1-Ethylbutanol: A Down-to-Earth Look at a Versatile Chemical
Tracing the Roots: Historical Development of 1-Ethylbutanol
Looking back, 1-Ethylbutanol didn’t leap onto the chemical scene overnight. Chemists first began experimenting with higher alcohols like it toward the dawn of the 20th century as the world grew more curious about organic compounds beyond simple ethanol. During those years, the petroleum industry’s rapid expansion meant ancillary by-products from refining could sometimes serve as raw materials. Industrially, as synthesis methods matured, 1-Ethylbutanol turned from a lab curiosity to a useful industrial candidate, appreciated for its molecular flexibility and adaptability. Over generations, researchers have tinkered with better ways to produce and purify it. Refineries searched for ways to capture more value from petroleum, so 1-Ethylbutanol found its spot both as a solvent and as a building block in more complex syntheses. That history shaped not only how it’s made, but also how folks began to see its possibilities beyond the chemistry bench.
Getting to Know the Product: Overview
1-Ethylbutanol, often known as 1-ethyl-1-butanol or ethylbutyl alcohol, counts among the family of six-carbon alcohols. In its pure form, it brings a faint, sweetish smell, signaling its organic roots. Commercially, it doesn’t show up in every hardware store aisle, but it forms a small backbone for many industrial mixes. A variety of chemical producers pump out this product, narrowing in on applications ranging from coatings to intermediate steps in chemical production lines. If you flip through technical trade catalogs, you’ll often spot it as a specialist’s chemical rather than one you knock over in your kitchen cabinet. Yet, for those who use it, trace impurities, consistent physical properties, and a clear supply chain matter quite a bit.
Physical and Chemical Properties
This alcohol melts at about -79°C and boils close to 137°C, showing clear signs it won’t freeze or vaporize easily in typical working conditions. Its density hovers near 0.82 g/cm³—noticeably lighter than water—and as with others in the butanol group, it stays mostly immiscible with water. Dissolving power makes it interesting for blending both polar and non-polar substances, which matters a lot in lube, surfactant, and flavor syntheses. Chemically, the primary alcohol group allows reactions folks expect: oxidations, esterifications, and more. Handling it in a lab or plant brings that familiar alcohol “bite,” but without the volatility headaches you find with lighter molecules. Safety teams keep watchful eyes on its flashpoint; it ignites above 41°C, so it doesn’t pose quite the same risk as some faster-evaporating cousins, but you still can’t get careless with open flames or careless storage.
Technical Specifications and Labeling
Every drum or bottle needs the right info on the label, not just for shipping but also for those opening the seal. Suppliers must note the compound’s CAS number (137-32-6), lot/batch identity, purity—often above 98% for industrial grades—and major impurities if any. Labels require warning icons for flammability and irritant effects, plus storage guidelines. Material Safety Data Sheets (MSDS) accompany shipments, spelling out reactivity hazards and medical steps in the event of skin or eye contact. Regulatory compliance with standards from agencies like OSHA, REACH, and local environmental authorities avoids headaches for both buyers and producers. On the digital side, barcodes and QR codes link back to data sheets or certifications for traceability, a practice that emerged as global trade tightened oversight on specialty chemicals.
Preparation Method
The most common way to make 1-Ethylbutanol involves the Guerbet reaction, where two molecules of a lower alcohol, like ethanol, come together in the presence of an alkali metal catalyst. The process advances by first dehydrogenating the starting alcohol, then condensing it, and finally hydrogenating the result to form a longer, branched-chain alcohol. Petrochemical strategies have also tried using olefin hydroformylation, where butene reacts with synthesis gas (a mix of CO and H₂) to yield aldehydes, which then undergo hydrogenation to the desired alcohol. Home chemistry doesn’t really lend itself to production here, as the methods need tight control over temperature, catalysts, and by-product handling, so larger chemical plants dominate production. Efforts have ramped up to swap traditional fossil feedstocks with bio-researchers eyeing renewable routes through fermentation, though scaling up remains tricky outside of lab pilots.
Chemical Reactions and Modifications
As a primary alcohol, 1-Ethylbutanol offers a reactive spot ripe for modification. Esterification with acids kicks out fruity esters, giving fragrance mixers reason to keep it handy. Oxidation, performed with agents like potassium dichromate, yields the corresponding aldehyde or acid—a pathway environmental analysts sometimes exploit when tracking breakdown products in spills. Chlorination, sulfonation, and etherification unlock specialty surfactants or solvents for niche uses. Alkoxylation can build block copolymers needed in paints, while hydrogenolysis fragments longer chains for lubricant production. Ultimately, the functional group does the heavy lifting, letting chemists design molecules as needs shift. Local regulations control what kind of downstream reactions get the green light, especially if waste streams might harm local water or air quality, so development teams tune each process to both chemistry and compliance needs.
Synonyms and Product Names
Chemists rarely limit themselves to a single name for any compound. 1-Ethylbutanol sometimes pops up as ethylbutyl alcohol, 1-ethyl-1-butanol, or even as 1-butyl ethanol in certain older texts. CAS registry number 137-32-6 eliminates any room for confusion. In trade directories, you might stumble over code numbers or brand-specific monikers, especially as different countries or companies carve their own pathway through product catalogs. To avoid messy handovers and costly mistakes, most regulatory filings stick with the systematic name and attach all common synonyms in technical appendices.
Safety and Operational Standards
Safe handling starts with training and reliable equipment. Splash goggles protect eyes from the sting of accidental splashes, while nitrile gloves keep hands free from persistent skin irritation. Local fire codes dictate storage in flammable cabinets, neat and away from sunlight or ignition points. Workers log exposures and ventilation systems hum to clear out vapor build-ups, since inhaling high concentrations can leave you with headaches or worse—respiratory irritation. Safety checklists include spill plans, eyewash stations, and chemical-resistant containers to keep accidental releases small and manageable. As regulations tighten under authorities like OSHA and the European Chemicals Agency, companies perform regular audits to confirm every drum and pipe fits the latest rules, using accident data and near-miss reports to update training. Building a strong safety culture means more than ticking boxes—it’s about workers leaving at the end of the shift just as healthy as they started.
Application Areas
The versatility of 1-Ethylbutanol runs the gamut. Paint and coatings makers tap into its solvent power to fine-tune drying times and enhance flow without leaving heavy solvent odors that linger like those from aromatic compounds. In plastics, its molecular structure helps craftizers improve the workability of resins, boosting their usefulness in car panels, electronic casings, and furniture coatings. As a starting material, it sits at the beginning of chemical syntheses for pharmaceuticals, surfactants, agrochemical additives, and even custom flavors for foods, though no one pours it directly onto consumer goods. The quest for bio-based fuels also leads to research into modifying its structure so it blends well with gasoline, reducing dependency on fossil fuels. Specialty lubricants benefit from tailored versions, allowing machinery to run smoother and longer. Wastewater treatment plants sometimes use it as a carbon source to accelerate biological processing, showing how a relatively obscure chemical can end up supporting everything from cleaner air to more efficient farming.
Research and Development Efforts
Research teams don’t sit still with a chemical like 1-Ethylbutanol. University labs and industrial consortia push forward on better catalysts and new feedstocks so production gets cleaner and cheaper. Interest among green chemists drives investment in microbial engineering, hoping genetically tweaked organisms can churn out higher alcohols from plant sugars instead of petroleum. Process control engineers test new reactor designs in search of better yields and energy savings. Collaborations sometimes bridge competitors, pooling patents so everyone advances together on safe handling or emerging uses. Environmental scientists keep an eye on eventual breakdown products, since local rules now hold firms responsible for any persistent pollution or toxicity. Open publication of test results and frequent industry conferences help spread best practices, so positive results in one corner of the globe can be adopted elsewhere without starting from scratch.
Toxicity Research
The health picture around 1-Ethylbutanol remains under constant review. Acute inhalation brings headaches, dizziness, and respiratory distress—much like other mid-weight alcohols. Longer exposures can irritate eyes or skin, so workers track every accidental splash or inhalation event. Chronic effects aren’t as well documented as with more common solvents, so regulatory agencies call for ongoing studies. Animal models help map out dose thresholds, checking for changes in lung, liver, or kidney tissues after extended contact. Regulatory limits for workplace air rarely rest on outdated data; instead, they build in safety factors until new studies clarify the risks. As endocrine disruption and developmental toxicity raise red flags for peer compounds, researchers now look closely for subtle, long-term health patterns in workers and nearby communities. Sampling protocols and environmental fate studies cover groundwater, rivers, and soils, given past experience with persistent, bioaccumulative chemicals.
Future Prospects
Looking ahead, shifting supply chains and tougher environmental targets will shape the future for 1-Ethylbutanol. Green chemistry stands ready to play a bigger part, trimming fossil fuel reliance and waste. Investment in fermentation or hybrid bio-petrochemical routes shadows the broader movement to cut industrial emissions. Specialty applications in high-value coatings, lubricants, or eco-friendly fuels drive research into better reaction controls and minimized side products. Market demand will ebb and flow, tied to shifts in consumer habits, trade barriers, and stricter product labeling worldwide. Partnerships between startups, established chemical firms, and regulators could open up educational tracks, safer operations, and more transparent toxicity data. New testing protocols draw on real-world monitoring rather than lab-only scenarios, painting a more honest picture of long-term effects. With open data sharing and cross-border collaboration, the story of 1-Ethylbutanol still waits for new chapters as fresh challenges push the limits of what this workhorse molecule can offer.
What is 1-Ethylbutanol used for?
How Industry Finds Value in 1-Ethylbutanol
Walk through any major paint manufacturing plant or visit a chemical supplier’s warehouse, and the story of 1-ethylbutanol comes alive. While it might not make headlines the way a new tech gadget will, this chemical gets things done behind the scenes. Its power shows up in places you might not expect, adding value in ways that are easy to overlook.
The Role in Paints and Coatings
Decades ago, I watched my uncle mix paints in his workshop and learned not every can on the shelf holds the same story. 1-ethylbutanol shows up in many paint formulas, not because it’s flashy, but because it keeps the mixture smooth and workable. The chemical has a knack for controlling how fast paint dries. That simple trait can cut streaking and help even amateurs achieve a more professional finish. The solvent power of 1-ethylbutanol helps pigments and other additives dissolve right where they belong. This translates to better coverage and longer shelf life for the paint.
Powering Production Beyond the Surface
It’s not just the final coat that benefits. Large-scale adhesive production relies on chemicals that maintain consistency, especially in sticky situations—literally. 1-ethylbutanol helps many adhesives stay uniform during mixing, so products work the same way each time. My old engineering notebook is full of marks from test labels that either stuck too fast or faded too soon; controlling the blend makes all the difference.
Supporting Plastics and Cleaning Up
Out in the plastics labs, researchers keep tapping 1-ethylbutanol for its ability to tweak process temperatures and support smooth extrusion. This property allows operators to get repeatable results, reducing waste and lowering costs. In cleaning products, it breaks up greasy residues without overwhelming users with harshness. Hospitals and restaurants often count on solutions with this chemical to clear up messes that regular soap can’t touch.
Protecting Food and People
Quality assurance doesn’t get any tighter than in the food packaging world. Here, 1-ethylbutanol comes in handy to help inks used on packages dry quickly and stick well. That way, labels don’t smear and branding stays clear. This small role can actually support food safety and prevent confusion on store shelves. Industry regulators track chemical residues carefully, but with responsible handling, the benefits tip the scales toward better protection for consumers.
The Environmental Question
One point people keep asking about is the environmental impact. Volatile organic compounds, including 1-ethylbutanol, can add to air pollution if used carelessly. In my experience, bigger companies have adopted collection and recycling systems to capture emissions. Local ordinances keep small shops in check too, with paint strippers or cleaners offering lower-impact alternatives that minimize harmful off-gassing. That’s a trend worth keeping, since cleaner air benefits all of us.
Moving Forward with Facts and Care
Professionals who use 1-ethylbutanol rarely talk about it at dinner parties, but for manufacturers, it represents reliability and efficient production. Industry keeps evolving with better handling and smarter substitutes where possible. For now, this chemical keeps showing its worth across all sorts of products people use every day, painting, gluing, cleaning, and packing, often without a second thought.
What are the safety precautions when handling 1-Ethylbutanol?
A Close Look at 1-Ethylbutanol
Handling chemicals can seem routine, but mistakes leave little room for error. 1-Ethylbutanol, a colorless liquid with a strong odor, gets used in labs, paints, coatings, and sometimes even pharmaceuticals. The safety sheet looks intimidating, and there's a reason. Personal experience in the lab taught me respect doesn’t come from reading the label—it comes from understanding what this solvent can do to your eyes, skin, and lungs.
Personal Protection Always Comes First
Caring for your safety never means skipping the basics. Working with 1-Ethylbutanol means grabbing chemical-resistant gloves—nitrile or neoprene do the job. Regular lab gloves feel slick after a spill; with solvents like this, a chemical leak chews right through the wrong glove. Wearing safety goggles matters, too. Splash a bit of this compound in your eye, and you won't forget the burning. Lab coats, closed-toed shoes, and long sleeves aren’t negotiable. Even on a quick job, full coverage gives peace of mind.
Ventilation Makes a Difference
Strong odors signal danger. It only takes fifteen minutes in a stuffy lab with no exhaust fan for headaches to start, so working in a well-ventilated area isn’t an extra. Fume hoods aren’t just for show—set one up before you start, and keep the sash low. That keeps vapors moving up and away. I learned the hard way that breathing in solvent vapors once or twice doesn’t make you invincible. Chronic exposure chips away at your health silently.
Knowing the Risks Helps You Stay Ready
Contact with 1-Ethylbutanol dries out skin fast, causes redness, and leads to cracking. Inhaling it leaves you dizzy and gives your lungs a workout they don’t want. My own reaction to a splash reminded me that washing with soap and water isn’t enough—an emergency eyewash station and working safety shower can save the day. Cutting corners never pays: backup plans and preparedness go beyond a checklist.
Storage: One Simple Rule—Keep It Cool and Locked Away
A fire hazard hides with a flammable liquid. Storing 1-Ethylbutanol away from heat sources, sparks, or open flames makes sense. Metal safety cabinets marked for flammables improve peace of mind. One time, a co-worker left a container open near a window in July. The heat made vapors rise fast, and the air in the room thickened. That near-miss stuck with all of us. Keeping the storage area cool, dry, and fully labeled avoids confusion or accidents, especially during high-stress moments.
Disposal: No Shortcuts
Lab work piles up empty bottles fast, but pouring solvents down a sink poisons water systems and causes fines. Every workplace taught me to collect waste in assigned containers, tightly sealed, and then send them to certified hazardous waste management. Skipping this step isn’t just illegal—it’s dangerous for anyone downstream. Always label everything. Sharing spaces with others drove home the point—no one can read your mind, so don’t hide waste or cut corners.
Training and Clear Communication
Training saves lives. New team members shadowing old hands makes a real difference. Written safety protocols, drills, and reminders keep everyone on track. Mistakes happen when someone assumes a shortcut is harmless or skips a refresher. Open conversations about near-misses and honest records help everyone stay sharp. My safest workplaces encouraged speaking up—even about forgotten goggles or unlabeled containers. Everyone learns by example and stories.
Final Thoughts on Respecting the Risks
Experience inside and outside the lab shows that risk isn’t about the rarity of an accident—it’s about the impact. Protecting yourself and your team, keeping storage smart, disposing of waste right, and sticking to the basics keep a standard: everyone goes home safe, every time. Safety becomes a habit, and it matters every single day.
What is the chemical formula of 1-Ethylbutanol?
Looking at the Structure of 1-Ethylbutanol
1-Ethylbutanol grabs attention from folks working in labs, classrooms, and even food chemistry. The chemical formula for 1-Ethylbutanol is C6H14O. Each letter and number in that formula means something. It’s a six-carbon alcohol, where an ethyl group hangs off the first carbon of butanol’s chain. Behind this formula sits a mix of hydrogen, carbon, and oxygen — the bread and butter of organic chemistry.
Real-World Connections
Getting to know why formulas like C6H14O matter stretches beyond just passing exams. Over the years, I’ve spent time in labs hoping not to break another glass pipette. Breaking chemicals down by their formulas helped me navigate projects, write safer protocols, and talk clearly with colleagues. In an industry setting, knowing the right formula can mean the difference between safe reactions or a messy bench, especially when scaling up processes.
The details tucked into a formula guide how folks handle and use compounds. For instance, C6H14O points to a higher boiling point than its smaller cousins. Having six carbons and an oxygen makes it useful in solvents, blending fluids, and even niche food flavorings after rigorous testing.
Why Clarity in Naming and Formula Matters
If the name “1-Ethylbutanol” sounds like a tongue-twister, you’re not alone. Naming tells us about the shape and connections between atoms. When companies, educators, or regulators get sloppy with names or formulas, mistakes happen. Someone might order the wrong chemical. That’s costly. Worse, safety takes a hit if storage or reactivity gets misjudged.
In a world that cares about transparency, facts, and proven expertise, getting the formula right is a show of respect for science and the people handling these substances. The correct formula is also the first thing checked by regulators, researchers, and buyers working across continents.
The Impact of Accurate Chemical Knowledge
Accurate formulas shape day-to-day safety and long-term product development. I have seen chemists double-check labels because even a one-atom difference can turn a mild compound into something explosive or toxic. For students, learning to crack the code of chemical names and formulas builds habits that ripple through the rest of their careers.
Sharing information correctly isn’t just for textbooks. It helps those living near labs, factories, or waste sites trust the professionals running the show. Reliable data also gives smaller companies a fair chance to compete, since they aren’t left guessing about what’s really in a barrel or drum.
Looking Towards Better Solutions
Mistakes still crop up. Dropping handwritten notes for digital inventories cuts down errors — a homemade spreadsheet never caught all my typos, but software can flag them. Training lab staff to check formulas before ordering or mixing chemicals works better than hoping someone catches a slip later.
Pushing for easier-to-understand labels and digital tracking will help reduce costly mix-ups. We can’t make every chemistry classroom perfect, but getting the basics right — like nailing down that 1-Ethylbutanol means C6H14O — keeps everyone safer and makes communication stronger.
How should 1-Ethylbutanol be stored?
Real Risks Behind the Bottle
Handling chemicals like 1-ethylbutanol has real stakes. I remember my early days working in a chemical warehouse, where a single careless step could throw both health and inventory down the drain. This alcohol brings its own dangers—flammability, fumes, and the threat of chemical burns. News stories echo my experience: accidents happen where storage falls short or the rules are ignored. These stories stick because they’re a real reminder—chemicals aren’t forgiving. Take shortcuts with storage, and small errors turn into workplace emergencies.
Why Proper Storage Matters
According to CDC and OSHA resources, 1-ethylbutanol produces flammable vapors at room temperature and forms explosive mixtures with air. The health impacts from inhaling these vapors or getting the liquid on your skin are no joke: headaches, dizziness, even unconsciousness if things get out of hand. Anyone responsible for chemical stock—lab, factory, or even a training workshop—has an obligation not just to the bottom line but to co-workers and the surrounding community. It sounds obvious, but too many incidents come from skipping the basics.
Smart Storage Practices Learned on the Floor
Every bottle and drum should find a spot in a cool, well-ventilated storage area, far from flames or sparks. Too often, I’ve seen flammable chemicals shoved on shelves in cluttered rooms. That’s a recipe for disaster. Set clear zones for different hazard classes—don’t stash 1-ethylbutanol next to acids, oxidizers, or heat-producing equipment. Even static electricity from plastic pallets has set off fires when someone got lazy during drum transfers.
Metal safety cans or properly labeled glass bottles should hold 1-ethylbutanol tight with a fitting lid. Use signage that’s unmistakable: “Flammable,” “Toxic Vapors,” and so on. I’ve seen new hires mistake clear chemicals for water or general solvents. Good labeling keeps everyone alert to the real risk.
Temperature and Ventilation Count
Air movement matters just as much as temperature. The vapor from 1-ethylbutanol wants to drift and linger, leading to fire hazards and health issues. That means relying on more than just a window cracked open: exhaust fans and chemical fume hoods do the job right. I once watched a careless storage decision force a costly evacuation; it’s never worth saving a few bucks on air handling.
Facility managers should keep their storage areas cool—preferably below 25°C. Warm rooms mean higher vapor pressure, which means more fumes and higher fire risk. Never let direct sunlight hit tanks or bottles, as it speeds up the reaction and evaporation.
Training and Practice Create Safety
Experience taught me that even a perfect setup falls apart without ongoing training. Every new staff member, no matter their background, should walk through proper storage, handling, and emergency protocols. Fire extinguishers, spill kits, and emergency eyewash stations must be visible and fully stocked. Incident drills can feel tedious until you face a real leak or fire; then everyone is grateful for the muscle memory.
Keeping Accountability Front and Center
Checks and maintenance keep storage safe. Routine inspections shouldn’t get skipped because everyone feels too busy. Check for leaking lids, corrosion, or cracked containers, and deal with any issue immediately—not after the weekend. A slip in vigilance could send fumes into shared spaces or, worse, ignite a blaze. Documenting each inspection and reporting every close call creates a culture where everyone watches out for their own safety and their coworkers'.
Is 1-Ethylbutanol hazardous to health or the environment?
Getting to Know 1-Ethylbutanol
1-Ethylbutanol isn’t a household name. Yet, this chemical often pops up in factories and research labs. It’s a clear liquid with a sharp, alcohol-like smell. Most often, you’ll find it used as a solvent or in the manufacturing of flavors and fragrances. With chemicals like this, people want straight answers on safety—for themselves and the planet.
Health Risks Deserve Attention
Anyone handling 1-ethylbutanol should avoid skin contact or breathing in its fumes. This alcohol can irritate eyes, skin, and the respiratory tract. Direct contact tends to bring mild but uncomfortable symptoms—think redness, stinging, or coughing.
Spilling some on your hands doesn't mean a trip to the ER, but repeated exposure starts to add up. Over time, in high enough concentrations, this stuff can damage your liver and nervous system, similar to other alcohols in industrial settings. Inhaling too much can make you dizzy or nauseous, and workers stuck without proper ventilation do run risks.
The U.S. Occupational Safety and Health Administration (OSHA) sets strict guidelines for solvent exposure. 1-Ethylbutanol hasn’t gotten as much study as bigger names like methanol, but many experts agree—common-sense precautions keep you safer. Gloves, eye protection, and fresh air do a lot here.
Environmental Impact: More Questions than Answers
Spilling 1-ethylbutanol in a lab sinks into the bench or evaporates fast, thanks to its chemical makeup. Released in larger quantities out in the world, the impacts turn less predictable. It doesn’t hang around in soil or water for long because it breaks down pretty fast. That’s good. Still, during its short stay, it poses risks to aquatic life, especially smaller creatures that can’t break down chemicals as quickly as fish or mammals can.
Studies on aquatic toxicity show negative effects start showing up at moderate concentrations. Aquatic invertebrates and algae often react first, disrupting the food chain beneath the surface. Localized spills can kill off bugs and microscopic plants that bigger animals need to survive. Even low doses over time can stack up, especially in places where other solvents get dumped together.
The Environmental Protection Agency advises against draining chemicals like this into sinks or toilets. Rinsing containers haphazardly puts stress on water treatment plants and can slip into rivers or lakes. Landfills aren’t safe either, since chemicals can seep straight into groundwater. Responsible disposal involves high-temperature incineration or handing over to a hazardous waste handler—options that cost more but cut down on accidents and long-term pollution.
Solutions and Individual Choices Matter
It’s easy to view chemical safety as a job for scientists and regulators. Experience tells me that small choices make a big difference. I’ve seen accidents stem from skipping gloves or ignoring fume hood warnings just to save ten minutes. Shortcuts bite back. Taking chemical hazards seriously keeps more than just workers safe. Everyone along the supply chain benefits, from the plant mixing the solvent to the river downstream from a treatment facility.
Communities pushing for better toxic chemical reporting get results. Businesses switching to greener replacements keep raw materials under control and limit hazardous waste. Consumers can support brands that disclose their chemical practices—transparency drives safer habits.
At the end of the day, 1-ethylbutanol won’t wipe out a town, but its hazards pile up with poor habits and bad disposal practices. Treating it with healthy skepticism, proper training, and real accountability goes a long way for health and the local environment alike.
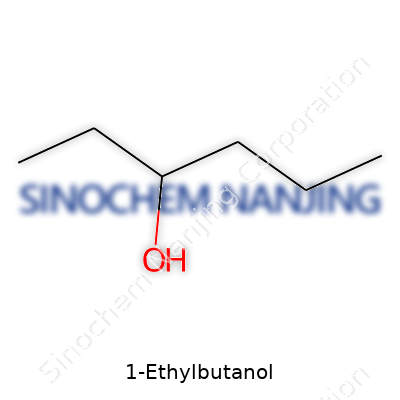
| Names | |
| Preferred IUPAC name | 3-Methylhexan-1-ol |
| Other names |
1-Ethyl-1-butanol 1-Ethylbutan-1-ol Ethyl n-propyl carbinol |
| Pronunciation | /ˌwʌnˈiːθɪlˌbjuːtənɒl/ |
| Identifiers | |
| CAS Number | 100-51-6 |
| 3D model (JSmol) | `C4H9CH2CH2OH` |
| Beilstein Reference | 1631328 |
| ChEBI | CHEBI:77717 |
| ChEMBL | CHEMBL43030 |
| ChemSpider | 74913 |
| DrugBank | DB14006 |
| ECHA InfoCard | ECHA InfoCard: 100.007.173 |
| EC Number | 203-990-0 |
| Gmelin Reference | 80538 |
| KEGG | C03041 |
| MeSH | D000073420 |
| PubChem CID | 11812 |
| RTECS number | EL8225000 |
| UNII | 75T64DKC1A |
| UN number | UN1128 |
| Properties | |
| Chemical formula | C6H14O |
| Molar mass | 102.18 g/mol |
| Appearance | Appearance: Colorless liquid |
| Odor | mild, vinous |
| Density | 0.815 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 0.97 |
| Vapor pressure | 0.642 mmHg (at 25 °C) |
| Acidity (pKa) | 15.0 |
| Basicity (pKb) | pKb ≈ 5.2 |
| Magnetic susceptibility (χ) | -74.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.409 |
| Viscosity | 2.965 mPa·s at 25 °C |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 339.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -368.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3735.7 kJ/mol |
| Pharmacology | |
| ATC code | J02AX13 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 68 °C |
| Autoignition temperature | 285 °C |
| Explosive limits | 1.0% - 8.6% |
| Lethal dose or concentration | LD50 oral rat 2,960 mg/kg |
| LD50 (median dose) | LD50 (median dose): **2,750 mg/kg (rat, oral)** |
| NIOSH | ELU |
| PEL (Permissible) | 100 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | IDLH: 800 ppm |
| Related compounds | |
| Related compounds |
1-Butanol 2-Butanol tert-Butanol Isobutanol 1-Pentanol |

