The Story and Science of 1-Chloro-1-Nitropropane
Understanding the Path from Discovery to Use
Some chemicals have stories interwoven with chapters of hard science and real-world trade-offs. 1-Chloro-1-nitropropane doesn’t draw headlines, but its tale stretches over decades, with its roots tangled in both laboratory ingenuity and evolving safety culture. From the early days, chemists hunted for new intermediates, especially those that could both donate and withdraw electrons in organic synthesis. The rise of nitroalkanes and their halogenated cousins in the mid-20th century made 1-chloro-1-nitropropane worth a look, especially for folks chasing new synthetic routes or searching for compounds that might shape better insecticides, explosives, or even pharmaceutical building blocks.
Examining Its Physical and Chemical Traits
1-Chloro-1-nitropropane comes across as a pale liquid, moderately volatile, often with an odor that turns heads in any lab where handling isn’t careful. It weighs a touch more than water, so it hangs low in a vessel. It doesn’t mix with water, due to both the chlorine and nitro groups, both making it more comfortable hanging out in organic solvents. Quick to react if you give it a strong base or heating, it often serves well as an alkylating agent, sliding its carbon chain or chlorine onto other molecules in the right hands—or under the wrong fume hood.
Highlights from the Lab Bench: Specs and Label Know-How
Chemists usually judge 1-chloro-1-nitropropane by color, boiling point, density, and purity level, but the real focus remains on how tightly it’s sealed, how carefully it’s labeled, and whether the shelf—preferably locked—protects it from both heat and the curious. It can break down or evaporate if left alone too long, and proper hazard labels warn users of toxic vapors and the risk of fire. On any shipment, marks for flammability and acute toxicity can’t be skipped. No one working with it on a large scale throws caution to the wind, especially as safety culture now demands more than a rag and a guess.
Getting to the Root: How the Stuff Forms
The common route for making 1-chloro-1-nitropropane runs through the halogenation of nitroalkanes like nitropropane, with classic methods using hydrochloric acid or thionyl chloride to swap a hydrogen for chlorine on the terminal carbon atom. Some labs favor phase-transfer catalysts, which help juggle the reactants and nudges up yields. Reaction conditions must be tight; over-chlorination or side reactions can fill the flask with other stuff that’s tricky or pricey to separate. People working these reactions focus hard on containment, good ventilation, and effective cleanup, because spillage or vapors pose a direct health threat and a regulatory headache.
Straightforward Chemistry, Surprising Twists
On the reaction front, 1-chloro-1-nitropropane offers chemists a reliable alkyl source. Pop it into a reaction with a nucleophile, and it hands off its chlorine or nitro group, shaping new carbon-nitrogen or carbon-oxygen bonds. It comes in handy during ring formation, conjugate additions, or making more complex nitro compounds. If left to hydrolysis or strong bases, it can spit out nitrites, alcohols, or related byproducts. Each reaction introduces its own risk, as nitro compounds infamously detonate if mishandled. It’s not TNT, but no one discounts the hazard.
Aliases and Chemical Names
Chemists call this liquid by several names. Some prefer 1-chloronitropropane, while others stick to IUPAC-style terms like 1-chloro-1-nitropropane. Database searches might throw up “chloro-nitropropane” or even straight formulas like C3H6ClNO2. These aliases help tie together old papers and data sheets from across continents, improving safety and reducing confusion, especially for emergency responders or researchers digging through archives.
The Hard Facts on Safety: Working with and Around It
Handling chloro-nitro compounds means a steady eye on the risks. 1-chloro-1-nitropropane lets off fumes that sting the nose and can irritate the lungs if exposure builds up; gloves, goggles, and fume hoods aren’t extra steps, they’re the bare minimum for safe handling. Drop it on your skin or breathe deep nearby and you risk chemical burns or headaches, possibly worse with chronic exposure. Regulations change as more toxicity data rolls in, so engineers and safety officers keep updating their protocols in both academic and commercial settings. Waste storage matters, since accidental mixing with other chemicals or basic solutions can spark ugly reactions. Decades of hard-learned lessons shape how labs enforce training, spills drills, and disposal practices.
Looking at Where It Matters
1-Chloro-1-nitropropane matters most in the synthesis of specialty chemicals. Medicinal chemists reach for it when nothing else fits the reaction they need, perhaps fashioning a pharmaceutical precursor or a new pesticide candidate. It has found some space in making certain dyes, stabilizers, and laboratory standards for method development, particularly where a nitroalkane needs a heavier functional handle for future reactions. Its reactive balance—neither too stable nor wildly unpredictable—makes it an intermediate that turns up in patents more often than production barns. Its shelf time shortens as bigger firms chase higher-volume, lower-hazard routes for the products at the end of the chain.
Pushing Chemistry Forward: R&D and Innovation
Ongoing research pokes at safer and more efficient ways to use halogenated nitro compounds. There’s hope for catalytic systems that slice out waste and drive up atom economy. Some labs have poured years into finding substitutes with less toxicity and better biodegradability. Academic papers chase ways to retool its basic structure, spin out new analogues, or deploy it in fewer reaction steps for medicinal scaffolds. The environmental impact of chlorinated nitro products pressures some users to switch to greener alternatives, especially as new regulations roll in on emissions, disposal, or reporting.
Watching the Horizon: Toxicity and What Comes Next
The toxicity of compounds like 1-chloro-1-nitropropane shapes where, how, and why it gets used. Studies point to both acute and chronic health effects, from respiratory distress to suspected organ effects, mostly in industrial or accidental high-level exposures. Regulatory agencies keep reviewing animal and environmental toxicity data before allowing broader commercial applications. Many chemists now want less hazardous feedstocks, so there’s real momentum behind switching to molecules with recognized metabolic fates or validated antidotes if accidents happen. Green chemistry’s growing voice in the field isn’t satisfied with “manage the risk”—it seeks to “design out the risk.” Future prospects for 1-chloro-1-nitropropane probably won’t sit in mass market manufacturing, but in specialized research, diagnostics, or teaching labs, where the stakes call for precision and the professionals can weigh risk against reward.
What is the chemical formula of 1-Chloro-1-Nitropropane?
The Basics Behind the Chemistry
I’ve learned through working closely with scientific data that each chemical formula holds a story about how atoms tie together, impacting safety, applications, and environmental risks. 1-Chloro-1-nitropropane, for example, carries the formula C3H6ClNO2. Those letters and numbers show more than just a code. They map out a framework of three carbons, six hydrogens, one chlorine, one nitrogen, and two oxygens, all joined in a very specific way. This molecule’s shape and parts open up many conversations around safety and chemistry.
Why 1-Chloro-1-Nitropropane Matters
Some chemicals never leave the research lab, but others stick around because they do a job nobody else can. 1-Chloro-1-nitropropane slots in here. It’s not a household staple. You won’t find it on supermarket shelves, but chemists have good reason to notice it. With a chloro group and a nitro group off the same carbon, this molecule sits at the crossroads of both reactivity and stability. The blend of a halogen and a nitro group makes it a great demonstration piece in organic chemistry classes, where students learn how different functional groups affect reactions.
What the Science Says About Its Use and Risks
Every so often I read accident reports from research labs, and many accidents come from underestimating a small, unfamiliar compound. The nitro group brings explosiveness, and the chloro group can make things toxic or reactive under the right light. People sometimes underestimate these secondary chemicals, letting “low profile” chemicals hide real dangers. The molecular structure taught me early on that the shape of a chemical changes how it reacts and how safe it’ll be to handle. A small molecule like C3H6ClNO2 might look simple, but those functional groups can turn sour really quickly in the wrong conditions—like heat or friction.
Safety, Regulation, and Practical Handling
This field runs on both knowledge and routine. After seeing how chemicals like this one can surprise people, I believe strong training trumps complicated gear every time. Proper glove use, fume hoods, and clear storage labels make all the difference. Chemists need to know not just what’s in the bottle, but how the atoms are stuck together and what can go wrong. Regulatory bodies such as OSHA and the European Chemicals Agency list guidelines that keep researchers honest and careful about labeling and storing these kinds of compounds. Ignoring those rules or getting comfortable spells trouble faster than you’d think.
Pathways to Safer Laboratory Chemistry
A safer lab culture grows from honest conversations about chemical risk. I’ve made it a rule in my own work: never assume familiarity means safety. Even compounds that pop up during organic synthesis experiments can punch above their weight in risk. I’d argue for a few changes—regular training updates, better real-world accident case studies for students, and clear databases for quick chemical risk checks.
Scientists and students should feel comfortable asking questions. Even a formula like C3H6ClNO2, which looks neat and unremarkable at first, opens doors to bigger lessons about careful work, respect for chemistry, and the protection of everyone in the building.
What are the main uses or applications of 1-Chloro-1-Nitropropane?
Where 1-Chloro-1-Nitropropane Finds Its Place
Some chemicals slip under the radar, but 1-Chloro-1-Nitropropane shouldn’t get overlooked. My own background in chemical safety brings this out—handling reagents like this demands not just respect but a clear understanding of their purpose. This compound pops up in more places than most folks realize, far beyond a label on a drum in a storage building.
Synthesizing Pharmaceuticals and Agrochemicals
Everyday medicines and crop protectants rely on solid chemistry workhorses. 1-Chloro-1-Nitropropane shows up during the early reaction steps that create core structures for active pharmaceutical ingredients. Chemists use it to introduce certain molecular groups, making it easier to build up more complicated drugs. I learned from running bench-scale tests how even a single synthetic building block like this can open doors for safer, more effective treatments or bump up the efficiency of common pesticides.
Creating Fine Chemicals and Advanced Materials
Paints, dyes, and specialty compounds don’t just appear from thin air. Behind the colors on a car bumper or the sheen on a coated wire, detailed chemistry takes place. 1-Chloro-1-Nitropropane steps in as a reagent when teams aim to stitch together molecules in just the right way. In coatings R&D, we tested several chlorinated nitro compounds, and this one responded best for adding stability or introducing specific colors to polymers. That adaptability pays off, especially in applications where strong bonds and predictable performance matter.
Fuel Additive and Explosives Research
In fuel technology labs, even minor ingredients can change the way an engine runs. Additives based on nitroalkanes like 1-Chloro-1-Nitropropane help control combustion, reduce knocking, or tweak fuel properties. The same backbone that supports controlled burning attracts explosives researchers. They use this compound to craft intermediates with high-energy content. As a hazard trainer, I stress the need for cautious protocol—these applications mean any misstep has real consequences.
Potential Risks and How to Address Them
The promise of 1-Chloro-1-Nitropropane comes with questions about safety and environmental impact. Direct exposure risks range from respiratory irritation to chemical burns. Having seen accidents stemming from poor chemical labeling or improper storage, it’s clear that safe handling practices and strong regulatory oversight make a difference. Any site using the compound should include robust ventilation, protective gear, and spill control strategies.
Beyond immediate hazards, disposal draws scrutiny. Chlorinated nitro compounds, if mishandled, can persist or break down into bioaccumulative toxins. Working toward greener alternatives—using less toxic precursors and recycling solvents—offers a meaningful path forward. Companies can partner with academic labs to develop safer synthetic routes or biodegradable substitutes, keeping the benefits while shrinking long-term risks.
Looking Ahead
Chemicals like 1-Chloro-1-Nitropropane keep industrial and research labs moving. Their impact shows up in smaller environmental footprints, better crop yields, and even smarter engines. By staying grounded in safety, transparency, and new technology, the field stands to keep using these tools responsibly, meeting today’s standards for trust and expertise.
What safety precautions should be taken when handling 1-Chloro-1-Nitropropane?
Know What You’re Dealing With
Anyone who spends time in the lab knows you can’t treat all chemicals the same. I’ve worked with volatile organics, and the truth is: 1-Chloro-1-Nitropropane belongs on the list of things nobody should handle casually. A quick glance at its chemical structure hints at trouble—when chlorine hooks up with a nitro group, you’re essentially looking at an irritant with more than one unpleasant trick up its sleeve. Its vapors attack the nose and eyes. On the skin, you get stinging, burning, even lasting damage if you don’t wash fast or suit up properly. Inhaling can bring on headaches, respiratory irritation, and worse with long exposures. You’re not doing this work for fun, so respect is the cornerstone.
Work the Controls: Airflow and Access
The old-timers like to say, “If you can smell it, you’re exposed.” I go straight to a fume hood every time something like this needs handling. Exhaust fans backed by good maintenance set the gold standard. Never skip local ventilation. In smaller setups, portable extraction fans can knock exposure down, but never treat them as a permanent fix. Opening a window alone doesn’t cut it. Only bring out the container when ready. Return it once finished, no exceptions. The chemical should never sit on a bench waiting for someone to trip or knock it over. Clear lab benches save time and skin alike.
Suit Up Like You Mean It
I’ve seen accidents turn a good day into a nightmare because someone decided to “just pop the cap off.” I go with a face shield and goggles, not just safety glasses. Latex gloves melt under harsh solvents; go with thick nitrile or butyl rubber gloves. If there’s any risk of splashing, I pull on a full-length lab coat. My ankles and wrists stay covered at all times. Contaminated clothing goes straight into a sealed bag—no sense tracking hazardous material through hallways or break rooms. Taking shortcuts leads to burns or long-term health problems. The small effort up front always beats the aftermath.
Storage and Disposal: No Room for Guesswork
1-Chloro-1-Nitropropane likes to break down in light and heat. I stick with fire-resistant cabinets, cool shelves, and clearly labeled containers. In my experience, clear hazard signs and up-to-date inventories save surprises. Never reuse containers or mix with incompatible substances—mixing this with common cleaning chemicals or acids lands you in the ER. Disposal routines make or break safe practice. I always hand off unused portions to trained waste management contractors. Local environmental rules can make things complicated, but this is not a time to improvise. Pretending a drainpipe is a chemical waste chute invites legal and health trouble.
Training Makes Safety Real
The best masks and gloves mean nothing if folks skip the basics. Every new lab member in my shops runs through hands-on safety drills before touching anything remotely toxic. I quiz teams on spill kits, eyewash use, and emergency shut-offs. I stay ready for the worst-case scenario; even veterans need refreshers at least yearly. No one works alone with these nasties—buddy systems ensure there’s help fast.
Real Results Come From Respect
Rushing leads to scars, lost research, and sick colleagues. Good habits are tough to build, easy to lose, and essential every day. Anyone working with 1-Chloro-1-Nitropropane owes it to themselves and their team to take things seriously every single time.
What is the physical appearance and odor of 1-Chloro-1-Nitropropane?
Physical Appearance
Anyone crossing paths with 1-chloro-1-nitropropane in the lab for the first time finds it quite unremarkable in terms of looks. This chemical usually pours out as a clear or pale yellow liquid. It carries a slight oily sheen that’s not hard to spot under normal lab lighting. Unlike powders or crystals that stick to every surface in reach, this liquid keeps a low profile—no crystallization at the edge of a bottle, no telltale clumps in the cap. Holding a vial up to a light reveals almost no cloudiness, signaling a pretty good level of purity. For those who rely on appearance to catch problems, even a yellow tinge alerts them to potential breakdown or contamination. Those who spend time around chemicals know to take a step back and let their eyes scan for color shifts, because even the smallest change can point to something worth checking before use.
Odor Profile
Meeting 1-chloro-1-nitropropane by scent delivers a different kind of message. Odor can be the early warning system that things aren't right, especially in chemical safety. In my experience, this compound gives off a not-too-pleasant, sharp, acrid smell. It's the kind that makes you naturally pull your head back without thinking. The scent isn’t overwhelmingly strong at a distance, but up close, it brings to mind other nitro- and chloro-compounds—somewhere between harsh cleaning solvents and burned plastic. Anyone who handles organic chemicals knows how important it is to avoid sniffing too deeply, and always check a chemical’s documentation before breaking a seal in an enclosed space. For 1-chloro-1-nitropropane, the odor is a reminder to use proper ventilation and personal protective equipment before transferring or mixing it.
Why It Matters
Chemicals like 1-chloro-1-nitropropane play quiet but essential roles in research, synthesis, and manufacturing. Appearance and smell might seem like trivial details, but they’re signal flags for professional chemists and industry workers. Changes in color or odor can warn about contamination, age, or the start of decomposition. In the worst case, ignoring these changes can mean introducing a hazardous material into an otherwise controlled process. Years spent around labs taught me fast: what the Material Safety Data Sheet says matches up to what’s actually in your hand, unless you notice something strange—then it’s time to investigate.
Safety Considerations
Handling 1-chloro-1-nitropropane properly starts with respect for its potential hazards. It’s not just about the possible health effects from inhalation or skin contact, though those matter a lot. It’s also about containing fumes and preventing long-term exposure. Good airflow makes a big difference—fume hoods and well-maintained ventilation systems become allies when dealing with volatile liquids. Chemical-resistant gloves and eye protection follow as the next lines of defense. The best labs make safety standard practice, not just a checklist item.
Possible Steps Forward
Anyone responsible for training new staff or students learns quickly that hands-on walkthroughs beat lectures. A bottle of 1-chloro-1-nitropropane serves as more than just another reagent—it’s a teaching opportunity. If the bottle looks a little cloudier than yesterday, or the odor seems harsher, it’s worth pausing for a closer inspection. Quick identification and proper storage keep everyone safer. Investing in regular safety briefings, easy-to-read labeling, and clear incident reporting keeps mishaps rare and workplace confidence high. Consistent monitoring and respect for these signals—sight and smell—go a long way in supporting a healthier lab environment.
How should 1-Chloro-1-Nitropropane be stored and transported?
Understanding the Risks of 1-Chloro-1-Nitropropane
Some chemicals demand more respect than others, 1-Chloro-1-Nitropropane falls right into that category. It’s used in chemical synthesis, so folks working with it rarely encounter it outside of a lab or specialty plant. Still, every chemical handler deserves real-world advice, not just jargon-packed sheets that everyone ignores until trouble hits.
Tough Lessons from Mishandling
Many people skip the “fine print” with chemical storage and move straight to doing things the quick way. Years of seeing accidents that could have been avoided taught me never to treat rules as red tape. 1-Chloro-1-Nitropropane carries health hazards—skin and eye irritation, possible respiratory harm, not to mention that mix of chlorine and nitro groups makes it a legitimate fire risk.
The chemical gives off toxic fumes if burned. Nobody ever wants to chase an ambulance because a fire started in the wrong drum or because someone forgot about static discharge. That reality hits harder than any regulation.
Storage—not Just “Somewhere Out of the Way”
Keep 1-Chloro-1-Nitropropane away from sunlight, sparks, and open flames. Don’t just rely on general rules for organic solvents. Get a proper flammable liquid cabinet, the kind designed for these jobs, and check the seals—not every old drum passes muster.
Tempting as it is to slap it on the back shelf, the storage area should be cool, dry, and have solid ventilation. That ventilation keeps vapors from building up, plain and simple. After seeing cracked containers cause headaches and costly cleanups, I recommend checking everything weekly, not yearly.
Label every container. Use real hazard signage, not just an ugly sticker nobody knows how to read. Separate it from incompatible chemicals like strong oxidizers, acids, or even strong bases—think about what happens if a leak pools underneath and finds the wrong neighbor.
How the Pros Move It Safely
Nobody wheels 1-Chloro-1-Nitropropane around like a box of nails. Only trained staff should handle the loading. Certify anyone transporting it knows what they’re moving. Wear chemical-resistant gloves and goggles, not hand-me-down gear from the janitor’s closet.
Move it in containers rated for both corrosion and pressure changes; don’t count on old barrels. If moving big loads, companies use temperature-controlled trucks and inventory tracking. I’ve seen how GPS tracking and real-time sensors take out a chunk of uncertainty. Regulations from the Department of Transportation in the US or ADR guidelines in Europe spell out exactly what vehicles, placards, and paperwork are needed. Skipping steps lands both companies and drivers in real legal and financial trouble. It’s more than fines—mistakes can ruin lives.
What Smart Facilities Do
The best setups never put all their trust in hope. They run emergency drills, have spill kits nearby, and double-check exit routes. Old hands in chemical plants tell you that cutting corners with hazardous materials always backfires. Training beats a couple of laminated safety posters any day. Local fire services appreciate clear site maps and access to up-to-date chemical lists; sharing information saves time if the worst ever happens.
To keep everyone safer—store with respect, transport by the book, train like you mean it, fix problems before they grow. The lessons don’t change, no matter how advanced the world gets.
| Names | |
| Preferred IUPAC name | 1-chloro-1-nitropropane |
| Other names |
1-Chloro-1-nitropropane 1-Nitro-1-chloropropane 1-Chloro-1-nitro-propane |
| Pronunciation | /ˈklɔːroʊ ˈnaɪtroʊ ˈproʊpeɪn/ |
| Identifiers | |
| CAS Number | 600-25-9 |
| Beilstein Reference | 1209222 |
| ChEBI | CHEBI:86971 |
| ChEMBL | CHEMBL42497 |
| ChemSpider | 12316 |
| DrugBank | DB12701 |
| ECHA InfoCard | 12be85c1-2e06-48c6-8de0-a046959bda82 |
| EC Number | 214-477-6 |
| Gmelin Reference | 9793 |
| KEGG | C19243 |
| MeSH | D016234 |
| PubChem CID | 10915 |
| RTECS number | TF9275000 |
| UNII | W78314PQ5D |
| UN number | UN1609 |
| CompTox Dashboard (EPA) | DTXSID2067322 |
| Properties | |
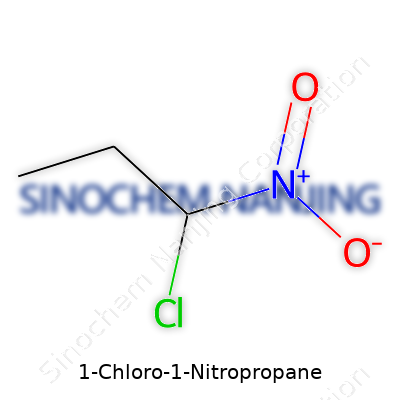
| Chemical formula | C3H6ClNO2 |
| Molar mass | 122.54 g/mol |
| Appearance | Clear colorless to pale yellow liquid |
| Odor | sweet odor |
| Density | 1.206 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 0.96 |
| Vapor pressure | 2.8 mmHg (20°C) |
| Acidity (pKa) | 11.2 |
| Basicity (pKb) | pKb = 9.37 |
| Magnetic susceptibility (χ) | -12.96×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.422 |
| Viscosity | 1.347 cP (20 °C) |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -147.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4466.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H226, H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 96 °C (Closed cup) |
| Autoignition temperature | 250 °C (482 °F; 523 K) |
| Explosive limits | Explosive limits: 2.1–11% |
| Lethal dose or concentration | LD50 oral rat 816 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 480 mg/kg |
| NIOSH | SN1225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Chloro-1-Nitropropane: Not established |
| REL (Recommended) | REL: 1 ppm (5 mg/m³) |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Chloronitromethane Chloronitroethane 1-Bromo-1-nitropropane 1-Iodo-1-nitropropane 1-Chloro-2-nitropropane |

