1-Bromo-3-Methylbutane: A Close Look at its Role and Future in Chemistry
Historical Development
Chemists started exploring alkyl halides over a century ago, curious about how simple modifications could shake up molecular behavior. 1-Bromo-3-methylbutane quickly drew interest for its straightforward structure and strong reactivity. In the labs of the late 1800s, researchers discovered its use as a reliable alkylating agent. Early synthetic organic chemistry workhorse compounds, like this one, laid the groundwork for many pharmacological and agricultural breakthroughs. By the 1950s, with petrochemical and pharmaceutical revolutions running strong, scientists counted on halogenated butanes as key steps in the assembly of more elaborate molecules. Over decades, teams dialed in purification and production; manufacturers responded by tightening standards and scaling up operations.
Product Overview
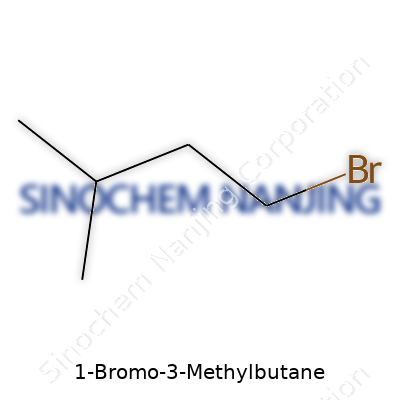
1-Bromo-3-methylbutane, also called isoamyl bromide, packs a punch in a compact molecule. Thanks to a stable carbon backbone and a reactive bromine end, the compound earned a spot in synthetic chemists’ toolkits. Users in the lab pick it for its reliability as an alkylating agent, especially in Grignard reactions or for introducing isopentyl groups. Its straight-forward formula, C5H11Br, means no one gets caught guessing its composition. Most bottles on the shelf come as a clear, colorless to pale yellow liquid, allowing for quick visual checks on purity.
Physical & Chemical Properties
In daily lab use, 1-Bromo-3-methylbutane reveals itself by a distinctive odor—not exactly pleasant, but unmistakable. Boiling near 118°C, with a density around 1.20 g/cm³, it straddles the line between volatility and manageable handling. Its refractive index helps techs confirm identity, while low solubility in water highlights its affinity for organic phases. Reactivity centers on the bromine atom, which often departs under the right circumstances, replaced by all sorts of functional groups. Useful for SN2 substitution, its moderate steric hindrance provides a dependable balance of selectivity and speed, making it a preferred candidate during multi-step syntheses.
Technical Specifications & Labeling
Every container of 1-Bromo-3-methylbutane comes with a set of numbers and warnings. Labs may spot purity grades ranging from 95% for general use up to 99% for high-stakes projects. Labels flag flammability and skin irritation risks, thanks to its halide nature and liquid form. Standard storage calls for tight seals, cool places, and clear hazard symbols. For researchers worried about regulatory compliance, each jug displays CAS number 5162-09-6, ensuring seamless record-keeping during audits or cross-referencing with global inventories.
Preparation Method
Turning out 1-Bromo-3-methylbutane isn’t especially tricky, but it does call for patience and careful planning. A standard approach uses isoamyl alcohol, hydrobromic acid, and sometimes sulfuric acid as a catalyst. The process consistently brings organic chemistry students face-to-face with caution signs: the fumes bite, and the reactions demand respect for temperature controls. After distillation, clean-up steps shunt unreacted alcohols and water out of reach. Chemists finishing this prep often learn practical skills in extraction and purification along the way.
Chemical Reactions & Modifications
Hands-on chemists reach for 1-Bromo-3-methylbutane during nucleophilic substitution reactions. Alkali metals, magnesium, and strong bases coax the bromine atom away, leading to Grignard reagents that build carbon-carbon bonds—a foundation in drug and fragrance synthesis. More advanced applications bring in cyanides, amines, or thiols to form nitriles, amines, or thioethers. Every swap expands the molecule’s reach. Its moderate branching reduces elimination side-reactions, giving more predictable results than straight-chain analogs. Learners quickly grasp why the compound appears in textbooks and GPCR ligand libraries alike.
Synonyms & Product Names
Browsing catalogs, researchers stumble across familiar aliases: isoamyl bromide, 1-bromoisoamyl, or even 3-methyl-1-bromobutane. Chemists with their heads in patent literature spot entries under these names, ensuring broad recognition. These variations don’t change the chemistry but emphasize its global presence across suppliers and regulatory agencies.
Safety & Operational Standards
Using this compound safely means listening to experience. Skin contact burns, inhalation irritates: gloves and fume hoods become mandatory, not optional. Fire risk rises with its low flash point—storage next to ignition sources or heat can turn a routine day into a disaster. Disposal standards reflect a broader shift in lab culture, requiring halogenated wastes to funnel into specialty containers. More often now, labs train staff beyond the basics, knowing that a moment’s inattention leaves lasting consequences. Suppliers usually pack 1-Bromo-3-methylbutane in robust bottles and ship according to hazardous material protocols.
Application Area
Walk into a modern research lab and the uses for this halide jump out. Synthesizing novel pharmaceuticals, flavor and fragrance intermediates, or stepwise chain extensions during material science projects—1-Bromo-3-methylbutane frequently helps bridge small-scale exploration and industrial-scale output. At analytical benches, its non-aqueous nature allows rapid phase separations. Fragrance houses count on it to build banana- and pear-scented isoamyl derivatives, while pharmaceutical groups value its role in constructing complex scaffolds that explore new therapeutic spaces.
Research & Development
Research teams keep searching for greener synthesis methods, hoping to use fewer hazardous reagents and generate less waste. One approach relies on phase-transfer catalysis to bump up yields and ease separation. Analytical chemists run finer purity checks, pushing detection limits lower and flagging impurities before scale-up. There’s a collective push toward process safety and environmental consciousness. In classrooms, new learning modules use this compound to illustrate the balance between reactivity and selectivity, helping students understand why small tweaks in structure change whole reaction pathways.
Toxicity Research
Toxicologists know halogenated organics introduce challenges to safe handling. In animal tests and workplace monitoring, data suggest 1-Bromo-3-methylbutane can cause nervous system symptoms at high exposures; skin absorption risks prompt warnings about even minor spills. Regulatory agencies continue monitoring long-term risks, including breakdown product effects on soil and water. Recent studies lean into chronic exposure, evaluating risks for laboratory workers who handle the substance regularly. In response, companies update exposure limits and recommend shorter open-bench times to limit risk.
Future Prospects
Looking ahead, green chemistry will shape the story of 1-Bromo-3-methylbutane. Biobased feedstocks and recyclable catalysts offer more responsible choices for preparing halides. Regulatory landscapes keep tightening, demanding lower emissions and stronger protections for users. Universities and manufacturers realize that training future chemists on safety and sustainability isn’t just good ethics—it’s necessary for long-term viability. Adoption of alternative alkylating agents may rise, but 1-Bromo-3-methylbutane isn’t bowing out yet; it remains a benchmark for developing better tools, safer workflows, and smarter synthetic routes.
What is the chemical formula of 1-Bromo-3-Methylbutane?
A Look at 1-Bromo-3-Methylbutane
Chemistry class often brought on headaches with names like 1-Bromo-3-Methylbutane. People who work in labs handle complex names every day, but outside those walls, words like these feel intimidating. Still, these compounds touch our lives in ways we hardly notice. Unpacking what goes into a name like this, you find both the origins of the substance and how deeply structure defines what it becomes and what we use it for.
The Structure Hides in the Name
Let's strip down the name: 1-Bromo-3-Methylbutane. “Butane” tells you there are four carbon atoms in the main chain. Counting starts from the end that makes the rest of the name fit best. “1-Bromo” means a bromine atom attaches to the first carbon. “3-Methyl” adds a methyl group (CH3) onto the third carbon. If you ever pieced together a LEGO set, this sort of building block chemistry rings a bell.
Writing it out, the formula comes to C5H11Br. Count those up: five carbons, eleven hydrogens, one bromine. No hidden rules—just careful placement. Details shape function, and in the world of organic chemistry, swapping out even one atom changes everything.
Real-Life Importance: Where Science Leaves the Classroom
In day-to-day work, knowing the formula for a compound like 1-Bromo-3-Methylbutane means understanding its risks and uses. Brominated hydrocarbons like this one see life in industries from pharmaceuticals to pesticides. Sticking a bromine atom on a molecule can crank up a drug’s performance, or change how a pesticide stays stable under sunlight.
The chemical’s properties matter for everyone from factory workers to end customers. Exposure to brominated substances brings health risks—eye and skin irritation, even bigger problems if a lot of the chemical gets into groundwater. People deserve to know what’s being made in their neighborhoods, and scientists have to account for long-term safety. Transparency comes from sharing exact formulas, allowing independent reviews for both performance and risk.
Challenges and Next Steps
Anyone working around 1-Bromo-3-Methylbutane can feel the push to handle and dispose of it responsibly. The Environmental Protection Agency and other health agencies have warned about improper disposal of halogenated organics. These rules write safety into the workflow, so mistakes become less likely. But rules alone don’t keep disasters from happening—industry insiders need solid training, and everyone should share findings, both good and bad, to spot trouble before it spreads.
Sustainable chemistry isn’t just a slogan; it adds value by keeping air, water, and people safer. Research points toward using greener chemicals where possible, and for essentials like 1-Bromo-3-Methylbutane, improving how we recycle or neutralize byproducts lessens the long-term impact.
Knowledge is Power, and Power Means Responsibility
Knowing C5H11Br at a glance means seeing both opportunity and responsibility. The structure tells chemists how to build it and break it down, but the rest of us get value by keeping science honest. Safety, transparency, and a drive for greener methods can move industry forward without ignoring risks. It starts with understanding what a formula means and what it can do once it leaves the bottle.
What is the primary use of 1-Bromo-3-Methylbutane?
Grasping the Backbone: Where Does 1-Bromo-3-Methylbutane Fit?
For anyone who’s poked around a synthetic chemistry lab, the name 1-Bromo-3-Methylbutane rings a bell. This compound, a colorless liquid with a faint, sweet smell, doesn’t show up in headlines or your medicine cabinet. Its presence in the lab tells a deeper story. My years juggling test tubes and textbooks in the world of organic synthesis kept showing me one thing: intermediates like this are the unsung workhorses of modern chemistry.
One Main Use: Building Blocks for Bigger Ideas
Ask a chemist, “What’s the big deal with 1-Bromo-3-Methylbutane?” Most folks will mention its role as an alkylating agent. In real terms, this simply means it helps stick pieces together. To make new carbon-carbon bonds, you need molecules that will deliver their carbon chain to another. 1-Bromo-3-Methylbutane brings a butyl group—a chunk of four carbons—ready to attach where the chemist wants it.
The big stage for this is something called Grignard reactions. Picture magnesium getting thrown into the mix with 1-Bromo-3-Methylbutane. They form a Grignard reagent that acts like a charged-up construction worker. Now you’ve got a tool that snaps onto carbonyl compounds—aldehydes, ketones—and lets makers build up bigger, more interesting molecules. I can’t count how many pharmaceutical research projects I’ve watched that rely on this. Even if you never hear the compound’s name again, its fingerprints end up on painkillers, agrochemicals, and fragrances.
Beyond the Bench: Industry and Research
In industry, time means money. Companies need to crank out specific molecules with as little waste as possible. 1-Bromo-3-Methylbutane fits the bill for efficiency. It boils at a manageable temperature, so separation and purification steps run smooth. Labs don’t wrestle with stubborn side products, so the process keeps moving. This ability to slip into reactions and deliver a usable chunk of carbon streamlines production for specialty chemicals.
As a teaching assistant, I saw undergrads use this compound in practice sessions to understand nucleophilic substitution. There’s no replacement for hands-on trial and error: watch how quickly a real reaction runs, see what happens if you skip a step or use too much heat. Chemistry is a contact sport, and tools like 1-Bromo-3-Methylbutane bring theory out of the textbook.
Handling Hazards and Seeking Greener Paths
No one should work with 1-Bromo-3-Methylbutane without real safety precautions. Bromoalkanes don’t play nice with skin, eyes, or lungs. Chronic exposure to chemicals like these has been linked to liver and kidney stress, so smart labs invest in fume hoods and gloves. It’s a running conversation in academic circles and on technical forums—how can we find or invent less toxic substitutes?
Chemists keep tinkering with ways to avoid halogenated reagents like this one. Some new research turns to alternative alkylation methods—moving to greener solvents, picking safer leaving groups, and embracing electrochemical techniques. Progress isn’t always fast, but each year shows new approaches that limit waste or use renewable materials.
The Takeaway
1-Bromo-3-Methylbutane rarely grabs attention, but it forms the backbone of many chemical syntheses. Its most important job is playing matchmaker in the creation of more complex organic molecules. I’ve seen this compound in action on countless bench tops, carrying ideas from the drawing board to real-world application—and along the way, it’s pushed chemists to aim for safer, cleaner, and smarter science.
What are the safety precautions when handling 1-Bromo-3-Methylbutane?
Understanding the Risks
Working with chemicals like 1-Bromo-3-Methylbutane brings a few responsibilities. This compound can irritate the skin, eyes, and lungs. The smell hints that it’s not something to take lightly. Anyone who’s opened a bottle without proper ventilation knows the heavy, pungent odor that lingers. The hazards don’t stop at the immediate discomfort. Long-term exposure could harm your health, and spills can cause headaches long after the mess is cleaned up.
Personal Protective Equipment: Not Just for the Lab
The best way to avoid trouble starts with good habits. Nitrile gloves handle this chemical better than latex. Safety goggles keep accidental splashes away from your eyes. Most folks usually ignore lab coats or aprons, but after seeing a lab partner’s ruined jeans during a spill, no one skips protective clothing anymore. Wearing long pants and closed shoes also keeps accidents in check.
Ventilation: Breathing Easier
Anyone who has handled solvents in a small room knows what poor ventilation feels like. Breathing becomes harsh and the nose burns. Good airflow cuts down risks fast. Working under a fume hood or in a well-aired space means less chance for fumes to build up. This is something the American Chemical Society and OSHA emphasize for good reason: irritation and toxic effects from inhalation can develop before you even notice.
Spill Control and Clean-Up
Spills happen, even if precautions are in place. Absorbent pads and spill kits should always sit nearby. Pouring slowly and using less whenever possible goes a long way. Once saw someone try to mop up a chemical spill with paper towels and no gloves. They ended up with irritated skin and a lesson not soon forgotten. Use tools, not hands, to wipe up. Place waste in a labeled, sealed container for disposal—don’t toss it in regular trash.
Storage: Not All Shelves Work
This chemical shouldn’t mix with oxidizers or open flames. Storing bottles in a designated cabinet, away from heat and sunlight, limits degradation and vapor buildup. Tight lids on containers help too. In one shared lab space, an unlabeled bottle left open ended with a reeking storage area and a costly clean-up. Clear labeling and secure lids prevent confusion and accidents.
Training and Supervision
Instruction matters. A step-by-step run-through and active supervision, especially for newcomers, keeps everyone safer. I learned most safety routines from older coworkers who weren’t shy about pointing out mistakes. Checklists and daily reminders can feel repetitive until you watch them stop a problem before it starts. Safety information and first-aid protocols should sit close at hand—to me, they work better posted on the wall than buried in a binder.
Recognizing Symptoms and Responding Fast
Pay attention to your body: headache, dizziness, coughing, or a burning sensation should never be ignored. Quick rinsing after a splash, fresh air for a cough, and prompt medical attention for serious exposure make a difference. In every chemical handling class and lab, the advice stays the same—don’t push through discomfort, step away and report it.
Building a Safer Practice
Respect for chemicals like 1-Bromo-3-Methylbutane builds from seeing the effects of poor habits. No shortcut replaces gloves, goggles, and fresh air. Safe handling is a culture that grows out of practice, not just rules. Until safety feels like part of the process rather than a set of hoops to jump through, incidents keep happening. Solutions exist in the shared commitment to careful handling, honest reporting, and ongoing education.
What is the boiling point of 1-Bromo-3-Methylbutane?
Digging Into 1-Bromo-3-Methylbutane
Chemistry brings a lot of curiosity, especially when you come across names like 1-Bromo-3-methylbutane. For anyone who spends time working with chemicals, either in the lab or in industry, knowing something as specific as a boiling point isn’t just trivia. 1-Bromo-3-methylbutane, a colorless organic liquid, pops up in many experiments and manufacturing processes. Its boiling point sits around 91–92°C. That might sound like just another number in a long list, but the science and the uses behind this point carry real-world weight.
Why Boiling Points Matter
The boiling point of a compound drives choices in storage, handling, and the design of production equipment. A boiling point right under 100°C means this compound will vaporize fairly easily. Ventilation and temperature controls go from nice-to-have to must-have in spaces where this chemical appears. In college, I remember working with a fellow student who underestimated the volatility of a low-boiling halide. Our flask popped its stopper, sending fumes around the room. That episode stuck with me—always respect a compound’s physical properties.
Real Chemists, Real Risks
Even seasoned chemists sometimes trip up without a clear picture of what happens at certain temperatures. With this brominated butane, proper PPE and hood use matter. They aren’t just reminders in a safety video. On one project, a colleague switched out a solvent for an alkyl bromide in a synthesis, thinking the new setup was risk-free. Only later did lingering vapors slow down productivity for days. The lesson: boiling points drive more than lab trivia—they affect workflows and safety records in labs and factories.
Industrial Value and Challenges
1-Bromo-3-methylbutane isn’t just sitting on a shelf for fun. It’s used for making other chemicals, especially where you need to add a butyl group or insert a halogen atom. It also comes up in pharmaceutical intermediates. During production, careful distillation steps take place—often under inert atmosphere—to avoid runaway evaporation or accidental ignition. Mishandling these steps leads to lower yield, product contamination, and higher risk of exposure for workers.
Finding Solutions
Addressing boiling point challenges isn’t just about stricter rules—it’s about working smarter. Chemists regularly use closed systems with real-time temperature monitoring. Fume hoods and local exhaust keep air quality safe. Labels and hazard sheets go on every container, cutting down confusion. Newer digital records make safety information easier to share, especially as younger lab staff come up against volatile chemicals for the first time. In teaching labs, repeated drills and transparent incident debriefs set up better habits from the start. Industry players also choose greener, less volatile alternatives when possible, reducing overall risk.
Final Thought on Chemical Know-How
It’s easy to treat numbers like boiling points as forgettable details. Years of bench work reinforce one truth: clear information prevents accidents. Knowing the boiling point of 1-bromo-3-methylbutane means keeping people safer, saving time, and getting reliable results. It’s a small number holding up a big part of daily laboratory and industrial practice.
How should 1-Bromo-3-Methylbutane be stored?
Reality of Working Around Chemicals
Plenty of people don’t think twice before grabbing a chemical in the lab and pouring it out, but the trouble starts if the right storage habits aren’t part of the routine. 1-Bromo-3-Methylbutane isn’t something anyone wants spilling or turning volatile indoors. I’ve seen firsthand, from years chasing down strong lab odors and cleaning up spills in tight storerooms, how improper storage can lead to split-second emergencies.
What 1-Bromo-3-Methylbutane Demands
1-Bromo-3-Methylbutane rolls onto the scene as a halogenated compound, so it calls for practical measures. It loves to evaporate and release vapors that irritate the skin and eyes. Because inhaling its fumes or dealing with accidental splashes leads to more hospital visits than a simple cut, the safest bet is keeping it under lock and key in a cool place. Glass bottles or tightly sealed containers offer best protection.
I’ve stored my share of nasty stuff in flammable storage cabinets, but this chemical prefers storage away from direct sunlight down in the temperature-controlled sections of the lab. Ignoring this risks decomposing the contents, increasing pressure inside bottles, or even turning a minor leak into a full-on hazardous cleanup job.
Avoiding the Recipe for Disaster
Every time I see a bottle perched on an open shelf or getting too cozy with acids, I remember that even the most experienced folks get burned by comfort. 1-Bromo-3-Methylbutane reacts with strong oxidizers. If someone stores it near cleaning chemicals like hydrogen peroxide or bleach, things can turn explosive, literally. Dedicated shelving — not next to the glass cleaner or the acids — eliminates the cross-contamination problem. Clearly labeled areas help everyone, novice or expert, find the right spot without confusion.
Storage Isn’t Only About Temperature
Humidity wrecks containers over time, so dry spaces slow down leaks caused by corrosion. Spill trays under containers offer a backup for those moments someone fumbles a bottle or a cap fails. These little steps, from my own experience, have kept Friday afternoon surprises to a minimum.
Ventilation also plays a quiet hero. Uncork a bottle in an unventilated storeroom, and even small spills leave the air heavy and foul. Chemical fume hoods at the point of use help, but adding exhaust fans near storage cabinets means lingering vapors don’t build up to dangerous levels.
Accountability and Education Matter Most
Locking up hazardous chemicals isn’t enough. I’ve watched brand new interns and crusty old lab techs struggle with the difference between storing alcohols and halogenated species. Roll out regular safety training that includes real photos and stories of what happens after a storage mistake. People remember lessons tied to a near-miss.
Inventory tracking — not just jotting numbers on a clipboard, but someone actually checking each bottle’s seal, label, and expiration date every month — keeps dangerous surprises out of reach. Publicly visible logs produce results, because no one wants their name associated with the most recent spill.
Protecting People, Not Just Property
In my years inside chemical storerooms, the best labs set up redundant layers of safety. Backup containers, visible hazard signs, and proper personal protective equipment nearby transform storage from a box-checking exercise into a genuine shield. Responsible storage of 1-Bromo-3-Methylbutane means fewer accidents, cleaner air, and peace of mind for everyone clocking in.
| Names | |
| Preferred IUPAC name | 2-Bromo-3-methylbutane |
| Other names |
isoamyl bromide isopentyl bromide 3-methyl-1-bromobutane 1-bromoisoamyl |
| Pronunciation | /waɪˈbrəʊmoʊ θriː ˈmɛθɪlˌbjuːteɪn/ |
| Identifiers | |
| CAS Number | InChI=1-1-bromo-3-methylbutane-3724-82-3 |
| 3D model (JSmol) | CC(C)CCBr |
| Beilstein Reference | 1361005 |
| ChEBI | CHEBI:81217 |
| ChEMBL | CHEMBL15639 |
| ChemSpider | 32237 |
| DrugBank | DB02229 |
| ECHA InfoCard | 100.008.225 |
| EC Number | 208-801-3 |
| Gmelin Reference | 1201491 |
| KEGG | C07338 |
| MeSH | D016690 |
| PubChem CID | 11810 |
| RTECS number | EO1575000 |
| UNII | Z1W2M8K5JP |
| UN number | UN2344 |
| Properties | |
| Chemical formula | C5H11Br |
| Molar mass | 137.04 g/mol |
| Appearance | Colorless liquid |
| Odor | penetrating |
| Density | 1.225 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 2.89 |
| Vapor pressure | 3.3 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 50 |
| Magnetic susceptibility (χ) | -71.0e-6 cm^3/mol |
| Refractive index (nD) | 1.439 |
| Viscosity | 2.39 mPa·s (20 °C) |
| Dipole moment | 2.30 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -139.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2447.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P271, P301+P312, P304+P340, P312, P403+P233 |
| NFPA 704 (fire diamond) | NFPA 704: 1-2-0 |
| Flash point | 48 °F |
| Autoignition temperature | 410 °C |
| Explosive limits | Upper: 8.3% ; Lower: 1.0% |
| Lethal dose or concentration | LD50 oral rat 760 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 1600 mg/kg |
| NIOSH | PB6125000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
1-Bromobutane 2-Bromobutane 3-Bromopentane 1-Chloro-3-methylbutane 3-Methyl-1-butene |

