1-Aminoethanol: A Chemical with a Story
Looking Back: Historical Context
Chemistry has its own set of unsung heroes, and 1-aminoethanol certainly belongs in that camp. This compound, which goes by the chemical formula C2H7NO, isn’t exactly a household name. That said, 1-aminoethanol has been part of organic chemistry conversations for over a century. It drew attention early on as chemists mapped out the complexities of hydroxyamines and their reactivity. Researchers in the twentieth century, hunting for intermediates in amino alcohol syntheses, stumbled upon it and quickly realized its potential in making more advanced molecules. For many, the compound evokes stories of benchtop trials, glassware lined up along fume hoods, and the pursuit of better, safer, and more efficient synthetic routes.
Simple but Not Boring: What It Is
At its core, 1-aminoethanol mixes the functionality of an alcohol with the reactivity of an amine. You get a molecule built off a two-carbon chain, with one carbon holding a hydroxyl group and the other binding to an amino group. The most common way to picture it is a high-energy intermediate or a fleeting product you might capture in a cold flask. It’s not flashy or particularly stable under standard room conditions, and yet this simplicity makes it an important subject in both academic and industrial contexts. Chemists recognize its value not because it's exotic, but because it gives them the building blocks for deeper discoveries.
Physical and Chemical Properties
1-aminoethanol comes across as a colorless liquid under standard conditions, but it's sensitive to both heat and air. Handling it requires preparation since its two reactive groups interact with moisture and oxygen over time, which can transform the compound or degrade it. Its boiling point sits lower than most three-carbon alcohols, reflecting the increased volatility introduced by the amino function. Solubility in water isn’t a major hurdle, since the molecule's polar groups make it highly miscible with aqueous solutions. Where things get trickier is with other solvents. Traditional organics might work, but careful handling is always needed to avoid unwanted side reactions, especially in the presence of strong acids or bases.
Technical Details and Labeling
As with many specialized organic chemicals, quality control for 1-aminoethanol means checking purity, water content, and possible traces of related amines or alcohols. Batch certificates frequently include melting and boiling points as well as refractive index and spectroscopic data, ensuring researchers handle what they expect. Most bottles carry hazard markings due to the molecule's reactivity and (in larger quantities) its irritant qualities. From years of reading labels and safety sheets, clear hazard pictograms and concise risk statements help remind lab users to glove up and keep the solution capped tight when not in active use.
How the Pros Make It: Preparation Method
The textbook route to 1-aminoethanol involves reacting acetaldehyde with ammonia or an ammonium salt under controlled conditions. The reaction starts fast but needs temperature and pH adjustments to favor the right product. Some researchers improve yield by tweaking ammonia concentration or using catalysts, but you rarely see massive scale production in industry—it's more of a small-batch, lab-scale material. The main challenge is avoiding the formation of 2-aminoethanol (ethanolamine), since both are possible. This pushes chemists to optimize steps with careful distillation and select crystallization or extraction techniques. Over time, advancements like flow chemistry and microreactor technology have nudged yields higher, but lab prep remains deliberate and detail-driven.
Chemical Reactions and Modifications
Chemists value the dual reactivity baked into 1-aminoethanol. The primary amine group allows classic amide bond formation, enabling the synthesis of various new molecules through acylation. The alcohol end will jump into etherification or oxidation reactions, often producing aldehydes, ketones, or carboxylic acids depending on the reagent. In my own experience, modifying 1-aminoethanol means keeping reaction times short and choosing solvents that won’t compete or react with either functional group. Its unique position offers a starting point for chiral amine synthesis, making it a useful intermediate toward biologically active compounds and pharmaceuticals. You can't call it a “universal” precursor, but it definitely cracks open doors to new branches of synthetic chemistry.
Other Names Chemists Use
Spending time with chemical catalogs, you notice that 1-aminoethanol wears a handful of aliases. Some call it alpha-aminoethanol, referencing the group’s location on the carbon chain. Others use ethylolamine or 1-hydroxyethanamine. These synonyms matter because they hint at where and how the compound has turned up in patents and academic research. It's a reminder that the same fundamental structure can show up under different guises, depending on the tradition of the particular lab or the naming conventions of the era.
Priority on Safety: Handling and Standards
Any chemist who spends time around volatile amino alcohols understands the importance of solid operational standards. Spills pose skin and eye hazards, and sensitive respiratory tracts can react to vapors. Wearing gloves, goggles, and operating under a fume hood quickly becomes habit, not just suggestion. Storage calls for sealed glassware, with labels tracking dates and purity checks, and careful waste management to keep byproducts from pouring down the drain. Regulatory expectations insist on up-to-date safety data sheets and standardized training for anyone tasked with using or disposing of the compound. Years in the lab have taught me that shortcuts only multiply problems—work smart, keep things clean, and handle with respect.
Who’s Using 1-Aminoethanol? Application Area
1-aminoethanol doesn’t headline major commercial processes, but it occupies a critical spot as a research intermediate. Medicinal chemists look to it when assembling complex molecules, particularly small-molecule drugs and compounds modeling amino acid derivatives. Its structure offers a useful probe for studying reaction mechanisms and stereochemistry. Some teams investigating biodegradable polymers or bio-inspired scaffolds incorporate it as a bridge between the simple and the sophisticated. No, you won’t find it by the drum in a warehouse, but you will find it in the small vials of a synthetic chemistry lab, helping graduate students and researchers follow new hypotheses.
Pushing the Science: R&D Directions
Academic labs continue to look for smarter, greener ways to make and use 1-aminoethanol. Research groups examine its potential to serve as a platform for new catalysts, particularly chiral variants that could influence reaction stereochemistry. Projects looking for alternatives to fossil-derived chemicals take an interest, aiming to synthesize it from biorenewable feedstocks. Advances in analytical chemistry, such as high-resolution NMR and mass spectrometry, allow scientists to track every phase of the molecule’s reactivity and breakdown. These data-driven efforts drive more targeted, efficient applications, letting chemists fine-tune each step of the synthetic route. The trend in chemical research leans toward efficiency, reduced waste, and carbon-consciousness, and 1-aminoethanol offers a real proving ground for these principles.
Toxicity Research: What the Evidence Says
1-aminoethanol isn’t known for high toxicity, at least not on the scale of many other laboratory chemicals. That said, caution is always wise. Animal studies suggest mild irritation in concentrated forms, and nobody recommends large-scale exposure or ingestion. Chronic exposure data remains rare, which stands as both a data gap and a motivator for further toxicology programs. Lab teams take the sensible route—minimize contact and exposure any time you’re not sure of the long-term effects. Decision-makers in regulatory agencies lean on available data, but always push for deeper, longer-term studies before approving broader applications in food, drugs, or personal care products.
Glimpses Forward: Future Prospects
Chemists who work with amino alcohols see a slow but steady expansion in how and where 1-aminoethanol can serve science. Renewed interest in fine chemicals, especially those involved in pharmaceutical and green chemistry, keeps it relevant. As synthetic biology and enzyme engineering mature, this molecule might play a larger role in chemoenzymatic synthesis or novel polymer development. Researchers keep an eye out for new catalytic processes and greener preparation methods that could reduce cost and environmental impact. Its story isn’t static, and the next innovations will build off the hard-won advances accumulated through years of careful, systematic chemistry.
What is the chemical formula of 1-Aminoethanol?
The Building Blocks
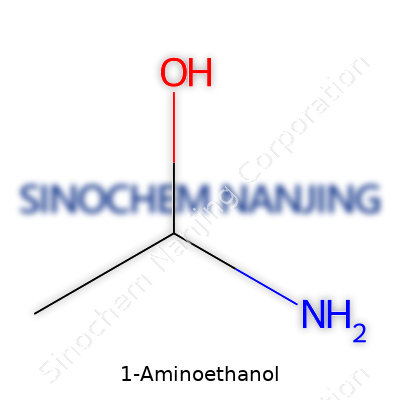
1-Aminoethanol isn’t a household name. It slips past conversations in most chemistry classrooms, but it has a simple story. This molecule comes with two important groups on its structure—a hydroxyl group (OH) and an amino group (NH2)—both attached to the same carbon backbone. Its chemical formula reads as C2H7NO, a tight cluster of two carbons, seven hydrogens, one nitrogen, and one oxygen atom.
You might expect something with “ethanol” in the name to bring to mind the alcohol found in disinfectants or drinks. The difference here comes from the “amino” group. Picture adding a dash of nitrogen to the party, making the molecule a bit more versatile. The subtle shift between a standard alcohol and an amino-alcohol opens doors in medicinal chemistry and laboratory work. This combination gives the structure some new chemical reactions and behaviors that straight-up ethanol can’t perform.
Why Care About 1-Aminoethanol?
I remember my first organic chemistry lab, struggling with naming conventions and molecular shapes. Spotting patterns helped bring clarity. Molecules like 1-aminoethanol matter not just for memorization, but for the way they expand the chemical ‘alphabet.’ Lab techs rely on these types of compounds when piecing together larger, complex drugs from more basic scaffoldings. 1-Aminoethanol serves as a building block in creating pharmaceutically active substances.
This molecule appears in intermediate stages of some industrial syntheses. For students grinding out reaction mechanisms, spotting a C2H7NO in the middle of a transformation often signals a key step in forming something more complicated—a bridge between the raw starting materials and clinically useful end products. Everyday folks might not see these transformations, but they play a role behind the scenes in the medicine cabinet or in cleansers that keep surfaces free from bacteria.
Supporting Better Chemistry in Practice
Understanding these formulas brings more than just exam points. Strong foundation in chemical notation, like what you get from simple structures such as C2H7NO, strengthens critical thinking. When someone learns about molecules that easily trade a hydrogen here or an oxygen there, troubleshooting in the lab goes smoother. There’s less wasted material, fewer hazardous byproducts, and a smaller environmental footprint. Fact: chemical industries worldwide produce millions of tons of specialty molecules a year, and one miscalculation can amplify waste streams.
Precision in studying this formula builds safer habits. I’ve watched a careless mistake turn a calm bench into a bubbling mess. Getting structures right—starting with something as (seemingly) modest as 1-aminoethanol—protects against hazardous surprises. Organizations like the American Chemical Society press for this exact kind of detail-mindedness, reminding us that accurate chemistry makes for safer, more effective products.
Forward Steps
Misinformation about chemical names or structures sometimes floats around online. Fact-checking has become more vital, especially for learners just stepping into the field. Reliable open-access databases like PubChem and textbooks reviewed by experts help keep the information straight and trustworthy for students, teachers, and professionals. Every time we double-check a formula, especially those that seem obscure, we strengthen the bonds—no pun intended—between science and public trust.
What are the main uses of 1-Aminoethanol?
Putting 1-Aminoethanol to Work in the Lab
I remember the first time I came across 1-Aminoethanol during a university practical—I was surprised how often this simple compound pops up once you know what to look for. Its structure might seem pretty basic, just an amino group hooked to an ethanol backbone, but don’t let appearances fool you. Under the hood, it offers a ton of flexibility in organic synthesis and industrial chemistry. Chemists appreciate its two reactive spots—the amino and hydroxyl groups—which makes it a valuable building block for combining with other chemicals to create new molecules. You’ll run into it mostly in labs as a starting point for crafting more complicated amines or for threading nitrogen into larger organic structures.
Intermediate in Pharmaceutical Manufacturing
Medicinal chemistry leans on compounds like 1-Aminoethanol for stepwise building of bigger, complex frameworks. I’ve seen it used as an intermediate, bridging raw materials and finished drugs. Because it carries both an alcohol and an amine, it can head off in different directions depending on what functional groups you need to bolt onto the drug candidate. You’ll see researchers testing out different substitutions using 1-Aminoethanol as a sort of scaffold, tweaking the molecule to improve how a drug interacts with the body or to dodge side effects. Having easy access to this compound can speed up the route from lab bench to clinical trials. The global pharmaceutical market continues to rise, and efficient routes to intermediates like this pay off both in time and cost. According to recent market analysis, the push for new medicines only underscores the importance of reliable chemical intermediates.
Improving Synthetic Methods in Organic Chemistry
Synthetic chemists take advantage of 1-Aminoethanol to introduce both nitrogen and oxygen into new compounds without juggling too many protections or extra steps. I’ve seen it appear in the literature as a chiral auxiliary—meaning chemists use its unique shape to bias reactions and create just the right mirror-image version of a compound. This matters for products like pharmaceuticals and agrochemicals, where the “wrong” isomer doesn’t just fail to work, it might actually cause harm. Directing chemical reactions using 1-Aminoethanol can cut down on waste and help reach the target molecule faster. The Green Chemistry trend places emphasis on routes that use fewer raw materials and produce less hazardous waste, and 1-Aminoethanol can contribute to that goal.
Moving Toward Safer and Greener Industrial Processes
You won’t find big bottles of this compound in every factory, since it’s mostly a niche raw material, but its ability to act as a chelating agent or stabilizer shows up in certain specialty sectors. For instance, it can bind with metal ions and help keep them in solution, playing a role in making catalysts more effective or extending the life of chemical baths used in plating and surface treatment. Companies want fewer emissions and hazardous byproducts in their processes, so safer alternatives matter. Better sourcing, monitoring, and closed-loop processes help minimize risks for workers and the surrounding community. Responsible handling and personal protective equipment (PPE) remain non-negotiable in workspaces using active chemicals like this.
Broadening Future Applications
Even as 1-Aminoethanol mostly serves as a behind-the-scenes player in research and industry, new uses could emerge as green manufacturing takes off and chemists hunt for more sustainable routes. Collaboration between academic researchers and industry leaders encourages responsible innovation—an approach backed by experience and substantial evidence. Regulatory oversight from agencies like the EPA and OSHA reinforces safer handling, disposal, and reporting, cementing trust from those of us who care about the impact of chemistry beyond the lab.
Is 1-Aminoethanol hazardous to health or the environment?
Digging into What 1-Aminoethanol Really Is
1-Aminoethanol isn’t a household name. You might stumble onto it listed in old chemistry textbooks or see it as a footnote in advanced organic synthesis articles. The molecule shares a family tree with better-known chemicals—take ethanol, or switch out a functional group, and you get compounds with very different properties. 1-Aminoethanol has the formula C2H7NO, with both an amino and a hydroxyl group, which means you’re dealing with something that can change behavior based on its environment.
Health Risks: Real and Hypothetical
Here’s the twist. You won’t find vast regulatory records on 1-aminoethanol. It pops up most as a transient intermediate in laboratories, not in bulk manufacturing or food processes, so the average person won’t cross paths with it. Toxicological databases don’t feature a flood of warnings about it. Still, chemical cousins—like ethanolamine or simple amines—are irritating to skin, eyes, and the respiratory tract if mishandled. If you spill 1-aminoethanol, expect similar consequences: burning, redness, coughing. Even without exhaustive testing data, chemists assume the molecule demands gloves, goggles, good ventilation, and respect.
I’ve handled plenty of small-molecule amines and alcohols over the years, and there’s a rule that keeps coming up: treat any unfamiliar compound with caution. The reason is simple: missing data on long-term effects. We all hope chemicals like this won’t linger in the bloodstream or trigger mutations, but nobody should gamble. Rather than ignoring that knowledge gap, responsible workspaces lean on established hazard protocols, even if databases stay thin.
Environmental Footprint: What Happens After Disposal?
Environmental impact depends on both chemical stability and use patterns. Unlike big-volume solvents or pesticides, 1-aminoethanol rarely enters rivers, soil, or air in large amounts. In a lab, workers dilute leftovers, treat them, and send them to hazardous waste handlers. Still, if this molecule slipped into groundwater in a big spill, the amine group carries a risk of raising the water’s pH or feeding nitrogen cycles. Amines and alcohols don’t hang around like heavy metals, but they can stress bacteria or aquatic life at high doses. Regulations in major countries insist on proper waste management, not just for legal reasons but because unpredictable chemistry can ruin more than an experiment.
Public records on industrial-scale leaks or contaminations involving 1-aminoethanol do not exist. That absence doesn’t mean safety; it points to the rarity of its use, at least outside niche research facilities. I’ve watched what happens when more common amines get loose—fish kills, weird algae blooms, and expensive clean-ups. It teaches a lesson: even a one-off compound deserves consideration.
Better Safe than Sorry: Solutions Going Forward
The world doesn’t always need another cautionary tale. Safe lab practices—closed systems, splash guards, ample ventilation—handle the basic threats. When students enter the lab for the first time, mentors talk through safety data sheets in plain language. They learn that even a friendly-looking structure like 1-aminoethanol can bite if you let your guard down. Disposal isn’t just about compliance. It’s about respect for the folks downstream—waste handlers, water treatment workers, communities living near disposal sites.
Safer substitutes play a big part in modern chemistry. If research demands a molecule similar to 1-aminoethanol, chemists look for alternatives with established safety records. Where that’s impossible, education becomes the shield. The labs I trust never shrug off small volumes or odd chemicals. It’s never worth the shortcut.
Wrap-Up Thoughts
No chemical stays harmless just because it’s obscure. 1-Aminoethanol sits in the shadows, but labs treat it with the same respect they show to better-known hazards. Any risk—no matter how rare—calls for attention, solid training, and a backup plan. That’s how you protect people and the planet, regardless of the compound’s fame.
How should 1-Aminoethanol be stored and handled safely?
Connecting with the Realities of 1-Aminoethanol
Working with chemicals takes more than just ticking boxes; it requires respect for what you’re handling. 1-Aminoethanol offers plenty of use in both industrial and research labs, but that doesn’t mean it can be treated carelessly. Anyone who’s been around chemical storage rooms knows that poor habits often lead to bigger problems down the line. Following science-backed guidelines isn’t optional—it’s about health, wellbeing, and everyone’s peace of mind.
Getting Storage Right Isn’t Rocket Science
Put 1-Aminoethanol in a cool, well-ventilated spot—straightforward and practical. Temperature swings only invite trouble. Many chemicals become unpredictable around heat sources or in direct sunlight; this one’s no different. Keep it away from oxidizers and acids. Stuff gets messy fast if incompatible materials end up side by side on storage shelves. This means labeling shelves, setting clear zones for different substances, and not rushing when unpacking shipments.
People sometimes get relaxed with “temporary” storage. That’s a fast route to confusion or even dangerous mistakes. Use containers designed for chemical safety. Glass with secure lids or safety-approved plastics avoid unnecessary risk. Leaks can do much more than damage labels—the wrong spill could cause a real emergency. If you store chemicals in glass, make sure it’s away from high-traffic areas. Besides, nobody wants a shelf of glass jars above a workspace.
Handling: Trust What’s Proven Safe, Not What’s Convenient
Growing up around home workshops and later working in labs, I’ve seen small shortcuts end badly. Gloves and goggles should come out every single time someone picks up a bottle of 1-Aminoethanol. Even if someone claims confidence from years of no accidents, memory fades and mistakes happen. The CDC and OSHA both recommend personal protective equipment—no one should skip it. Spills on the skin can cause irritation; eye exposure doesn’t bear thinking about.
Use in a fume hood beats working out in the open. Chemical vapors may not always seem strong, but exposure adds up. Many assume they’ll “just be quick,” but air movement in a hood takes extra risk out of the equation. Measuring and transferring are best done with pipettes or dispensers, not pouring from heavy bottles. Over time, the extra caution pays off, because there’s no second chance with chemical burns or lung irritation.
Keeping People and Environments Safer
It helps to train regularly. Even those who have handled chemicals for decades benefit from reminders. New staff should never go it alone on their first day with materials like this. I’ve found real value in walk-throughs and safety drills. It surprises some people how forgotten basic steps come flooding back in those settings.
Spill kits work best if everyone knows where they are and what’s inside. Label them clearly. Most kits use absorbents, pH neutralizers, and proper disposal bags—these should sit within arm’s reach of storage areas. In the rush to clean up, people sometimes forget that certain spills interact badly with water or standard cleaners. Sticking to industry guidance matters more than instinct.
Disposing of leftover or degraded 1-Aminoethanol calls for more than dumping it down the drain. Companies and universities now have hazmat contacts and safe transport agreements. Encouraging staff to speak up if they’re not certain about disposal helps the whole group. This chemical world keeps changing, so staying current on regulations matters just as much as old-fashioned care.
Moving Toward Smarter Habits
Smart storage and handling habits keep everyone safer—there’s no downside to thinking before doing. Bringing in outside audits and letting fresh eyes look at processes can highlight blind spots. I’ve seen first hand how a culture of care makes labs and workplaces not just safer, but more productive. In the long run, sticking to evidence-based best practices saves costs, prevents injuries, and makes sure that everyone heads home at the end of the day healthy.
What is the difference between 1-Aminoethanol and 2-Aminoethanol?
The Importance of Small Chemical Changes
In chemistry, even the shift of a group from one atom to another can lead to major changes in how a compound behaves. Looking at 1-aminoethanol and 2-aminoethanol is a good way to see this. Both have an amino group and a hydroxyl group stuck onto a two-carbon backbone, but the difference in their arrangement shapes more than just a name.
Structure Changes Everything
The structure of 1-aminoethanol puts the amino group and the alcohol on the same carbon. This means the formula looks like CH3CH(NH2)OH. 2-aminoethanol, on the other hand, has them on different carbons—HO-CH2-CH2-NH2, also known as ethanolamine. This little swap drives more than you might think.
Stability Makes a Big Difference
From my experience in the lab, 2-aminoethanol is the one you see everywhere—from gas treatment plants to pharmaceuticals or cosmetics. The reason comes down to stability. 2-aminoethanol doesn't fall apart easily, so it can handle storage and tough conditions. 1-aminoethanol, by contrast, tends to break down quickly, making it tough to find in any bottle labeled in a stockroom. If a chemical can't sit on a shelf for long without turning into something else, companies don’t want to use it. That explains why you probably only spot 2-aminoethanol in household products or industrial settings—never the 1-version.
Applications Go Where Stability Goes
Industries need materials they can trust to stay the same during transport and use. Because of that, 2-aminoethanol gets tapped for scrubbing acidic gases out of natural gas streams, boosting cleaning product formulas, and even stabilizing pharmaceuticals. It can act as a building block for everything from emulsifiers to drugs. 1-aminoethanol, lacking that same reliability, rarely shows up on ingredient lists. If a factory wants to invest in equipment and guard worker safety, running a process with a stable and well-understood molecule wins every time.
Safety is Tied to Structure
Handling any amino alcohol needs respect for safety, and ethanolamine—2-aminoethanol—has been studied for decades. People have sorted out exposure risks, and companies provide safety data sheets up front. For 1-aminoethanol, that lack of commercial use means its health effects are less mapped out, adding extra uncertainty. Regulatory agencies prefer dealing with risks they can predict, so familiar compounds with proven records get the green light more often.
Getting the Right Chemistry into Products
Choosing between similar molecules comes down to practical chemistry: stability, cost, and safety. If a chemical can't last or risks unpredictable reactions, it doesn’t fit the bill outside a carefully managed research setting. The path from academic curiosity to everyday utility is shaped by much more than a name. Small structural details force big decisions in chemistry labs and on production floors. Knowing the difference helps avoid costly mistakes and boosts trust in the safety and reliability of everyday products.
| Names | |
| Preferred IUPAC name | 2-Aminoethan-1-ol |
| Other names |
1-Hydroxyethanamine Ethylolamine Aminoacetol Ethylene amino alcohol |
| Pronunciation | /ˌwʌn.əˌmiː.nəʊˈɛθ.ə.nɒl/ |
| Identifiers | |
| CAS Number | 616-38-6 |
| 3D model (JSmol) | `load =C(CO)N` |
| Beilstein Reference | Beilstein Reference: 635639 |
| ChEBI | CHEBI:16044 |
| ChEMBL | CHEMBL25310 |
| ChemSpider | 68270 |
| DrugBank | DB04211 |
| ECHA InfoCard | The ECHA InfoCard of product '1-Aminoethanol' is: **"03bb7e18-0fec-4b84-bde7-2c573b6629ea"** |
| EC Number | 1.1.1.102 |
| Gmelin Reference | 26202 |
| KEGG | C02236 |
| MeSH | D016205 |
| PubChem CID | 16710 |
| RTECS number | KE3675000 |
| UNII | 40DXH31UR6 |
| UN number | UN2672 |
| Properties | |
| Chemical formula | C2H7NO |
| Molar mass | 61.08 g/mol |
| Appearance | Colorless liquid |
| Odor | amine-like |
| Density | 0.945 g/cm³ |
| Solubility in water | soluble |
| log P | -1.2 |
| Vapor pressure | 0.539 mmHg (at 25 °C) |
| Acidity (pKa) | 14.8 |
| Basicity (pKb) | The pKb of 1-Aminoethanol is 4.14 |
| Magnetic susceptibility (χ) | -51.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.441 |
| Dipole moment | 1.525 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 81.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -229.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -393.6 kJ/mol |
| Pharmacology | |
| ATC code | N06BX08 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H302, H319 |
| Precautionary statements | P261, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 50 °C |
| Autoignition temperature | 460 °C (860 °F; 733 K) |
| Explosive limits | Explosive limits: 2.6–12.4% (in air) |
| Lethal dose or concentration | LD50 (oral, rat): 1000 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral Rat 1000 mg/kg |
| NIOSH | KN7200000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 – 1 |
| Related compounds | |
| Related compounds |
Ethanol Ethylamine 2-Aminoethanol Methamphetamine |

