Getting to Know 1-Butyn-3-Ol: A Look at Its Journey and Role in Chemistry
Historical Development
1-Butyn-3-ol didn't land on the scientist’s bench by accident. Early research into small alkynols started as the search for new building blocks in organic chemistry picked up speed in the mid-20th century. Chemists chasing versatile, reactive molecules soon realized that combining an alcohol group with a triple-bonded carbon chain unlocks options you just don’t get from simpler alcohols or alkynes. Over decades, this compound moved beyond pure curiosity; researchers adopted it into several synthesis strategies, especially where the need for sequential modifications arises. My years spent in labs made it easy to see why: chemists love tools that pack multiple reactive handles. It’s no surprise to see journals from the '60s and '70s piling up with papers featuring this molecule, especially as folks looked to create more complex pharmaceuticals, fragrances, and intermediates.
Product Overview
1-Butyn-3-ol is more than a niche chemical. Markets regard it as a valued intermediate, meaning a substance not often found in final products at the store, but essential in their creation. The compound’s appeal comes from its unique chemical combination—a triple bond sandwiched with a hydroxyl group. This setup lets it anchor itself in an ever-growing number of synthesis pathways. As someone who worked with intermediates in both petroleum and fine chemical industries, I’ve seen how one small bottle can stand behind towering stacks of finished products. Technology companies and pharmaceutical labs keep a close eye on supplies, knowing a snag in the availability of a compound like this can disrupt entire projects.
Physical & Chemical Properties
1-Butyn-3-ol comes across as a colorless liquid at room temperature, sporting a faint, sharp scent that never quite lets you forget it belongs in a fume hood. Its molecular formula is C4H6O, with a triple bond between the first and second carbons and an alcohol group fixed at the third. That triple bond doesn’t just look good on paper; it brings notable reactivity, setting the molecule up for addition reactions with electrophiles, hydrogenation, and other classic laboratory tests. The boiling and melting points land in a range that’s workable for most labs, and its solubility in common organic solvents adds a practical edge for scale-up operations. The compound’s instability around strong bases and oxidants pushes chemists toward careful planning and layered safety precautions.
Technical Specifications & Labeling
Standard chemical suppliers label 1-Butyn-3-ol with accurate hazard symbols, including those marking its flammable and irritant nature. Labels rely on the molecule’s established nomenclature—including synonyms like 3-hydroxy-1-butyne and sometimes the trivial name ethynylcarbinol. The proper handling protocols show up right on the label, and most bottles come with purity specified in percentage points—typically above 95 percent for reliable use in synthesis. The labeling approach aims to catch the attention of any chemist or technician handling the substance who may underestimate its volatility or toxicity.
Preparation Method
Laboratories and manufacturers don’t treat 1-Butyn-3-ol as a starter material; they synthesize it from simpler compounds. One common process starts with propargyl alcohol, extending the carbon backbone using alkylation with appropriate sources. Catalysts—often copper-based—play a central role in driving the reaction, with temperature and solvent control critical at each stage. Another method involves handling acetylenic compounds directly and then introducing the hydroxyl selectively, with careful exclusion of water reacting with the alkyne. From my own lab experience, reactions like these require a tight grip on atmosphere and glassware cleanliness to crank up yields without dangerous byproducts. Because of the safety hazards surrounding strong bases and alkynes, the best labs avoid cutting corners.
Chemical Reactions & Modifications
The unique pairing of the hydroxyl and terminal alkyne in 1-Butyn-3-ol calls out to chemists drawn to complex reaction networks. The molecule’s triple bond opens doors to functionalization—hydrogenation produces saturated alcohols, while halogenation steps in to create a host of valuable derivatives. Electrophilic addition reactions enrich possibilities in both laboratory and industrial syntheses. Protective group chemistry takes on critical importance here to navigate the competing reactivity of the hydroxyl moiety during multi-step syntheses. My time slugging through multi-step organic syntheses in graduate school taught me that nothing tests your patience like balancing competing reactivity patterns. 1-Butyn-3-ol tests skillful hands with the twin allure and challenge of its functional groups. Analysts and process chemists often use it not just as a building block, but as a probe for new catalytic cycles.
Synonyms & Product Names
Chemists often know a molecule by more than one name. For 1-Butyn-3-ol, you’ll see it listed as 3-hydroxy-1-butyne in catalogs and research articles. The systematic IUPAC name and casual references like ethynylcarbinol float through patents, protocols, and production records. This variety plays a practical role: mislabeling leads to delays, costly errors, or unsafe mix-ups. Experience in chemical inventory management cemented the importance of cross-referencing names, especially for globally sourced reagents.
Safety & Operational Standards
1-Butyn-3-ol falls squarely in the class of flammable, acute toxicants that warrant robust controls. The flammability risk isn’t just theory—spilled material in poorly ventilated spaces can create genuine hazards. Skin and eye contact bring irritation, calling for gloves, eye protection, and lab coats every time the bottle comes off the shelf. Industrial handling leans heavily on local fume extraction, strict climate and atmosphere control, and rigid adherence to disposal standards. Training and experience count for everything; stories swap around research groups about close calls with chemicals like this, and regulatory agencies regularly update handling practices to reflect ongoing research into toxicity. Keeping chemical waste streams separate and updated Material Safety Data Sheets nearby is less about bureaucracy and more about protecting lives and reputations.
Application Area
1-Butyn-3-ol finds its way into pharmaceutical ingredient synthesis, often as an intermediate on the winding route to more complex active molecules. Specialty chemicals and agrochemical manufacturers also make use of its reactive backbone for formulating compounds that fight pests or pathogens, or that help in material modification. My time consulting with process improvement teams highlighted just how many new materials in coatings and adhesives trace their origins back to small, specialized intermediates. Sometimes, the presence of an alkyne in a structure acts as a handle, waiting for late-stage transformations or cross-linking. Its reach creeps into research laboratories, where investigators try to modify its structure for new biological or catalytic properties.
Research & Development
For those digging into chemical innovation, 1-Butyn-3-ol rarely sits idle. Synthetic chemistry teams toy with new routes to increase yields and tame hazardous byproducts, with green chemistry pushing for safer and more sustainable methods. Computational chemists model its interaction with advanced catalysts, while materials scientists explore its utility in fabricating functional polymers. Academic research serves the dual purpose of deepening understanding and opening commercial opportunities, with several recent patents seeking improved methods for preparing derivatives or integrating them into broader reaction cascades. As industry and academia keep ratcheting up standards for efficiency and safety, 1-Butyn-3-ol frequently comes under the microscope, both as an old standby and a candidate for replacement by less hazardous alternatives.
Toxicity Research
Questions about toxicity always trail chemical development. Scientific studies flag acetylenic alcohols as possessing moderate acute toxicity; exposure through inhalation or skin contact leads to symptoms ranging from mild skin irritation to central nervous system effects at higher doses. Animal studies and cell culture assays report that long-term exposure needs strong mitigation measures, especially for workers in production and laboratory roles. Routine toxicology screens and workplace monitoring protect users, and new research programs continually re-assess old conclusions as analytics develop more sensitive detection methods. I’ve watched research institutes coordinate with regulatory agencies, nudging chemical companies toward improved hazard labeling and data transparency. The push for safer substitutes still meets resistance when performance takes a hit, but mounting evidence makes re-examination an industry-wide priority.
Future Prospects
The future paths for 1-Butyn-3-ol reflect both its inherent chemical appeal and external pressures on the chemical industry. Demand won’t fade away anytime soon—industries need building blocks that lend themselves to reactivity, and this compound’s dual-functional nature fits that bill. Trends in green chemistry could open new preparation methods that sidestep hazardous reagents. Efforts in synthetic biology may one day bring fermentation or enzymatic methods to the table, replacing traditional chemical synthesis with cleaner, renewable feedstocks. Regulatory standards, ever sharper, will push suppliers and users to account for cradle-to-grave impacts, nudging adoption of safer practices or substitute molecules. Progress relies on transparent data, collaborative standards, and embracing new tools without losing sight of the safety lessons learned through decades of chemical work.
What are the main uses of 1-Butyn-3-Ol?
Introduction to 1-Butyn-3-Ol
1-Butyn-3-ol doesn’t feature on billboards, but it quietly supports several important chemical sectors. With its formula C4H6O, this compound brings together an alkyne and an alcohol group. That mix brings a set of properties that keeps it in demand across different industries, especially pharmaceuticals and chemical synthesis.
Supporting Pharmaceutical Innovation
Drug development relies on building blocks with versatility, and 1-Butyn-3-ol offers exactly that. Scientists use it to build molecules containing triple bonds and alcohol groups, which helps in synthesizing drugs that tackle diverse conditions. For instance, some antiviral and anti-inflammatory agents start with small molecules like 1-Butyn-3-ol. The structure of this compound makes it easier to create complex ingredients, giving researchers more flexibility in designing active pharmaceutical ingredients.
Synthesis of Specialty Chemicals
In chemical manufacturing labs, demand for molecules that can do multiple things stays high. 1-Butyn-3-ol offers both a reactive alcohol and an alkyne, making it a go-to choice for making specialized ingredients—including flavors, fragrances, and chemical intermediates. Its triple bond can handle various coupling reactions, and its alcoholic group makes it compatible with transformations that add value. Synthetic chemists often appreciate the efficiency it brings to reaction steps, speeding up production cycles and unlocking routes unavailable with less flexible starting materials.
Building Polymers and Advanced Materials
Polymer scientists find use for 1-Butyn-3-ol in fine-tuning molecular chains. By incorporating a small amount into the process, it influences the final properties of specialty plastics or rubber compounds. In electronic materials and coatings, its unique structure allows for new surface characteristics or enhanced bonding, keeping products competitive in high-tech markets. I’ve seen colleagues in materials labs use it to improve adhesive strength or to develop protective films for sensitive components.
Agrochemical Pathways
Crop protection calls for precision. In the agricultural sector, product makers use 1-Butyn-3-ol as a building block for certain pesticides or plant growth regulators. Its chemical features lead to molecules that interact in specific ways within plants or insects. This selectivity helps develop safer and more effective products, reducing the need for high dosages and cutting environmental impact, supporting the farmers who rely on these solutions.
Improving Manufacturing Processes
In my experience, efficiency in manufacturing often comes down to reliable intermediate steps. 1-Butyn-3-ol serves as one such dependable intermediate. It provides a base from which chemical plants can efficiently craft new molecules with diverse applications. By optimizing reactions around this compound, facilities can manage costs and energy use. This supports cleaner production and higher-quality outcomes. The compound’s safer handling profile, compared to some other alkynes, also contributes to worker safety—a benefit that matters every day on the job floor.
Potential for Broader Use
Looking forward, the need for adaptable building blocks grows as industries face new challenges. With its reactivity and track record, 1-Butyn-3-ol could see wider use in both research and industrial practice. Efforts to improve supply chains and lower production costs will help more industries access the benefits it supports. By keeping an eye on advancements in synthesis and greener chemistry, I see 1-Butyn-3-ol staying relevant as demand shifts toward sustainability and better health outcomes.
What is the chemical formula of 1-Butyn-3-Ol?
Getting to the Structure
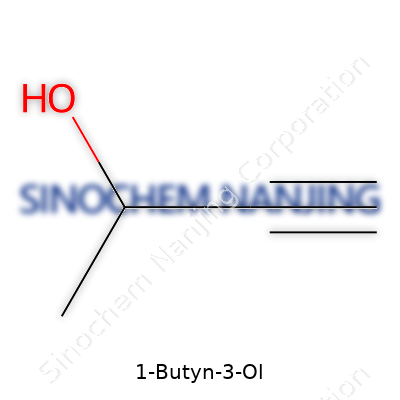
1-Butyn-3-ol doesn’t usually show up in casual conversation. Its name points straight to its chemical roots. You get “butyn” from the four-carbon chain with a triple bond on the first carbon. Add “3-ol” and you place an alcohol group (-OH) on the third carbon in that chain. Understanding the structure means getting a grip on how this stuff acts, reacts, and ends up used in labs and industry. The formula for 1-Butyn-3-ol clocks in at C4H6O.
Why People Care About 1-Butyn-3-ol
The detail in naming reflects how serious chemists are about placing every atom. 1-Butyn-3-ol might look simple, but on a workbench, it’s all about what you can build from it. For students, this molecule shows up as a classic example in organic synthesis courses. Its practical side comes out during the making of pharmaceuticals, specialty solvents, and chemical intermediates. Experimental chemists and engineers use it for the triple bond’s reactivity and the alcohol group’s ability to stick to other molecular pieces. I’ve seen it help craft more complex molecules step by step, where being off by one carbon can wreck an entire process.
The Argument for Chemical Literacy
Plenty of folks glaze over when talk turns to chemical formulas. The thing is—small mistakes create costly setbacks, especially in drug development or chemical manufacturing. If a worker can spot that C4H6O belongs to 1-Butyn-3-ol, the risk of mixing up reagents or misunderstandings drops. Mislabeled chemicals cause accidents. A clear chain of understanding saves time and prevents real harm. I remember a time in labs when someone grabbed a bottle with the wrong formula on the label; that afternoon nobody got much work done, and management had to conduct a safety review. Getting the formula right means staying safe and smart about resources.
Challenges with Access and Sharing
Schools and work sites don't always teach or label thoroughly. One big hurdle shows up in developing regions or smaller companies that skip on training. Inconsistent labeling and patchy safety data put extra pressure on workers. 1-Butyn-3-ol looks unassuming but has flammable potential and can irritate skin. Shortcuts or mistakes on the formula—C4H6O—gum up emergency responses. Even in modern labs, folks need tight protocols and clear documentation.
Building a Smarter System
Good habits save lives: double-checking formulas, enforcing labeling standards, and keeping up with ongoing training. Bringing in digital systems that verify and track chemicals from delivery to storage keeps dangerous confusion to a minimum. I’ve favored digital chemistry databases for quick lookups, but old-school laminated charts next to storage cabinets matter just as much. Sometimes tech fails and having the right answer in plain sight means everything. Real safety culture involves everyone, from the guy unloading supplies to the team analyzing results at the computer.
Staying Grounded with Chemistry
Straightforward chemical names and formulas simplify life for scientists, teachers, and warehouse staff alike. 1-Butyn-3-ol, C4H6O, shows how even small details shape real-world safety and success. Clarity keeps people safe, improves learning, and helps entire industries run smoother. Tidy information makes confident decision-making easier; it can turn chemistry from abstract mystery to everyday know-how.
Is 1-Butyn-3-Ol hazardous or toxic?
Getting a Closer Look at 1-Butyn-3-Ol
Workers in labs or factories come across a wide range of chemicals, and 1-Butyn-3-ol sometimes shows up on the ingredient list. This organic compound, with the structure HC≡C–CH₂–CH₂OH, brings together an alkyne and an alcohol group. It gets used in chemical synthesis, research, and sometimes small-scale specialty manufacturing. Looking at safety sheets, curiosity rises about whether this is a chemical people should fear or simply handle with common sense.
Potential Hazards in the Shop or Lab
Direct information about human toxicity of 1-Butyn-3-ol remains limited. That tends to happen with lesser-known chemicals. What jumps out: it’s flammable, giving off vapors that can catch fire if sources of ignition lurk nearby. Flammability matters a lot in workspaces with poor ventilation or lots of electrical equipment. Spills, even small ones, can produce vapors heavier than air. A worker might not even see the danger before a spark lights up the room.
As for personal risks, 1-Butyn-3-ol can cause skin and eye irritation. Inhalation of high concentrations means headaches, dizziness, or downright makes people feel sick. It can compared, at least in irritation risk, to similar short-chain alcohols or alkynes. Researchers tested acute toxicity in lab animals and found mid-range effects, with rats showing symptoms at moderate doses. Chronic effects, carcinogenicity, and mutagenicity don’t have much data to go on, but lack of evidence doesn’t promise safety.
Comparing to Industry Standards
Using any chemical requires checking workplace exposure limits. Agencies like OSHA or NIOSH have not set a permissible exposure limit specific to 1-Butyn-3-ol, which pushes companies to use the best available data for workplace exposure. Following a substance’s Material Safety Data Sheet makes sense. Workers in chemical handling or R&D already know gloves, eye protection, and good ventilation form the everyday armor against exposure. For spills, the usual non-sparking tools and absorbent materials work well—never ignore even a splash.
Chemicals with close relatives, like propargyl alcohol or butyne, have poisoning reports from accidental ingestion or major exposure, showing symptoms from respiratory distress to central nervous system depression. So even if 1-Butyn-3-ol isn’t as studied, it’s smart to treat it with the same respect as its chemical cousins.
Managing Risks and Looking Ahead
At the user level, the story stays the same: treat it as you would any other lab reagent not meant for casual contact. Keep it away from heat, flame, and sources of static electricity. Safe storage, in cool and dry locations, reduces the risk of vapor buildup. If a spill or exposure happens, flush with plenty of clean water and seek medical advice quickly. Wearing the right gear turns a dangerous job into a routine task.
There’s a patchwork of research on many lesser-known chemicals, including 1-Butyn-3-ol. Workers find themselves relying on both personal habits and company policies. More thorough long-term studies could give teams clearer answers about subtle health effects and set better exposure standards down the road. Until then, good habits and the right equipment matter more than anything written on a label.
How should 1-Butyn-3-Ol be stored safely?
The Real Risks in Handling 1-Butyn-3-ol
Anyone who has spent time in a research lab knows that the more obscure chemicals tend to cause the biggest headaches. 1-Butyn-3-ol stands out as a good example. While the bottle label might not alarm most people, this stuff brings together the hazards of both an alkyne and an alcohol. Add flammability and the potential for vapor build-up, and safety measures become non-negotiable.
Why Moisture Matters with This Chemical
From past experience, allowing water or even humid air near 1-Butyn-3-ol can spark unpredictable results. The alkyne group reacts readily, and the alcohol group pulls in water. That spells trouble if you keep it in a normal cabinet or with an ill-sealed cap. Moisture can trigger polymerization or decomposition, which not only damages the batch but also creates real danger for anyone nearby. Keeping the container tightly sealed and using a desiccator or inert gas blanket helps shut out humidity—this action doesn’t take more than a few extra seconds and can prevent hours of panic later.
Flammable Liquids Belong in the Flammables Cabinet
Fire safety rules have saved more than one lab from disaster, and ignoring them with 1-Butyn-3-ol just escalates the risk. Flammable storage cabinets serve a real purpose here—they keep accidental ignition sources at bay, control temperature, and contain leaks. Some folks get lazy and let such bottles sit on open shelves close to electrical outlets or hot plates. One spilled bottle, and an electrical spark too close by, would mean more than a ruined experiment. Flammable storage offers at least a fighting chance to keep people safe.
Ventilation: The Overlooked Layer of Protection
I once worked in a lab where someone stored all the solvents in regular wooden cabinets, and the air vents ran only at half power. It didn’t take long before the chemical odors became sharp—an easy warning sign that vapors were accumulating. 1-Butyn-3-ol has a volatile edge, sending fumes into the air. These vapors not only boost the fire risk but also pose inhalation hazards. Decent ventilation, as simple as working under a fume hood and keeping general airflow going, pulls vapors out before they reach dangerous levels. Investing in local exhaust for storage spaces offers one more shield against accidents.
Labeling and Segregation: Common Sense Steps
Even the most careful protocols fail if a bottle goes missing or mixes with incompatible chemicals. I have seen colleagues pull out unmarked bottles from the back of storage cabinets and spend precious minutes guessing what lies inside. Mislabeling or poor organization can lead to mixing something like 1-Butyn-3-ol with acids or oxidizers, setting off hazardous reactions. Clear, bold labels showing both the full name and hazard warnings, along with consistent records, keep mistakes from turning into emergencies. Segregating alkynes and alcohols from incompatible materials, not just for compliance but for real safety, means those who share your workspace can count on finding hazards before they find you.
A Culture That Values Speaking Up
People like to talk about PPE and storage cabinets, but safe work with 1-Butyn-3-ol ultimately depends on the habits of the people next to you. If you spot a leak or a bottle out of place, calling attention to it right away, instead of letting it slide, can keep everyone out of harm's way. I have found that regular training sessions and informal check-ins mean way more than posters on the wall. When you treat safe storage as part of the day-to-day routine, chemicals like 1-Butyn-3-ol become tools, not time bombs.
What is the purity and grade of the 1-Butyn-3-Ol product?
Getting to Grips With 1-Butyn-3-Ol Quality
Anyone working in chemical research or manufacturing knows the stress that comes from receiving a fresh barrel of raw materials. You want confidence that the bottle in front of you lives up to what the label claims. 1-Butyn-3-ol is no different. It’s a handy little molecule, often used as a starting material in pharmaceuticals, agrochemicals, and fragrance synthesis. Its triple bond and alcohol group open plenty of doors for creative chemistry, but those benefits rely on the product living up to the right level of purity.
Purity Means More Than a Big Percentage
Suppliers will typically list purities like 98% or 99%, usually backed up by a gas chromatography report. The remaining sliver—that unspoken percentage—actually makes a big difference for what comes next. I’ve stood in labs where a supposed 99% reagent consistently delivered dodgy results, only to discover an undocumented impurity was jamming up reactions or poisoning catalysts. Not all impurities show up. Color and odor sometimes tell a story labs ignore, especially with less common compounds like 1-butyn-3-ol. Some producers offer “HPLC grade” or “analytical grade” promises, which sound reassuring but can mask variations batch-to-batch.
Why Purity Matters in the Real World
Imagine scaling a reaction for a pilot program. You go with bulk 1-butyn-3-ol marked 98%. If the contaminant is a closely related alkyne or a leftover from synthesis—something reactive—it throws off yield or introduces a side product that’s hard to purify out. Teams on shoestring budgets sometimes gamble with tech-grade supplies, tempted by lower costs. That cheaper up-front price can turn into a mountain of analytical work and failed lots down the line.
Pharmaceutical regulations bring serious scrutiny. If 1-butyn-3-ol tags along in a drug intermediate, auditors want a clear record tracking contaminants at every stage. If trace impurities escape early characterization, developers risk failing validation—potentially losing millions. Working alongside colleagues in synthesis, I’ve seen whole projects stall over the “last one percent.” For new molecules destined for tight regulatory oversight, anyone using this compound must demand a complete report, not just a surface-level GC trace.
How To Secure the Right Grade
It makes sense to drill down on supplier transparency. Certifications like ISO 9001 help, but I want batch-specific certificates of analysis and spectra. In analytical development, it’s common to re-check supplier data with in-house NMR and GC-MS. If the project leaves little room for error, I always run a few test reactions and measure actual conversion. Sometimes that simple trial does more to reveal hidden trouble than a certificate ever could.
Collaboration offers a practical path forward. Bringing chemists, analysts, and buyers together at the purchase stage can catch red flags early. End users should provide feedback to suppliers if hidden contaminants crop up. Lab-tested standards and open communication help clamp down on mysterious product failures—saving serious time and cash for everyone involved.
| Names | |
| Preferred IUPAC name | But-1-yn-3-ol |
| Other names |
Ethynylcarbinol 3-Hydroxy-1-butyne But-1-yn-3-ol |
| Pronunciation | /ˈwʌtɪnˌθriː.ɒl/ |
| Identifiers | |
| CAS Number | '927-74-2' |
| Beilstein Reference | 1209221 |
| ChEBI | CHEBI:51736 |
| ChEMBL | CHEMBL16506 |
| ChemSpider | 71263 |
| DrugBank | DB02175 |
| ECHA InfoCard | 100.021.769 |
| EC Number | 201-102-1 |
| Gmelin Reference | 84135 |
| KEGG | C01734 |
| MeSH | D001419 |
| PubChem CID | 78435 |
| RTECS number | EO5950000 |
| UNII | 0W7RRY6P9Y |
| UN number | UN1993 |
| Properties | |
| Chemical formula | C4H6O |
| Molar mass | 70.09 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | sweet |
| Density | 0.857 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | 0.28 |
| Vapor pressure | 0.9 mmHg (at 20 °C) |
| Acidity (pKa) | 13.78 |
| Basicity (pKb) | The pKb of 1-Butyn-3-ol is 15.46 |
| Magnetic susceptibility (χ) | -27.3e-6 cm³/mol |
| Refractive index (nD) | 1.416 |
| Viscosity | 2.8 mPa·s (20 °C) |
| Dipole moment | 1.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -53.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4536 kJ mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | H225", "H319", "H335 |
| Signal word | Warning |
| Hazard statements | H226, H301, H311, H331, H373 |
| Precautionary statements | P210, P261, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | 46 °C |
| Autoignition temperature | 225 °C |
| Explosive limits | 1.8% - 12.2% |
| Lethal dose or concentration | LD50 oral rat 620 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 1-Butyn-3-Ol: **1230 mg/kg (rat, oral)** |
| NIOSH | KW8225000 |
| REL (Recommended) | 250 mg/mL in Methanol |
| IDLH (Immediate danger) | IDLH: 200 ppm |
| Related compounds | |
| Related compounds |
Propargyl alcohol Methylbutynol But-2-yne But-1-yne But-3-yne |

