all compounds like to sit on shelves under the same conditions. Take 1-Hydroxycyclobut-1-Ene-3,4-Dione, for example. This chemical brings its own quirks, and it’s best to treat it with respect. My own time in a research lab taught me that good habits pay off when handling materials that can degrade or react under normal room conditions. Temperature, light, air, and moisture can each fuel a disaster—or keep a substance safe.
Why Stability Matters
Storing sensitive chemicals like this dione isn’t only about keeping numbers in a logbook. It’s about safety, reliability, and science that holds up over time. 1-Hydroxycyclobut-1-Ene-3,4-Dione tends to break down if exposed to warm or humid environments. Decomposition can release gases or lead to messy byproducts, which complicate measurements, wreck instruments, and risk personal health. It pays off to learn from others’ mistakes: long ago, I saw a project delayed for weeks by material gone bad through poor storage. That sort of avoidable trouble doesn’t just slow down work, it puts people at risk.
Temperature and Light
Cool and dry conditions go hand-in-hand with preserving this compound. Keeping it in a dedicated refrigerator — not one shared with food — keeps the temperature steady, around 2–8°C. Cycling temperatures from frequent fridge door openings can shorten the shelf-life, so a separate scientific fridge always works best. Light also plays the villain’s role for molecules like this. At work, we always wrap amber vials in foil or use dark glassware to block UV rays. UV and visible light both drive unwanted changes in the molecule. Once, after a weekend with a sample left out in the open, what was left barely resembled what we’d started with, showing again how damaging even a little sunlight can be.
Keeping Out Air and Moisture
Air brings in both oxygen and water vapor, and both can lead to trouble. The dione can react slowly with oxygen, building unstable products. Moisture from the air gets in through careless bottle-top openings, even if the room seems dry. To fight back, I always use a desiccator with fresh drying agent or add molecular sieves to sample containers. Sealing vials tightly and minimizing time open to the air makes a difference, especially in seasonal changes when humidity spikes. Purging containers with an inert gas like nitrogen helps, too. Even just a short shot of nitrogen protects the compound for weeks longer.
Labeling and Safe Practice
Smart labeling prevents confusion. Knowing what’s in a bottle, when it was stored, and under what conditions lets researchers avoid the guessing game. I stick a small label with date, fridge box number, and opening history right on the cap. Routine checks of all stocked chemicals prevent surprises, so nothing dangerous gets used by mistake. A careful record-keeping culture, built on teamwork, boosts everyone’s safety and confidence.
Solutions That Work in Real Labs
The best practice borrows from pharmaceutical and fine-chemical storage routines: cool, dark, sealed, and inert. That means a reliable fridge, amber or foil-wrapped vials, fresh drying beads in the storage box, and routine visual checks. Nothing beats hands-on attention and learning from those who work with sensitive chemistry each day. Creating a system that everyone trusts and understands helps the whole team handle these specialized compounds with care, protecting both people and research quality.
What is the chemical structure of 1-Hydroxycyclobut-1-Ene-3,4-Dione?
Looking Up Close at the Structure
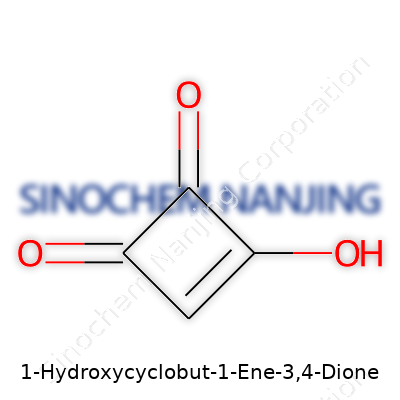
Take a simple cyclobutane ring. Now, imagine carving into that structure and sticking on some high-impact groups. That gives you something a bit like 1-hydroxycyclobut-1-ene-3,4-dione. Its formula, C4H2O3, doesn’t look like much at first glance. But the details tucked inside are where real insight sits. You’re staring at a four-membered hydrocarbon ring, cramped but holding its shape. At spot number one on the ring, there’s both a hydroxyl group (OH) and a double bond—a rare combination, each pulling electron density in different ways. Right next to that, at positions three and four, you expect to find two carbonyl groups. Double-bonded oxygens on neighboring ring carbons load stress onto the system, setting it up for reactivity.
Anyone who has wrestled with organic chemistry knows small rings, especially cyclobutene, strain at the seams. They push atoms together in awkward ways, forcing bond angles far from comfortable 109-degree tetrahedral geometry. This strain gets amplified with each new oxygen you bolt onto the ring. You can expect the molecule to be on the edge, always looking for a way out—through reaction, breakdown, or transformation.
Why This Small Structure Packs a Punch
That tension in small oxygen-rich rings isn’t just an academic point. In practice, these kinds of molecules often act as powerful intermediates in synthetic chemistry. They don’t stick around for long but pass along their energy and react quickly. I remember watching a postdoc in my lab try to isolate a compound with a similar skeleton. He struggled—every time we cooled the solution, something new formed, as if the original compound felt too cramped to survive for more than a few hours.
With two adjacent carbonyls, the pattern you see is called a 1,2-diketone setup. In nature and in the lab, this part of the molecule grabs attention because it loves to take part in reactions. Adding the hydroxyl makes it tautomerize or shift its double bonds and hydrogens back and forth. For those designing drugs, materials, or pigments, that kind of reactivity makes these building blocks useful, though handling them takes care and patience.
Staying Cautious in the Lab
A lab partner once mixed something similar, hoping for stability. Instead, the solution changed color and composition in minutes. This mirrors what many chemists spot with small, strained systems—change can come quickly, and reaction pathways branch in ways textbooks barely mention. If you’re planning to work with 1-hydroxycyclobut-1-ene-3,4-dione, don’t count on it sitting quietly on the shelf.
With great reactivity comes handling risk. Volatility in small dione systems means safety protocols matter, from eye protection and fresh gloves to proper ventilation. You won’t see these compounds featured in every undergraduate lab. Only researchers comfortable with rapid transformations and careful analysis tend to seek them out. Analytical tricks like rapid-injection NMR or cold-column chromatography help catch these fleeting molecules before they jump into something else. There’s satisfaction in managing that challenge, even if most days you’re left with a messy flask and a story about how chemistry never goes as planned.
Where Solutions Might Start
One way teams have worked with similar molecules is by trapping them as derivatives, protecting the sensitive spots until it’s time to let them loose. Building partnerships across departments—organic, analytical, even computational chemistry—helps predict and interpret wild behavior. Digital models and short-loop experiments shed light on possible breakdown pathways, letting researchers plan better syntheses or storage conditions. Persistent curiosity and careful technique keep you in the running, even against molecules that seem determined to rearrange themselves at every turn.
Is 1-Hydroxycyclobut-1-Ene-3,4-Dione hazardous or toxic?
Understanding the Chemical
Scientists keep pushing the limits of chemistry, looking for new compounds that might help in everything from medicine to materials science. 1-Hydroxycyclobut-1-ene-3,4-dione stands out because it has an unusual ring structure, squeezed tight by four carbon atoms. That ring can store a lot of chemical strain, which in turn makes this molecule very reactive. In the lab, high reactivity sometimes means risk. Reactions can go fast and hard when they involve strained rings, and that usually gets chemists double-checking for safety first.
Safety Data Is Scarce—But We Can Learn From Similar Compounds
The big snag is a lack of thorough data on 1-hydroxycyclobut-1-ene-3,4-dione itself. No big health agency or chemical group has published a full review. For those of us who spend time scanning safety sheets, that’s always a red flag to slow down and think. We know other cyclobutenediones — close chemical relatives — can cause trouble for the skin and lungs. Some cyclobutenones have shown signs of moderate toxicity in animal testing, including respiratory irritation and even signs of affecting the liver with repeated exposure. There’s no proof that 1-hydroxycyclobut-1-ene-3,4-dione behaves the same way, but it’s fair to expect at least some similar risks.
Hazard Signs and Worker Experience
In university labs, young researchers are taught to be careful with unfamiliar molecules. Experienced chemists treat new diones and cyclobutenes with respect, always using gloves, splash goggles, and fume hoods. With compounds like this, it’s too easy to underestimate how little skin exposure it takes to set off a chemical burn, or how quickly vapors escape those small glass flasks. I remember a time cleaning up a seemingly innocent dione compound — we made the mistake of skipping the fume hood, and the sharp odor filled the whole room. No one expected sore throats and headaches to follow so fast, but that’s what happened. The lesson stuck: treat new strained rings with extra caution.
Potential Environmental Risks
Waste from these chemicals also matters. Highly reactive small molecules can break down into unpredictable or persistent by-products. Some even end up being more toxic than the starting material. Cyclobutene derivatives break down in wastewater slowly, and the by-products can become hard for municipal water treatment systems to handle. Every facility that deals with ring-strained organics needs to have a plan for capturing and destroying effluent safely, not just dumping chemicals down the drain.
Practical Tips and Safer Paths Forward
Any shop or laboratory handling 1-hydroxycyclobut-1-ene-3,4-dione safely should build on what we know from structurally similar compounds. This means monitoring air quality, keeping emergency eyewash and skin wash stations ready, and not leaving containers open except behind a ventilated hood. Education goes a long way—clear facts on how and why these chemicals can cause harm stick with workers. Working alongside regulatory scientists, researchers can help fill the missing toxicology data by publishing detailed case studies and transparent incident reports. Before anyone brings a brand-new compound into routine use, they need to pressure suppliers for safety testing data and keep up strong habits that protect both people and the world outside the lab.
Bottom Line
Risk often hides in the details with unusual chemicals. Respect for ring strain and good lab sense help avoid surprises. Until research fills the gaps, everyone handling 1-hydroxycyclobut-1-ene-3,4-dione should go slow, focus on safety, and never assume that what looks harmless will act harmless.
What is the purity and available packaging size for 1-Hydroxycyclobut-1-Ene-3,4-Dione?
Understanding Real Purity Talk in the Lab
Anybody working in synthesis or research knows what a headache it is to hunt down solid sourcing information for rare compounds. 1-Hydroxycyclobut-1-ene-3,4-dione falls square into this camp—a pretty specialized molecule. You can’t just walk into a store and grab it off a shelf. Purity ends up being the deciding factor every time. For this chemical, vendors typically offer material with purity levels ranging between 97% and 99%. Sometimes an HPLC or NMR readout will confirm this. Even with a fraction of a percent in question, side reactions or analytical inconsistencies can sneak into a project, so being certain about the numbers pays off.
These purity figures are not just marketing fluff. The presence of extra water, solvent leftovers, or unknown isomers in a sample ruins scale-up attempts or complicates basic organic analysis. I’ve run more than one synthesis where an “almost pure” lot meant losing a week to trouble-shooting. So, the devil is in the details—check for a certificate of analysis from the supplier, and don’t take vague claims at face value.
Packing Conditions: How Size Shapes the Workflow
Most chemical suppliers stick to industry patterns—offering specialty compounds like this one in packaging starting from a few milligrams up to several grams. Common package options include 50 mg, 100 mg, 250 mg, or 1 gram glass vials with secure seals that block out moisture and light. The price jumps steeply as the quantity increases and the purity tightens. What you pick depends on your project’s real demands. If you’re screening reactions, the 50 mg vial usually makes sense—enough to run a handful of tests and stretch your funding. Folks doing scale-up or kinetic analysis order larger vials, but should only commit after confirming that material really matches the supplier report.
In my experience, smaller vials help keep waste down and save budget, especially when chemists don’t know how reactive a compound might get with air or light. I once watched a colleague waste half a gram of an analog, all because nobody wanted to split up the main stock into new containers. Smaller vials show their value there—the material stays fresh and doesn’t get ruined by repeated opening. Labs ordering in bulk often ask for custom packaging, like nitrogen-purged ampoules, especially if the molecule has a reputation for going off quickly.
Staying on the Safe and Smart Side
Jumping into any project with exotic molecules takes a certain amount of background work: secure certificates, ask the supplier for current NMR or HPLC spectra, and always clarify the specific container type coming your way. Too many times, academic labs receive something in a plastic screw-top tube, only to discover the substance stuck to the sides. I’ve learned to insist on amber glass packaging for sensitive reagents. The shipping journey itself poses risks—heat and vibration sometimes degrade fragile samples, so smarter vendors include cold packs or fast shipping.
Clear, honest communication from the supplier helps avoid research delays and wasted funds. Checking the current packaging options and confirming the actual purity ensures projects start on solid ground. The more people share accurate sourcing notes, the less time gets lost to surprise problems, and the more sustainable research feels—especially when budgets and patience are thin. Always look past the brochure: what matters most is real technical support on details like purity, packaging material, and proper storage.
| Names | |
| Preferred IUPAC name | cyclobutene-1,3,4-trione |
| Other names |
cyclobuta-1,3,4-trione hydrate 1-hydroxycyclobutene-3,4-dione |
| Pronunciation | /waɪˈdrɒk.si.saɪ.kləʊˈbjuːt.wʌn.iːn.θriː.fɔː.dʌɪˈoʊn/ |
| Identifiers | |
| CAS Number | [930-30-3] |
| 3D model (JSmol) | `3Dmol('CC1(=O)C=C1O')` |
| Beilstein Reference | 1638737 |
| ChEBI | CHEBI:139492 |
| ChEMBL | CHEMBL1234297 |
| ChemSpider | 20232772 |
| DrugBank | DB07642 |
| ECHA InfoCard | 03b7c807-7b7a-4d09-a6b3-dee730dd8a2a |
| EC Number | 3.1.2.23 |
| Gmelin Reference | 37780 |
| KEGG | C19611 |
| MeSH | D016700 |
| PubChem CID | 14177085 |
| RTECS number | GU4375000 |
| UNII | W0I8HW8A9D |
| UN number | 3261 |
| CompTox Dashboard (EPA) | DTXSID60146093 |
| Properties | |
| Chemical formula | C4H2O3 |
| Molar mass | 112.07 g/mol |
| Appearance | Yellow solid |
| Odor | odorless |
| Density | 1.68 g/cm³ |
| Solubility in water | soluble |
| log P | -0.77 |
| Vapor pressure | 0.0182 mmHg (25°C) |
| Acidity (pKa) | 7.03 |
| Basicity (pKb) | pKb = 11.68 |
| Magnetic susceptibility (χ) | -53.75 × 10^-6 cm³/mol |
| Refractive index (nD) | 1.507 |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -297.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -427.8 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | There is no ATC code assigned. |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05 |
| Signal word | Danger |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-1-ن |
| Flash point | 120 °C |
| NIOSH | KWJ87710C3 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Hydroxycyclobut-1-Ene-3,4-Dione is not specifically established by OSHA or other major regulatory agencies. |
| REL (Recommended) | 0.05 mg/m3 |
| IDLH (Immediate danger) | NIOSH does not currently list an IDLH value for 1-Hydroxycyclobut-1-ene-3,4-dione. |
| Related compounds | |
| Related compounds |
Cyclobutane-1,2,3,4-tetrone Squaric acid 2-Hydroxycyclobut-2-ene-1,4-dione |

