1-Bromo-2,4-Dinitrobenzene: A Detailed Commentary
Historical Development
Coming across 1-Bromo-2,4-dinitrobenzene takes me back to early days in organic chemistry labs, where compounds like this shaped our approach to understanding aromatic substitutions and halogenations. Back in the late nineteenth century, organic chemists started tinkering with nitroaromatic compounds, chasing new synthetic dyes and explosives. Through decades, researchers leaned into dinitrobenzene derivatives, eager to manipulate electron density and reactivity. The addition of a bromine to the ring, specifically at the 1-position, solidified 1-Bromo-2,4-dinitrobenzene’s place in labs building intermediates for further molecular tweaks. That legacy stays in textbooks, patents, and modern syntheses.
Product Overview
You don’t see flashy marketing about this compound, but its importance sits in factories and research departments across specialty chemical sectors. 1-Bromo-2,4-dinitrobenzene acts as a stepping stone, letting scientists install or swap groups on benzene rings with surgical precision. The core appeal isn’t just in its yellow, crystalline nature, but in the way it opens doors to more complex molecules. Firms ordering this substance know it isn’t the end product—they bank on its reliability as an electrophilic partner. Uses in dyes, pesticides, and as a marker for amino acid analysis reflect this sturdy versatility.
Physical & Chemical Properties
The crystals can look striking under a glass dish—rich yellow, classically aromatic. With a melting point often near 89–91 °C, it resists gentle warmth but gives way to moderate heating. Solubility in ethanol draws researchers during extraction or purification, while water shuns it almost completely. Chemical structure includes a bromine at the 1-position, and nitro groups at the 2- and 4- positions, shaping the molecule’s dense electron atmosphere. That drives a unique reactivity profile—a kind of ‘personality’ that savvy chemists quickly learn to respect. A formula of C6H3BrN2O4 and molecular weight tipping just past 260 g/mol might seem routine, but it’s a useful checkpoint through NMR, IR, and UV-Vis for anyone confirming a fresh batch.
Technical Specifications & Labeling
Every drum or glass bottle ships with clear hazard codes and a label most commonly reading “1-Bromo-2,4-dinitrobenzene.” Regulatory frameworks in both the US and Europe demand thorough lot numbers, safety symbols for toxicity, and storage instructions. Keeping this compound away from heat and direct sunlight isn’t just about protocol—improper storage affects not only purity but other materials nearby. Companies aiming for pharmaceutical or fine chemical markets keep testing standards tight, with HPLC or NMR verification for every shipment. Certificates of analysis travel with each sale. For those concerned with environmental and worker safety, hazard statements and protective guidance must stand out on every batch.
Preparation Method
Making 1-Bromo-2,4-dinitrobenzene from scratch rewards careful timing and a steady hand. The process typically starts by nitrating bromobenzene, though some routes work backward from dinitrobenzene itself. Old literature points to mixed acid nitration—think concentrated sulfuric and nitric acids—adding just enough heat to drive substitution without churning up tar or wasteful byproducts. Industrial practitioners control the temperature and acid feed rates, often under glass-lined vessels. Proper isolation calls for repeated washing, neutralization, and sometimes a touch of recrystallization in ethanol or acetone to reach the yellow, sharp-melting product found in research catalogs. Waste handling remains a real concern, with nitroaromatics requiring thorough neutralization and strict stewardship during disposal.
Chemical Reactions & Modifications
Years in synthetic labs taught me that 1-Bromo-2,4-dinitrobenzene demands respect during substitutions. The bromo group acts as a useful leaving group, making it ideal for nucleophilic aromatic substitution, especially with strong bases or amines. The presence of nitro groups at 2 and 4 positions cranks up the ring’s susceptibility to incoming nucleophiles. Swapping the bromine for oxygen, sulfur, or nitrogen-based substituents extends its utility far beyond the textbook example. Reductive modifications use iron filings and acid to take a nitro group down to an amine, forming new building blocks quickly. Analytical chemists have tapped it for labeling thiol groups in amino acids, marking proteins in early days of peptide sequencing.
Synonyms & Product Names
Researchers and suppliers list this compound under several names, sometimes leading to confusion if catalogs are outdated. “1-Bromo-2,4-dinitrobenzene” is most direct, but “Bromodinitrobenzene,” “BDNB,” or even “Bromide of Dinitrobenzene” may crop up. I’ve seen CAS number 585-79-5 used as a shorthand by regulatory officials and procurement teams tracking controlled substances. It pays to check a catalog or safety data sheet twice, as isomers with bromine or nitro groups in other positions behave remarkably differently in chemical reactions or safety.
Safety & Operational Standards
Nobody who’s handled nitrobenzene derivatives forgets their hazards. 1-Bromo-2,4-dinitrobenzene packs a punch—exposure risks run from acute toxicity to skin irritation. Safety goggles, gloves, and plenty of fresh air make a difference during weighing, mixing, and disposal. Absorption through skin remains a hidden risk, so splash-prone operations call for extra attention. Material safety data sheets advise against storing it with reducing agents or open flames. Spills need quick cleanup with inert absorbents, and local environmental rules often treat waste from nitroaromatics with heightened scrutiny. Regular audits and basic training keep labs accident-free, which matters more than any time or cost savings.
Application Area
While it’s not the face of consumer goods, behind the scenes 1-Bromo-2,4-dinitrobenzene stays influential in synthetic organic chemistry, pharmaceutical development, and polymer science. Its role in characterizing amino acids put it in earlier clinical labs, while dyed textiles point to its history in pigments. Agrochemical firms use its intermediacy for developing new pesticides and herbicides. Engineers building specialty monomers lean on its reactivity, crafting resins with demanding performance specs. Even as more eco-friendly molecules enter the scene, its niche persists where nothing else fits quite as well.
Research & Development
Chasing improvements for existing processes means researchers keep returning to 1-Bromo-2,4-dinitrobenzene. Green chemistry initiatives look to cut acid use and hazardous waste during synthesis. Automated reactors and flow chemistry have trimmed human error and improved yields on pilot scales. Universities run projects exploring new catalytic systems, hoping to swap out aggressive reagents for milder ones. Analytical chemists probe ways to use it in novel tagging strategies for proteomics, driven by advancements in mass spectrometry. Data from these efforts continues to fill peer-reviewed journals, industry white papers, and trade publications.
Toxicity Research
Toxicologists have studied 1-Bromo-2,4-dinitrobenzene for generations, setting exposure limits and chronic toxicity profiles. Even small doses can cause nausea, dizziness, headaches, with long-term exposure leading to more serious health effects—liver and kidney damage rank as chief concerns. Studies on aquatic organisms show real threats to water quality if waste streams escape treatment plants. Medical surveillance in workplaces involving this substance became common practice, reflecting a wider cultural push toward safe chemistry. Modern toxicologists now use in vitro and computational models to minimize animal testing, but the basics remain—treat it with seriousness, and invest in controls.
Future Prospects
Looking out at the future of specialty chemicals, there’s no denying pressure from green chemistry and stronger regulations. Markets drift toward safer, biodegradable alternatives, but legacy compounds rarely disappear overnight. Companies who stick with 1-Bromo-2,4-dinitrobenzene face mounting expectations for transparency, safety, and greener waste treatment. Researchers tinker with catalyst design and greener solvents, with one eye on cutting environmental impact. The journey probably won’t end with this molecule, but its deep-rooted utility in synthesis guarantees a role in classrooms and production sites for years to come.
What is the chemical formula of 1-Bromo-2,4-Dinitrobenzene?
Looking at Structure to See the Big Picture
Names of chemicals can look intimidating. Take 1-Bromo-2,4-Dinitrobenzene—this one packs plenty of syllables but breaks down simply for those who took high school organic chemistry. The name gives a roadmap: a benzene ring, one bromine at position one, and two nitro groups at positions two and four.
The formula for 1-Bromo-2,4-Dinitrobenzene is C6H3BrN2O4. The structure matters for more than showing off in a chemistry class. Organic compounds with a benzene ring often end up as ingredients or byproducts in dyes, pharmaceuticals, or agrochemicals.
The Real World Behind the Formula
I spent enough hours in a university lab to know how much time goes into taking a chemical from bottle to reaction flask. Having the correct formula on the bottle can save the day. Misreading a label, grabbing a bottle of 2,4-Dinitrobromobenzene (instead of something slightly different), and the next thing you know—hours lost and maybe a whiff of something that peels paint.
C6H3BrN2O4 starts with the ring—six carbons bonded into that hexagon known throughout organic chemistry. Bromine takes the top spot on the ring, bringing reactivity and a certain weight to the molecule. The two nitro groups bring a new level of chemical drama, both with their explosive history and with the way they change the electron character of the benzene ring.
Potential Hazards and Health Implications
Safety comes with knowledge. Nitro compounds catch the eye of anyone responsible for handling chemicals, since they frequently show up in explosives and dangerous industrial byproducts. Introducing bromine adds another layer of caution, because it can increase toxicity and environmental persistence. Safety data sheets show that 1-Bromo-2,4-Dinitrobenzene can irritate eyes, skin, and lungs. It's not the compound you want to spill or inhale.
Recognizing potential risks, old-school lab managers tend to double- and triple-check chemical identifiers before starting a reaction. Plenty of health risks come from poor labeling or outdated knowledge, and specialty chemicals like this one have a reputation that travels ahead of them in many research communities.
Environmental Concerns and Possible Solutions
Chemicals with halogens and nitro groups often linger in the environment longer than many would expect. They resist breaking down and can bioaccumulate. Years ago, I watched a university project look at breakdown pathways for nitro- and bromo-benzenes with some neural skepticism. Natural decomposition feels painfully slow in cold environments.
Waste management remains a point of frustration in labs across the country. Incineration with strict filtration can help manage dangerous byproducts, but greener chemistry methods offer a better way forward. Newer research combines low-impact synthesis with efficient breakdown routes, keeping hazards at bay for students, workers, and everyone downstream. Regulatory pressure plus green innovation makes proper labeling, documentation, and disposal of chemicals like 1-Bromo-2,4-Dinitrobenzene not just a rulebook item but a shared responsibility. Keeping good records, staying current with hazard assessments, and participating in lab safety programs add up, protecting both people and the places outside the lab doors.
What are the main applications or uses of 1-Bromo-2,4-Dinitrobenzene?
Tangible Applications in the Laboratory
1-Bromo-2,4-dinitrobenzene has earned a firm place in my work as a research chemist. Labs use it in diverse ways, although most people outside the field never hear about it. One of the biggest uses pops up in classic protein chemistry. Back in school, I ran across the compound as a reagent for identifying the amino acid sequences in peptides. The process isn’t fancy: treat an unknown peptide with 1-Bromo-2,4-dinitrobenzene, let it tag the terminal amino group, then use hydrolysis and analysis to figure out which amino acid sits at the end. The compound’s strong yellow color makes the resulting derivative easy to spot. Frederick Sanger, the two-time Nobel Laureate, used this technique when he made major breakthroughs in protein sequencing.
Supporting Evidence Through Medical and Environmental Science
New methods for protein identification exist today, but the old chemical reactions continue making classrooms and basic laboratories more accessible for students and researchers. Step into a biochemistry class anywhere, you’ll probably see some classic experiments involving 1-Bromo-2,4-dinitrobenzene for hands-on learning. Using stable, easy-to-detect reagents like this helps students grasp fundamental concepts, bringing theory to life before they move on to complex high-end equipment like mass spectrometers.
Beyond the teaching labs, this compound has cropped up in environmental chemistry as a kind of testing agent. Some groups use it as a marker or reference standard in analytical methods. For example, analysts working on environmental pollution sometimes need a solid reference to validate their equipment’s sensitivity. 1-Bromo-2,4-dinitrobenzene gives a strong, reliable signal for that job, helping ensure data’s accuracy in studies with large scientific or policy impact.
Risks and Responsibilities in Handling
The usefulness of this compound does not mean it’s safe to treat lightly. Both the bromine atom and the nitro groups make it much more reactive than everyday chemicals. The nitro groups give it toxic qualities. If someone asked me about handling, I’d say: keep it away from skin and eyes, use a fume hood, and never neglect proper disposal. Reports have linked related dinitrobenzenes to skin irritation, allergies, and harm to aquatic life. Regulatory agencies set limits for workplace exposure. These rules exist to keep both chemists and the environment safe from long-term harm. Anyone who uses this compound carries a duty to follow those rules to the letter.
Room for Improvements and Emerging Solutions
With experience comes a sense that research can always improve safety and environmental impact. A popular discussion topic among colleagues addresses whether labs can adopt greener tagging reagents. Some newer synthetic tags promise lower toxicity or easier disposal. Advanced analytical techniques may someday make older reagents like 1-Bromo-2,4-dinitrobenzene obsolete, but those shifts take time. Support for cleaner chemistry isn’t just about swapping chemicals—it’s about training, investment, and open communication in the global scientific community. Until substitutes fully replace it, responsible use and handling will remain important for both research and teaching labs.
What are the safety and handling precautions for 1-Bromo-2,4-Dinitrobenzene?
Knowing What You're Working With
1-Bromo-2,4-dinitrobenzene isn’t something you'd want in your lunchbox. Its sharp odor and yellow crystals signal danger. This compound falls into the same group as some old-school explosives and synthetic dyes, but its main role today sits in specialty chemistry labs and niche manufacturing. It reacts aggressively and readily latches onto the body’s proteins, which often spells bad news for skin and airways.
How Exposure Can Trip You Up
I've seen coworkers rush through setup and end up regretting it. This chemical burns the skin and eyes, irritates the lungs, and even a whiff can trigger coughing fits or sore throats. Worst-case, long exposure brings in headaches, nausea, or even more serious symptoms. The big issue comes from its ability to stick to surfaces or clothes, raising the chance for accidental, secondary contact.
Precautions Always Matter
The best plan starts before you even open the container. Every time I handled 1-Bromo-2,4-dinitrobenzene, I kept my gear checked and ready: snug-fitting nitrile gloves, splash-proof goggles, a sturdy lab coat, and closed shoes. That’s the baseline—no shortcuts. Goggles must cover the whole eye area; not just glasses but something that feels like a face shield, especially if splashing could happen.
Good fume extraction saves lungs. Modern labs run proper hoods and maintain airflow. The funny thing about fumes is that sometimes you don’t smell them until it’s too late. One faulty extractor led to a coughing session that proved the point. Even with short tasks, pulling open a sash for extra protection sets the right tone.
Spills and Cleanup: Acting Fast
No one likes facing a spill, but everyone should know how. Any powder or solution on the bench gets covered with damp disposable towels—not dry, which only stirs dust. I’ve used soda or specialized absorbents for acids in the past, but for this chemical, dedicated spill pillows and hazardous waste bags work best. Don’t just chuck these in the regular trash; treat all cleanup waste as hazardous and label the bags.
Anything reusable—think glassware or tools—must soak in an appropriate decontaminant. Never trust a quick rinse. Even microscales of residue carry risk, so wash till you’re confident, then send through the dishwasher or autoclave if possible.
Storage: Keeping Trouble Locked Down
Safe storage calls for a dry, cool spot, away from direct sunlight and sources of ignition. Being near strong reducers, bases, or other reactive chemicals only invites trouble. My routine has always included clear bottle labeling, tight screw-caps, and separate bins for all energetic or hazardous powders, with a log tracking every time I took some out.
Safety data sheets never collect dust. Keep them taped inside the cabinet. If the room ever smells odd or a color change pops up on the bottle, stop and call the lab supervisor. Small details like this make a difference.
Building Smarter Habits
Training helps, but habits matter more. I’ve made sure new folks follow the buddy system, especially when using dangerous chemicals. No one works alone, and no one skips over PPE. Safety reminders, like visible signs or walkthrough refreshers, keep procedures fresh in mind.
For labs handling this compound often, setting up spill drills brings peace of mind. Having eyewash stations and emergency showers open and tested builds trust—everyone knows what to do without thinking twice.
Why It’s Worth the Effort
Cutting corners opens up risk, both for yourself and everyone nearby. Early lessons taught me that taking a few extra minutes pays off every time. Working safely with 1-Bromo-2,4-dinitrobenzene isn’t about fear, it’s about respect for the compound, the workspace, and your own future health.
How should 1-Bromo-2,4-Dinitrobenzene be stored?
Understanding the Risks
Handling chemicals like 1-Bromo-2,4-Dinitrobenzene isn’t something most people bump into every day. The compound shows up in research labs, specialty manufacturing, and sometimes teaching spaces. It’s a yellow crystalline powder, bright enough to attract attention, but that’s not the reason safety matters here. This molecule brings real health and environmental concerns, from toxic dust to the risk of fire when mixed with the wrong substances.
Real-World Storage Practices
The moment you pull a bottle of 1-Bromo-2,4-Dinitrobenzene off the shipment box, you can smell the difference compared to table salt or sugar. The powder irritates the nose—sometimes, just opening a bottle triggers a sneeze. The material’s MSDS calls out its ability to harm eyes, skin, and airways, and it can hang in the air as a fine dust. It can also irritate the liver and kidneys with repeated exposure. Years of working in academic labs mean I’ve seen what happens when someone cuts corners—containers left unsealed, powders spilled near bench edges, and brown stains trailing from careless hands. Cleaning up after these mistakes isn’t fun, and nobody wants to learn about toxicity through itchy skin or a trip to the nurse.
A locked chemical cabinet with good labeling, dry shelves, and away from incompatible chemicals like alkalis or strong acids protects everyone in the building. The biggest problem starts when bottles sit next to each other with faded or half-torn labels. Once, in a shared storage space, I spotted a bottle with a handwritten note, “Nitro stuff, be careful.” That’s not enough to keep someone safe. Real storage happens in containers that close tightly—glass works best, since plastic sometimes sparks static—and a clear label tells you exactly what’s inside.
Temperature, Light, and Environmental Hazards
Temperature swings in a storage room push the chemical toward breakdown. Moisture or sun exposure may degrade 1-Bromo-2,4-Dinitrobenzene faster than most realize. My first boss used to keep an analog thermometer in every cabinet, not just relying on central air. Consistent, cool temperatures—usually below 25°C (about 77°F)—keep things stable. Direct sunlight acts like a slow bully, gradually breaking bonds in the chemical structure, so a dark shelf or cupboard helps preserve the material for long-term use.
Spill control means more than just stacking bottles upright. A tray underneath catches stray grains and makes cleanup possible if a wall shelf gets bumped. Lining the tray with absorbent material worked best in one grad student lab. Not everyone enjoys taking inventory, but opening every jar, checking for leaks, and updating logs prevents surprises down the road. Proper logs also help firefighters or hazmat crews understand what’s inside a room if alarms go off.
Solutions and Improvements
Standardized labeling, regular audits, and training beat out expensive equipment if budget feels tight. I’ve found that regular short safety meetings—just ten minutes once a month—lowered accident rates far more than another round of emails ever did. Workers remember techniques like working with gloves, using a mask, and washing hands after each step, especially when instructors connect the advice to real events. Upgrading old glassware and investing in secondary containment trays costs less than one emergency cleanup.
Every person who stores or handles 1-Bromo-2,4-Dinitrobenzene plays a part in building safety for themselves and everyone around. Responsible storage protects health, reduces waste, and lowers the risk of environmental spills—the kind that shut down labs or spark fines. Good practice here goes beyond avoiding trouble; it turns basic storage into a sign of respect for knowledge and for life itself.
Is 1-Bromo-2,4-Dinitrobenzene available in different purity grades?
Why Purity Matters in Chemical Purchase
Every chemist knows the frustration that comes from getting unreliable results. Plenty of us have watched hours of work go down the drain because a reagent turned out less pure than expected. Whether it’s a research group or a quality control lab in a pharmaceutical company, the choice of chemical purity determines experiment quality, even the safety of the team handling those chemicals.
1-Bromo-2,4-Dinitrobenzene: What’s On Offer
About 1-Bromo-2,4-dinitrobenzene—for many, this chemical acts as a valuable building block in synthesis and analytical chemistry. Vendors offer it in multiple purity grades. The two main categories you’ll run into are technical grade and analytical (often called reagent grade or ACS grade). Technical grade describes material with sufficient purity for industrial processes, often hovering below 95%. On the other hand, analytical and reagent grades usually guarantee purity levels above 98%, backed by specific testing protocols and certificates of analysis.
My Lab’s Experience with Purity Choices
I remember my team running multiple reactions where trace contaminants sabotaged our results. Only after checking the batch data did the culprit pop up: a lower grade of 1-Bromo-2,4-dinitrobenzene that brought in unwanted byproducts. The high-purity stuff fixed the problem. The price? Of course, higher, but lost time in failed reactions cost a lot more.
Not every job needs the cleanest chemical available. In bulk applications, like dye manufacturing or explosives, companies might settle for technical grade to cut costs. These processes often tolerate a few extra percent of unspecified material. Academic and pharmaceutical research can’t take those gambles. An extra tenth of a percent impurity could shoot down months of hard work if it interferes with spectra or causes unexpected side reactions.
Risk of Impurities in 1-Bromo-2,4-Dinitrobenzene
Many folks, especially students, underestimate the risks contaminants pose. Certain byproducts might trigger skin reactions on handling, or even worse, become toxic if they end up in a finished compound. Errors in reporting purity not only mess up data—they increase risk for anyone working down the line.
How to Pick the Right Grade
It pays to ask for certificates of analysis. A good supplier documents not just purity percentage, but also lists potential trace byproducts from their manufacturing process. I always tell new students to cross-check this paperwork before signing off on an order. Also, regularly calibrate equipment and run reference checks with standards from different suppliers. Lab audits might be tedious, but bloated data from bad reagents stays in the literature forever.
The Pursuit of Reliable Results
People trust published results, drug companies depend on precise outcomes, and environmental safety regulations keep tightening. Inconsistent results often trace back to the basics: what goes in. Skimping on purity causes more headaches than it saves dollars. Maybe some can gamble in industry-scale vats, but research and product development thrive on reliability.
Possible Solutions: Better Sourcing, Smarter Testing
Suppliers could tighten batch testing and provide transparent trace impurity data. Labs can pool resources to buy higher grades when needed, and regulators can enforce clearer labeling. As someone who has seen entire projects derailed by murky chemical data, I believe improved transparency and a little more scrutiny on reagent purchases dramatically raises the standard. Never hurts to stay paranoid about the basics—in chemistry, those basics run the show every time.
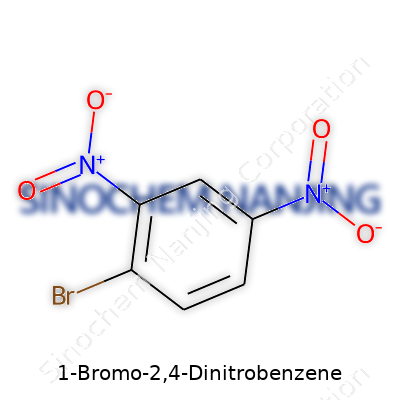
| Names | |
| Preferred IUPAC name | 1-bromo-2,4-dinitrobenzene |
| Other names |
m-Bromodinitrobenzene 2,4-Dinitrobromobenzene |
| Pronunciation | /waɪ ˈbroʊmoʊ ˌdaɪˈnaɪtroʊ ˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 585-71-7 |
| Beilstein Reference | 1461875 |
| ChEBI | CHEBI:51470 |
| ChEMBL | CHEMBL14736 |
| ChemSpider | 162143 |
| DrugBank | DB08274 |
| ECHA InfoCard | 100.012.586 |
| EC Number | 208-188-5 |
| Gmelin Reference | 71585 |
| KEGG | C14568 |
| MeSH | D001930 |
| PubChem CID | 7417 |
| RTECS number | CU8925000 |
| UNII | 8B51O9A1G7 |
| UN number | UN1992 |
| Properties | |
| Chemical formula | C6H3BrN2O4 |
| Molar mass | 246.01 g/mol |
| Appearance | Yellow crystalline powder |
| Odor | Odorless |
| Density | 1.90 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 0.000086 hPa (25°C) |
| Acidity (pKa) | 13.8 |
| Basicity (pKb) | 1.01 |
| Magnetic susceptibility (χ) | -29.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.682 |
| Viscosity | 115 mPa·s (20 °C) |
| Dipole moment | 3.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 339.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -22.9 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1739 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335, H411 |
| Precautionary statements | P261, P280, P305+P351+P338, P301+P312, P337+P313, P501 |
| NFPA 704 (fire diamond) | 2-3-1-✕ |
| Flash point | 60 °C (closed cup) |
| Autoignition temperature | Autoignition temperature: 537°C |
| Lethal dose or concentration | LD50 oral rat 175 mg/kg |
| LD50 (median dose) | LD50 (median dose): 283 mg/kg (oral, rat) |
| NIOSH | CP8925000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Bromo-2,4-Dinitrobenzene: Not established |
| REL (Recommended) | 0.15 mg/m³ |
| IDLH (Immediate danger) | IDLH: 15 mg/m3 |
| Related compounds | |
| Related compounds |
1-Chloro-2,4-dinitrobenzene 2,4-Dinitroanisole 2,4-Dinitrochlorobenzene 2,4-Dinitrophenol 2,4-Dinitrotoluene |

