Poking Into 1,5-Dinitronaphthalene: Origins, Value, and Where It Might Go
Historical Development
People have a knack for stretching the potential of molecules. That story holds for 1,5-Dinitronaphthalene. Its roots go back to the mid-19th century, around the same time chemists made headway with aromatic nitro compounds. The world watched chemists transform coal tar into fundamental building blocks. Naphthalene became a workhorse for more complicated aromatic chemistry, picking up importance as researchers learned that swapping hydrogen atoms for nitro groups could offer new reactivity. The nitro-naphthalenes—especially the 1,5 isomer—pulled special attention for reasons that go beyond typical curiosity. People saw these were not just intermediates but stepping stones for bigger discoveries, paving the way for advances in dyes, explosives, and organic synthesis. As time moved forward, commercial interests and safety regulations shaped how labs and industries managed the compound.
Product Overview
Curious minds ask what makes 1,5-Dinitronaphthalene stick out. The answer lies partly in its unique structure: two nitro groups spaced on the naphthalene ring at the 1 and 5 positions. That placement tweaks its chemical behavior, making it distinct from other dinitro isomers. Industries don't grab this chemical for its showy appearance but because of what it can do for synthesis, particularly for crafting specialty dyes and advanced organic compounds. Academic and industrial chemists treat it as a trusty intermediate, giving them a direct path to more complex targets or specialized reagents.
Physical & Chemical Properties
Most folks working in chemical labs recognize the yellowish crystals and solid form of 1,5-Dinitronaphthalene. It tends to show up as a stable solid at room temperature, not prone to evaporate or decompose unless someone turns up the heat or exposes it to strong bases. Its molecular weight clocks in around 218.13 g/mol, and even though it doesn't melt at everyday temperatures, pushing it toward 180–185°C unlocks that transition. Unlike some energetic materials out there, this chemical doesn't explode when nudged, but mishandling can still lead to trouble. Its poor solubility in water means cleanup accidents can get persistent, while organic solvents handle it more gently, dissolving it for further transformations.
Technical Specifications & Labeling
People labeling or shipping 1,5-Dinitronaphthalene have rules to follow. Packing it means clear indication of its chemical identity, purity, and batch history because regulators pay tight attention to nitro compounds. Purity speaks volumes—trace impurities might change its usefulness or introduce hazards. Chemists, in my experience, go after high-purity batches to keep their processes reliable.
Preparation Method
Synthesis isn't a random toss-up. Most of the world's supply comes from controlled nitration of naphthalene using concentrated nitric and sulfuric acids. Targeted temperature control and careful drip feed of reagents reduce the chance of unwanted isomers or side products. Success really leans on achieving selective dinitration: too much heat or acid can send things in the wrong direction, birthing various isomers instead of the prized 1,5 variant. Those missteps sound small, but they swell up the cost of separation and purification.
Chemical Reactions & Modifications
Few who have worked with it forget 1,5-Dinitronaphthalene’s knack for reactivity. Those nitro groups can be swapped, reduced, or replaced in a variety of chemical transformations. People chasing diamines love this starting point, since hydrogenation in the right setup turns both nitro groups into amines, yielding 1,5-diaminonaphthalene. That step powers up later syntheses for dyes and high-temperature polymers. Chemists also use it to explore cyclization and condensation reactions for generating heterocycles. The fun comes with tradeoffs; improper conditions can lead to decompositions or, in rare cases, sensitive mixtures that pose handling risks.
Synonyms & Product Names
This molecule doesn’t hide behind many names. People recognize it as 1,5-Dinitronaphthalene, but older literature might call it Naphthalene-1,5-dinitro or simply DN-15. Some catalogs drop the naphthalene for “dinitro,” but savvy chemists track the numbers and positions—the details matter far more than the label.
Safety & Operational Standards
Anyone with hands-on experience knows safety trumps curiosity. 1,5-Dinitronaphthalene brings moderate toxicity and irritation risks, especially with repeat handling or airborne dust. Direct inhalation or skin contact should be avoided; goggles, gloves, and local ventilation aren’t just suggestions—people should view them as bare necessity. In my own lab work, spills meant weeks of deep cleaning, not just a quick swipe. Waste handling also commands respect, as the nitro groups break down slowly and pollute water or soil if not treated before disposal. Storage away from sunlight or strong oxidizers keeps surprises off the table. The risk profile has led workplaces and material handlers to adopt rigid standard operating procedures, combining personal vigilance with legal mandates.
Application Area
The real-world draw of 1,5-Dinitronaphthalene comes in its role as an intermediate. Dye makers turn to it, since the naphthalene core throws off vivid shades after chemical modification. Polymer chemists use the diamine byproducts when laying the groundwork for robust, high-temperature plastics. Some energetic materials research still circles this molecule, but the lower volatility and stability keep it out of the hands of most explosive-makers. I’ve come across its use as a stopover in multi-step laboratory syntheses, where its predictable behavior beats some of the more temperamental alternatives.
Research & Development
Academic circles don’t just chase established pathways—they push the molecule into new chemical spaces. Over the past decade, cleaner nitration methods, including use of ionic liquids and recyclable acid catalysts, have appeared in the literature. These fresh approaches draw interest for reducing waste and sidestepping harsh reagents, aligning with broader goals for greener chemistry. A handful of medicinal chemistry groups have looked at modified derivatives in antitumor research, banking on the nitro group’s ability to mess with metabolic pathways, though so far commercial translation sits out of reach. The molecule’s rigid framework also attracts researchers digging into charge-transfer materials and molecular switches, though the field waits for a breakthrough there.
Toxicity Research
Living with chemicals in the workplace means living with uncertainty—especially about long-term exposure. Studies on 1,5-Dinitronaphthalene sketch out a clear picture of moderate toxicity: laboratory animals exposed to large doses showed blood and organ disruptions, though outcomes depend on exposure route and frequency. Chronic exposure can lead to symptoms like headaches, nausea, and blood disorders, so standards push hard for limited, controlled usage. Environmental researchers have flagged incomplete waste degradation, highlighting potential bioaccumulation. These findings have fueled the call for research into rapid, on-site detoxification methods and better personal protective equipment for daily handlers.
Future Prospects
Everyone invested in organic synthesis watches shifts in regulations and technologies. Growing pain points around waste generation, green chemistry mandates, and worker safety mean that handling of 1,5-Dinitronaphthalene faces legitimate scrutiny. That said, as a reliable stepping stone molecule, demand isn’t vanishing soon. Upgrades to cleaner, safer preparation and reduction methods make a serious difference both for health and environmental impact. Improved analytical tools could pare down impurities and cut costs, boosting its reputation for reliability. Maybe years from now, safer nitration and reduction technologies will tip the scales further, opening doors to new applications in smart materials and pharmaceutical research. For now, the story continues—one foot in tradition, one eye on smarter ways of making and using molecules like this.
What is 1,5-Dinitronaphthalene used for?
The Role of 1,5-Dinitronaphthalene in Explosives Manufacturing
1,5-Dinitronaphthalene doesn’t spark recognition in most conversations. Still, this compound carries real weight in explosives production, especially in military and mining sectors. It’s a yellowish solid, not something you stumble across at the local hardware store. But if you dig into the world of synthesis, you’ll notice it’s important as an intermediate for creating more complex energetic compounds.
Digging deeper, I learned that manufacturers rely on 1,5-Dinitronaphthalene for making 1,5-diaminonaphthalene, which stands out in producing certain high-performance explosives like naphthalene-based nitramines. Materials like these end up in products such as plastic explosives, detonators, and propellants. The safety and performance of these devices matter—lives can depend on stability, reliability, and predictability. The synthetic process uses the two nitro groups on the naphthalene ring, which open pathways for modifications that give better control over sensitivity and energy release. Chemists argue over the details, but the demand remains.
Beyond Explosives – Other Applications
I’d guess not many people realize some dyes and pigments also depend on byproducts from this chemical. In the manufacturing of azo dyes, for example, 1,5-Dinitronaphthalene and related compounds help build deep, lasting color for fabrics and inks. The process is controlled, but traces of explosives chemistry make their way into things as common as a shirt or a printed poster. Naphthalene derivatives, created using the scaffold that 1,5-Dinitronaphthalene provides, play a quiet but crucial role in these industries.
Safety and Environmental Challenges
Handling toxic chemicals isn’t just another step in the laboratory. I’ve read stories about careless handling in small factories causing toxic spills or fires. 1,5-Dinitronaphthalene is no exception. Its toxicity calls for tight safety measures and sharp training. Inhalation or skin contact can cause significant health problems. The substance can contaminate water and soil, and accidental release may persist longer than people expect.
What comes next? Smarter containment and waste management. Chemistry doesn’t happen in a vacuum, so strong ventilation, sealed storage, and serious personal protective gear keep workers safe. Regulations direct how much of this compound can be stored, what to do in emergencies, and how to dispose of leftovers in a way that doesn’t threaten the environment. In larger plants, on-site incineration or neutralization processes help breakdown hazardous leftovers. I’ve seen some companies invest in closed-loop systems to keep chemicals in and pollution out. R&D teams also look for greener alternatives, though substitutes with the same performance and stability are not always available or affordable.
The Broader Picture
Even though 1,5-Dinitronaphthalene isn’t exactly a household name, it sits at a fork between science, industry, and public health. Every time we hear about new building projects or defense systems, chances are that compounds like this were in the mix behind the scenes. The push towards better safety, environmental care, and search for substitutes underscores the need for balance. Science drives progress, but responsibility shapes the future. Each link in the chain matters, from researchers fine-tuning a synthesis to workers suiting up in a chemical plant.
What are the safety precautions when handling 1,5-Dinitronaphthalene?
Why It Matters: Real Risks on the Table
1,5-Dinitronaphthalene isn’t a chemical you find by mistake. This compound has a long history in the manufacture of dyes and sometimes explosives, and it brings serious hazards. Workers exposed to its dust or vapors won’t just get a mild cough; the risks stretch from skin burns to potentially deep lung injury. The yellow crystalline powder looks harmless at a glance, but it packs enough punch to demand respect each time you handle it.
Respect Starts With Understanding the Hazards
Trust in a checklist won’t protect you on its own. You have to know why gloves, goggles, and proper ventilation aren’t extra steps—they’re your best defense. 1,5-Dinitronaphthalene irritates skin, erodes eye tissue, and can set off a chain reaction in your lungs after inhalation. If it makes its way inside, nausea and headaches won’t just ruin your day; repeated exposure increases the risk of dangerous blood disorders.
Let’s Talk About Good Gloves and More
Nitrile or neoprene gloves do the heavy lifting in protecting skin. Relying on thin latex won’t cut it here. If you work with open containers or clean up spills, full splash goggles and a face shield keep those yellow crystals out of your eyes and nose. A simple clothing lab coat only holds up if you launder it after every use and never take it home. Contaminated gear can take this hazard into your kitchen or car seat.
No Shortcuts With Ventilation
A dusty shop or poor airflow quickly multiplies your risk. Local exhaust ventilation—ideally a fume hood—keeps airborne particles at bay. Basic fans just push the problem sideways. Regular filter checks keep this gear working, and the numbers matter: aim for at least six air exchanges per hour in your work zone, based on NIOSH research.
Let the Data Make Your Decisions
Published studies back up the necessity for constant vigilance. The German MAK Commission, for example, lists strict airborne limits based on documented cases of methemoglobinemia—a blood disorder triggered by chemicals like 1,5-Dinitronaphthalene. Long-term effects aren’t speculative. The chemical can hang around in dust, sticking to surfaces and remaining dangerous after hours.
Labels, Training, and the Human Factor
A locked cabinet and a dated label don’t help if no one reads them. Each person in the lab or factory needs to understand the chemical’s story—and what it does inside the body. OSHA has fined firms for skipping briefings or letting new techs “learn as they go.” Solid hazard training and accessible safety data sheets turn near-misses into stories, not news reports.
Emergencies and Everyday Cleanups
Take spills seriously, no matter the size. Keep spill kits designed for nitroaromatic compounds within reach, including absorbent pads that actually bind the powder. Don’t grab paper towels or try to sweep with a dry broom. Use a vacuum fitted with a HEPA filter to avoid kicking dust back into the air. For eye or skin contact, multiple minutes of flushing with water can mean the difference between temporary pain and lasting injury.
Seeing the Bigger Picture: Better Design, Stronger Habits
The right infrastructure clears a path for safer work. Labs with segregated chemical storage and clear pathways keep mistakes rare. Fixing leaks and signs of corrosion on storage containers should happen before anyone comes near with a scoop or pipette. Trust is good, but double checks and clear records keep everyone honest—and healthy. Ultimately, taking 1,5-Dinitronaphthalene seriously gives each person the freedom to focus on the work, not the worry.
What is the molecular formula of 1,5-Dinitronaphthalene?
Peeking Inside the Molecule
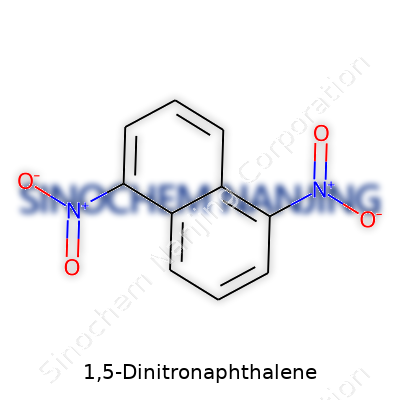
Asking about the molecular formula for 1,5-dinitronaphthalene might seem like a trivia question from high school chemistry, but this little detail opens up an entire world of science, industry, and even social responsibility. The formula—C10H6N2O4—looks simple on paper. That arrangement means a naphthalene ring with two nitro groups stuck at positions one and five. This structure defines its use, safety profile, and the way people interact with it in labs or factories. Looking up close, C10H6N2O4 is more than a chemical equation; it’s a story of molecules built for purpose.
Real Uses Demand Real Precision
My first encounter with dinitronaphthalenes took place at a university lab bench. We wore gloves for safety and worked under ahead of hoods because it quickly became clear that substances with nitro groups don't mess around. The arrangement of atoms in a molecule like 1,5-dinitronaphthalene determines its reactivity. Factories putting this compound to work in dyes, pesticides, or explosives can't afford to get their formulas muddled. That level of precision becomes a bedrock for safety protocols. Getting a single number wrong in the formula turns into an expensive, sometimes dangerous mistake.
Health and the Production Chain
Chemical safety comes straight out of understanding molecular structure. Inhaling or handling nitro-derivatives often involves health risks. The 1,5 variant sits right in that group. Proper labeling—complete with accurate molecular formulas—keeps workers from confusing one isomer with another, which can create toxic byproducts. People trust the numbers printed on safety data sheets. It’s more than paperwork; it can keep breathing air clean and groundwater safe. With so many manufacturing processes using naphthalene derivatives, a slip in formula can mean environmental or health issues. Those small details have a real-world impact when an accident happens.
Why Knowledge Stays Useful
Knowing why a molecular formula matters shifts the conversation from “what is this?” to “how do I handle this?” Sharing correct chemical information is a basic but crucial way to support E-E-A-T—experience, expertise, authority, and trust. Giving clear and accurate chemical facts shows respect for both the people in the shop and those living downstream. Teaching young scientists about formulas in the classroom, from memory drills to hands-on models, builds habits that stick. It also helps reinforce where these numbers show up outside a textbook, whether in pharmaceutical design or troubleshooting contamination.
Raising the Bar on Safety and Innovation
Chemical innovation lives by its details. Down-to-earth, the right formula builds a ladder for safer engineering, more effective research, and even compliance with hard regulations. Questions about 1,5-dinitronaphthalene's formula may look basic, but each answer connects to bigger choices in the lab, the plant, or the classroom. Open access to this kind of information can keep doors open for smarter questions and safer decisions moving forward.
How should 1,5-Dinitronaphthalene be stored?
Staying Practical About Chemical Hazards
Scientific research labs and chemical plants handle all kinds of substances, and some can stir up trouble if stored carelessly. 1,5-Dinitronaphthalene stands out on the list. This chemical, common in dye and pharmaceutical production, can present dangers beyond simple mishandling. My own early lab days taught me one thing: a small mistake with certain nitro compounds creates big headaches, and nobody wants an evacuation or an accident hanging over their head.
Understanding Real-World Risks
A molecule with two nitro groups on a naphthalene ring looks harmless in a glass jar. Reality proves otherwise. Nitroaromatics like 1,5-Dinitronaphthalene have a knack for thermal instability—meaning heat, friction, and direct sunlight can create hazardous situations in a hurry. Anyone who has ever seen a degraded sample of an old nitroaromatic will tell you—keep heat far away.
Storage Recommendations Backed by Experience
My chemist mentors often repeated the basics: keep such substances cool, dry, and protected from light. Sounds simple, but it saved me from learning tough lessons the wrong way. Shelves on the north wall, away from steam pipes and radiators, worked best. I always used tightly sealed amber glass bottles, placed on sturdy racks, inside secure cabinets that never got humid or warm.
Materials safety data from sources like PubChem and Sigma-Aldrich underscore these habits. They recommend locked chemical cabinets with ventilation, out of reach of unauthorized personnel. Fire-resistant storage never felt like overkill, especially with larger quantities. Double checking labels and avoiding storage near acids or reducing agents kept everyone calm and confident, which goes a long way in busy workspaces.
Environmental and Legal Reasons for Care
Disposal crews regularly remind us that spills or leaks involving 1,5-Dinitronaphthalene aren’t easy fixes. Traces in the wrong wastewater can cause headaches for entire communities. Many institutions face serious penalties under EPA rules for careless storage or disposal. This keeps everyone aware that safe storage goes well beyond personal safety. It protects colleagues, neighbors, and the company’s bottom line from fines or worse.
Solutions for Busy Workplaces
Busy labs can stretch people thin, but sound organizational habits help everyone breathe easier. Regular inventory checks, up-to-date safety training, and clear signage make all the difference. I have seen places where a lab manager posts a short checklist above every storage cabinet: temperature log, last inspection date, and a quick rundown of “never store these together.” This not only kept the rules in plain sight but also helped new staff ask the right questions before opening a bottle or moving containers.
Oversight by trained professionals—never students or interns alone—closes the loop on proper storage. Double-layered containment (such as secondary trays or tubs under containers) traps leaks from broken jars before they spread. Emergency contacts posted nearby make sure every staff member knows how to act if something does go wrong.
Moving Beyond Minimum Standards
Some folks see chemical regulations as red tape. My own experience tells a different story. Procedures built from small missteps or, worse, serious incidents, shape safer workplaces. Treating 1,5-Dinitronaphthalene with respect in storage isn't just about following the rules. It’s one way seasoned professionals show care for their team and their community, every day.
Is 1,5-Dinitronaphthalene hazardous to health or the environment?
Looking at the Risks
1,5-Dinitronaphthalene, a yellowish nitroaromatic compound, shows up in labs and factories as an intermediate for more complex chemicals. Most people won’t run across this powder at the grocery store. Still, anyone working with explosives, dyes, or specialty chemicals might find a bag of the stuff nearby. Exposure can happen, though, and that means the risks aren’t just theoretical.
What Happens to the Body
Breathing in 1,5-dinitronaphthalene dust can irritate the nose, throat, and lungs. That’s not unique among chemical powders, but nitroaromatics like this carry extra baggage. If particles get into the bloodstream, the nitro groups tend to cause red blood cells to lose their oxygen-carrying power. That leads to a condition called methemoglobinemia. People hit by this may notice blue lips or fingers, headaches, fatigue, or dizziness—classic signs their body isn’t getting the oxygen it craves. It’s not some remote chemistry concept. Emergency rooms see blue babies from contaminated well water or accidental poisonings with similar compounds several times a year.
Touching the powder leads to similar trouble. Skin exposure often ends in reddish or brown stains on hands or under nails. More dangerous, though, is the potential for these chemicals to slip through the skin and add to a person’s total exposure. That's what worries industrial hygienists—where protective gloves become an absolute must.
Environmental Fallout
Factories don’t always keep every molecule locked up. Leaks happen, as history has shown with chemicals like dioxins and other nitroaromatics. 1,5-Dinitronaphthalene doesn’t dissolve well in water, but it sticks to soil and sediments, where it can hang around for years. Fish and small creatures lurking in muddy rivers and ponds may take in more than their share. Researchers at the EPA found related compounds building up in the food chain, crowding out safer substances.
Breaking down nitroaromatics in nature usually takes specialized bacteria. Sunlight and oxygen can chip away, but the process drags on. It brings to mind problems near military test sites, where explosive residues trapped in soil keep showing up in water samples decades after weapons research chilled out.
Ways Forward
Prevention beats cleanup every time. In factories, that means sealed systems, dust control, and safety training. Workers deserve modern gear that protects against skin contact and inhalation, not just an old pair of safety glasses. Quick decontamination stations and good ventilation go a long way toward making an unsafe process less frightening.
On the environmental side, monitoring rivers and soil near manufacturing plants gives an early warning for trouble. Bioremediation — relying on bacteria that eat up nitroaromatics — holds promise but still needs more field tests. Real progress often stems from replacing risky chemicals with safer alternatives. Back in the 1990s, some dye plants switched raw materials rather than pay for elaborate waste handling.
Personal Takeaway
Anyone who has ever dealt with chemical safety paperwork knows just how easy it can be to overlook the slow dangers—chronic exposure, gradual buildup, small leaks that seem trivial until problems catch up. 1,5-Dinitronaphthalene offers a sharp reminder that what helps in industry may threaten health and nature if not handled with respect. A bit of vigilance and investment in protection can spare a mountain of trouble for workers and the ecosystem both.
| Names | |
| Preferred IUPAC name | 1,5-dinitronaphthalene |
| Other names |
1,5-Dinitronaphthalin 1,5-DNN Naphthalene, 1,5-dinitro- |
| Pronunciation | /ˌwaɪˌɛn.faɪˌdaɪˌnaɪ.trəʊ.næfˈθæliːn/ |
| Identifiers | |
| CAS Number | 602-31-1 |
| Beilstein Reference | 138388 |
| ChEBI | CHEBI:76244 |
| ChEMBL | CHEMBL22213 |
| ChemSpider | 198622 |
| DrugBank | DB02587 |
| ECHA InfoCard | 03b8c8f8-3572-4f1a-8e79-9b72438d2168 |
| EC Number | 210-594-5 |
| Gmelin Reference | 64868 |
| KEGG | C14134 |
| MeSH | D008384 |
| PubChem CID | 8662 |
| RTECS number | QQ9625000 |
| UNII | KFO4VGN1Q3 |
| UN number | UN1660 |
| Properties | |
| Chemical formula | C10H6N2O4 |
| Molar mass | 258.16 g/mol |
| Appearance | Yellow crystalline solid |
| Odor | Odorless |
| Density | 1.41 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.96 |
| Vapor pressure | 0.000017 mmHg (25°C) |
| Acidity (pKa) | 13.40 |
| Basicity (pKb) | 12.87 |
| Magnetic susceptibility (χ) | -81.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.658 |
| Dipole moment | 3.11 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 197.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 77.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3216 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 160 °C |
| Autoignition temperature | 410 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 2830 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2820 mg/kg (rat, oral) |
| NIOSH | SN10500 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
1,8-Dinitronaphthalene 1,3-Dinitronaphthalene 1,5-Diaminonaphthalene Naphthalene Nitronaphthalene |

