Looking Closer at 1-Diethylamino-4-Aminopentane: An Editorial Perspective
Historical Development
Chemical curiosity has always pushed scientists to explore odd corners of molecules and substances, and 1-Diethylamino-4-Aminopentane falls right into that category. The roots of this compound, sometimes called N,N-Diethyl-1,4-pentanediamine, trace back to the surge in organic and medicinal chemistry in the last century. Back when synthetic amines were taking over labs in the hunt for better drugs, adhesives, and intermediates, chemists learned to appreciate both the challenges and opportunities posed by flexible molecules like this one. During the industrial expansion era, when new polymers and pharmaceuticals came out almost every year, researchers looked for secondary and tertiary amines with controllable properties. Out of that crucible came the process refinements and application insights that now put 1-Diethylamino-4-Aminopentane on the map for both research and specialty manufacturing.
Product Overview
1-Diethylamino-4-Aminopentane often draws attention for its practicality and versatility, as it bridges a gap between pure laboratory chemicals and more mainstream industrial intermediates. The linear pentane backbone and dual amine groups give it a unique advantage in synthetic schemes, where other diamines often run into reactivity or stability issues. Scientists prize its cleaner reactivity profile and its ability to slot into multi-step processes with fewer side products. Some see it as a go-between, able to tweak physical properties in end products or serve as a clever building block for custom molecules. Its use hinges less on headline-grabbing applications and more on the quiet, essential work of systematic chemistry.
Physical & Chemical Properties
With a straightforward molecular structure—five carbons in a chain, wrapped with two amine groups at each end—1-Diethylamino-4-Aminopentane stands as a solid exercise in controlled molecular design. It appears as a clear liquid under ambient conditions and carries a noticeable amine odor, which is no surprise given the diethylamino functionality. Its boiling point, miscibility with water, and relative solubility in various organic solvents stem directly from its moderate chain length and the amine groups’ polarity. Handling this compound, folks have noted it resists oxidative breakdown better than many aliphatic amines, making it sturdier for storage and repeated sampling. This chemical stability, combined with predictable protonation and base strength, speaks to its value in both synthesis and product formulation.
Technical Specifications & Labeling
Down in the trenches, details like purity, residual moisture, and amine value matter more than fancy marketing. In most laboratory and pilot plant situations, the compound needs to be at least 98% pure, and that bar rises for pharmaceutical and electronics work. Labels from reputable vendors stick to concise identification, proper hazard pictograms, and common naming conventions to avoid confusion. From personal experience, the more precise the labeling and documentation, the fewer headaches down the pipeline, especially during scale-up or regulatory review.
Preparation Method
Those with a background in synthetic organic chemistry understand the satisfaction of a well-executed amination. Creating 1-Diethylamino-4-Aminopentane typically involves controlled alkylation of a suitable pentanediamine precursor, often by introducing diethyl groups using selective alkylating agents. The trick, learned after a few failed runs, lies in keeping conditions mild enough to avoid over-alkylation and side reactions, which can ruin yield and purity. Modern practices favor batch reactors with inert atmospheres and calculated stoichiometry to drive reactions cleanly, followed by fractionation or extraction to strip away incompletely reacted feedstock. Waste minimization and worker safety now weigh in as much as product purity in any forward-thinking operation.
Chemical Reactions & Modifications
If there’s a Swiss Army knife in the world of smaller amines, this one competes. The diethylamino and primary amino groups both offer anchor points for chemical modifications. Researchers explore acylation, alkylation, and coupling with aromatic systems to build larger molecules—ranging from pharmaceutical candidates to specialty surfactants. The ability to form stable salts or participate in cross-linking reactions has led to experiments in polymer science, creating blocks within advanced materials with targeted mechanical or electrical properties. Outside of the lab, chemical engineering teams keep testing how these reactions play out at different scales, always chasing higher yields and greater selectivity.
Synonyms & Product Names
Industry and academia both love giving compounds as many names as they have uses. 1-Diethylamino-4-Aminopentane often pops up under its systematic name, N,N-Diethyl-1,4-pentanediamine, but folks searching for “diethylaminopentanediamine” or related descriptors usually land at the same door. This web of synonyms ensures cross-compatibility in the literature yet sometimes causes confusion when ordering or interpreting data sheets. Double-checking CAS numbers has become second nature for anyone sourcing chemicals for sensitive research or regulated manufacturing.
Safety & Operational Standards
Working safely with amines calls for habits born out of experience, not just following checklists. 1-Diethylamino-4-Aminopentane, like other medium-chain amines, poses risks of skin and respiratory irritation and should be kept away from incompatible oxidizing agents or acids. Teams who work day in and day out with these substances invest in good ventilation and personal protective equipment and keep emergency showers and eyewash stations close to hand. Safety data sheets flag its flammability under certain conditions, so fire prevention demands respect. From a regulatory standpoint, keeping process residues and emissions in check remains a focus, especially with stricter environmental controls now shaping industrial practice.
Application Area
Those outside the chemical sector might never hear about 1-Diethylamino-4-Aminopentane, but dig a little deeper, and its fingerprint turns up in places ranging from fine chemical synthesis to polymer science and even experimental drug development. In research, it often acts as a precursor for tailored ligands and as a component in designing molecules meant to cross biological membranes or deliver active pharmaceutical ingredients. Some specialty adhesives and coatings gain better flexibility or adhesion when built around this compound’s core structure. Polymer chemists experiment with it to shape the performance of specialty materials, taking advantage of its bifunctional nature to control cross-linking or end-group functionality.
Research & Development
Active labs put 1-Diethylamino-4-Aminopentane through its paces, looking for fresh angles in synthesis, improved compatibility with enzymatic reactions, or ways to use it as a scaffold for medical research. Graduate students and industrial chemists both have stories about tweaking its base or salt forms to unlock new solubility profiles or reactivity patterns. Several published studies dig into its behavior in peptide coupling, while others look at how structured amines might enhance delivery molecules in gene therapy or diagnostic agents. Research often must weigh regulatory oversight and material cost against creative ambition.
Toxicity Research
Information about toxicity doesn’t spread as fast as synthetic protocols, but the responsible workplace always demands safety before scale. Like most lower molecular weight amines, 1-Diethylamino-4-Aminopentane raises concerns for its acute irritation potential. Animal studies and in vitro research show patterns of irritation and toxicity similar to those of related chemicals, but data on chronic exposure or long-term environmental impact still lags behind. This points to the need for new investigations—not only to secure worker health but also to future-proof any industrial roles this molecule may take on. Anyone serious about R&D invests time into reading full toxicity profiles and pushing for more complete toxicological data.
Future Prospects
With green chemistry and new drug development raising the bar on efficiency, safety, and sustainability, 1-Diethylamino-4-Aminopentane stands at a crossroads. Chemists with an eye for innovation keep poking at its unexplored reactivity and potential to serve as a modular unit in larger synthetic schemes. Industry shifts toward renewable feedstocks might bring new synthetic routes for amines like this, further reducing environmental footprints and dependencies on petrochemicals. As regulatory scrutiny tightens, the only way forward will be through a blend of smarter process design, deeper toxicity research, and open collaboration across the sciences. This compound’s story is far from over, and those tuning in from bench or boardroom can look forward to its next chapter unfolding alongside the relentless pace of chemical innovation.
What is 1-Diethylamino-4-Aminopentane used for?
What Stands Behind This Chemical
1-Diethylamino-4-aminopentane belongs to a group of compounds known to chemists as amines. These amines show up in a lot of everyday products people might not suspect. The structure of 1-Diethylamino-4-aminopentane gives it some interesting properties. Both the diethylamino and amino parts bring flexibility to the molecule. Plenty of manufacturers and researchers lean on that flexibility for various applications.
Where People See It Put to Use
A lot of chatter around this compound comes from the supplement world. Fitness circles occasionally mention it in the same breath as other “stimulant” ingredients. Some products push energetic blends that promise quick results, and sometimes this chemical finds a spot on those labels.
Beyond the supplement space, the pharmaceutical world pays attention to chemicals like this for their potential to change how the body sends certain signals. The amine backbone acts as a skeleton for a whole family of medicines. Chemists often tweak molecules like 1-Diethylamino-4-aminopentane while searching for new drugs, especially for use as stimulants or agents that wake up the central nervous system.
Potential Dangers and Oversight
Nothing pushes a chemical into the spotlight faster than loose regulation blended with big promises. 1-Diethylamino-4-aminopentane sometimes lands in a gray area. Not every country or market has clear rules about its safety or quality, yet the fitness industry often works faster than regulators. In practice, this means that consumers might see strange names on a pre-workout blend without knowing what long-term exposure does.
If it sounds familiar, it’s because there have been waves of concern about similar amines in the past, like DMAA and DMBA. These compounds wound up banned in several places once the risks — high blood pressure, rapid heartbeat, possible neurological harm — came into focus. The same attention should fall on any newcomer. Risk sneaks in when companies substitute one untested compound for another, claiming it’s “natural” or “safe.” Peer-reviewed studies on this particular molecule barely scratch the surface, and self-experimentation produces stories, not science.
Shifting Toward Evidence and Accountability
In my experience with sports supplements and medicine regulation, the gap between what goes on a label and what researchers know gets wider each year. Retailers and online shops tend to follow trends before researchers catch up. That stretches the time it takes to spot a real danger. There’s nothing new about companies jumping on the next big ingredient, but regulators, manufacturers, and consumers each share some responsibility.
One step people can take is pushing for transparency. Easy access to batch testing and full ingredient lists helps keep companies honest. Independent researchers also play a role by publishing findings, even when they raise doubts rather than deliver clear answers. Doctors and fitness coaches can stay informed by reading trusted journals instead of following influencer buzz. Education always beats guesswork — real harm comes from treating speculation like fact.
Building a Safer Future for Novel Compounds
Everyone benefits when health claims stand on solid ground. Products containing little-known chemicals demand good science before boasting benefits. As regulators pick up on new ingredients, clear rules pave the way for better consumer protection. Responsible companies can set the bar by investing in honest research instead of riding marketing hype.
1-Diethylamino-4-aminopentane may not dominate headlines now, but it shares a story with many chemicals before it. Until facts catch up with commerce, caution makes better sense than blind trust.
Is 1-Diethylamino-4-Aminopentane safe for human consumption?
Understanding the Chemical
Hearing about 1-Diethylamino-4-Aminopentane, people might wonder if they’ve stumbled onto the next generation of dietary supplements. This compound pops up in some supplement blends, often with bold claims about boosting energy or focus. The name isn’t catchy or familiar like caffeine or B vitamins, so skepticism is healthy. My own experience scrolling through supplement forums shows that people are looking for edges in workouts or studying, sometimes without pausing long enough to wonder about safety.
Transparency in Supplement Formulation
Supplements don’t go through the same rigorous process as prescription drugs. The FDA doesn’t vet every ingredient before it appears on store shelves. In the case of 1-Diethylamino-4-Aminopentane, there’s very little reliable clinical data on its effects in humans. Search for studies in peer-reviewed journals, and you won’t find much. That lack alone should give pause. Some folks might dismiss these concerns and trust marketing language, but the gap between label and evidence seems wider here than with older, better-studied compounds.
Lack of Safety Data
Safety reviews usually involve months or years of observing what happens to people taking a chemical, tracking not just intended effects but any harm. Without quality research, no clear picture emerges about short-term or long-term risks. Sometimes, companies list this ingredient by one of its many synonyms, making it even harder for consumers to know how much they’re taking, what it might do, or how it interacts with other substances.
Unknown Side Effects
Most reports from users come in the form of anecdotes online — someone says they felt more alert, while someone else got a headache or felt a racing heart. These stories pop up on bodybuilding forums or social media groups, not medical journals. Without broad studies, there’s no way to spot patterns or compare rates of adverse events.
Why Trust and Testing Matter
Trust in a product’s safety comes from transparency and strong science. Caffeine, for example, has gone through decades of study. Doctors know what too much looks like and understand how it moves through the body. The same can’t be said about 1-Diethylamino-4-Aminopentane. Unsanctioned compounds have landed in the news before for causing unexpected health scares. DMAA, which once flooded the supplement market, caused heart attacks and strokes, leading regulators to pull it from shelves. 1-Diethylamino-4-Aminopentane shares some chemical similarities, which should prompt extra scrutiny.
A Smarter Consumer Path
The solution isn’t complicated — demand evidence before putting trust in any new substance. Reading beyond flashy product claims and asking tough questions can save lives. Industry and regulators should shoulder responsibility, but consumers can push the conversation forward by seeking out transparent brands and verified ingredients. An open dialogue, backed by solid science, does more to protect health than hoping for the best with an untested compound. If a company can’t provide robust, independent research on a supplement’s safety, turning to time-tested alternatives often works out far better in the long run.
What is the recommended dosage of 1-Diethylamino-4-Aminopentane?
Looking Beyond the Label
Most people researching 1-Diethylamino-4-Aminopentane, sometimes popping up as "AEA" or similar names, run up against a wall: official dosage information simply doesn’t exist. Let’s clarify that point from the top. This compound hasn’t received approval from regulatory bodies like the FDA or EMA for medicinal or supplement use. So, if you’re here after spotting the name on a supplement label or in a gym chat group, know that there’s no government-endorsed answer to “how much is safe?”
No Substitute for Clinical Research
For any substance intended for human consumption, health authorities demand research. The story with 1-Diethylamino-4-Aminopentane doesn’t feature controlled human studies or peer-reviewed clinical trials. Nobody has completed proper work charting its effects in the body. Anecdotes float around forums — some claim energy boosts, others report the opposite, describing side effects that sound like rapid heart rate, headaches, or worse. None of these stories should guide medical decisions.
Sourcing Cloudy Origins
This compound sometimes appears in products labeled as pre-workout supplements. Manufacturers often swap out or hide stimulant-like chemicals in their formulas. Because there’s little oversight, buyers don’t know if a serving size of one “scoop” is safe or dangerous. Problems don’t just come from obvious overdose. Impurities, mislabeling, or combining with other stimulants can raise the risks.
Widely Recognized Red Flags
Almost every trusted health source—Mayo Clinic, Cleveland Clinic, WebMD—says the same about unapproved designer stimulants: avoid use. Anything with a blank spot where dosage instructions should go deserves double scrutiny. The World Anti-Doping Agency (WADA) and US military have taken steps to ban or warn about novel compounds like this, especially after stories of adverse reactions.
Why People Even Try It
The ingredient shows up because people are always looking for shortcuts to more energy, faster fat loss, or better workouts. I’ve met plenty of folks who roll the dice on untested chemicals, hoping for an edge. The drive often comes from internet hype or clever marketing, not any scientific backing. I get that impatience, especially with slow, grind-it-out progress in training. But short-term gain doesn’t outweigh long-term risk, and crossing into the territory of hazardous supplement ingredients makes nobody stronger in the end.
Smart Approaches
Instead of searching for a dosage, talk to a healthcare professional before touching anything new. If no one in the scientific community, medical field, or supplement industry can say how much of a substance is safe, steer clear. Supplements with a “proprietary blend” usually count on customers not asking questions. Every day, experts say that proven changes—improving diet, sleep, hydration, and natural exercise methods—give far better, longer-lasting returns. Looking for an edge from research-backed ingredients like caffeine or creatine, at recommended doses, makes sense for most.
Key Point
An unknown compound like 1-Diethylamino-4-Aminopentane belongs in the “do not touch” category until science catches up. Your health doesn’t grow stronger through guessing games.
Are there any known side effects of 1-Diethylamino-4-Aminopentane?
What People Ought to Know About This Compound
Many people scan ingredients on supplement labels or research chemicals, and stumble across names like 1-Diethylamino-4-Aminopentane. Some call it DEAP or DAEA. The chemical appears in certain pre-workouts and nootropics, favored by folks chasing sharper focus or greater physical output. It’s understandable to feel uneasy about unfamiliar names in your daily routine.
Most stories about DEAP trace back to its similarity with DMAA—a stimulant dropped from many markets due to safety complaints. This background sets off alarm bells for some, and rightly so. While DEAP looks like it might mimic stimulants found naturally in the body, like epinephrine, it's not the same. Just because something fires up your focus or energy, that doesn’t guarantee safety. The science behind this specific ingredient runs thin. You'd be hard-pressed to find trustworthy clinical trials that examine DEAP in real-world settings.
Known Side Effects and Potential Risks
People using supplements that list DEAP often report familiar issues: headaches, jitteriness, rapid heartbeat, or high blood pressure. These match up with what you’d expect from strong stimulants. Some users complain about irritability, nausea, or trouble sleeping, too. These stories don’t come from polished medical journals, but peer-to-peer supplement forums or manufacturer disclaimers.
The real problem lies in the lack of open, peer-reviewed studies on this compound. Most side effect lists stick to reports from other similar substances, crossing their fingers that DEAP doesn't cause anything worse. There’s no oversight from authorities like the FDA on these ingredients, and safety testing sits behind marketing claims. From my time around gym regulars and supplement enthusiasts, some wind up mixing products without realizing the dangers stack up—like hypertension or unintentional dehydration. Stimulant overload leads to emergency room visits more often than supplement ads suggest.
Checking the Facts on 1-Diethylamino-4-Aminopentane
Facts about DEAP remain scarce because most research sticks to animal models or cell cultures, which might not match what happens in a real body. For folks managing heart conditions, blood pressure, or anxiety, any new stimulant poses unnecessary risks. Even for otherwise healthy people, side effects can go from annoying to alarming—especially with large doses, or when mixed with caffeine or other pre-workout stimulants.
Since regulation lags behind the industry, smart shoppers always dig for independent third-party testing or quality certification. If a company won’t share ingredient sources or safety data, that signals a problem. Supplement manufacturers rarely spend time proving safety unless forced by regulators. So the onus shifts to users, gyms, and doctors to connect the dots when bad reactions happen.
Steps Toward Safer Supplement Habits
Before buying anything with DEAP or similar chemicals, checking your own health comes first. If you’re not sure how your body responds to new compounds, talk it through with a healthcare provider—preferably one who keeps up with sports medicine or nutrition science. Friends at the gym often share stories about what gave them a boost, but every body handles things differently.
Better regulations would help—mandating proper clinical studies, clear labelling, and post-market surveillance. In the meantime, sticking to reputable brands with solid safety history, paying attention to your own side effects, and limiting the stacking of stimulants provides a safer path. Every supplement carries some risk, but knowledge helps folks make better choices for their bodies and lifestyles.
Where can I purchase 1-Diethylamino-4-Aminopentane?
Understanding What You’re Looking For
Curiosity runs high around 1-Diethylamino-4-Aminopentane and how people go about buying it. This compound doesn’t pop up on retail pharmacy shelves or in the supplement aisle. Folks searching for this are often doing research for a university, a lab, or the chemical industry. It’s not something you’ll grab off Amazon for a home project.
Legitimate Supply Channels
My own time in research labs taught me a lot about sourcing specialty chemicals. Most people go through certified chemical suppliers. These are companies that ask for business login, proof of credentials, or detailed paperwork. Sigma-Aldrich, Fisher Scientific, and TCI America have long lists of chemicals for research, and they ask the right questions before shipping anything out. Industrial buyers have reps to talk them through safety and regulations. Private individuals looking to buy compounds like this are almost always out of luck unless they partner with a verified institution.
Risks of Shady Online Sellers
Not every listing on the wilds of the internet provides real chemical products, and not every supplier cares about the law or safety. Chemistry forums and social media sometimes buzz with names of gray-market sites shipping everywhere with no questions asked. It’s an easy trap: slick websites, no business credentials needed, attractive prices. The risk here goes beyond getting scammed; you might land chemicals that aren’t what they claim, cut with who knows what, or worse, flagged in customs. That’s a call no one wants.
Safety and Legal Considerations
Expect most countries to regulate sales of structures even remotely tied to pharmaceuticals or research chemicals. For instance, the US keeps a close eye on compounds with stimulant potential or links to controlled substances. Buyers—even with the purest intentions—can fall into legal gray areas. My work with campus procurement officers drove home how much compliance eats into process. No purchase gets approved without checking laws, SDS documentation, end-use declarations, and handling plans.
Why Testing and Verification Matter
Even if you get the compound from a trusted seller, there’s still the question of quality. Every shipment at my old lab needed matching certificates of analysis and purity testing. This protects everyone from research disasters—or worse, harmful exposure. Commercial labs, not mail-order mystery sources, provide traceable paperwork.
Alternative Solutions
People interested in this compound for research or innovation should start with institutional help. University procurement teams can vet suppliers and handle the paperwork. Industry partners can unlock access through established contracts. If you don’t have a lab and the paperwork seems overwhelming, talking directly to a chemist or a university chemistry department might spark the right partnership. Many breakthrough projects start this way, rather than through the back channels of ecommerce.
Final Thoughts on Safe Access
Working within the rules protects you, your research, and your reputation. Online shortcuts carry high risks in this field. Focusing on certified suppliers, transparency, and documentation gives you the best shot at reliable, safe, and legal access.
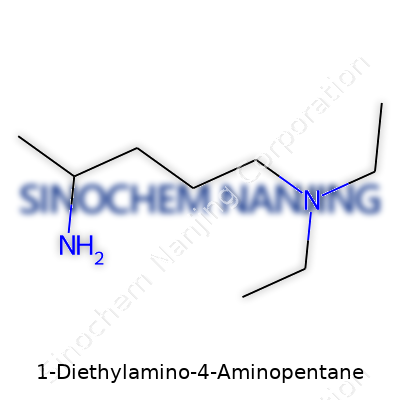
| Names | |
| Preferred IUPAC name | N-ethyl-N-(3-aminopentyl)ethanamine |
| Other names |
1,5-Diamino-3-diethylaminopentane N,N-Diethyl-1,5-pentanediamine |
| Pronunciation | /ˈwʌn daɪˌɛθɪl əˌmiːnoʊ fɔːr əˈmiːnoʊ ˈpɛnteɪn/ |
| Identifiers | |
| CAS Number | 3938-88-9 |
| 3D model (JSmol) | `CCC(CN(CC)CC)N` |
| Beilstein Reference | 82140 |
| ChEBI | CHEBI:16643 |
| ChEMBL | CHEMBL4045731 |
| ChemSpider | 11042787 |
| DrugBank | DB14065 |
| ECHA InfoCard | 03ba4a99-aa9e-414a-99fb-03b53fa35f7a |
| EC Number | 211-207-0 |
| Gmelin Reference | **Gmelin Reference:** 83651 |
| KEGG | C08325 |
| MeSH | D015410 |
| PubChem CID | 134748037 |
| RTECS number | YK8575000 |
| UNII | N6TMJ26537 |
| UN number | UN2711 |
| CompTox Dashboard (EPA) | DTXSID90968725 |
| Properties | |
| Chemical formula | C9H22N2 |
| Molar mass | 131.24 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Amine-like |
| Density | 0.84 g/cm3 |
| Solubility in water | Slightly soluble |
| log P | 0.98 |
| Vapor pressure | 0.157 mmHg at 25°C |
| Acidity (pKa) | 10.73 |
| Basicity (pKb) | 2.92 |
| Magnetic susceptibility (χ) | -72.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.432 |
| Viscosity | 2.33 cP (25°C) |
| Dipole moment | 3.0904 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 403.23 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. H335: May cause respiratory irritation. |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P260, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P330, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 77 °C (closed cup) |
| Lethal dose or concentration | LD50 oral rat 273 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 320 mg/kg |
| NIOSH | UY4375000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.7 mg/m³ |
| Related compounds | |
| Related compounds |
1,4-Diaminopentane 1-Diethylamino-2-pentanol 1-Diethylamino-4-nitropentane Diethylamine Pentylamine |

