1-Chloro-2-Bromopropane: Digging Deeper Into a Niche Yet Notable Chemical
Tracing the Origins and Development
1-Chloro-2-bromopropane has its roots dug deep into the broader world of halogenated organic compounds. Early research into alkyl halides paved the way for discoveries tied not just to chemistry itself but to changes in industrial practices across the last century. Chemists first stumbled onto compounds like 1-chloro-2-bromopropane by exploring the effects of substituting different halogens onto hydrocarbon chains. The evolution of organic synthesis in academic labs and later in industrial production plants saw this compound move from a curiosity to something useful both as a reagent and as an intermediate for making other chemicals. Cursory glances at old journals reveal an evergreen scientific curiosity: what happens when you swap out chlorine for bromine or vice versa in a propyl backbone? That lens carried the field from basic research to a point where today’s researchers and manufacturers can take these discoveries for granted and build from them.
Overview From a Product Perspective
Any conversation about 1-chloro-2-bromopropane can draw from direct experience working in university glassware—its slightly pungent odor, its slick behavior in separatory funnels, the steady hum of its virtues as both a building block and sometimes as a solvent. This compound, with its dual halogenated structure, catches the eye among similar small-molecule organics. It stands not just as an intermediate in organic synthesis; it has grown to find practical value in research, analytical chemistry, and niche areas of industrial formulation. Its market footprint lags behind bulk chemicals like chloroform or dichloromethane, but ask a synthetic chemist about halogen exchange reactions or the preparation of labeled compounds and you may get a seasoned nod.
Physical and Chemical Properties
Focusing on its essence, 1-chloro-2-bromopropane is a colorless liquid at room temperature, giving it away as neither too volatile nor tricky to handle in normal laboratory setups. It sinks below water, a hint at its density and the influence of the halogen atoms. Its boiling point sits within reach for distillation in a standard organic lab—a practical feature for those who need to purify it or use it as a solvent in a pinch. The molecular structure, balancing between the larger, heavier bromine atom and the slightly smaller chlorine, packs three carbons and enough polarity to blend into many non-polar and slightly polar organic solvents. The reactivity stands a notch above simple alkanes but below more aggressive halogenated compounds, making it cooperate in substitution or elimination reactions with the right approach. From a sensory angle, a whiff of it in the air tells veterans to turn up the fume hood.
Technical Specifications and Labeling Realities
Chemicals do not come off the shelf without their labels and paperwork, but in the trenches, what matters is clarity on percent purity and contaminant profile. Laboratory stocks of 1-chloro-2-bromopropane most often show purity levels topping 97 percent, sometimes creeping up to reagent-grade ratings of 99 percent, depending on the manufacturer’s process. Impurities often track back to downstream halogen exchange products or leftover solvent traces from preparation. Reliable bottles tell you at a glance whether your compound is fit for precise synthesis work or if a quick distillation is worth your time. Those metrics matter less for a large industrial scale, but even there, technical specs become a workaround for avoiding unwanted surprises in sensitive downstream processes. Take it from anyone on the laboratory floor—misreading purity levels leads to stopped reactions, failed experiments, and wasted hours.
How It’s Made
The preparation of 1-chloro-2-bromopropane can be seen as a lesson in controlled substitution chemistry. Conventional routes favor starting from 1,2-dichloropropane, replacing a chlorine atom with a bromine under carefully moderated conditions. Others capitalize on the easier availability of 2-bromopropanol, converting this alcohol with thionyl chloride or phosphorus-chlorinating agents to install the chlorine while keeping the bromine in place. These preparations echo through undergraduate organic labs: pick your precursor, control your addition of halogenating agents, monitor for side products, and keep your reaction environment dry to prevent unwanted hydrolysis. On the industrial level, continuous flow reactors and careful distillation strip away byproducts and contaminants, producing usable amounts under stricter controls to keep cost and waste down.
Reactions and Modifications in the Lab
Working with 1-chloro-2-bromopropane in organic synthesis brings both a sense of challenge and familiarity. Its structure invites nucleophilic substitution, a reaction that most chemists know as SN2. That single carbon flanked by both halogens makes it accessible for tailored substitutions, allowing the chlorine or bromine atom to be swapped out depending on the incoming nucleophile. It plays nicely in the preparation of a range of other organohalides and, in the hands of experts, enables the synthesis of more complex compounds with selective halogen placement. Eliminations, particularly in the presence of strong bases, offer yet another route, bringing about alkenes that can further extend a reaction sequence. For those who practice the sometimes messy art of organic chemistry, 1-chloro-2-bromopropane keeps its place as a reaction chameleon and a ready source of functional diversity.
Synonyms, Product Names, and Trade Realities
Naming conventions for 1-chloro-2-bromopropane come from the systematic world of IUPAC but spread into older, industry-rooted language. Some refer to it as 2-bromo-1-chloropropane, flipping the order but not the substance. The presence of both “propane” and different halide prefixes means you bump into alternate names such as 1-bromo-2-chloropropane or 2-chloro-1-bromopropane, usually depending on where synthesis traditions set the numbering. On product listings, these distinctions can mean the difference between getting exactly what you need and finding something half right. That misstep can eat a week of productivity, as anyone who has ever waited for a reorder cycle knows all too well.
Keeping the Work Safe and Standardized
Safety with 1-chloro-2-bromopropane starts before the reagent even leaves its bottle. Anyone who ever loaded it into a reaction vessel recalls the sharp, pungent fumes—fast to warn you to respect your fume hood. Like a lot of halogenated organics, it causes headaches or dizziness if inhaled at high concentrations, and it does not respond kindly to skin or eye contact. Spills mean slick floors and sticky gloved hands. Proper practice demands not only gloves and goggles but a sense of discipline—keeping bottles capped, glassware clean, and waste properly contained. Each run-through reminds those in the lab why standards from organizations like OSHA and good laboratory practice guidelines carry real-world weight. Training new researchers on how to handle, store, and dispose of halogenated materials reduces risks and keeps work running. Lapses bring not just personal harm but lost research and regulatory red tape.
Applications: Real Uses in the Real World
You do not find 1-chloro-2-bromopropane in household products or public consumer goods, but its value in research and specialist production stands out. Research chemists harness its reactivity in the pursuit of new reaction pathways and as an intermediate for more elaborate organic molecules. Analytical labs turn to it when calibrating certain detection equipment or synthesizing isotopically labeled standards. Every so often, it features in pilot studies for pharmaceuticals or fine chemical production, acting as a building block for those who must install both chlorine and bromine functionalities with precision. In these environments, accuracy counts—not just for product yield but for ensuring that downstream chemistry works as expected.
Keeping Pace With Research and Development
Academic and industrial research treats 1-chloro-2-bromopropane as more than a static entity; it often serves as a benchmark for understanding the impact of halogen substitution on reactivity and mechanism. Newer fields like green chemistry look to lessen the environmental footprint of halogenated synthesis, driving researchers to develop more efficient, less wasteful ways of preparing and using this compound. Analysts working at the boundaries of detection methods use it as a case study for understanding halogen interference in measurements. There is an ongoing push to engineer better catalysts and milder conditions that maintain the useful parts of this molecule’s reactivity profile while reducing the safety and disposal headaches that traditionally follow halogen chemistry.
Toxicity: What the Data Shows
Toxicological research on 1-chloro-2-bromopropane follows two main paths—understanding its acute risks in lab and industrial contexts, and its longer-term effects on health and the environment. Acute exposure, especially through inhalation or direct contact, definitely puts workers at risk for mucous membrane irritation, headaches, and possible central nervous system effects if exposure runs high enough for long enough. Studies on related halogenated organics show a tendency for liver and kidney effects after chronic exposure, and this compound sits squarely in line with that family profile. Data remains limited in terms of carcinogenic potential, so regulatory agencies set handling and exposure thresholds based on analogous chemicals and observed short-term toxicity. To researchers who have handled it, the warnings do more than decorate a data sheet—they remind you to close your bottle, check your gloves, and respect the rules for safe disposal.
Looking Forward: Future Prospects and Challenges
Innovation never stands still around specialty organics. For 1-chloro-2-bromopropane, the challenge and opportunity both stem from its halogenated structure. Growing concern about persistent, bioaccumulative chemicals pressures researchers and manufacturers to minimize waste and look for clean-reacting alternatives or improved preparation routes. There is a clear appetite for developing catalysts and synthetic methods that get more out of every molecule with less environmental fallout. The bridge between pure academic research and practical application continues to shape how this and similar chemicals get used. Expectations for better monitoring, tighter exposure limits, and more robust safety protocols keep increasing as new data emerges. Ultimately, the future of 1-chloro-2-bromopropane reflects the broader march of chemistry: drive for efficiency, value in targeted research, no shortage of respect for safety, and a growing focus on the impact of our choices inside and outside the lab.
What is the chemical formula of 1-Chloro-2-Bromopropane?
Understanding 1-Chloro-2-Bromopropane
1-Chloro-2-bromopropane sounds like a mouthful, but breaking down the name helps make sense of its structure. Chemists use names that actually tell you where each atom sits in the molecule. In this case, “propane” gives away that the main backbone contains three carbon atoms. Then, chlorine and bromine each attach to the chain at different spots: chlorine sticks to the first carbon, bromine joins the second. Lay it out visually and you get C3H6BrCl.
Why Structure Matters
Small changes in where atoms hook onto carbon chains make big differences. Swapping the positions of chlorine and bromine or changing which carbon each attaches to leads to a totally different compound. The real world proves this point. Think about how isomers like glucose and fructose both have the formula C6H12O6 but act differently in your body. For 1-chloro-2-bromopropane, function depends on its exact structure, too.
Everyday Chemistry—Why Does the Formula Matter?
Chemical formulas aren’t just classroom trivia. Whether you work in a lab or tinker at home, knowing a molecule’s make-up keeps people safe and helps avoid expensive mistakes. Take it from someone who’s watched the cleanup after a small lab fire—misunderstandings about what’s in a bottle can mean bad news. The formula also guides how this compound reacts with others. For example, once you know it’s C3H6BrCl, you can figure out the byproducts when it reacts with sodium hydroxide. These details shape decisions in real chemistry, from industrial factories to research.
Safety Is Not Just Fine Print
Compounds with halogens like chlorine and bromine deserve respect. Breathing in fumes, or spilling the liquid, isn’t harmless. Chlorinated and brominated hydrocarbons often bring health risks—think nerve effects or long-term problems if handled carelessly. Those labels covered in pictograms and warnings? They tell the truth. Proper gloves, eye protection, and good ventilation are as important as knowing the formula.
Supporting Trust: Facts and Transparency
Reliable information makes for better decisions. Researchers double-check their sources and always look for trusted databases—like the National Institutes of Health or PubChem—when tracking down formulas. Building confidence with readers or students means pointing to these sources instead of guessing from memory or cutting corners. You’ll see C3H6BrCl listed the same way across reliable references. Lab reports and safety sheets back it up, echoing the need for accuracy and honesty in chemistry education.
Moving Forward with Confidence
Chemistry isn’t just about big discoveries or breakthroughs. Everyday attention to basic facts—like the formula of 1-chloro-2-bromopropane—protects health, budgets, and experiments. Solutions start with education: teachers, lab managers, students, and curious learners gain from clear explanations and verified facts. Training, labeling, and easy-to-find data stop simple misunderstandings from turning into bigger problems. The next time someone asks about a formula, double-check. It's not just science—it’s smart practice.
What are the main applications or uses of 1-Chloro-2-Bromopropane?
Lab Reagent for Chemical Synthesis
In the lab, speed matters, but so does accuracy. Chemists looking for a selective alkyl halide often pick 1-chloro-2-bromopropane. The compound’s double halogen structure makes it ideal for studying reaction mechanisms, testing nucleophilicity, and exploring substitution or elimination routes. Many undergraduates get their hands on it in advanced organic classes, and seasoned researchers rely on it to train new team members. It’s this utility that helps labs probe fundamental principles, especially in teaching experiments comparing the behavior of different halides.
Building Specialty Chemicals
Anyone working in specialty chemicals needs building blocks that deliver reliability and control. 1-chloro-2-bromopropane serves here as a versatile intermediate. Pharmaceutical and agrochemical manufacturers see it as a starting point for crafting complex molecules, including active pharmaceutical ingredients or crop protection agents. Its mix of chlorine and bromine lets chemists swap out these atoms under mild conditions, which is useful for creating molecules that mimic or block natural signals in organisms. Rather than reinventing the wheel each time, companies count on compounds like this for efficiency and flexibility.
Testing Instrument Calibration and Methods
Instrument calibration might sound boring, but without it, results drift and trust erodes. 1-chloro-2-bromopropane’s stable and predictable behavior helps labs validate gas chromatography and mass spectrometry methods. Analytical chemists often spike samples with this compound, checking the accuracy, precision, and sensitivity of their instruments. Quality control professionals want assurance that their data reflects reality, not just hope. That confidence is essential for companies working under regulatory pressure, especially in pharmaceuticals or food testing labs.
Safety in Handling and Environmental Impact
Anyone who has managed a chemical storeroom knows the importance of safety. 1-chloro-2-bromopropane poses hazards because of its halogens and volatile organic profile. It can irritate skin, eyes, and lungs, so gloves, goggles, and good ventilation are non-negotiable. Waste disposal isn’t just a headache; it’s a legal and ethical requirement. Treating any spent material as hazardous and arranging proper incineration or specialized disposal cuts the risk of environmental leaks. In my own time coordinating research projects, I've seen strict tracking and staff training make the difference between routine work and accidents.
Potential Solutions for Safer and Greener Use
Industry and academia face mounting pressure to reduce environmental footprints. Many companies now look for less harmful substitutes or design greener processes that generate less waste or swap in safer solvents. Some labs invest in fume hoods with advanced filtration, which help reduce emissions. Others partner with waste management firms to recycle solvents or decompose hazardous byproducts responsibly. Professional development—teaching safe handling and disposal—remains a cornerstone, especially for new team members. Champions for sustainability aren’t just managers talking about policy; they’re front-line workers adopting safer habits day after day.
Final Thoughts
1-chloro-2-bromopropane isn’t a household name, but it plays a quiet role in labs, manufacturing, and safety programs. For anyone who cares about science and public health, using this compound wisely speaks to a bigger commitment: trusting the process, protecting people, and minimizing risks for the next generation.
What are the storage and handling precautions for 1-Chloro-2-Bromopropane?
Understanding the Real Hazards
Anybody with a chemical background knows that handling reagents like 1-Chloro-2-Bromopropane involves more than ticking boxes. This compound carries some clear hazards: flammability, toxicity, and the risk of skin or respiratory exposure. I’ve seen what a simple mistake with volatile halogenated solvents can do—rashes, breathing trouble, or even bigger accidents if they catch fire. Getting too comfortable around these chemicals often leads to close calls.
What Makes 1-Chloro-2-Bromopropane Risky?
This compound doesn’t forgive sloppy handling. Its vapors catch fire easily. Splashing it can irritate skin and burn eyes. Inhaling its fumes digs into the lungs, leaving you coughing and lightheaded for hours. Nobody wants to rush a colleague to the sink or the emergency room—we read enough warning labels to know these are not empty threats.
Storing It Right
Proper storage starts with picking the right spot. At every lab I’ve worked, we use ventilated flammable storage cabinets, away from direct sun and heat. Placing it at the right temperature makes a difference; keep it cool but never freezing, and far from anything reactive like oxidizers or acids. I always check bottle seals because I’ve once found a cracked cap that let fumes leak into the whole storage unit. It’s not difficult to prevent, but you need to pay attention.
Personal Protection Always Comes First
Every time I open a bottle of this stuff, I reach for gloves, goggles, and a fresh lab coat—no exceptions. Even a small spill gets through regular fabric fast. Adequate ventilation matters too: if you’ve ever caught the stinging whiff of this compound inside a closed room, you know open benches won’t cut it. Chemical fume hoods are a must; no shortcuts. Splash-proof face shields make sense for those who transfer larger amounts or prep solutions, since one sudden jerk can drench your face. Having an emergency shower and eyewash nearby isn’t just for inspection visits. We’ve all seen chemical accidents happen faster than anyone expects.
Tackling Spills and Accidents
If you spill 1-Chloro-2-Bromopropane, don’t grab a mop. Use an absorbent specified for halogenated organics, scoop up the material, and dump it into a sealed waste container. Ventilate the area straight away—turning on a fan or increasing airflow in the fume hood reduces risk. I make sure to train new staff to call for help, not to fix spills alone. Emergency numbers must stay taped above the workstation. Once, I saw hesitation cause more harm than the chemical itself.
Waste Isn’t Garbage
Down the drain solutions tempt in a pinch, but this practice pollutes water and breaks the law. Our approach takes a clear line: solvent waste container plus proper labeling. Disposal teams know exactly what’s in that waste when they arrive. Reactivity happens quickly with the wrong combination in shared waste. Avoiding surprises keeps the whole building safer.
Building Safer Habits
Smart handling of 1-Chloro-2-Bromopropane relies on sticking to protocols every time, not just during inspections. The best safeguard is honest communication—flagging risks, watching out for improper storage, and practicing clean-up drills. I encourage every chemist in training to respect the stuff—not fear it, but treat every bottle like it’s their responsibility. Good habits limit accidents, protect the team, and keep research moving forward.
What is the CAS number for 1-Chloro-2-Bromopropane?
Why the CAS Number Matters in Chemistry
People working in labs or running chemical supply businesses know that finding the right compound often feels like searching for a needle in a haystack. Many chemicals have names that sound similar or have slight variations, so one digit off in a name or formula could mean mixing up harmless solvent with something dangerous. This is where the CAS number comes in handy. The Chemical Abstracts Service Registry Number, or CAS number, acts a bit like a digital fingerprint. For 1-Chloro-2-Bromopropane, that identifier is 4219-73-4.
Personal Experience in the Lab
Working with chemicals daily teaches you to double-check everything. I remember a time a friend of mine ordered the wrong chemical for a synthetic reaction because two compounds had close names—just one prefix different. His project got delayed by weeks. The only surefire way to avoid that kind of mistake is to pay attention to the CAS number. The number 4219-73-4 for 1-Chloro-2-Bromopropane isn’t just bureaucratic red tape; it’s a lifesaver.
Trust and Accountability in Chemical Sourcing
Most suppliers run inventory with CAS numbers, not just names. Scrupulous manufacturers include CAS numbers on labels, safety data sheets, and certificates of analysis. This level of detail builds trust, cuts down on counterfeit products, and keeps people safe. Imagine working in pharmaceuticals—nobody wants any confusion over which ingredient ends up in a drug batch. Using 4219-73-4 as a standard gives everyone a single reference point.
How CAS Numbers Help in Research and Regulation
Journals, scientific databases, and chemical catalogs all catalogue entries with these numbers. Reducing ambiguity cuts through a lot of jargon. Google Scholar or Scopus pulls up research articles tied to 4219-73-4 much faster than fiddling with all the ways “1-Chloro-2-Bromopropane” could get spelled or abbreviated. Regulators depend on the same codes when they assess safety risks, import/export controls, or hazardous chemical registries.
Real Challenges and Solutions
Mistakes happen. Shipping departments sometimes grab the wrong drum. Interns may not notice a swapped barcode when restocking. Problems like these waste money and endanger lives. Simple workarounds—like requiring a CAS number on every order sheet, safety label, and project summary—keep operations smoother. Software that syncs with chemical databases using CAS numbers avoids mistakes and saves headaches.
Adopting CAS numbers as the default language in science doesn’t just help big pharmaceutical firms or chemical giants. Small research groups, teaching labs, and local manufacturers also gain a strong tool for accuracy, efficiency, and transparency. It pays off to be scrupulous—spending an extra minute verifying a number like 4219-73-4 beats weeks of troubleshooting a failed reaction or explaining a regulatory violation.
A Sense of Reliability
CAS numbers bring order to the crowded world of chemicals. Keeping the right one for each compound gives everyone from students to seasoned chemists peace of mind. Countless hours and resources get saved just by referencing that nine-digit code. The lesson sticks: in labs and factories, double-check the CAS number, always.
Is 1-Chloro-2-Bromopropane hazardous to health or the environment?
A Closer Look at 1-Chloro-2-Bromopropane
People working in labs or factories probably recognize 1-Chloro-2-Bromopropane as a colorless liquid with a mild, sometimes sharp odor. This chemical isn’t as famous as solvents like acetone, but it turns up in a surprising range of chemical reactions, especially when scientists want to swap atoms around on carbon skeletons.
Now, the question isn’t just about how useful this chemical can be. The bigger issue comes from what it does to our bodies and our home ground — both at work and outside the lab.
Health Effects Aren’t Always Obvious
Years ago, during a summer job in a research lab, I learned early on to respect anything with a “halo-” prefix — halogenated hydrocarbons, like 1-Chloro-2-Bromopropane, don’t play nice with people. Even brief skin contact can trigger redness or a burning feeling. Vapors from chemicals in this group, if people breathe them regularly, often cause headaches or dizziness. There's also a lingering uncertainty about what happens after months or years of exposure.
Chlorinated and brominated chemicals tend to collect in fatty tissues. This isn’t just theory. Workers in manufacturing who have handled similar compounds for years often walk away with liver or kidney complaints. A paper from the International Programme on Chemical Safety notes that many haloalkanes can damage the central nervous system and even disrupt heart rhythms at high exposures. Chronic contact, as seen in industrial settings, can set off even bigger problems.
Impact on the Environment
Getting rid of 1-Chloro-2-Bromopropane doesn’t mean it just disappears. Waterways downstream from chemical plants have shown traces of compounds like this, partly because they don’t break down quickly. Once they slip into the soil or get washed into rivers, it can take years for microbes or sunlight to fully finish their work.
Animals living near industrial outflows pay the price—fish that swim through contaminated waters can suffer from lower reproductive rates and strange mutations. Insects and small creatures like worms often vanish first, which tips the balance for birds and bigger animals who eat them. I’ve seen local streams go quiet after chemical releases from factories up the road—a sign that even minor leaks matter.
Bigger Picture: Responsible Handling and Solutions
The risks aren’t reasons to panic, but they point to the importance of clear safety standards. Gloves, goggles, and well-sealed containers should be the basics. Good ventilation makes a noticeable difference, since once a chemical gets into the air, the chance for exposure multiplies. At one job, switching from open buckets to closed systems cut the number of headaches and skin rashes among workers in less than a month.
Regulation can’t step in for plain common sense on the ground. Most labs in Europe now require material safety data sheets and proper waste disposal logs before opening a single bottle. Incinerators designed for halogenated wastes keep byproducts out of the soil and air. Even at small scales, people soak up knowledge by seeing clear rules in use—lessons that spread fast on shop floors and classroom benches.
Some chemists have started looking for alternatives that break down more quickly and don’t stick around in the body or water systems. But substitutes don’t always have performance or price on their side just yet.
If handling of 1-Chloro-2-Bromopropane stays sharp and routine checks hold everyone accountable, the risks shrink. The best outcomes usually follow when companies and schools teach real respect for what’s in those bottles, not just a quick warning on a label.
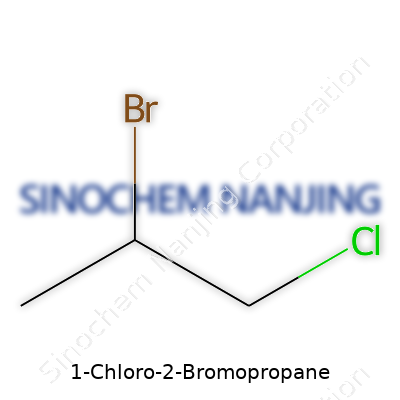
| Names | |
| Preferred IUPAC name | 2-Bromo-1-chloropropane |
| Other names |
2-Bromo-1-chloropropane 1-Chloro-2-bromopropane Isopropyl bromide chloride 1-chloro-2-bromopropane |
| Pronunciation | /ˈklɔːrəʊ-ˈbrəʊməʊ-prəˈpeɪn/ |
| Identifiers | |
| CAS Number | 6307-59-1 |
| 3D model (JSmol) | `/JSmol/3d/1/1,1-chloro-2-bromopropane.mol` |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:132673 |
| ChEMBL | CHEMBL1698860 |
| ChemSpider | 167369 |
| DrugBank | DB11651 |
| ECHA InfoCard | ECHA InfoCard: 100.008.969 |
| EC Number | 203-299-6 |
| Gmelin Reference | 106954 |
| KEGG | C18919 |
| MeSH | D015242 |
| PubChem CID | 13339 |
| RTECS number | TX9625000 |
| UNII | NY83V45R2O |
| UN number | UN2346 |
| CompTox Dashboard (EPA) | DTXSID9059575 |
| Properties | |
| Chemical formula | C3H6BrCl |
| Molar mass | 156.44 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | sweet odor |
| Density | 1.34 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 2.6 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 50 |
| Magnetic susceptibility (χ) | -74.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.471 |
| Viscosity | 1.821 cP (20°C) |
| Dipole moment | 2.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 350.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -67.5 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4666.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P312 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 58 °F |
| Autoignition temperature | 590°C |
| Explosive limits | Explosive limits: 2.5–10% |
| Lethal dose or concentration | Lethal dose or concentration: **LD₅₀ (oral, rat): 2350 mg/kg** |
| LD50 (median dose) | LD50 (median dose): 1900 mg/kg (oral, rat) |
| NIOSH | CN8400000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL : NIOSH considers 1-Chloro-2-Bromopropane to be a potential occupational carcinogen. |
| Related compounds | |
| Related compounds |
1-Bromo-2-chloropropane 1-Chloro-2-propanol 2-Bromopropane 1,2-Dibromopropane 1,2-Dichloropropane |

