Understanding 1,5-Dihydroxy-4,8-Dinitroanthraquinone: Journey of a Unique Chemical
Historical Development
Chemists have chased discovery and color for as long as the discipline has existed, so it’s no surprise that 1,5-dihydroxy-4,8-dinitroanthraquinone found fame within the pages of dye chemistry. The late 19th and early 20th centuries saw a flourishing of organic dyes, with researchers continually searching for anthraquinone derivatives that brought resilience, vivid pigments, or unique reactivity to textile industries. This molecule, with its specific pattern of hydroxyl and nitro substitutions, stood out early for offering strong chromatic potential and chemical adaptability. Beyond color, the structure hinted at deeper possibilities: electron-withdrawing nitro groups and electron-donating hydroxy groups working together in a framework that proved both stable and versatile. This kind of discovery shaped not just clothing and fabric, but also advanced the understanding of organic electronic effects and conjugation in aromatic systems. Over time, research branched out from its roots, as these compounds began cropping up in organic photovoltaics and sensors, showing how historical development keeps guiding science down new paths.
Product Overview
Take 1,5-dihydroxy-4,8-dinitroanthraquinone, and most specialists think of a dense, often reddish-orange to brown solid, depending on purity and crystalline form. The attention it draws comes not only from color, but from the robust backbone of anthraquinone—a well-studied skeleton in organic chemistry. For decades, the scientific crowd used it mainly as a dye intermediate, but developments in analytical chemistry opened new windows. By the early 2000s, it started finding small but meaningful roles in research labs and specialty applications, especially where electron transfer and redox behavior needed harnessing. A blend of color chemistry, molecular stability, and capacity to accept or donate electrons, keeps this compound relevant for more than 100 years after its first synthesis. Even today, its story grows as researchers dig for answers in material science and biological signaling.
Physical & Chemical Properties
Anyone handling 1,5-dihydroxy-4,8-dinitroanthraquinone quickly notes a high melting point, pointing to a sturdy molecule with tightly packed aromatic rings. Most common anthraquinones resist breakdown, and this one follows suit, resisting decomposition up to several hundred degrees Celsius. It doesn’t dissolve well in water, reflecting its mostly nonpolar character, but it interacts better with polar aprotic solvents like dimethylformamide or dimethyl sulfoxide, traits it shares with other heavily substituted aromatics. Nitro groups add not just color but create potential for hydrogen bonding and even limited solubility in especially strong acids or bases. The molecule combines rigidity with just enough reactivity at its functional groups—a duality researchers learned to appreciate while seeking combinations of stability and modifiability.
Technical Specifications & Labeling
Chemists and laboratory suppliers classify 1,5-dihydroxy-4,8-dinitroanthraquinone by purity, particle size, and occasionally crystalline form, depending on intended use. The nitro and hydroxy functional groups must meet analytical thresholds; too much leftover precursor or unchanged anthraquinone reduces utility for sensitive experiments. Labels usually carry molecular weight and exact formula, with hazard notations tied to nitroaromatic toxicity. Any reputable source provides a certificate of analysis for lab use, backed up by spectrophotometric or chromatographic verification. Handling calls for personal protective equipment, given the risk from nitro compounds, and safe disposal practices center on controlling environmental exposure.
Preparation Method
Manufacturers usually start with anthraquinone or a related derivative, introducing hydroxy groups by directed substitution, while nitration reactions position nitro groups in the desired spots. Temperature controls and careful choice of reagents make a difference; too much heat or acid concentration results in unwanted over-reactions or side products. Once formed, purification becomes a challenge—a test of patience and skill—to remove tars or incompletely substituted molecules. Recrystallization and solvent extraction separate the finished product from impurities, drawing on generations of techniques honed in dye factories and fine chemical plants. Steps like these sound routine on paper, but it’s real hands-on work for lab techs who know just how sensitive nitroaromatic reactions can be.
Chemical Reactions & Modifications
The two hydroxy groups along with the nitro substituents open doors for further modification. Through standard chemistry, the hydroxy sites allow etherification, acylation, or even cross-coupling if advanced catalysts enter the mix. On the other side, nitro groups serve as launching points for reduction, amination, or cyclization, paving the way to synthesize whole families of related molecules. Chemists studying electron transport, redox reactions, or biological interactions prize these handles for creating targeted derivatives, often in research into antitumor agents or as probes for electron transfer mechanisms. Each reaction in the lab turns a simple pigment into a functional piece of a much wider scientific puzzle.
Synonyms & Product Names
Depending on the scientific source or product catalog, you see names like Dantron yellow, C.I. 58235, or its full systematic label under IUPAC rules, 1,5-dihydroxy-4,8-dinitro-9,10-anthraquinone. Old dye handbooks sometimes gave it color index numbers, reflecting its place among anthraquinone pigments, while organic chemists stick to concise chemical nomenclature. Some research papers refer to it by trade names no longer in common currency, but specialty manufacturers and academic suppliers nearly always use the longest formal version or the C.I. shorthand when clarity matters most.
Safety & Operational Standards
Working with this compound means facing risks typical of nitroaromatics—namely, toxicity by inhalation, possible skin absorption, and environmental persistence. Over the years, institutions developed strict protocols: gloves, goggles, and fume hoods are mandatory, as the compound ranks as hazardous. Waste must be collected and treated rather than washed away, a measure that speaks to its chemical stability and slow breakdown in the environment. As someone who’s helped carry out nitro compound workups, proper ventilation and spill control become second nature, because careless handling led in the past to accidents or contamination. These lessons persist in modern lab training, echoing advice from decades-old manuals paired with fresh updates in material safety data sheets.
Application Area
For most of its life, 1,5-dihydroxy-4,8-dinitroanthraquinone went straight into dyes for textiles and plastics, prized for the depth and fastness of its color. More recently, application areas expanded, including roles as redox indicators in electrochemical research or as building blocks for organic semiconductors. Some research groups explore its properties in photovoltaic materials, looking to capture sunlight with organic frameworks less toxic or expensive than their heavy metal counterparts. Analytical chemistry sometimes brings it out for specific reactions involving electron transfer, where nitro and hydroxy group arrangements give unique response patterns. Even new medical studies sometimes repurpose the molecule for imaging or as a scaffold in drug development, proof that an “old” molecule can find a fresh job.
Research & Development
Research circles circle back to molecules like 1,5-dihydroxy-4,8-dinitroanthraquinone precisely because their versatility keeps inspiring new uses. Material scientists care about how such molecules interact with surfaces, stack in thin films, or shuttle electrons across devices. Chemists appreciate simple yet tunable frameworks, especially with hybrids that link classic dye chemistry to modern applications in electronics, catalysts, or biological assays. Academic labs continue to publish findings on derivatives made by modifying hydroxy or nitro groups, often tailoring properties for specific sensors or as test cases in quantum chemistry simulations. R&D also moves towards greener synthesis and safer handling; finding routes that reduce waste or avoid hazardous reagents supports both industrial and academic sustainability goals.
Toxicity Research
Not all anthraquinones are benign, and nitroaromatics draw special scrutiny for both acute and chronic toxic effects. Animal studies and in vitro tests show varying degrees of cytotoxicity, especially involving liver and renal function; these findings spurred more careful assessment before use in any product touching humans or wildlife. Environmental impact remains a talking point, since persistence and low biodegradability complicate cleanup. Ongoing research probes deeper: tracking metabolism in model organisms, measuring byproducts, and testing cellular responses in tissue culture. Calls for replacement in many dye applications led to lower reliance on nitro-substituted anthraquinones, but their value in research or advanced materials keeps them under the microscope—quite literally—in toxicology labs worldwide. In my experience, debate often doesn’t end with just the chemical itself, but extends to its manufacturing byproducts and downstream waste.
Future Prospects
Looking ahead, the prospects for 1,5-dihydroxy-4,8-dinitroanthraquinone rest on both old strengths and new challenges. Research into organic electronics and flexible solar panels invites renewed attention, as scientists hunt for robust, affordable solutions that skip rare metals and complex fabrication steps. Changes in environmental and safety standards push ongoing R&D to make cleaner, safer derivatives or to engineer complete life-cycle management—including better degradation pathways. Green chemistry approaches aim to cut toxic reagents, produce less waste, and recycle or neutralize byproducts, making the legacy of this compound compatible with tomorrow’s regulatory and ecological needs. Conversations about the future rarely stray from the question: how much value can a molecule deliver if it fits responsible production, use, and disposal? A focus on careful research, open reporting of risks, and commitment to greener chemistry will shape the next chapter for molecules like this one.
What is 1,5-Dihydroxy-4,8-Dinitroanthraquinone used for?
An Unseen Player in the World of Color
Most people pass by shelves of bright red plastics, glossy automotive finishes, and daily-use printed goods without pausing to consider the chemistry behind those bold, reliable colors. Out of sight for almost everyone, compounds like 1,5-Dihydroxy-4,8-Dinitroanthraquinone quietly support these industries. This mouthful of a compound forms part of the foundation for many synthetic dyes and pigments. Years back, I had a chance to work in a lab that specialized in color chemistry for plastics and textiles. That experience showed me firsthand how materials engineers rely on just a handful of stable, tough pigments. This anthraquinone derivative comes up every time the job calls for deep reds or violets that don’t fade or break down under sunlight or heat.
Why Manufacturers Pick This Compound
Companies don’t use 1,5-Dihydroxy-4,8-Dinitroanthraquinone simply for its name. Chemically, it’s prized because it delivers intense color and stands up to environmental stress. Demand for colorfast red pigments keeps climbing, especially in automotive coatings, outdoor signage, and plastic furniture. If you walk through a playground or notice a row of fire trucks, the resilience of that color owes a lot to compounds like this one. Its chemical backbone shrugs off UV light, resists washing out, and doesn’t leach into the environment as easily as cheaper, less stable options.
Textiles, Printing, and Compliance
Textile dye-houses turn to it for lasting shades. Take a simple cotton T-shirt. No one wants to spend money and have it look dull after a few washes. Pigments built on these robust anthraquinone frameworks help deliver clarity and saturation even after months of wear. In the printing business, it provides fade-resistant inks. Think about outdoor banners staying crisp and readable all summer, or packaging that survives shipping and supermarket shelves without losing its impact. The safety aspect matters, too. Many countries restrict the use of certain dyes due to concerns about toxicity or environmental impact. Products based on 1,5-Dihydroxy-4,8-Dinitroanthraquinone meet established health standards, provided the manufacturing process follows proper protocols. This attention to compliance reassures both industry buyers and consumers.
Environmental and Safety Concerns
Problems appear when companies cut corners to reduce costs, or sourcing turns to poorly regulated suppliers. I’ve seen first-hand how rogue pigments cause issues in recycling streams, or when mismanagement exposes workers to harmful dusts. Responsible production involves handling this compound under strict industrial hygiene. Wastewater treatment needs careful monitoring so residues don’t enter rivers and soil. Europe and regions in East Asia now mandate cleaner production lines and push manufacturers toward green chemistry—alternatives using raw materials with lower toxicity and better biodegradability. The chemical in question still shows up in many applications, but watchful regulators and buyers keep pressure on suppliers to maintain transparency and limit environmental harm.
Paths Toward Safer Colors
It always surprises me how fast industry adapts when pushed. Some newer pigment chemistries build on the lessons learned from anthraquinones but tweak the structure to lower environmental risks. Big brands now invest in certification schemes, full life-cycle tracking, and transparent labeling. This keeps both consumers and workers safer, and it helps clean up downstream recycling. Better filtration, closed-loop processes, and careful sourcing should keep the legacy of strong, stable dyes without repeating mistakes of the past. As demand for sustainable goods rises, expect to see alternatives take over—but for now, 1,5-Dihydroxy-4,8-Dinitroanthraquinone still plays a dependable part on the color palette.
What is the chemical formula of 1,5-Dihydroxy-4,8-Dinitroanthraquinone?
Real Value Behind a Chemical Formula
People who mess around with molecules know every detail matters. The smallest tweak on a chemical ring shifts the whole story — and that’s pretty clear with 1,5-Dihydroxy-4,8-Dinitroanthraquinone. Its chemical formula, C14H6N2O8, stands out for more than just the numbers or the letters. Anyone who’s worked in a lab understands: naming a compound points to where the sticking points are for reactivity, color, and usefulness.
The Power of Arrangement
This isn't just a tangle of carbons, hydrogens, nitrogens, and oxygens. That combination, set on an anthraquinone backbone, opens doors in materials science and dye technology. Anthraquinone compounds earned a reputation for making deep colors stick around. Here’s where real experience comes in—older colleagues talk about yellow stains from certain anthraquinones refusing to leave the bench, no matter which cleaner they try.
Throw in two nitro groups and two hydroxy groups at those very specific positions. Now, the molecule isn’t just a pigment; it's a lesson in tuning. Those nitro spots change electron flow, and the hydroxy groups open routes for hydrogen bonding. Even if you haven’t spent extra hours reading journals, using dyes with this structure makes it clear: certain shades and stabilities come from chemical architecture, not just luck.
Applications and Real-World Impact
C14H6N2O8 doesn’t just sit around as a research oddity. Anthraquinone derivatives form the backbone for a pile of industry dyes, especially in textiles and inks. Synthetic chemistry let humans stop relying on crushed insects or rare minerals. Instead, tailored molecules like this one gave us brighter colors and better resistance to washing or sunlight.
Working in a textile lab, I saw plenty of complaints about colors fading. Shifts from natural dyes to robust synthetic compounds made a huge difference. People often don't realize how clever molecular tweaks transformed not just style but durability—something that gets overlooked if you don’t handle the stuff yourself.
Health, Environment, and Better Practices
Not every synthetic dye gets applause. Nitro-anthraquinones, just like some other nitro aromatics, raise questions about toxicity and wastewater. The facts support caution—some nitro compounds break down into toxic bits, and stubborn dyes clog up water filters. Handling and disposal rules stick around for a reason, and ignore them at your own risk.
One solution sits in research on greener synthesis — swapping out harsh reagents or developing dyes that break down cleanly after use. Companies and labs push toward safer production cycles by listening to workers, reading real data on chemical exposure, and substituting risky molecules where they can. My own experience in a custom dye facility made it clear: supply chain transparency and updated safety protocols don't grow from wishful thinking; they grow from calling out hazards and trying better ways.
Wrapping Up the Significance
The formula C14H6N2O8 gives more than a snapshot of atomic makeup; it hints at decades of science packed with tradeoffs between performance, safety, and sustainability. The next time someone sees a vibrant synthetic color, remember that behind it stands a massive body of chemical know-how, experiments, and probably a few stained fingers.
Is 1,5-Dihydroxy-4,8-Dinitroanthraquinone hazardous or toxic?
Understanding the Risks
Curiosity about a chemical’s safety shows up quickly when its name runs several syllables deep, like 1,5-dihydroxy-4,8-dinitroanthraquinone. Plenty of sites call it a dye intermediate or pigment, but the story does not end with industrial use. Anyone who has watched the bright colorations of anthraquinones turning objects vivid red or orange has likely wondered if that jar on the shelf brings hazards along with color. Digging past the jargon, let’s look honestly at the questions people bring up about toxicity.
What Happens With Contact or Exposure?
People working in dye factories face dust from anthraquinones nearly every day. Eyes and lungs know it first. In my experience working near pigment production, nosebleeds and skin rashes came up if safety steps got skipped. 1,5-dihydroxy-4,8-dinitroanthraquinone, because of its nitro groups, falls into the pile of nitroaromatics, a family known for causing real problems if inhaled, touched, or—worse—ingested. Research points to nitroaromatic compounds sometimes breaking down in the body to form more reactive species, disrupting red blood cells and bringing on methemoglobinemia (a condition that keeps blood from carrying enough oxygen).
Laboratory studies on similar anthraquinones showed persistent skin and respiratory irritation. Eating the stuff stands as an obvious bad idea. Even dust in the air can be enough to cause nose and throat irritation. That matches what occupational health records show in long-term manufacturing: high rates of dermatitis, headaches, and sometimes riskier effects like dizziness and mild anemia, all tied to both skin and inhalation exposures.
Where Does the Cancer Risk Stand?
Some folks remember hearing about anthraquinone dyes showing up on health risk lists. That reputation begins in part with certain kinds of anthraquinones—especially those with nitro groups—being flagged as possible carcinogens. Studies on animal tests found particular nitro-anthraquinones increased the chance of cancer when taken in big amounts for a long time. The World Health Organization cases out chemicals this way before labeling them, and while not all anthraquinones carry the same weight of proof, enough share structural features to draw concern. In fact, a study published in Environmental Health Perspectives called anthraquinone derivatives “reasonably anticipated to be human carcinogens” if people get exposed regularly or in large doses. Nobody, whether in a research lab or on an industrial floor, should treat exposure as safe.
Safer Habits and Smarter Solutions
Handling 1,5-dihydroxy-4,8-dinitroanthraquinone safely means more than gloves. A decent fume hood matters if mixing or weighing powders. Respirators—those tighter-fitting kinds—made a clear difference in air monitoring results at pigment plants I watched. Dermal exposure can slip past thin gloves, so chemical-resistant gear and thorough washing routines come standard in companies that care about workers’ health. Good ventilation limits airborne fine particles. Continued air monitoring and regular health checks spot trouble before it grows.
Alternatives to nitroanthraquinone pigments exist. Biobased and less reactive pigments come out of labs each year, looking for the same shade without the same risks. Replacing high-hazard dyes with safer blends cuts the danger for everyone involved in production.
What It Means Beyond Industry
For the general public, 1,5-dihydroxy-4,8-dinitroanthraquinone does not end up in household products often, but older consumer goods and some textiles can still contain residues. Washing hands after handling dyed fabrics and skipping mouth contact with art materials helps. In workplaces, safety habits build a barrier between potential toxicity and long-term health. Factories that train and outfit workers properly, along with regular safety audits, make all the difference.
What is the solubility of 1,5-Dihydroxy-4,8-Dinitroanthraquinone in water?
The Science and Real-World Implications
I’ve worked with a fair share of stubborn compounds in the lab, and 1,5-Dihydroxy-4,8-Dinitroanthraquinone stands out as especially tough. Anyone expecting this compound to dissolve in water like salt lands quickly in a puddle of disappointment. This molecule, a derivative of anthraquinone, clings to its solid form and barely acknowledges water as a solvent. Academic records and chemical handbooks back up that notion: its solubility in water barely makes it to the measurable mark, clocking in at less than 0.1 mg/mL. Call it negligible—this is practically off the scale for most applications needing a dissolved compound.
Why Solubility Matters
As someone who has seen dyes, pigments, and molecular markers in action, solubility isn’t just a fancy term on a bottle. It decides if a compound plays well in biological testing, wastewater purification, and even industrial dyeing. For a chemical like this anthraquinone derivative, poor water solubility limits its use in any process that relies on even distribution.
Many researchers and industry professionals want to use vivid, stable dyes, but poor water-solubility forces a creative workaround. This compound’s chemical structure—packed with aromatic rings and topped off by nitro and hydroxy groups—locks it into a low-solubility state. Water molecules struggle to break apart these packed solids. Instead, this compound floats or settles rather than dissolves. That bodes fine for pigment formation, but for solutions and aqueous processes, it’s a real headache.
Evidence and Health Considerations
Younger chemists sometimes think they can strong-arm solubility with heat or stirring. In my early days, I tried that too—boiling, sonicating, even adjusting pH. The results never matched the ease of dissolving something like sodium chloride. The chemical structure simply resists. Facts from the Merck Index and chemical safety sheets agree: expect extremely low solubility. Beyond handling and application, low aqueous solubility usually signals limited bioavailability, which matters in environmental monitoring and health risk analysis. When a dye washes out into waterways, poor water solubility can limit the spread—sometimes making removal easier but possibly persisting as micro-particulates instead.
Looking Forward: Solutions for Low Solubility
Some researchers head for organic solvents—dimethyl sulfoxide (DMSO) or ethanol—to coax stubborn compounds into solution. Others try surfactants, which wrap around the molecule and smuggle it into water. In technical settings, these solvents or additives come with handling rules. Human health and environmental guidelines place real limits on solvent use, especially in manufacturing or biomedical work. Pure water solutions stay the safest, but with this dye, that’s off the table without creative chemistry.
Formulating powders, suspensions, or using nanoemulsions turns out to be a more realistic direction for such a compound. Some companies tackle tough solubility by grinding the substance to nanoparticle sizes, which increases surface area and gives water a fighting chance. You might see these in advanced inks and specialized sensors today. Careful process development and transparency about limitations mean safety and efficiency don’t have to give way to laboratory frustration.
Staying Grounded in Science and Experience
Working with chemicals that don’t want to dissolve often forces inventiveness. The case of 1,5-Dihydroxy-4,8-Dinitroanthraquinone makes solubility more than a textbook number—it’s a deciding factor in research, safety, and industrial progress. Documentation, facts, and practical handling tips rooted in experience make scientific work and product development both safe and effective.
How should 1,5-Dihydroxy-4,8-Dinitroanthraquinone be stored and handled?
Recognizing Real Risk
Working with 1,5-Dihydroxy-4,8-Dinitroanthraquinone, I’ve seen how overlooked chemical safety can have fast, costly consequences. This stuff, often used for pigments and dyes, brings both color and hazard into any lab or factory where it lands. Nitrated anthraquinones don’t mess around; their sensitivity to heat, friction, and even sunlight means lax storage invites trouble. That reminder stacks up quick in shared lab spaces where one careless shelf or leaky bag becomes a department-wide headache.
Mitigating Hazards Starts with Smart Storage
Leaving a reactive chemical on a shelf near oxidizers, acids, or solvents asks for disaster. I’ve stepped into storerooms thick with fumes—a sure sign someone lumped incompatible materials together. With 1,5-Dihydroxy-4,8-Dinitroanthraquinone, you separate it from potential reactants every time. Flammable cabinets do the trick, especially metal ones with clear, chemical-resistant labeling. I never toss it into a wooden drawer or leave it in sunlight. Direct light will start breaking down the compound, especially over long weekends or summer heat waves.
Controlling Environment Beats All Else
Humidity’s the sneaker here. Moisture in the air creeps in, making powders clump or liquefy, and can make clean-up a pain. I trust sealed glass jars, or tough HDPE containers with tight lids. A desiccant packet in the storage cabinet ups the odds of dry storage, and silica gel shows a visible sign when saturated. I’ve opened caked-together samples more times than I care to admit, all due to absentmindedly skipping this step. Keeping the temperature steady, well under 25°C, puts another buffer between you and an accident. Industrial fridges, kept separate from food, work well for this sort of chemical. Never pack too tight—good airflow in the storage area keeps it safer for everyone who steps in.
Personal Protective Gear Stands Between You and Regret
Accidents can happen fast, especially with powders. I put on gloves (nitrile or neoprene, not latex as it degrades), a dust mask, and tight-fitting goggles. A long-sleeved coat acts as a second line of defense against spills. The point? Even careful folks slip up. Eye-wash stations and emergency showers should stand close by, never tucked away behind stacks of boxes. Once, a mislabeled container cost a visiting researcher a whole morning flushing her eyes because a splash hit while moving samples at speed. She wore goggles, which made all the difference, but not everyone remembers to suit up every single time.
Clean-Up and Waste: The Overlooked Frontier
Disposal bins marked clearly for hazardous chemical waste stop mixing disasters before they start. Never rinse small spills down the sink or mop them into the floor. Adsorbent materials—vermiculite or powder spill kits—grab any loose product fast. I always double-bag waste, and tag it for pick-up by professionals. Local regulations shape the last leg of safe handling, but skipping a step—or worse, improvising—cuts corners in a way that often circles back. Direct training and signs posted at eye-level do more for safety than thick manuals gathering dust in a drawer.
Building a Culture of Caution
1,5-Dihydroxy-4,8-Dinitroanthraquinone won’t forgive mistakes. Good habits—double-checking labels, tracking expiry dates, never rushing transfers—pay off. Peer checks, real training, and straight conversations about near-misses keep risks visible. I’ve learned that it isn’t about stoking fear; it’s about respect for a material that transforms industry and research, but never without cost when treated carelessly. Treat storage and handling with focus. There’s always more riding on it than a simple sample.
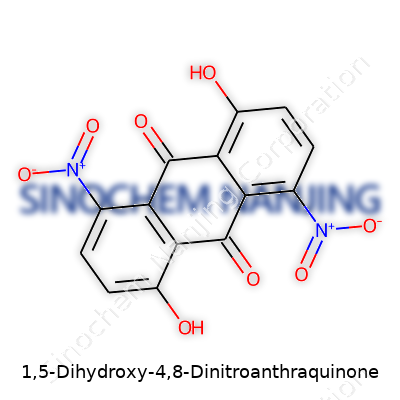
| Names | |
| Preferred IUPAC name | 1,5-dihydroxy-4,8-dinitroanthracene-9,10-dione |
| Pronunciation | /ˈwʌn.faɪv.daɪˈhɒk.si.fɔːrˌeɪt.dʌɪˈnaɪ.trəʊˌæn.θrə.kwiː.nəʊn/ |
| Identifiers | |
| CAS Number | 117-25-5 |
| Beilstein Reference | 124054 |
| ChEBI | CHEBI:9199 |
| ChEMBL | CHEMBL362277 |
| ChemSpider | 184792 |
| DrugBank | DB14049 |
| ECHA InfoCard | ECHA InfoCard: 100.023.444 |
| EC Number | 1.10.3.2 |
| Gmelin Reference | 147765 |
| KEGG | C15815 |
| MeSH | D014062 |
| PubChem CID | 68672 |
| RTECS number | CB5950000 |
| UNII | V6Z4T764CX |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C14H6N2O8 |
| Molar mass | 348.19 g/mol |
| Appearance | Red powder |
| Odor | odorless |
| Density | 1.76 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.61 |
| Vapor pressure | 4.1 x 10^-9 mmHg (25°C) |
| Acidity (pKa) | 7.92 |
| Basicity (pKb) | 13.75 |
| Magnetic susceptibility (χ) | -80.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.738 |
| Dipole moment | 6.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -492.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1397 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | D10BA01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P302+P352, P305+P351+P338, P337+P313 |
| Flash point | Flash point: 295.2 °C |
| Lethal dose or concentration | LD50 (oral, mouse): 4000 mg/kg |
| LD50 (median dose) | LD50 (rat oral): 2836 mg/kg |
| NIOSH | LH8225000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |

