Unpacking 1,8-Epoxy-P-Menthane: History, Science, and the Road Ahead
The Backstory of 1,8-Epoxy-P-Menthane
1,8-Epoxy-P-Menthane, better known by chemists and industries as Eucalyptol or cineole, has a history that traces back centuries, mostly through its starring role in the aromatic oils of eucalyptus trees. Before the age of synthetic chemistry, people would crush eucalyptus leaves or boil them to pull out the rich, camphor-like vapors that carried this compound. Interest grew rapidly in the nineteenth century as colonial botanists documented its practical uses, especially in Australia where eucalyptus trees are common. The world quickly caught on. Scientists began extracting and purifying the compound, uncovering unique properties that set it apart from other plant oils and monoterpenes. This hands-on approach established a foundation for exploring both its practical applications and the science behind the molecule itself.
Product Overview and the Pulse of Industry
Today, 1,8-Epoxy-P-Menthane lives many lives. Some know it as a key component in cough drops and mouthwashes, others prize it in the formulation of cleaning agents, and still more recognize its role in flavorings and perfumes. With a distinct, refreshing scent and a cooling effect, it pulls double duty in both consumer and industrial sectors. Its ready blending with oils, alcohol, and many organic solvents turns it into a staple behind the scenes in everything from air fresheners to pharmaceuticals. The volume of annual production pales compared to petrochemicals, but its importance to health and wellness, hygiene, and olfactory products means a steady place on the global chemical stage.
Digging Into Its Physical and Chemical Side
Eucalyptol stands out as a clear liquid at room temperature. It brings a strong scent that strikes most as crisp and vaguely medicinal—a signature hinting at the familiar aroma of a vicks vapor rub. As a member of the cyclic ethers, its molecular formula is C10H18O, and it has a boiling point hovering just above 170°C. It doesn't mix with water, but dissolves readily in common solvents. Stability ranks high under most indoor conditions, though exposure to strong acids or sunlight nudges it toward slow breakdown or color change. This resilience—in tandem with volatility—drives its use in cough syrups and aromatherapy formulations.
Technical Specifications & What Really Matters in Labeling
Laws and safety standards turn the fine print on a container of 1,8-Epoxy-P-Menthane into something more meaningful than chemistry alone. Products with high cineole content must stick to minimum purity requirements, especially when intended for food or pharmaceutical use. Labels require accurate concentration, storage guidance, and hazard symbols in most regions. International organizations like the European Pharmacopeia and U.S. Food and Drug Administration keep close tabs on the details, demanding strict purity and contamination levels. All these requirements exist because manufacturers and end-users count on predictability and safety far more than marketing claims.
How It’s Made: From Tree to Test Tube
Extraction techniques have shifted over the years. The oldest methods involved steam distillation of eucalyptus leaves, coaxing out the vapors and cooling them into a separable liquid. Improved techniques, such as vacuum distillation and fractional crystallization, came online as demand for purity and higher yields increased. Some large-scale producers use molecular sieves or advanced chromatographic separation to further refine the product. While the bulk of global supply still originates from the direct processing of eucalyptus oil, research into selective synthetic approaches continues in labs. These routes use basic starting materials like alpha-pinene, applying catalytic oxidation to construct the epoxide ring with minimal byproducts.
Chemical Reactions & Modifications
The structure of eucalyptol offers several interesting avenues for reaction. Epoxides generally carry a strained ring, making them targets for ring-opening attacks by acids or nucleophiles. Under the right conditions, the compound reacts to form glycols, ethers, and alcohol derivatives suited to more specialized uses. That flexibility drives much of the research aimed at turning eucalyptol into new drug candidates or functional materials for everyday life. Some chemists explore ways to attach additional functional groups, hoping to improve solubility or alter scent profiles for cosmetic products. Each change relies on a steady hand and a clear understanding of both the chemistry at play and the needs of real-world applications.
Names in Circulation: Synonyms Past and Present
Over the years, markets and scientific circles have attached several synonyms to this molecule. Cineole, the alternate name, appears in pharmacopoeias and industry documents. Eucalyptol remains the public-facing label in everything from vapor rubs to air sanitizers. The substance sometimes turns up in older texts as cajeputol or 1,3,3-trimethyl-2-oxabicyclo[2.2.2]octane, a mouthful better left to academic papers. These overlapping names reflect its deep ties to plant oils and evolving global regulations.
Safety & Operational Standards: Lessons from Experience
Working with 1,8-Epoxy-P-Menthane teaches respect for potential hazards. The compound poses risks through inhalation, skin contact, or accidental ingestion. Handling guidelines call for gloves, goggles, and well-ventilated workspaces. Concentrated forms can irritate mucous membranes and eyes. Industry reports and case studies point to rare but serious effects from overexposure, especially in poorly ventilated settings. Regulatory agencies classified it as a substance with moderate acute toxicity. Long-term exposure data remains more limited, but available findings push for cautious handling and routine risk assessments. Safety lessons come sharply into focus in labs and production plants, where routine stretches can turn familiar tasks into dangerous situations if protective measures slide.
Application Area: More Than Meets the Nose
Eucalyptol shows much more range than the average consumer expects. Pharmaceutical companies draw on it for cough drops, inhalers, and ointments targeting blocked sinuses or inflamed airways. Fragrance houses fold it into colognes, perfumes, and fresheners to deliver a clean, sharp olfactory note. Food and beverage companies trickle in microdoses to boost certain flavor profiles, especially in chewing gum, candies, and some hot beverages. Household cleaning products rely on its ability to cut through grease, reduce odors, and impart a sense of freshness. Insect repellents—both natural and synthetic—take advantage of its volatility and effect on certain pests. Even the world of agriculture and veterinary medicine carves out uses for the compound, particularly in anti-parasitic formulations and feed additives. These examples drive home its cross-sector appeal—one molecule, many solutions.
Research & Development: Where Curiosity Meets Demand
Interest in eucalyptol’s science runs deeper than simple utility. Laboratories probe deeper into how eucalyptol interacts with biological systems, from combating bacteria and viruses to modulating body temperature or reducing inflammation. Clinical researchers look for new ways to harness its properties—think anti-inflammatory agents for chronic conditions, or adjunct therapies that complement established drugs. Chemists continue to tinker with derivatives, coaxing out promising features that might shift its use beyond the current boundaries. Environmental scientists study how eucalyptol released from forests impacts atmospheric chemistry, seed germination, or pest populations, opening unexpected doors in climate and ecological studies. This ongoing R&D helps ensure the compound remains in step with future needs and new regulatory hurdles alike.
Toxicity Research and Lessons Learned
Toxicology has never been a back-burner issue with eucalyptol, especially given its common use in over-the-counter medicinal products. Research shines light on both the risks and the safe dosages. Acute toxicity studies in rodents point to relatively low risk when used at recommended concentrations. Human reports, largely tied to excessive ingestion or misuse, warn against casual use of concentrated oils. There’s growing interest in the subtler risks—possible allergic reactions, metabolic effects, or interactions with other medications. Regulatory agencies set strict limits for these reasons, shaping not just the formulation of final products, but also the design of safety studies and post-market surveillance. While the reputation leans favorable, scientists caution that natural doesn’t always mean harmless. The lessons from animal and human studies underscore vigilance, robust labeling, and continued education for anyone who handles, packages, or uses these products.
The Road Ahead: Future Prospects in Chemistry and Commerce
Forecasting the future for 1,8-Epoxy-P-Menthane blends optimism with realism. The appetite for plant-based and sustainably sourced ingredients continues its steady rise. This trend points to more innovation in extraction, purification, and waste minimization. Consumer demand for evidence-based remedies carves out more space for clinical research on its therapeutic effects, especially as antimicrobial resistance, chronic airway disease, and environmental allergies put health systems under pressure. Green chemistry continues to push boundaries, seeking more energy-efficient or less polluting ways to generate not just eucalyptol, but also valuable derivatives. Regulatory scrutiny sharpens year by year, nudging the industry toward greater transparency and direct research collaborations. The compound’s enduring value, carved out through centuries of empirical use and modern science, promises a place in medicine cabinets and laboratories for years to come—so long as curiosity, caution, and creativity keep pace.
What is 1,8-Epoxy-P-Menthane used for?
Where You’ll Find 1,8-Epoxy-P-Menthane
Step into a household cleaning aisle. Many of those fresh citrus scents blasting from bottles trace back to 1,8-Epoxy-P-Menthane, better known as limonene oxide. It comes from natural sources like citrus peels, providing that sharp, clean aroma many people love. But beyond the scent, this compound plays a big part in modern manufacturing.
Cleaning Products and Deodorizers
For the average shopper, this ingredient pops up in surface cleaners, air fresheners, and degreasers. Its biodegradability and low toxicity separate it from harsh chemical alternatives. Companies use it not just for smell but also because it acts almost like a low-grade solvent, cutting through grease and leaving kitchens sparkling. Eco-friendly brands tout it, banking on buyers who are tired of choking on strong, synthetic fumes.
Paints, Adhesives, and Industrial Uses
Factories add 1,8-Epoxy-P-Menthane to paints and coatings, counting on its properties to help paint flow more smoothly or dry faster. It helps thin formulations, making products easier for pros and weekend warriors alike to apply. It doesn’t just mask odors, it actively breaks them down, so folks painting a nursery or family room don’t have to air out the space for days afterward. In adhesives, it sometimes acts as a safer alternative to standard solvents that carry health warnings.
Food Flavoring (and Some Safety Considerations)
Manufacturers sometimes slip 1,8-Epoxy-P-Menthane into food and drink as a flavoring. Think lemon candies, sodas, or baking extracts. The U.S. Food and Drug Administration lists it as “generally recognized as safe” for these applications. No one should think of it as a health booster, but regular exposure from food or household products hasn’t raised red flags for most people.
Personal Care and Cosmetics
This compound supports bath bombs, lotions, and soaps in two ways. It brings that signature citrus note and also works as a skin conditioner. Some products use it to help blend oils with water, giving creams or sprays a smoother feel. There’s a reason citrus-inspired body washes feel refreshing in the morning shower.
Environmental Impact and Sustainable Choices
For those weighing environmental impact, 1,8-Epoxy-P-Menthane stands out because it breaks down pretty easily in water and soil. Stories about pollution from cleaning products push more people toward plant-based solutions like this one. Large companies tout their use of it as a badge for sustainability.
Concerns and Looking Forward
Not everything is perfect. A small slice of people can have skin reactions to citrus-derived ingredients. Rules on labeling aren’t always clear, so sensitive shoppers need to read ingredient lists closely. Also, most of this ingredient comes from massive citrus farms, raising questions about pesticide use and working conditions for farm laborers. Tougher regulations and better transparency from manufacturers would help everyone make informed choices.
What’s Next?
Demand for greener, safer ingredients looks steady. Researchers keep looking for ways to pull out more useful compounds from food waste like orange peels, so 1,8-Epoxy-P-Menthane might soon become even more widely available at a lower cost. Whether in your kitchen cabinet or your tool shed, this little molecule shows up everywhere — often quietly making life cleaner and a bit more fragrant.
Is 1,8-Epoxy-P-Menthane safe for human contact?
1,8-Epoxy-P-Menthane: Everyday Uses and Exposure
Products you’d find around the house or at the supermarket often include 1,8-epoxy-p-menthane, better known as d-limonene oxide. Manufacturers like its fresh citrus smell and use it in cleaning products, air fresheners, cosmetics, and even some food flavorings. Touching those items, breathing the scent, or eating food flavored with it all mean most folks encounter this compound at least now and then.
Potential Risks and Real-World Effects
People want peace of mind about everyday chemical exposures. Safety data shows 1,8-epoxy-p-menthane breaks down pretty quickly in the environment, and does not build up in animal tissue. My own routine includes scrubbing counters with citrus-based cleaners and using lotions that list this ingredient. I've never faced a reaction personally, but the picture broadens beyond anecdotes.
Scientific studies reveal most people do not experience issues from small to moderate contact. The compound enjoys a GRAS (Generally Recognized As Safe) status for food flavoring by the FDA, which speaks to its history of safe use. Usually, irritation only shows up with higher concentrations or exposure over long periods. One study by the European Chemicals Agency flagged it as a mild skin and eye irritant, especially when someone handles pure or concentrated forms. Mild rashes or red eyes might occur, nothing like the harsh burns you risk from bleach or strong solvents.
A few folks who work with 1,8-epoxy-p-menthane often, like factory workers making cleaning products, can get allergic reactions over time. Symptoms mainly act up in those sensitive to citrus fragrances or essential oils already. Proper gloves and eye protection lower those risks. In home use, diluted products rarely cause trouble, though anyone with allergies should stay alert.
Weighing Benefits with Sensible Precaution
1,8-epoxy-p-menthane offers real advantages as an alternative to harsher chemicals. It gets oils and sticky messes off surfaces without the heavy chemical smell. Biodegradability means it won’t linger in landfills, unlike some older cleaning agents. Still, “natural” doesn’t guarantee zero risk, so clear labeling and easy access to ingredient lists help everyone decide what’s worth buying.
From a practical angle, the most important piece is how you use products containing d-limonene oxide. At home, stick with intended uses: don’t mix products or use them on skin unless the instructions say so. Wash your hands after heavy cleaning, and keep sprays off your face. If a rash develops, switching brands usually solves the issue.
Workplaces with high exposure should rotate jobs to limit direct contact and swap out gloves regularly. Companies ought to train staff on chemical safety and post reminders where everyone can see them. Regular medical checkups catch allergies before they turn chronic.
What Research Still Needs to Uncover
Scientists keep running tests to see exactly what happens after long-term, low-level exposure to many chemicals, including 1,8-epoxy-p-menthane. Insights matter for communities, especially those living near factories or waste sites. Modern studies mostly support the continued use of d-limonene oxide in everyday products—at the same time, new research could always lead to stricter guidelines if better evidence of harm emerges.
What has stood out from my personal experience and the bulk of the evidence—read product labels, keep chemicals away from kids, and look for transparent brands. Trust builds through companies sharing what’s inside the bottle, so shoppers can make informed calls about what’s safe for their families.
What are the storage requirements for 1,8-Epoxy-P-Menthane?
Why This Chemical Draws Attention
Most people know 1,8-Epoxy-P-Menthane as limonene oxide. You’ll find it in flavors, fragrances, and as an intermediate in manufacturing. With most chemicals, storage is often treated as an afterthought until something goes wrong. Limonene oxide, on the other hand, asks for a bit more care. It’s a colorless liquid that carries a strong scent but its real challenge hides in its reactivity and sensitivity to conditions.
Direct, Evidence-Backed Storage Rules
I’ve worked in labs on and off for years, and it’s common to see containers sweating on shelves in rooms where temperatures fluctuate too much. With limonene oxide, even small oversights can trigger problems. Heat speeds up its oxidation. Sunlight doesn’t play nicely either, sometimes spurring chemical changes that can degrade its quality or turn it into something more hazardous. So, the right move is to keep it cool and shaded. The National Institute for Occupational Safety and Health (NIOSH) recommends storing similar epoxides around room temperature, away from direct light.
Container choice impacts safety. Limonene oxide won’t stay stable in metal containers over long periods—corrosion or reactions with certain metals may lead to unpleasant surprises. Glass and high-quality plastic containers, tightly sealed, prevent air or moisture from creeping in. That air and moisture speed up degradation, so every time you open the container, you’re letting in things that shouldn’t be there.
Ventilation and Fire Prevention
Volatile organic compounds like this one evaporate fast. You’d think a screw cap provides all the protection needed but fumes can build up, especially during summer. I’ve walked into storerooms that smelled like oranges only to discover a leaky seal. Proper ventilation pulls those vapors away before they collect. Without good airflow, fire risk rises. The National Fire Protection Association (NFPA) tags limonene oxide as a flammable liquid. That means no open flames or sparks anywhere near storage areas. Static discharge sets off more fires in chemical storage than most care to admit, so antistatic flooring matters if you handle a lot.
Avoiding Mix-ups and Cross-Contamination
Storing chemicals side by side can seem tidy but limonene oxide doesn’t mix well with acids or strong oxidizers. Store it away from nitric acid, hydrogen peroxide, and similar agents to prevent dangerous chemical reactions. Labels must stay clear and legible. As a rule, I always double-check that dates and hazard warnings show on every bottle. Misread or faded labels have caused more than their share of accidents.
The Human Factor
Most storage accidents don’t come down to faulty equipment but people in a rush. Routine checks and training on material safety data sheets (MSDS) go a long way. In my experience, reviewing handling guidelines every quarter helps catch forgotten details. Even the cleanest storeroom gains a rogue bottle or sticky shelf without care. Hands-on checks and honest feedback between team members cut down on problems, and nothing beats reporting a concern as soon as you notice it.
Toward Better Safety
Regulations serve as a minimum standard. But in places committed to safety, people treat the storage of limonene oxide as more than rule following—they tie it into their work culture. Regular audits, temperature logs, and investment in proper containers all prevent waste and risk. The effort put in now means fewer headaches and a healthier environment, for both workers and the communities nearby.
What is the chemical composition of 1,8-Epoxy-P-Menthane?
A Look at the Compound
1,8-Epoxy-p-menthane pops up more often than most folks realize, particularly under a different name: 1,8-cineole or simply eucalyptol. This clear liquid usually shows up in essential oils, especially those from eucalyptus leaves. The chemical formula—C10H18O—shows a compound with ten carbon atoms, eighteen hydrogen atoms, and one oxygen atom, but these numbers only tell part of the story. Looking closely, scientists see a bicyclic ether with that distinct oxygen atom bridging carbon atoms one and eight, giving the molecule its “epoxy” character and its notable ring structure.
Why Structure Shapes Function
The arrangement makes a huge impact: 1,8-epoxy-p-menthane’s aroma stands out, crisp and camphor-like. Essential oil specialists and pharmacists gravitate to this compound because of that same ring layout. Something about the way the oxygen atom sits in the ring means the molecule resists breaking down under normal heating and light. So, you’ll often spot it in medicines, vapor rubs, mouthwashes, and even in food flavorings. The physical properties—like a boiling point around 176°C and limited solubility in water but strong solubility in alcohol or fats—come right from that chemical structure.
How This Matters for Daily Life and Health
The benefits aren’t just for the lab. Eucalyptol, as it’s called on most ingredient lists, shows real promise. Several studies published in journals like the Journal of Agricultural and Food Chemistry and Phytomedicine highlight its antimicrobial and anti-inflammatory actions. Doctors have looked into its use for airway diseases, like asthma and sinus infections, since it helps with mucus breakdown and has a soothing effect.
Walk through a pharmacy, pick up cough drops or cold rubs, and there’s a strong chance you’ll find 1,8-epoxy-p-menthane on the label. From personal experience fighting off winter colds, a blend with a dash of this compound seems to clear the nose and soothe the cough better than plain menthol alone. Families have used steam inhalations, topical salves, and teas brewed with eucalyptus for generations.
The Importance of Transparency and Safety
Not everything with a plant source comes risk-free. Research from the National Institutes of Health urges caution: 1,8-epoxy-p-menthane can trigger toxic reactions if swallowed in large amounts or used undiluted on the skin. Small children and pets sometimes face more risk. Knowing the chemical composition lets healthcare workers and end-users measure and control exposure properly. The presence of that single oxygen atom tells professionals what to expect during metabolism or breakdown in the environment, too.
Pathways to Safer Use
Experts recommend labeling dosage strength in over-the-counter products containing 1,8-epoxy-p-menthane. Education matters on all sides. Pharmacists, natural wellness stores, and even online ingredient suppliers should make chemical information front and center for customers. A step further, food producers align their flavoring choices with regulatory rules—authorities such as the FDA monitor concentration levels quite closely.
Scientific transparency means more than a passing glance at a label. Background knowledge about the actual atoms and bonds inside 1,8-epoxy-p-menthane drives safer, more effective choices. As research details continue to grow, a deeper understanding of what really sits inside common formulas continues to matter—at the lab bench and the home medicine cabinet alike.
How should 1,8-Epoxy-P-Menthane be handled in case of a spill?
What Happens If 1,8-Epoxy-P-Menthane Spills
In many workplaces, 1,8-Epoxy-P-Menthane—also called d-limonene oxide or limonene epoxide—pops up in cleaning solutions or as a flavor and fragrance ingredient. This clear, oily liquid can seem pretty harmless, especially since it has that strong citrus smell. Realistically, though, it acts as an irritant and can harm people’s lungs, eyes, or skin. Once this particular chemical leaks onto a floor, a bench, or flows toward a drain, a mess grows out of what started as a minor accident.
What Makes This Chemical a Concern
In my own years working around big drums of chemicals, strong cleaning agents, and paint solvents, the slickness of something like 1,8-Epoxy-P-Menthane on a tile floor causes alarms to go off. Not just for the slip hazard—the vapors quickly fill a room, especially if there isn’t enough ventilation. According to the National Institute for Occupational Safety and Health, skin contact with epoxides can spark allergic reactions or dermatitis. Inhaling enough vapor might give someone headaches, dizziness, or worse.
People often forget that a simple citrus scent can mask these real risks. Statistics published in the Journal of Occupational and Environmental Hygiene point out that accidental exposures to solvents rank as one of the most frequent reasons for chemical-related hospital visits in light industry. The real takeaway: just because something smells like an orange, nobody should treat it like orange juice.
What to Do Right After a Spill
Clear thinking saves the day. Spills don’t wait for shift supervisors or safety officers to show up, so workers nearby turn into first responders. The priority—get anyone who’s not part of the cleanup away from the area. Open windows or doors to let air move through. Some workplaces keep exhaust fans ready for emergencies just like this.
Handling the spill itself means plugging on gloves made of nitrile or butyl rubber, not thin latex. Eyes need protection—a full face shield or tight-fitting goggles stop splashes from finding their way in. If a lab coat is close, button up, since chemicals can soak through street clothing in minutes.
Containing and Cleaning the Chemical
Stopping the spread comes first. Spill-control pads or inert materials like sand or vermiculite create a dike. Kitty litter works in a pinch. Never let this chemical slide toward drains—1,8-Epoxy-P-Menthane breaks down slowly in wastewater and hurts aquatic organisms. Companies that focus on environmental health, like the European Chemicals Agency, call for immediate containment and separation from water sources.
Once contained, scoop up everything, including the material used for containment. Transfer waste into labeled buckets made of glass or compatible plastic. Tightly seal and store these containers in the designated area for hazardous chemicals. Nobody likes filling out disposal paperwork, but improper storage can lead to bigger environmental or financial headaches.
Prevention and Training Matter Most
One thing chemicals like this taught me: the time spent on safety training never gets wasted. Spills shrink in consequence when everyone on a shift already expects how to tackle them. Regular drills and reviews boost muscle memory. Employers who keep up-to-date safety data sheets in reach—for substitutes, too—give their teams the best shot at fast, safe responses. Generalizing, people tend to pay attention if shown real-life numbers and stories about previous accidents.
Companies should store 1,8-Epoxy-P-Menthane in tightly closed containers, away from heat sources and direct sunlight. Cabinets marked for solvents stay organized, with absorbent spill kits nearby where production lines work fastest. The future may bring safer alternatives, but right now, diligence and respect for chemicals remain the most reliable strategies at hand.
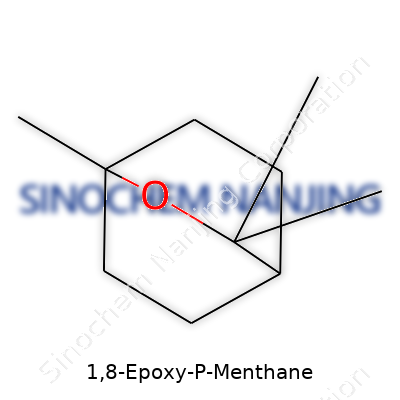
| Names | |
| Preferred IUPAC name | 1,3,4,7,7a-Hexahydro-4,7-methano-1H-inden |
| Other names |
1,8-Cineole Cineole Eucalyptol Cajeputol 1,3,3-Trimethyl-2-oxabicyclo[2.2.2]octane Eucalyptolum |
| Pronunciation | /ˈwʌn.eɪt.iˈpɒk.si pi ˈmɛn.θeɪn/ |
| Identifiers | |
| CAS Number | 470-82-6 |
| Beilstein Reference | 1033665 |
| ChEBI | CHEBI:25515 |
| ChEMBL | CHEMBL31700 |
| ChemSpider | 12047 |
| DrugBank | DB09534 |
| ECHA InfoCard | 03f7ccb1-6019-41b6-8e43-175faaa18d8e |
| EC Number | 208-912-8 |
| Gmelin Reference | 8227 |
| KEGG | C09822 |
| MeSH | D003077 |
| PubChem CID | 7216 |
| RTECS number | GO8925000 |
| UNII | K8RFC5P8T7 |
| UN number | UN2319 |
| Properties | |
| Chemical formula | C10H16O |
| Molar mass | 154.25 g/mol |
| Appearance | Colorless liquid |
| Odor | pleasant; characteristic; orange-like |
| Density | 0.922 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.7 |
| Vapor pressure | 0.32 mmHg (25°C) |
| Acidity (pKa) | 15.6 |
| Basicity (pKb) | 8.06 |
| Magnetic susceptibility (χ) | -70.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4630 |
| Viscosity | 0.921 mPa·s (25°C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 326.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -291.9 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -5896.3 kJ/mol |
| Pharmacology | |
| ATC code | R05CA02 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H317, H411 |
| Precautionary statements | P210, P261, P273, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1,2,0 |
| Flash point | > 110°C |
| Autoignition temperature | 237 °C |
| Explosive limits | '1.3% - 7%' |
| Lethal dose or concentration | LD50 oral rat 4000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 4000 mg/kg (oral, rat) |
| NIOSH | RG0875000 |
| PEL (Permissible) | PEL: 100 ppm (TWA) |
| REL (Recommended) | REL (Recommended Exposure Limit): 50 ppm (290 mg/m³) |
| IDLH (Immediate danger) | IDLH: 1,800 ppm |
| Related compounds | |
| Related compounds |
p-Cymene Terpinene Terpineol Menthol Eucalyptol (1,8-cineole) |

