Unpacking 1-Chloro-2,3-Epoxypropane: In-Depth Reflections on Its Role, Risks, and Research
Historical Development
Some chemicals become so tightly linked to industry and science that it’s easy to forget they weren’t always sitting on lab shelves. 1-Chloro-2,3-Epoxypropane, widely recognized by its common name epichlorohydrin, turned into a workhorse during the rapid growth of industrial chemistry throughout the twentieth century. Before synthetic polymers caused the plastics boom, chemists seeking tough, chemical-resistant materials started tinkering with small molecules like this one. Epichlorohydrin’s long-standing use in creating epoxy resins, which took off after World War II, came from its unique reactivity and versatility. Though first prepared in the 1800s, large-scale manufacturing and commercial demand picked up steam alongside the rise in modern construction, electronics, and automotive industries that leaned heavily on epoxies. Seeing how something with such a simple structure can drive whole economies always puts things in perspective.
Product Overview
People often associate 1-Chloro-2,3-Epoxypropane with just "epoxy glue," but its reach stretches far wider. As a colourless and somewhat pungent liquid, this chemical finds roles in making not only adhesives and coatings but also water treatment agents, pharmaceuticals, and even paper additives. Having watched the manufacturing process in person, it struck me just how demanding quality control can be. Manufacturers focus on purity, minimizing byproducts, and adjusting properties to fit each downstream use, whether it’s making something hard as rock or carefully drug-compatible. The drive for safety and performance improvements constantly reshapes how companies handle and package this material. That kind of rigor links back to the sometimes harsh lessons learned through decades of trial, error, and—occasionally—disaster.
Physical & Chemical Properties
A clear, mobile liquid, 1-Chloro-2,3-Epoxypropane boasts a boiling point under 120°C, which means it evaporates quickly in a warm room. Its low viscosity makes pouring easy but also calls for care: spills become hard to control. The chlorine atom in its structure lends strong reactivity, making it corrosive to skin and mucous membranes. Its distinctive ether-epoxide ring is highly strained, almost begging to react with any nucleophile, driving much of its value in synthesis but also posing a challenge for storage and transport. The trick with any reactive aldehyde or epoxide comes down to temperature control and moisture exclusion, so storage facilities look more like high-security vaults than simple chemical cupboards. Flammability and the ability to generate toxic gases under improper conditions demand robust ventilation and constant vigilance.
Technical Specifications & Labeling
The world of technical specs can look dry on paper, but for those handling the real stuff, it’s a matter of health and compliance. Common standards require specific gravity, purity levels above 99 percent, and rigorous assaying of chloride impurities. Labels need to warn of inhalation hazards, carcinogenic potential, and the dangers of skin contact. In practice, these standards help avoid the tragic accidents that marred earlier eras, when chemists worked without gloves and fume hoods. Regulatory shifts have sharpened the focus on precise batch documentation, clear hazard icons, and tamper evidence, which means every shipment now lands with a paper trail as substantial as its contents.
Preparation Method
Synthesizing 1-Chloro-2,3-Epoxypropane usually involves an indirect chlorohydrin route, starting from propylene. Chlorination under controlled conditions forms the chlorohydrin intermediate, which is then treated with a base to close the epoxide ring. Some sources use alternative synthetic routes, but each brings its own set of tradeoffs in yield, waste generation, and safety management. Lab equipment must resist halogen corrosion—think stainless steel or special polymers for gaskets and pipes. Operating these reactors can be nerve-wracking, as both temperature spikes and runaway reactions have the potential for explosions or toxic releases. My own time in a pilot plant left a lasting impression: alarms on the process control panel become second nature, and no shortcut ever feels worth the risk.
Chemical Reactions & Modifications
The reactive epoxide ring and the attached chlorine atom set 1-Chloro-2,3-Epoxypropane apart. Reacting with everything from amines and alcohols to water and acids, it acts like a Swiss Army knife for synthetic chemists. This versatility makes it central to building long-chain epoxy resins or as a stepping stone for pharmaceuticals. Outside industrial settings, its aggressive chemistry leaves little margin for error—a brief, uncontrolled release can trigger irritation, fires, and worse. The years spent refining its reactivity also led to a host of derivatives with tailored properties. Whether cross-linked polymers or fine-tuned intermediates for agritech, every modification brings new possibilities—and new safety headaches to solve. That balance between creativity and caution never goes away in the lab.
Synonyms & Product Names
Few chemicals wear as many badges as 1-Chloro-2,3-Epoxypropane. Besides epichlorohydrin, the literature and old catalogues throw around names like ECH, chloromethyloxirane, and glycidyl chloride. Certain trade names still appear on older stock, causing headaches for supply chain managers chasing down regulatory paperwork. Industry veterans usually settle on epichlorohydrin, but regional habits die hard, and documentation sometimes lists the full IUPAC name just for clarity’s sake. Spending time with cross-border shipments, I’ve seen firsthand how synonym confusion can play havoc with customs and legal audits. Standardization now mostly keeps supply chains running smoothly, but legacy systems still trip up the unwary.
Safety & Operational Standards
Few topics stir stronger feelings among facilities staff than chemical safety, and 1-Chloro-2,3-Epoxypropane raises the stakes. Strict rules cover everything from minimum air exchange rates to backup spill containment. Personal protective gear means full-face respirators, chemical-resistant gloves, and double-layer clothing. Ventilation systems, monitors, emergency showers, and evacuation routes turn from plans on paper into practiced drill routines. Decades of incident reports led to tougher local and international rules, with agencies like OSHA, ECHA, and various state regulators keeping a close watch. Plant managers and line workers share responsibility: even a moment’s distraction with line connections or valve settings can cause real harm. As more stories surface about chronic exposure dangers—sometimes stretching back to family members—it’s clear that vigilance and training deliver benefits that echo far beyond the factory fence.
Application Area
No single market corners the use of 1-Chloro-2,3-Epoxypropane. Mainstream uses revolve around epoxy resin production for composite materials, floor coatings, and adhesives. Water treatment plants turn to it for producing cationic resins that grab onto contaminants, while specialty paper and textile companies rely on additives derived from it for wet strength improvement. Over the years, pharmaceutical manufacturers tapped its structural motifs, building blocks for antiviral and anticancer experiments. Beyond industry, I’ve brushed shoulders with restoration artists fixing centuries-old artwork using epoxies derived from it, underscoring how research and art can intersect in surprising ways. Downstream users care as much about emissions and disposal as performance, and the push for greener alternatives continues to grow. Each application comes with its own set of operational protocols, from contained mixing rooms to rigorous post-use decontamination.
Research & Development
Scientific inquiry never really slows down where 1-Chloro-2,3-Epoxypropane is concerned. Research teams investigate process intensification, aiming to cut waste and energy inputs in synthesis. Green chemistry drives new projects exploring biobased propylene and recyclable solvents. Current literature pushes innovation for safer, higher-yielding derivatives and smarter analytical techniques to track trace contaminants. Cross-disciplinary groups team up to explore advanced composite materials for aerospace, automotive, and civil engineering applications, sometimes reimagining the structures from the molecular level up. Social and regulatory pressures also drive investment in improved remediation strategies for spills, with more sensitive early-warning sensors, encapsulation methods, and neutralization protocols. The deep bench of ongoing patent activity signals a field nowhere near maturity.
Toxicity Research
Hard truth cuts through any discussion about 1-Chloro-2,3-Epoxypropane toxicity. Studies in lab animals and occupational settings consistently link exposure to respiratory, skin, liver, and nervous system impacts. Its classification as a probable human carcinogen stems from long-term studies showing increased tumor rates in exposed rodents and decades of follow-up among workers. Inhalation and skin absorption remain prime routes of harm, making robust protective measures a must. Direct accidental releases—thankfully rare in better-regulated regions—bring risks of chemical burns, lung damage, and long-term chronic illnesses. Professional toxicologists spend their careers tracking emerging evidence, adjusting exposure standards, and improving biomonitoring efforts, while medical teams look for subtle early markers of harm. Advocacy for lower thresholds and improved environmental monitoring never lets up, especially as new data emerges about low-level, long-duration risks. For families living near chemical plants, the drive for cleaner production holds real-world consequences.
Future Prospects
Looking toward what comes next for 1-Chloro-2,3-Epoxypropane, the convergence of regulation, science, and sustainability creates both headaches and opportunities. Green chemistry advocates press hard for biodegradable or less hazardous substitutes. Industry, under pressure to cut emissions and solvent use, invests in process upgrades, tighter emission controls, and safer containment. Workforce training and public reporting grow more transparent, driven by both legislative changes and community activism. On the research side, smarter molecular design hints at safer derivatives with less environmental impact and reduced toxicity—with digital simulation tools speeding up what once took years of bench work. The broader market keeps evolving too: as composite materials head toward lighter weight and higher strength, and as electronics demand ever-higher purity and reliability, demand persists—though always chased by the specter of regulation. Having seen both promise and peril up close, it’s hard not to feel the stakes each time a new batch comes off the line. For everyone in the chain, from synthesis to cleanup, real progress means honesty, vigilance, and unflagging commitment to safety and innovation.
What is 1-Chloro-2,3-Epoxypropane used for?
The Basics of 1-Chloro-2,3-Epoxypropane
1-Chloro-2,3-epoxypropane, also called epichlorohydrin, draws a lot of attention in chemical circles because it sits at the crossroads of many essential manufacturing processes. Having spent years in labs and listening to plant operators talk about challenges, I can tell you, its importance goes far beyond its technical-sounding name.
Building Better Plastics and Resins
Factories rely on epichlorohydrin to make epoxy resins. These resins hold together everything from wind turbine blades to boat hulls. Walk through any hardware store and you’ll see the result: adhesives, floor coatings, and waterproof sealants. Epoxy’s grip keeps bridges safe and airplane parts solid. Without the unique structure that 1-chloro-2,3-epoxypropane brings, making high-performance polymers would be much tougher, if not impossible.
Take the automotive world. Here, lighter and tougher makes a direct difference in fuel economy. Epoxy-resin composites allow engineers to cut car weight, helping manage rising fuel prices and tighter emissions rules.
Drilling Down into Clean Water
Chlorinated epoxy compounds show up in water treatment as well. They help manufacture membranes and ion-exchange resins used to filter contaminants. My own time assisting with municipal water projects showed me how crucial these barriers are. One faulty resin batch or shortcut, and heavy metals or pesticides end up in household taps. Through proper chemical processes—using reagents like 1-chloro-2,3-epoxypropane—these safety nets stay tough and reliable, channeling cleaner water to cities and farms alike.
Agrochemicals and Pharmaceuticals
Crop protection may sound like a world away from industrial polymers, but the link lies in the chemistry. Makers of herbicides, fungicides, and certain medicines use 1-chloro-2,3-epoxypropane as a stepping stone to more complex molecules. One example: synthesizing active ingredients in plant-protection products. Farmers can then guard fields more effectively against pests and disease. The pharmaceutical sector also turns to this molecule for creating building blocks in drug development. It’s these kinds of connections that keep food and medical supply chains running.
Challenges and Safer Solutions
It’s not all smooth sailing. Epichlorohydrin can harm people and the environment without careful handling. Breathing it in or getting it on the skin may cause health problems. Regulators worldwide keep a close eye, setting exposure limits for workplaces and strict rules for disposal. When I spoke with a plant safety manager in Texas, he explained how extra monitoring and upgraded ventilation systems kept yearly exposure below mandated thresholds. This kind of vigilance comes from hard-won experience after incidents that made headlines and prompted costly shutdowns.
Researchers explore greener technology including safer alternatives, better protective gear, and closed-loop systems to cut waste. Chemists tweak production steps to use less energy or generate fewer byproducts. These changes chip away at risks while keeping up with the modern world’s demand for advanced materials and clean resources. Every person—from chemical engineer to maintenance tech—carries responsibility to handle this compound with respect and foresight.
What are the safety precautions for handling 1-Chloro-2,3-Epoxypropane?
Understanding the Risks
1-Chloro-2,3-epoxypropane, better known to some as epichlorohydrin, does a lot behind the scenes in making resins and glues. It's not a chemical to treat lightly. Even brief exposure to the fumes can irritate your eyes, skin, and throat, with the risk of leading to something more serious. Letting the liquid make contact with your skin can cause burns, while getting it in your eyes is even more dangerous. Inhaling its vapors can affect breathing, and over time, people exposed in factories have found themselves dealing with chronic health problems.
Personal Safety Practices
Anyone handling this chemical needs solid habits, backed by evidence and experience in industrial settings. I remember folks in my lab always suiting up like they meant business, because they knew one slip could cost them their health. Gloves aren’t just a suggestion — only nitrile or other chemical-resistant materials keep it from soaking through. The same deal goes for goggles and a face shield, since accidents happen when splashes are least expected. Lab coats or chemical-resistant aprons protect against spills that could soak through regular fabric.
Never trust basic ventilation. Exhaust hoods should pull vapors away from your face. Those vents work overtime, especially in tight spaces. A good system saves more than a headache; it’s there for the lungs.
Clean-Up and Spills
Spills happen even for the careful. Folks I’ve worked with grab absorbent pads specifically rated for chemical spills. Sprinkling inert material like sand helps, but never let anyone sweep or mop up bare-handed. Bags need to be sealed tight, labeled, and moved out as hazardous waste right away. Letting material linger allows vapors to build up, which no one wants. Wash any skin contact right away. I’ve seen co-workers dash for the sink the second they felt a splash or irritation.
Storage and Transport
This chemical stays safest in air-tight containers, parked somewhere cool and dry with good airflow. Sunlight or high heat make it break down or build up pressure, so dark corners do better. Labels need to be big and clear so no one grabs the wrong stuff. Transport rules apply too; never throw a drum in the back of a truck unstrapped. It’s all about control, from shelf to shipping.
Health Monitoring and Training
Places where 1-chloro-2,3-epoxypropane gets used should run regular health checks. Workers talk about simple symptoms for a reason; early warning means a chance to avoid something permanent. Training counts just as much. Newcomers should watch veterans suit up, handle clean-ups, and ask questions whenever they aren’t sure. Supervisors need to update safety checks as new facts come out, since recommendations shift as science learns more.
Better Solutions Forward
Some labs and factories look for substitutes now, aiming to lower these health hazards. Until a safer replacement is found and adopted everywhere, strict personal safety routines remain the best defense. Sticking to these habits, backed by experience and the best data, means everyone makes it home in one piece—and that’s always the main goal.
What are the storage requirements for 1-Chloro-2,3-Epoxypropane?
Why Smart Storage Matters
1-Chloro-2,3-epoxypropane, better known as epichlorohydrin, plays a big role in making things like epoxy resins and water treatment chemicals. Every time I’ve worked around this stuff, I couldn’t ignore how fast even a small spill turns into an emergency. Out in the field, folks get nervous when anyone cuts corners. That uneasy feeling isn’t paranoia—it’s respect for something that can burn skin, pollute the air, or cause bigger trouble if it leaks.
Room for Error? Not Here
This chemical evaporates easily and attacks metals, plastic, and concrete if left unchecked. To cut down risks, people turn to tightly sealed steel drums with special coatings or lined tanks. Good ventilation can’t be optional. Shoving containers in the back of a shipping container or next to an office is asking for problems. I’ve watched warehouses scramble after somebody thought stacking barrels three high without straps would “save space”—only to find a toppled mess, an acrid smell spreading, and three people racing for eyewash stations.
Fire Risk: Plan for the Worst
Epichlorohydrin is flammable. Folks storing this material pay attention to every electrical spark, static discharge, or even a parked forklift running too close. In some facilities, I’ve seen storage areas grounded with heavy-duty copper wire and beefy fire suppression setups standing by. Storing far from acids, amines, and strong bases isn’t just being picky—it’s about stopping unwanted chemical reactions. Nothing wakes you up like hearing the fire alarm in a plant where incompatible chemicals mixed thanks to careless storage.
Personal Safety: It’s Not Optional
Stories stick with you: once, a maintenance worker—no respirator—walked by an open drum, didn’t make it three steps before his throat closed up. You can’t smell or see every hazard, but a tight-fitting mask, gloves, and protective goggles shield a person from the stuff you can’t predict. Epichlorohydrin can seep through some plastics, so double-checking every label on gloves and goggles makes a huge difference. Rigorous training, checklists, and drills don’t waste anybody’s time—they keep people out of the emergency room.
Tracking and Labeling: Small Habits, Big Impact
Over half the near-misses I’ve witnessed come down to missing or unreadable labels. A habit as simple as checking for leaks or faint whiffs of chemical smell before unloading pays off. Digital tracking systems aren’t just there for inspectors. They help people see which containers are due for rotation, which ones are nearly empty, or which ones should never sit in the sun.
Solutions That Last
Practical solutions go beyond paperwork. Regular audits catch wear and tear or rust before it spells disaster. Investing in good warehouse racks and specialized containers pays for itself by keeping the product contained and people safe. It takes buy-in from everyone—managers willing to spend on better storage, workers trained to spot problems, and maintenance teams fixing small leaks fast. Real progress happens when companies go beyond the minimum rules and keep sharing lessons from close calls.
Is 1-Chloro-2,3-Epoxypropane hazardous to health or the environment?
What Is 1-Chloro-2,3-Epoxypropane?
1-Chloro-2,3-epoxypropane often shows up in factories as a chemical workhorse. You may also hear it called epichlorohydrin. Its job in industry covers everything from making epoxy resins to acting as a stabilizer in some plastics and rubbers. Most people pass this compound in their daily lives without ever seeing it labeled outright, but the effects reach far beyond the factory floor.
Direct Health Effects: Not Just for Workers
Plenty of reports, along with years of toxicology studies, point out the trouble this chemical brings. Folks exposed at work describe burning eyes, breathing problems, and skin reddening after handling it. Research from the US National Toxicology Program and similar bodies worldwide place epichlorohydrin as a likely carcinogen. Multiple studies link long-term exposure with higher risks for respiratory system cancers and blood disorders.
Acute exposure begins right away, and even small amounts can irritate the nose, lungs, and eyes, especially without good protection. I once toured a plant in Texas where the chemical's sharp scent lingered in the air. The plant manager told me about headaches and sore throats before they updated ventilation. The experience left its mark—good safety gear remains non-negotiable for those working near the stuff.
Hidden Reaches: How the Environment Pays the Price
Factories once dumped chemical waste straight into streams. Even now, spills and leaks pose risks to soil and water. 1-Chloro-2,3-epoxypropane doesn’t stay put. This chemical breaks down in sunlight, but not before causing serious trouble for fish and plants. Studies by the Environmental Protection Agency found reduced hatch rates and odd behavior in fish exposed to even low doses.
The chemical’s vapor rises into the air, leading to problems miles from the original factory. For communities living nearby, there’s no easy way to track low-level, long-term exposure. That lack of clarity feeds distrust of industry and discomfort at home. People deserve to know what’s in their water, soil, and food.
What Can Change?
Stronger regulations can help, but they only mean something if enforced. Workplace controls—tight-fitting masks, regular air checks, closed systems—reduce exposure. Factories must handle waste responsibly, investing in monitoring and quick clean-up plans. On the community side, better transparency builds trust. Posting real-time air and water monitoring results publicly gives everyone a fair shot at understanding the risk.
Alternatives already exist for some uses, such as safer stabilizers and resins. Shift doesn’t come fast, but the savings in health care and lost time are hard to ignore. Industry may balk at new costs, yet the stories of workers and their families, along with sickened wildlife, demand better solutions. In my view, once you see the downstream effects—both in people and the land—you can’t look away.
Choosing safety means more than just following rules. It takes listening to experience from the shop floor and evidence from researchers. Big change calls for bold steps. Without that, health and the environment keep taking the hit, even when the tools exist to fix it.
What is the chemical structure and molecular formula of 1-Chloro-2,3-Epoxypropane?
Looking at the Structure Up Close
1-Chloro-2,3-epoxypropane, known by many as epichlorohydrin, keeps things surprisingly simple—a three-carbon backbone, one chlorine atom, and an epoxy ring. Its molecular formula, C3H5ClO, tells the story plainly. Here’s the chemical structure: one end holds the chlorine attached to the first carbon, while the other two carbons form a three-membered ring with an oxygen, called an epoxide. Tossing a halogen into a small ring like this creates a strained, highly reactive molecule. It’s easy to see why this compound grabs the attention of chemists, both in laboratories and large-scale industry.
My Hands-On Experience with Epichlorohydrin
Working in a lab that used epichlorohydrin taught me to respect its peculiar nature. Holding a sample is much different than reading about it in a textbook. Even a small spill releases a pungent, sharp odor. This comes from its volatility and reactivity, traits that stem from that tiny epoxide ring.
Mixing it with water or alcohols often triggered energetic reactions. The molecule just wants to relieve the strain built up in that three-membered ring, and it’ll go through hoops to do it. Because of this, it becomes a building block for polymers, resins, and elastomers. Most people don’t realize that the durable plastics and coatings around cars and electronics often start with molecules as simple—and tricky—as epichlorohydrin.
Why Its Chemistry Matters
The blend of chlorine and epoxy in a single small molecule gives it a Jekyll-and-Hyde character. On one side, it enables the production of epoxy resins, used in paints, sealants, and stronger adhesives. On the other, it’s also dangerous to human health. The chlorine atom makes it more soluble and able to slip through membranes. Inhaling fumes or skin exposure can cause real harm, and there are strict workplace rules where this chemical shows up.
The Environmental Protection Agency lists epichlorohydrin as a probable carcinogen, and several countries have set clear limits on allowable exposure. In my early career, assessing ventilation in rooms where we handled this molecule felt just as important as mixing chemicals. There is no place for carelessness with compounds like this. Safety routines—gloves, goggles, well-maintained hoods—aren’t optional extras.
Pushing for Better Safety and Sustainability
Epichlorohydrin production still relies on propylene, a product from fossil fuels, which casts a shadow for anyone concerned with sustainability. In my view, chemists should look seriously at alternative green synthesis routes. Some research groups now use glycerol, a byproduct of biodiesel, to make epichlorohydrin. This approach cuts down waste and dependence on petroleum at the same time.
Industry and regulators both need to watch developments in the field carefully. Pushing for greener options while maintaining robust safety rules can save lives and shrink environmental damage. Training matters too—young chemists must learn to analyze risks and respect the hidden dangers behind a simple molecular formula.
What to Take Away
Looking at 1-Chloro-2,3-epoxypropane isn’t just an exercise in memorizing atoms and bonds. Knowing its real-world risks and uses brings the structure into sharper focus. If you handle or manufacture compounds like these, it pays to keep a close eye on new developments—both in safety and in sustainable synthesis. In chemistry, the smallest changes in structure or practice can have the biggest impact.
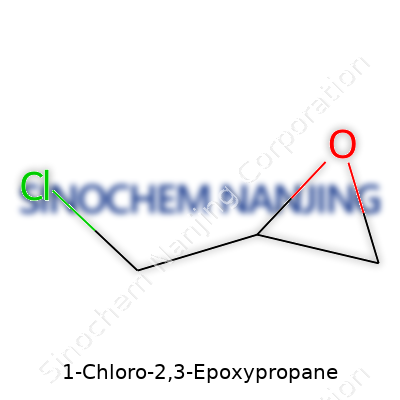
| Names | |
| Preferred IUPAC name | 2-(Chloromethyloxirane) |
| Other names |
Epichlorohydrin Glycidyl chloride 1-Chloro-2,3-epoxypropane Chloromethyloxirane 1,2-Epoxy-3-chloropropane |
| Pronunciation | /waɪnˈklɔːrəʊ tuː θriː ɪˈpɒk.si ˈprəʊpeɪn/ |
| Identifiers | |
| CAS Number | 106-89-8 |
| Beilstein Reference | 606043 |
| ChEBI | CHEBI:41470 |
| ChEMBL | CHEMBL1230900 |
| ChemSpider | 15702 |
| DrugBank | DB01956 |
| ECHA InfoCard | 03d4ad1b-1e81-4146-b686-65a1d8a1b322 |
| EC Number | 202-439-7 |
| Gmelin Reference | 7848 |
| KEGG | C00852 |
| MeSH | D002472 |
| PubChem CID | 7855 |
| RTECS number | GD2450000 |
| UNII | 04J7JA71ZK |
| UN number | 2023 |
| CompTox Dashboard (EPA) | DTXSID4020717 |
| Properties | |
| Chemical formula | C3H5ClO |
| Molar mass | 92.52 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet, chloroform-like |
| Density | 1.2 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | 0.4 |
| Vapor pressure | 3.7 kPa (at 20 °C) |
| Acidity (pKa) | 14.0 |
| Basicity (pKb) | – |
| Magnetic susceptibility (χ) | -0.000193 |
| Refractive index (nD) | 1.439 |
| Viscosity | 2.5 mPa·s (20°C) |
| Dipole moment | 3.74 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 267.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -122.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -682.8 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS05,GHS06,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H302, H315, H319, H330, H334, H341, H350 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P337+P313, P301+P310, P303+P361+P353, P304+P340, P308+P311, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 'Flash point: 33 °C (91 °F; 306 K) - closed cup' |
| Autoignition temperature | 416°C |
| Explosive limits | 3.8–23% |
| Lethal dose or concentration | LD50 oral rat 843 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1130 mg/kg (oral, rat) |
| NIOSH | TX4025000 |
| PEL (Permissible) | PEL: 5 ppm (19 mg/m³) |
| REL (Recommended) | 50 ppm (180 mg/m³) |
| IDLH (Immediate danger) | IDLH: 75 ppm |
| Related compounds | |
| Related compounds |
Epichlorohydrin 1,2-Epoxypropane 2-Chloropropane Glycidol Propylene oxide |

