1,8-Dinitronaphthalene: More Than Just a Chemical Curiosity
Historical Development
Chemical sciences move forward on the backs of molecules like 1,8-dinitronaphthalene—compounds that slip past popular notice but spark real interest behind the lab bench. Back in the late 19th and early 20th centuries, as the hunt for energetic materials heated up, organic chemists eyed the naphthalene ring not just for its stability but for its track record as a starting point in dyestuffs, explosives, and intermediates. Nitration of naphthalene gave birth to a whole family of dinitro derivatives, but 1,8-dinitronaphthalene emerged for its unique structure, which seemed to defy simple substitution patterns. Early research linked it to efforts in explosive synthesis and later to the formation of specialty intermediates that powered industries from pigment manufacturing to agrochemicals. It stood as an example of how persistent experimentation trumps luck, and how knowledge builds layer by layer, through both failed attempts and occasional breakthroughs.
Product Overview
The physical presence of 1,8-dinitronaphthalene—typically a pale yellow solid—doesn’t make it stand out from most other aromatic nitro compounds. It’s easy to overlook at a cursory glance. Yet, its arrangement of nitro groups on the naphthalene ring gives it unique reactivity, especially in terms of downstream modifications. Chemists track its purity closely, since small amounts of mono- or other dinitro isomers can change the outcome of later reactions, especially when used as an intermediate in high-value synthesis. This compound can be both a goal for method development (especially cleaner or safer nitration processes) and a rung on the ladder toward more complex targets.
Physical & Chemical Properties
The fascination with 1,8-dinitronaphthalene often comes down to the practical side of chemical handling. Its melting point tends to land in the range of 189 to 192°C. Most folks will note it doesn’t dissolve easily in water, but it finds a niche solubility in hot organic solvents such as acetone, benzene, and ethanol—a fact that’s made it especially useful for stepwise purification after synthesis. The presence of two nitro groups makes it far more electron-poor than the parent naphthalene, a trait that pushes it toward certain types of reactions (think nucleophilic aromatic substitution). The positional isomerism brings another twist, since the 1,8-arrangement builds in some key steric strain that’s harnessed in specific transformations, especially when developing more exotic nitroaromatic scaffolds.
Technical Specifications & Labeling
Quality standards tend to revolve around the percentage purity (often greater than 98 percent for any serious research or industrial work) and care in packaging, since the powder can cake if handled poorly. Its yellow color makes it easy to distinguish visually from some of its closely related relatives, which gives a simple but effective check on contamination. Most chemists working with it lean on established characterization markers—melting point, infrared and NMR spectra, and sometimes mass spectrometry—to rule out the possibility of unwanted isomers or incomplete nitration. Shipping and storage—especially at the industrial scale—need to respect its sensitivity to shock or friction, due to its relationship to energetics, even though it’s not nearly as hazardous as some related nitroaromatics.
Preparation Method
The journey to obtain 1,8-dinitronaphthalene usually starts with careful, slow nitration of naphthalene using sulfuric and nitric acids. Getting the dinitro groups into those stubborn positions on the ring isn’t a trivial exercise, since naphthalene prefers to send new groups to other positions. Success here comes from controlling temperature, nitric acid concentration, and timing. Often, the crude product requires repeated crystallization to isolate the 1,8-isomer from others, especially the 1,5-variety. With increasing calls for safer and greener chemistry, some labs aim to swap concentrated acids with milder conditions or solid acid catalysts, though scaling up such methods can bring its own headaches. For the working chemist, patience is the watchword—rushed reactions tend to pile on impurities that haunt later steps.
Chemical Reactions & Modifications
With two electron-withdrawing nitro groups, the molecule opens doors for many transformations. In organic synthesis, reducing only one nitro group sets up interesting paths toward amines or azo compounds. Both groups can also take part in cyclization reactions, and the ring often undergoes nucleophilic aromatic substitution, especially at the positions flanked by the nitro groups. My own time spent in a synthesis lab showed how small changes—solvent swap, slightly different temperature ramp—shift the balance between multiple possible products. The beauty of 1,8-dinitronaphthalene lies in its ability to act either as a synthetic dead-end or, with a clever idea, as a springboard to new chemistries.
Synonyms & Product Names
Within scientific literature and commercial catalogs, you’ll see 1,8-dinitronaphthalene hiding under a small set of synonyms: naphthalene-1,8-dinitro, 1,8-dinitro-naphthalene, or 1,8-DNN. Unlike more common industrial chemicals, it doesn’t usually parade under a sea of trade names. Most references stick to IUPAC or slightly abbreviated variants, partly because it doesn’t see broad use outside specialty or research-driven sectors—an honest reflection of its niche status.
Safety & Operational Standards
Anyone handling 1,8-dinitronaphthalene in a practical setting respects its hazardous reputation. Nitroaromatic compounds generally raise flags for their toxicity and environmental persistence, and this compound doesn’t buck that trend. Precautions start with personal protective equipment: gloves, safety goggles, and lab coats form the baseline. Avoiding contact with skin or inhalation of dust reduces risk; the compound can irritate the skin or mucous membranes on exposure. At a larger scale, explosion risks drop compared to more volatile nitrate esters, but mishandling in the presence of reducing agents or intense friction can cause issues. Waste material demands careful collection, as nitroaromatics resist easy breakdown and can persist in soils and groundwater. Many institutions outline clear standard operating procedures and enforce them during practical lab work, underscoring the real-world necessity for vigilance.
Application Area
Despite its specialty character, 1,8-dinitronaphthalene threads its way into several key application areas. In the pigment industry, it becomes a stepping stone to more complex dyes and pigments, contributing to hues that require high thermal stability and lightfastness. It also finds use in the synthesis of pyrotechnic substances and, in earlier eras, as an intermediate in some high-energy materials. More recently, chemists exploring new functional materials lean on its reactivity for developing advanced polymers, specialized intermediates, and as a scaffold in medicinal chemistry experiments where precise control over substitution patterns carries real value. Though you won’t find it gracing headlines, researchers know it as a silent workhorse.
Research & Development
R&D efforts surrounding 1,8-dinitronaphthalene rarely make the front pages of chemistry journals, yet its story inside the laboratory is full of quiet innovation. The modern push to reduce hazardous waste puts pressure on traditional acid-based nitration methods, spurring new research into alternative synthetic routes—sometimes involving phase-transfer catalysts, ionic liquids, or even enzymatic methods. Analytical chemistry teams tune methods for purifying and characterizing the compound at ever-decreasing quantities, responding to the need for trace-level detection in both environmental and quality control contexts. New techniques for directed substitution have expanded how chemists think about the naphthalene core, leading to new drugs and materials built on foundations first laid over a century ago. It’s a testament to how so-called “old” chemistry powers today’s cutting-edge projects.
Toxicity Research
Toxicologists raise measured concern about exposure to 1,8-dinitronaphthalene. Like many nitroaromatics, it carries risks if inhaled, ingested, or absorbed through the skin. Animal studies often reveal symptoms from methemoglobinemia—compromising the blood’s oxygen-carrying capacity—to liver and kidney effects at higher doses. Chronic exposure potentially leads to more serious outcomes, including neurological and reproductive issues, though the data pool remains limited due to low-volume use outside pharmaceutical or specialty chemical sectors. Regulations grow stricter as understanding of nitroaromatic toxicity deepens. The focus on occupational health translates into tight permissible exposure limits, rigorous air monitoring, and an ongoing search for safer alternatives or exposure minimization through engineering controls.
Future Prospects
Looking ahead, the future of 1,8-dinitronaphthalene may not bring blockbuster innovation, but it offers room for improvement in safety, environmental impact, and synthetic efficiency. Chemists who value sustainability will likely invest more effort into non-acidic nitration routes and improved waste management. Its utility as a building block in emerging materials science will keep it relevant for those chasing new dyes, advanced polymers, and molecular electronics. Toxicologists and industrial hygienists will keep pressure on introducing better controls and perhaps even phasing out applications where greener substitutes exist. As much as old molecules often seem consigned to the sidelines, the persistent push for rethinking production and application creates a quiet but valued role for 1,8-dinitronaphthalene in the next chapter of chemical science.
What is 1,8-Dinitronaphthalene used for?
Finding Real Value in Industrial Chemistry
Plenty of chemicals don’t make headlines, but their impact runs deep. 1,8-Dinitronaphthalene lands right in that group. In the broader map of specialty chemicals, its story starts with explosives and ripples out into a web of research and manufacturing roles. Many search for safe, effective methods of synthesizing components for explosives. Here, 1,8-Dinitronaphthalene stands out thanks to its specific structure—a pair of nitro groups fixed to a naphthalene ring at the 1 and 8 positions. This arrangement isn’t random. It sets the stage for building larger, more complex molecules.
From Explosives to Specialty Applications
I learned pretty fast in the lab that every molecule has a personality. 1,8-Dinitronaphthalene feels like the tough, quiet workhorse. It gets called up as a key raw material in producing 1,8-diaminonaphthalene, which sits at the core of some high-energy explosives—especially naphthalene-based ones. These explosives offer advantages in controlled demolition, defense, and mining. Companies look to chemicals like this for stability and reliability, not just power. Safety becomes a deal-breaker in this business. Every step from synthesis to transport demands sharp attention and real accountability.
The importance does not end at blowing things up. In specialty dye production, 1,8-dinitronaphthalene provides a precursor for pigments. As color chemistry moves toward greener, more lasting coloring agents, it gives pigment makers a solid starting block. Research into new dyes for textiles, printing, and electronics often circles back to these reliable intermediates.
Challenges in Handling and Regulation
I have seen how chemical plants must walk a tightrope between productivity and regulations. 1,8-Dinitronaphthalene shares a similar risk profile with other nitroaromatics. Its handling catches the attention of safety engineers and environmental watchdogs. One spill, one missed control measure, and the cost balloons in fines and remediation. Manufacturing and distributing such powerful molecules forces companies to rethink storage, training, and monitoring.
Laws like the REACH regulations in Europe and federal oversight in the US keep pressure on manufacturers. These rules do not just make paperwork piles—they steer companies to invest in safer processes and push for data-backed risk assessments. There’s a hard truth here: chemicals with explosive potential need clear-cut procedures and transparent tracking from the moment they leave the reactor to final use.
Pushing for Safer and Greener Chemistry
Sustainability feels more than a catchphrase when dealing with risky intermediates. Researchers and process engineers keep searching for routes to make and handle compounds like 1,8-dinitronaphthalene with less waste and lower carbon footprints. Developing better catalysts or milder reaction conditions isn’t just good PR—it saves lives and resources.
Real change will come when feedback from workers, communities, and regulators fuels smarter plant design and honest reporting. Smart sensors, better training, and upgraded emergency systems build a culture where safety tops speed or profit. I have watched innovation turn once-dangerous steps into routine work through automation and solid lab data.
Considering the Bigger Picture
It often takes small, under-the-radar molecules to deliver big industry advances. 1,8-Dinitronaphthalene shapes key parts of modern chemistry—fuel for explosives, color for products, and a yardstick for safety benchmarks. Firms that thrive make safety, research, and open communication their everyday tools. That’s how the industry keeps moving forward, one specialized molecule at a time.
Is 1,8-Dinitronaphthalene hazardous to health?
What’s Lurking in Industrial Chemistry
Most folks have never heard of 1,8-dinitronaphthalene unless they’re deep in the world of industrial chemistry. I’ve spent years writing and researching about workplace hazards, and this one keeps coming up in discussions with engineers and safety experts. Used as a chemical intermediate, 1,8-dinitronaphthalene appears in the manufacture of dyes, explosives, and sometimes in specialty chemical synthesis. Its potential dangers don’t jump out at first glance, but a closer look reveals clear risks that shouldn’t get brushed aside.
How Does 1,8-Dinitronaphthalene Affect People?
Stories from plant workers have put a face on these dangers for me more than any safety datasheet ever could. Direct skin contact sometimes means irritation, redness, or allergic responses. Breathing in its dust or fumes may lead to symptoms like coughing, headaches, or dizziness. Long-term exposure raises even bigger concerns. Many compounds in the nitroaromatic family have produced evidence of liver and blood toxicity during animal testing. Though there’s no mountain of long-term studies on 1,8-dinitronaphthalene itself, the chemical structure and logic from similar compounds suggest that chronic exposure carries a real threat.
It’s not just production workers facing these risks. I’ve seen accident reports describing improper waste handling or equipment leaks that brought office staff or cleaning crews into the danger zone. The scary thing is, symptoms can be slow to show up. By the time fatigue or anemia enters the picture, the chemical may have already quietly damaged red blood cells or organs.
Taking Responsibility in the Workplace
Talking to industrial hygienists, you quickly learn that personal protective equipment (PPE) only works as well as the training behind it. Gloves and respirators look great on paper, but the best protection comes from managing exposure at its source. Proper ventilation, sealed containers, and splash-proof equipment often keep airborne particles or direct contact in check. Routine air and surface monitoring have helped many companies find leaks before they cause harm.
Communicating these risks remains a constant challenge. In meetings, managers sometimes underestimate the importance of clear labels or safety briefings, especially during shift changes or when contractors visit. Actually, strong incident reporting and a willingness to listen to those who handle these chemicals daily keep everyone safer.
Solutions: Moving Beyond Band-Aids
Engineers and safety pros have worked together to replace hazardous chemicals with safer alternatives wherever possible. Substitution isn’t always easy—dyes and explosives have specialized requirements—but by now, green chemistry offers more options than ever. Companies investing in research sometimes manage to produce the same results with reduced toxicity.
Proper disposal closes the loop. Environmental damage from poor chemical disposal almost always circles back to human health, especially in communities near manufacturing sites. I’ve seen grassroots advocacy tip the scales more than once, leading to tighter oversight and better cleanup efforts.
The Takeaway from Experience
Risk rarely disappears just because a chemical isn’t household-famous. 1,8-dinitronaphthalene demands respect and close supervision. Learned lessons from other hazardous substances should guide how we handle not just the known threats, but the lesser-known ones with similar profiles. Whether you’re on the plant floor, managing a supply chain, or advocating for workers’ rights, the same principle applies: knowledge and vigilance save lives.
What is the chemical formula and CAS number of 1,8-Dinitronaphthalene?
Chemical Identity: The Facts
1,8-Dinitronaphthalene stands out to chemists by its distinct chemical formula: C10H6N2O4. Not everyone on the street walks around knowing a compound’s molecular makeup, but few can argue about the value of precision in science. In research, accuracy makes all the difference. This chemical goes by its unique CAS number 602-35-1. For scientists, that number isn’t just a catalog entry — it’s the fingerprint for tracing published research, sourcing authentic materials, and steering clear of mix-ups with other dinitronaphthalene isomers.
Why Do These Numbers Matter?
Chemical formulas may look cold and clinical, but they pack real meaning. C10H6N2O4 tells anyone familiar enough that this molecule holds two nitro groups attached to a naphthalene backbone. That arrangement cues certain chemical properties: sensitivity, color, possible health risks. The CAS number puts a precise label on it across languages and borders. I’ve seen lab teams lose hours (and money) from ordering the wrong variant by accident—and all because they skimmed on database checks.
Health, Safety, and Environmental Impact
Nitroaromatic compounds like 1,8-dinitronaphthalene do not belong on a kitchen shelf. In a laboratory, researchers stay aware of toxic effects. Long-term exposure can harm human health. Industrial releases can linger in soil and water, affecting more than just lab techs. The U.S. Environmental Protection Agency and similar organizations worldwide link CAS numbers to public safety data. Waste management, transport, and labeling rules lean on this system, so the right chemical never slips through the cracks.
Not everyone views chemicals with suspicion, but transparency and access to safety data prevent accidents. Over the years, I’ve noticed the gap narrows between researchers, regulatory agencies, and curious citizens. When students or officials ask about chemical safety, they don’t need jargon—they need facts locked to a verified identity. Sharing the CAS number directs them to reliable sources, published research, and government hazard statements.
Uses and Challenges
Engineers and scientists value 1,8-dinitronaphthalene mainly for its role in organic synthesis. Some research groups tap it while seeking novel drugs, dyes, or other specialty chemicals. The challenges keep changing, though. Sourcing pure material isn’t the end of the story—processing it safely, controlling emissions, and minimizing waste are growing priorities. Trade journals and patent offices check formulas and CAS numbers to avoid misidentifying ingredients. Seen from the ground up, mix-ups cost both credibility and cash.
So how do chemical companies and researchers stay ahead? Modern labs lean on electronic inventory systems. Regulatory compliance officers train staff to check chemical registry numbers before any experiment. Automation helps flag duplicates or expired stock. I remember one small lab that used color-coded tape for chemical bottles, but relying on digital records (linked to CAS) has rescued more than a few projects from unwanted surprises.
Building a Safe, Informed Future
Committing to clear labeling, safety, and education keeps workplaces and communities safer. Everyone from the warehouse worker handling shipments to the PhD synthesizing new compounds benefits from cross-checking CAS numbers before opening a bottle. By treating formulas and chemical identity as fundamental, not just trivia, researchers and companies show respect for health, science, and the public interest. The system may seem simple, but it’s a linchpin in chemistry’s push toward safer, smarter research and manufacturing.
How should 1,8-Dinitronaphthalene be stored?
Understanding the Risks
1,8-Dinitronaphthalene has earned a place on safety data sheets for good reason. It’s a solid chemical with explosive and toxic qualities. People working in labs or storage facilities have learned to take these warnings seriously—not because regulations say so, but because they’ve seen accidents happen when corners get cut.
In my early days in a chemical warehouse, I witnessed a minor fire caused by a mislabeled oxidizer. That ten minutes taught everyone at the facility how fast things escalate when proper storage isn’t enforced.
Temperature Control Matters
Storing 1,8-Dinitronaphthalene at the right temperature makes a huge difference. Heat starts chemical reactions that might not seem urgent until it’s too late. Keeping it in a cool, dry place away from sunlight has always been the gold standard. This doesn’t mean the nearest refrigerator will do. Dedicated temperature-controlled units, registered and checked, give an added layer of protection. If ambient temperatures climb above 25°C, risks multiply. Even small rises can push some sensitive materials toward unwanted reactions.
Keep Moisture and Sparks Away
Humidity pulls moisture into the container, changing the chemical’s properties over time. Metal shelving needs protection against rust, and floor drains should channel accidental leaks straight to containment. The best storage offers good ventilation, with units away from open flames and sources of static discharge.
Many companies use explosion-proof electrical systems near sensitive storage. At one facility, a humid and crowded storage area caused a mislabeled batch to degrade, costing thousands and risking even more. Dry, spark-free zones help keep disasters at bay.
Segregation Saves Lives
Mixing incompatible chemicals can be disastrous. I’ve seen colleagues get sick from fumes formed when containers failed and nitro compounds mixed with acids. Storing 1,8-Dinitronaphthalene far from acids, alkalis, strong oxidizers, and combustible substances makes sense—no cross-contamination, no chance of surprise reactions. Clearly marked shelves, color-coded bins, and regular training have become common in responsible workplaces.
Proper Containers Prevent Leaks
Containers with tight seals provide peace of mind. Glass works well for small quantities, while industrial stocks go in chemical-resistant, sealed drums. No single method offers perfect safety, so secondary containment barriers, like spill pallets, have become essential. Weekly checks help spot weak seals or slow leaks. During audits, inspectors want to see lids secure and containers free from corrosion or buildup.
Training and Documentation
Newcomers to chemical storage may learn about 1,8-Dinitronaphthalene from binders and safety sheets, but hands-on experience guides real changes. Companies invest time in fire drills, spill-response practice, and questioning every storage decision. Safety Data Sheets stay posted near storage rooms, and digital inventory logs flag expiration dates.
Some countries require companies to track hazardous chemicals from purchase to disposal. Responsible operators follow these laws, but those who know the risks firsthand have little trouble seeing their value.
Finding Better Solutions
In the long run, chemical suppliers aim for safer alternatives or packaging that reduces risk. Until then, real safety comes from diligence. Routine checks, honest reporting, and respect for every chemical’s potential hazard keep people and communities safe.
What precautions should be taken when handling 1,8-Dinitronaphthalene?
Respecting the Hazards
1,8-Dinitronaphthalene falls into a category of chemicals that demand more than a quick glance at a safety sheet. With a background in industrial chemistry, I’ve learned not to underestimate substances that carry the “nitro” group. This compound picks up its explosive reputation from the same stuff that makes TNT so dangerous, and even a small misstep can turn a quiet workbench into a chaotic scene. Proper handling isn’t about being dramatic—it’s critical for personal safety.
Personal Protective Equipment Comes First
PPE talks sometimes get brushed aside, but there’s no chance I’d ever touch 1,8-dinitronaphthalene without a decent lab coat, chemical splash goggles, and sturdy gloves made from nitrile. Standard latex gloves just don’t cut it. Even the smallest skin contact can mean trouble, from irritation to far worse if absorbed. Respiratory protection stays crucial, especially during weighing and mixing—dust and vapors have a knack for traveling where you least expect.
Keep Away From Heat And Sparks
This molecule brings sensitivity to heat and shock. I’ve heard quietly whispered stories of small accidents—nothing dramatic, but enough to send nervous glances around the lab. All work stays far from open flames. Hotplates and even the static from certain surfaces can become ignition sources. Having a grounded, non-sparking workspace might feel excessive, but it stops a crisis before it starts. Static control mats and anti-static coats go a long way to prevent unpleasant surprises.
Plan For Ventilation
I can’t count how many times proper airflow stopped a problem before it built up. Some forget that fumes aren’t always obvious—you won’t see danger coming. Fume hoods should always be running when handling nitro compounds. Negative pressure prevents any vapors from wafting around the room. Keeping doors closed and limiting foot traffic in and out of the workspace keeps the zone safer for everyone nearby.
Understand Storage Risks
Storing this stuff means choosing a spot that rules out sunlight, moisture, or temperature swings. Metal cabinets lined with secondary containment trays work well, and every container gets a clear, accurate label. Some labs keep small, clearly logged inventories, partly because larger amounts just aren't worth the risk. Monthly checks help spot any crusting around lids or discolored powder—both are early warning signs that call for careful and immediate disposal following hazardous waste protocols.
Solid Incident Planning
Training can feel dry, but it saves lives when things go sideways. Every person in the lab should know where the eyewash and emergency shower stations are. Spill kits get stocked with materials suited for energetic compounds, not just solvents. Detailed SOPs for small and large spills help everyone know exactly what steps to follow—panic fades when a checklist is right there on the wall. Emergency numbers need to be big and bold, not tucked away in a binder.
Knowledge Beats Complacency
Trying to save time or supplies backfires quickly with chemicals like this. I always push for refreshers on handling energetic materials—people forget, especially if they don’t see problems often. Building a culture where everyone double-checks each other and isn’t afraid to ask for help has kept my teams accident-free. Mistakes come when someone feels rushed or think caution only applies to bigger quantities. If one person speaks up, it raises the bar for everyone else.
Hurried Work Isn’t Worth the Cost
1,8-Dinitronaphthalene doesn’t give second chances. Good habits—PPE, ventilation, smart storage—turn a hazardous task into manageable routine. A bit more time spent each day on safety means never seeing a news headline with your lab’s name in it. Staying grounded in facts and treating safety as a shared responsibility keeps both people and science moving forward.
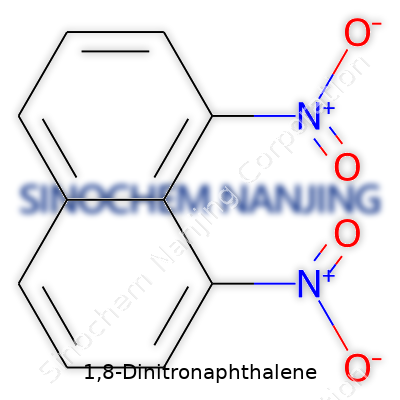
| Names | |
| Preferred IUPAC name | 1,8-dinitronaphthalene |
| Other names |
Naphthalene, 1,8-dinitro- 1,8-Dinitro-naphthalin 1,8-Dinitronaphthalin |
| Pronunciation | /ˈwʌn.eɪt daɪˌnaɪtroʊ næfˈθæliːn/ |
| Identifiers | |
| CAS Number | 602-28-4 |
| Beilstein Reference | 120776 |
| ChEBI | CHEBI:76905 |
| ChEMBL | CHEMBL125164 |
| ChemSpider | 154842 |
| DrugBank | DB07815 |
| ECHA InfoCard | 03be6c34-ea56-4866-bc6d-9e63717c4c77 |
| EC Number | 208-550-1 |
| Gmelin Reference | 774 |
| KEGG | C19429 |
| MeSH | D017929 |
| PubChem CID | 10405 |
| RTECS number | QJ9625000 |
| UNII | R9B6YQ263J |
| UN number | UN1321 |
| Properties | |
| Chemical formula | C10H6N2O4 |
| Molar mass | 258.17 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 1.57 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.85 |
| Vapor pressure | 1 mmHg (188 °C) |
| Acidity (pKa) | 13.10 |
| Basicity (pKb) | 13.60 |
| Magnetic susceptibility (χ) | -90.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.653 |
| Viscosity | 1.34 cP (131°C) |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 226.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3237.6 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06, GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 154 °C |
| Autoignition temperature | 360 °C |
| Lethal dose or concentration | LD50 oral rat 1560 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2830 mg/kg (rat, oral) |
| NIOSH | DN2625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 1.5 mg/m3 |
| Related compounds | |
| Related compounds |
1,5-Dinitronaphthalene 1,6-Dinitronaphthalene 2,6-Dinitronaphthalene |

